Abstract
Introduction
The control of infectious bacteria is greatly hindered by their propensity to develop resistance to antibiotics which is a major public health challenge globally.
1
The widespread misuse and overuse of antibiotics in both medical and agricultural settings have significantly accelerated the development of resistant bacterial strains. Among these,
Among the natural products studied for their antibacterial properties, beetroot (
Moreover, combining two or more plant extracts has been shown to enhance bioactivity against different types of bacteria.15–17 This effect could be due to either the combined action of different bioactive compounds without alteration of their chemical structure or the chemical interaction between multiple bioactive compounds resulting in the formation of new compounds or their derivatives which are more potent than their precursors. Studies on the chemical interactions that occur when plant extracts are combined for enhancing their bioactivity is scarce. These studies are crucial as they could provide insights into the potential mechanisms through which bioactive compounds exert their effects on specific pathogenic microorganisms. Organic solvents commonly used for extracting aromatic compounds which are common natural antibacterial compounds in guava and beetroot include alcohols (such as ethanol and methanol), esters, ketones, and hydrocarbons. Each solvent has unique properties that make it suitable for extracting specific types of compounds. Ethanol was particularly selected due to its wide application in extraction of biomolecules based on its polarity which allows it to dissolve a wide range of bioactive compounds. Since different biomolecules have different optimal concentrations, 70% was selected as a moderate concentration. This study, therefore, explored the effect of combining guava leaves and beetroot ethanolic extracts on their bioactivity against
Materials and Methods
Materials
An autoclave was used for sterilizing agar plates, glassware, and other equipment to prevent contamination. A laminar flow hood was used to provide a sterile work area for preparing agar plates and handling microbial cultures. An incubator was used to maintain a controlled environment for the growth of microorganisms at selected temperatures. A UV-Vis spectrophotometer (LAMBDA 265 UV/Vis Spectrophotometer, by PerkinElmer, Waltham, Massachusetts, USA) was employed to measure absorbance of the compounds present in the extracts before and after combining. A refrigerator stored culture media, reagents, and microbial cultures at appropriate temperatures. Sterile cotton swabs or inoculating loops were used for spreading the bacterial inoculum on agar plates. Petri dishes held the solidified agar medium during the disc diffusion assay. A glass spreader was utilized for spreading beetroot and guava leaf extracts on agar plates. An analytical weighing balance was used to measure the quantity of beetroot and guava leaves for the extraction process. A standard strain of bacteria
Methods
Sample and Reagents Preparation
The preparation of guava leaf and beetroot extracts was carried out according to the method described by Chaari et al with modifications. 18 Three kilograms of fresh guava leaves were collected, washed to remove dirt and impurities, and dried in a well-ventilated room until a constant weight was achieved. The dried leaves were then ground using a mortar and pestle to increase the surface area for extraction. Thirty grams of powdered guava leaves were measured using a weighing balance and placed into a sterile 250 mL Erlenmeyer flask, followed by the addition of 150 mL of 70% ethanol. The mixture was covered with aluminum foil to reduce evaporation and stored in a dark room for three days at room temperature (24 ± 2 °C). After three days, the extracts were filtered using Whatman filter paper No. 4, and the filtrates were concentrated using a rotary evaporator at 70 °C. The fresh beetroots were similarly washed to remove dirt and impurities, then chopped into smaller pieces and dried to constant weight. The procedure used for the preparation of the guava leaf extract was then repeated for the beetroot. The resulting filtrates were concentrated and stored at −18 °C until further use.
The media were prepared according to the standard methods 19 with slight modifications. Nutrient Agar (1.4 grams) was dissolved in 50 mL of distilled water, boiled with gentle swirling to completely dissolve the medium, and then sterilized in an autoclave for about 20 min at 121 °C and 15 psi. The medium was allowed to cool to approximately 45–50 °C while ensuring it remained in a liquid state before being poured into sterile petri dishes and allowed to solidify. Similarly, 15.2 grams of Mueller-Hinton Agar (produced by MilliporeSigma, Burlington, Massachusetts, USA) was dissolved in 400 mL of distilled water, boiled with gentle swirling to dissolve the medium completely, and sterilized in an autoclave for about 20 min at 121 °C and 15 psi. The medium was allowed to cool to around 45–50 °C while ensuring it remained in a liquid state before being poured into sterile petri dishes and allowed to solidify. The experiments were done in duplicates and average values used in plotting graphs. The sterile distilled water was used as negative control while ciprofloxacin (produced by Actiza Pharma, Gujarat, India) 30 µg/ml as positive control.
The
Determination of the Antibacterial Effect of Individual Guava Leaves and Beetroot Extracts on Staphylococcus aureus
The beetroot and guava leave crude extracts solutions were prepared separately by dissolving 5 g of the prepared extracts concentrate into 2.5 mL of sterile distilled water in separate sterile glass beakers. This stock solution was then used to prepare the serial dilutions of concentrations from 1000 mg/mL, 500 mg/mL, 250 mg/mL, 125 mg/mL, 62.5 mg/mL and 31.25 mg/mL of crude extract of beetroot and guava leaves. The filter paper discs of 6 mm were soaked in the measuring cylinders containing different concentrations of extracts (1000 mg/mL, 500 mg/mL, 250 mg/mL, 125 mg/mL, 62.5 mg/mL and 31.25 mg/mL) of the beetroot and guava leaves extract solutions. The discs were then left for 24 h to be thoroughly impregnated with the extract solution. The bacterial suspension was then swabbed on the surface of Mueller-Hinton Agar plates followed by the application of the impregnated filter paper disks containing different concentrations altogether with positive and negative control maintaining a standard distance between the discs to avoid overlapping of zones of inhibition. 21 The plates were incubated at 37 °C for 24 h and observed for zones of inhibition formed. The diameters of the zones of inhibition around each disc were then measured using a ruler and recorded in millimeters (mm). The effect of different concentrations and minimum inhibitory concentration was obtained by comparing the diameters of zones of inhibition at different concentrations with the negative and positive controls.
Determination of the Antibacterial Effect of Combined Beetroot and Guava Leaves Crude Extracts on S. aureus
The combined beetroot and guava leaf crude extracts were prepared in a 1:1 ratio by first creating a stock solution of 2000 mg/mL. This stock solution was then used to prepare serial dilutions with concentrations of 1000 mg/mL, 500 mg/mL, 250 mg/mL, 125 mg/mL, 62.5 mg/mL, and 31.25 mg/mL. The procedure used for individual extract was then repeated.
Determination of the Optimal Inhibition Time of the Combined Beetroot and Guava Leaves Extracts on S. aureus
To determine the time at which maximum inhibition is achieved, the procedure employed in determination of antibacterial effect was employed using combined guava leaves and beetroot crude extracts at 62.5 mg/mL while varying time from 1, 3, 6, 18 and 24 h. The development for zones of inhibition was measured after specified time up to 24 h. The measurements were recorded in millimeters and used to deduce the rate of inhibition of the combined extracts.
Determination of the Chemical Interaction Between the Bioactive Compounds in Combined Beetroot and Guava Leaves Extract
To determine the effect combining beetroot and guava leaves extracts on chemical interaction of the bioactive compounds in crude extracts of individual beetroot and guava leaves, a UV/Vis spectrophotometer (LAMBDA 265 UV/Vis Spectrophotometer, by PerkinElmer, Waltham, Massachusetts, USA) was used according to the standard methods22,23 with modifications, where, about 62.5 mg/mL of crude extract (guava leaves and beetroot) was subjected into the UV/Vis spectrophotometer and scanned for absorbance at different wavelength. The same concentration of the combined extract was used to establish changes in absorbance. Differences in the readings were used to deduce the possible chemical interactions in the bioactive compounds resulting from possible chemical interactions and possible compounds which play key role in the process.
Statistical Analysis
The experimental results for the bioactivity of the crude extracts used were recorded using mean values from duplicate data and calculated using MS Excel version 2021. The strength and direction of relationship between studied variables was determined by calculating the correlation coefficient using MS Excel version 2021.
Study Limitations
This study did not involve compound isolation and identification in the crude extract. This led to limit the interpretation and conclusions made. Moreover, the mechanisms of chemical interactions resulting in formation of new compounds or functional groups was not determined. In statistical analysis, this study opted for the use of correlation and data visualization using only average values.
Results
The Antibacterial Effect of Individual Guava Leaves and Beetroot Extracts on Staphylococcus aureus
The zones of inhibition observed indicate that as the concentration of guava leaf extract and beetroot extract increased from 31.25 mg/mL to 1000 mg/mL, the size of the zones of inhibition also increased from 6 mm to 18 mm and 16 mm, respectively (Figure 1) with correlation coefficient of 0.87. In comparison, the guava leaf extract, prepared using 70% ethanol, exhibited a higher inhibition than the beetroot extract. This finding indicates that the guava leaf extract was more effective in inhibiting the growth of

The antibacterial effect of guava leaves and beetroot extracts on
The Effect of Combining Beetroot and Guava Leaves Extract on Their Antimicrobial Activity Against S. aureus
Combining beetroot and guava leave extracts showed increased inhibition against

The effect of combined guava leaves and beetroot extracts on
The Optimal Inhibition Time of the Combined Beetroot and Guava Leaves Extracts on S. aureus
When the concentration of the combined extract was fixed at 62.5 mg/mL and the exposure time varied from 1, 3, 6, 18, and 24 h, there was a noticeable increase in the zone of inhibition, ranging from 6 mm to 10 mm (Figure 3). Initially, after 1 to 3 h of exposure, the zone of inhibition measured 6 mm, which could indicate that the antibacterial action of the combined extracts began to manifest after 3 h. This initial phase suggests that there might be a lag period where the bioactive compounds in the extracts start interacting with the bacterial cells, initiating the antibacterial process. As the exposure time increased to 6 h, the zone of inhibition expanded to 7 mm, indicating a steady progression in antibacterial activity. This gradual increase suggests that the extracts’ effectiveness against

The effect of time on antibacterial activity of combined guava leaves and beetroot crude extracts.
The Effect of Chemical Interaction Between the Bioactive Compounds in Combined Beetroot and Guava Leaves Extract
The guava leaf extract at a concentration of 62.5 mg/mL exhibited peaks ranging from 200 to 320 nm, in the UV/Vis spectrum with prominent characteristic peaks at 275 and 300 nm (Figure 4). The UV/Vis spectrometry curve for beetroot extract typically shows absorption peaks at various wavelengths as shown in Figure 5. The absorption peaks in the UV/Vis spectrum of beetroot extract observed around 245–280 nm is associated with aromatic amino acids like tryptophan and tyrosine. 8 Other absorption peaks around 300 to 365 nm are attributed to the presence of betalains in beetroot. 8 The combined extract spectrum revealed peaks ranging from 200 to 400 nm, indicative of tannins, aromatic flavonoids, and betalains present in the mixture. The absorption between 365 to 400 nm which was not present in the individual extracts could suggest the modifications in the phenolic flavonoids present in the individual extract resulting in formation of their derivatives. Moreover, the peaks at 245–280 nm showed decreased absorption in the mixture than in individual extracts (Figure 4–6) suggesting involvement of these compounds in the formation of new derivatives of betalains with absorption from 365–400, 475 and 675 nm. Additionally, new peaks at 475 nm and 675 nm were observed, suggesting the possible formation of carotenoids and fluorescent compounds such as fluorophores. The presence of these new peaks is therefore an indication that the mixture undergoes certain chemical transformations, enhancing the bioactivity of the extract. Such modifications could potentially be exploited in the development of novel therapeutic agents with improved efficacy. Furthermore, the increased absorption at higher wavelengths may reflect a broader range of biological activities of the studied materials, offering more versatile applications in pharmaceuticals and nutraceuticals. The formation of new derivatives also implies that the combined extract might possess weak synergistic effects, potentially providing enhanced health benefits compared to individual extracts underscoring the importance of combining extracts to exploit the full spectrum of bioactive compounds and their potential health benefits.
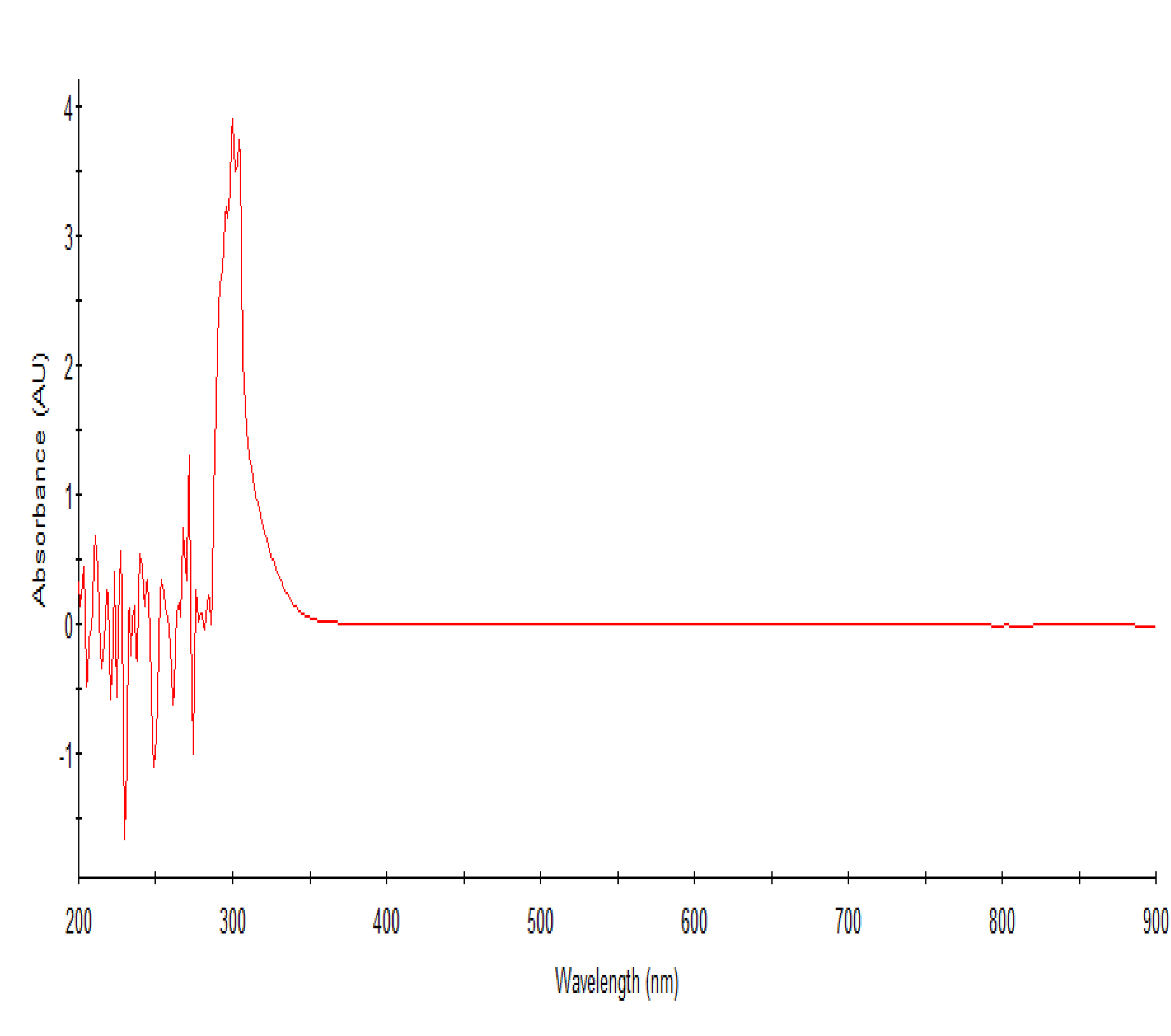
The UV/Vis spectrum of guava leaves crude extract.
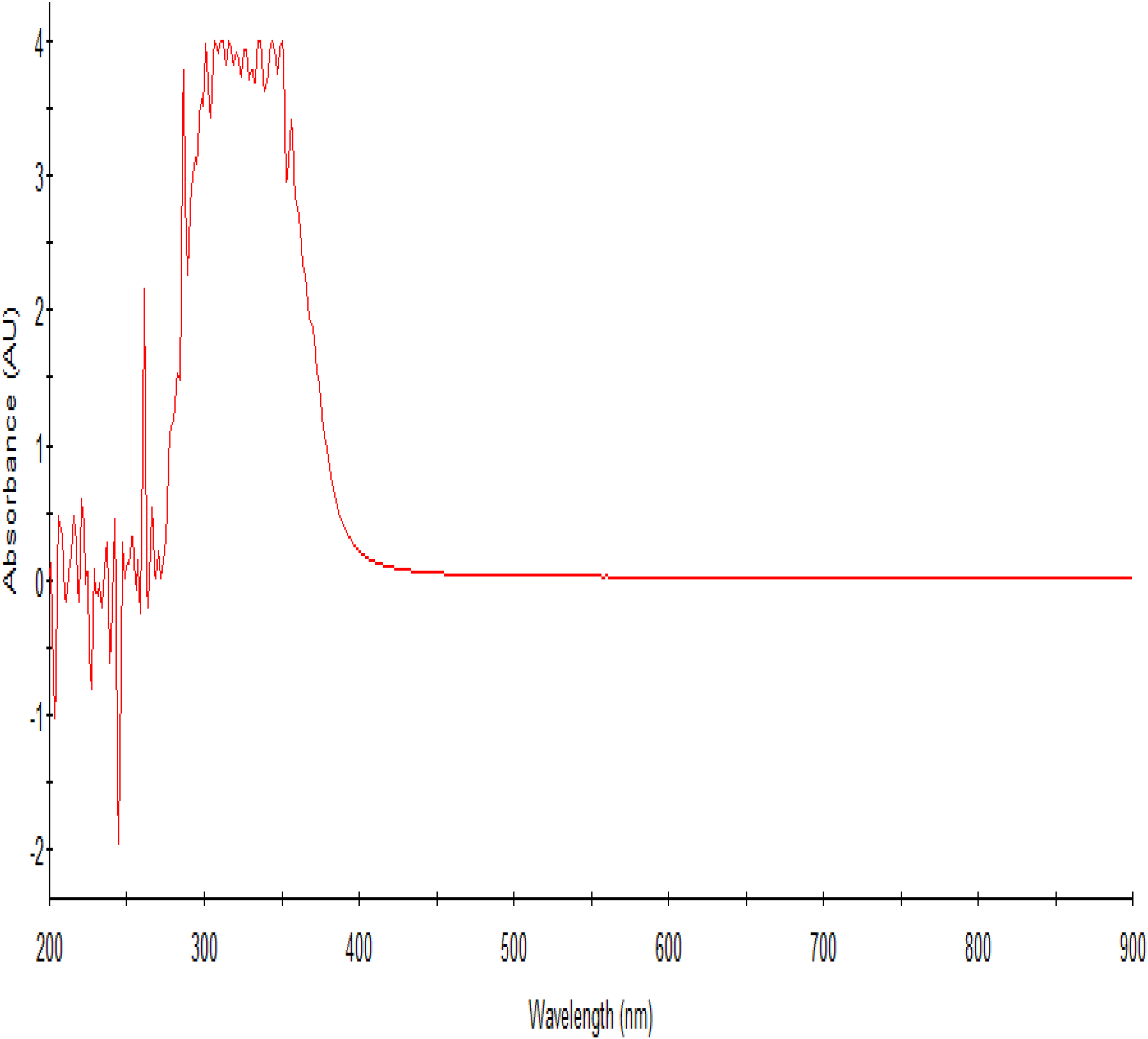
The UV/Vis spectrum of beetroot crude extract.

The UV/Vis spectrometry of combined guava leaves and beetroot crude extract.
Discussion
The efficacy of guava leaves extracts reported in this study was higher than what was reported by a scientist in South Africa 15 and lower than what was reported by a similar study. 7 The differences in the antibacterial activity observed could be attributed to the different concentrations of ethanol used in preparation of the crude extracts.7,15 This could further suggest that the concentration of ethanol used in extraction of crude extract is directly related to the antibacterial potency of the crude extract obtained. The active compounds in beetroot and guava are likely to be aromatic amino acids and flavonoids, such as tyrosine and quercetin which are highly extractable using alcohol as extractant. 7 This assertion could be supported by the absorbance at 220–250 nm, 270 nm, 280 nm, 320 nm, and 375 nm in the UV/Vis spectrophotometer readings (Figure 4 and 5). These peaks are characteristic for flavonoids and phenolic compounds, which are known for their antimicrobial properties. Flavonoids, in particular, are effective in precipitating microbial proteins and disrupting cell membranes, thereby inhibiting bacterial growth. The phenolic compounds, including flavonoids, have been shown to possess significant antimicrobial activity due to their ability to disrupt microbial cell walls and interfere with essential enzymatic functions. The increased zones of inhibition observed with higher concentrations of the extracts suggest a dose-dependent response, where the efficacy of the extracts in inhibiting bacterial growth increases with concentration.
Investigating on Figure 2, representing inhibition, and Figure 6, representing spectral shifts after combining indicates that the compound responsible for antimicrobial activity could be derivatives of betalains formed because of chemical interaction between flavonoids and phenolic amino acids in the mixed extracts. This could be further indicated by changes in absorbance at 225 to 320 nm and 330 to 400 nm between Figure 4, 5 and 6 and FIC calculated. 25
The increase in inhibition with increase in time demonstrates that the combined extracts’ antibacterial action continues to intensify with extended exposure and that the action is slow. The time-dependent nature of this response could suggest that the bioactive compounds within the extracts maintain their potency and continue to exert antibacterial effects over time. 6 This sustained activity could be due to the presence of various compounds within the extracts that work synergistically to inhibit bacterial growth. This gradual yet steady increase in antibacterial efficacy suggests that these extracts could be valuable in developing treatments that require sustained antibacterial effects, particularly in clinical and food preservation applications.
Absorbance at 200–320 nm are indicative of the presence of tannins, phenol-containing flavonoids, and carbonyl-containing aromatic rings within the extract (Figure 4). The presence of these compounds suggests that the bioactivity of the guava leaf extract may be primarily attributed to the aromatic flavonoids that were successfully extracted using ethanol. 26 Tannins are known for their ability to precipitate proteins and form strong complexes with metal ions, which can contribute to their antimicrobial properties. The phenolic nature of tannins allows them to disrupt microbial cell membranes and inhibit enzymatic activity, thereby providing a protective effect. 10 In addition, the presence of phenol-containing flavonoids enhances the extract's antioxidant activity. Furthermore, the carbonyl-containing aromatic rings detected in the guava leaf extract are often involved in various chemical interactions that could further enhance the antibacterial properties of guava leaves extract. The combination of these aromatic compounds, extracted using ethanol, creates a potent mixture capable of effectively inhibiting the growth of a wide range of bacterial pathogens.10,15,27,28 The peaks observed in the UV/Vis spectrum of the guava leaf extract provide valuable insights into its chemical composition and potential bioactive properties. The presence of tannins, phenol-containing flavonoids, and carbonyl-containing aromatic rings highlights the complexity and potency of the extract. These findings underscore the importance of using ethanol as a solvent in the extraction process, as it efficiently isolates these aromatic flavonoids, enhancing the extract's bioactivity.10,23,25
The observed new peaks and reduced absorbance in the other observable in Figure 4–6 can be attributed to the chemical interactions between the different bioactive compounds present in guava leaves with those in beetroot extracts. Since absorption between 245–280 nm is associated with phenolic compounds like tyrosine in beetroot extract, which are precursors of betalains, and flavonoids such as quercetin in guava leaves extract, it can be asserted that the tyrosine and flavonoids, whose absorbance decrease in the combined extract are converted to derivatives of the betalains in the combined extract which appear as new peaks.
The absence of these peaks in the individual extracts implies that mixing the beetroot and guava extracts may lead to the modification of alkenes into carotenoids through the formation of new bonds providing increased conjugation. Interestingly, these newly formed compounds are reported to have antibacterial activity.29,30 Their formation, therefore, like the formation of derivatives of betalains explained above, could have a role to play in enhancing bioactivity of the combined extract. This interaction between the bioactive compounds in guava leaves and beetroot may suggest the potential of such combinations in both clinical and food preservation applications.
Conclusion
This study provides valuable insights into the synergistic antibacterial efficacy of combined ethanolic extracts of guava leaves and beetroot extracts against
Footnotes
Acknowledgements
The authors are thankful to the research project panel in the Department of Applied Sciences for their valuable comments and technical advice given to researchers during study design, data collection and analysis.
Authorship Contribution Statement
Ester Vicent Mauya: Writing, Conceptualisation, Editing, Investigation and Data analysis. Hezron Timothy Mwakabona: Conceptualisation, Writing, Editing, Data interpretation, Reviewing.
Declaration of Conflict of Interests
The authors have no conflict of interest to declare in relation to authorship and publication of this article.
Ethics Approval
The study design and approach were in line with the Research Ethics policy and guidelines of the Mbeya University of Science and Technology Version 2023 and Tanzania Commission for Science and Technology First Edition, 2020 and approval for the design and approach was granted. Our institution does not require any other approval for this kind of study provided that the Departmental Research Panel approves the design.
Funding
The authors are thankful for the waiver for accessing laboratory services from the Mbeya University of Science and Technology during research work. Mbeya University of Science and Technology, (grant number 2024).
Statement of Human/Animal Rights
No human/animals were used in this study
