Abstract
Medicinal plants have long been sought after as a source of potentially valuable compounds to combat cancer. This review purposes that the phytochemicals found in Nepalese medicinal plants possess significant potential anticancer activity, which calls for methodical evaluation. The study explores the methodologies employed in screening the anticancer activity of medicinal plants found in Nepal. Full-text original research articles were searched on “Google Scholar” and “PubMed” using keywords “Anticancer” and “Medicinal plants”. The obtained plant species in the articles were looked over in the National Herbarium and Plant Laboratories (KATH) Plant Database to confirm their existence in Nepal. A list of 71 plant species showing anticancer activity was obtained. Active phytochemicals and screening methods of the plants reported in the articles were then analysed. The 71 plants contained 84 bioactive compounds, namely 7 alkaloids, 10 polyphenols, 1 saponins, 5 phytosterols, 29 terpenoids, 11 flavonoids, 3 polysaccharides, and 18 other miscellaneous compounds that showed anticancer activities. Likewise, 77.78% of the studies used cell line-based in vitro experiments while 6.94% of the methods employed animal-based in vivo methods, and the remaining 15.28% combined both approaches. In particular, 54.51% of the papers used multiple cell lines, while the remaining used single cell lines. These findings highlight the trend of screening methods, significance and potential of these plants in anticancer properties.
Introduction
Cancer, a global health challenge, is a disease characterized by uncontrolled cell growth and contribute significantly on morbidity and mortality rates around the world. The core of cancer treatment has long been conventional approaches like chemotherapy, radiation, and synthetic medications. 1 However, their serious side effects and limitations has shifted focus to alternative therapeutic options like plant derived compounds, often rich in phytochemicals such as polyphenols, flavonoids, alkaloids, tannins, and volatile oils, which exhibit pharmacological activities that can mimic essential endogenous compounds, hormones, and signal transduction molecules, providing promising avenues for cancer treatement. 2 As a result, the use of phytochemicals in anticancer research has gained tremendous momentum, evident with accelerating publications emphasizing their potential from 1992 to 2023. Bibliometric analysis shows that the primary research in this domain is concentrated in fields such as Pharmacology & Pharmacy, Plant Sciences, and Biochemistry & Molecular Biology. 3
Recent reviews have highlighted the increasing importance of phytochemicals in the treatment of cancer. For instance, Garcia-Oliveira et al collected information on anticancer agents derived from plants, emphasizing the effectiveness of flavonoids, camptothecin derivatives, and vinca alkaloids in clinical applications. 4 For evidence of the potential of natural products in the treatment of cancer comes from clinical trials conducted on a number of compounds derived from plants. Research on curcumin, resveratrol, taxol, genistein, epigallocatechin gallate such plant-derived compounds from traditional medicine are more frequently being used in clinical application. 5 The increasing prevalence of cancer worldwide has heightened activities in the search for alternative therapeutic options outside conventional treatment methods. Plants have gained special attention due to the wide variety of their bioactive compounds, which act on tumor growth at different stages. Unlike synthetic drugs, these plant-derived compounds, very often, act synergistically and through multiple pathways, such as oxidative stress reduction. DNA repair modulation, and immune system enhancement. 6 Several globally recognized plants, including Curcuma longa, Taxus species established the immense potential of natural products in oncology.
Nepal possesses an extensive range of medicinal plants with remarkable properties that have been utilized in traditional healing practices for centuries.7,8 In Nepal, there are 5309 species of flowering plants belonging to 1515 genera and distributed across 193 families. 9 Approximately 1515 to 2331 medicinal plants have been traditionally used for sustenance, home remedies, and traditional medicines.8,10 Natural compounds derived from medicinal plants are of particular interest due to their potential anticancer effects. To assess this potential, different screening methods are used to evaluate their anticancer properties. These screening methods range from in vitro cell-based assays to in vivo animal models and provide invaluable insights into these plant-derived compounds. These compounds, often possessing low toxicity and minimal side effects, are more biocompatible with the human body. 11 Therefore, this review hypothesizes that the phytochemicals derived from Nepalese medicinal plants have immense potential as anticancer agents. This study gives review on Nepalese plant species that demonstrated anticancer activity and their methodologies for their screening. This enables on the integration of Nepalese plants in the global initiative of anticancer drug discovery.
Methods
This study explores the methodologies employed in screening the anticancer activity of medicinal plants from Nepal. Comprehensive searches were conducted using online databases “Google Scholar” and “PubMed” with keywords “Anticancer” and Medicinal plants” in the title. The search focused on original research articles published between 2003 to 2023, excluding review articles and other non-original research articles for further analysis. The selected articles were reviewed to compile list of plant species studied for their anticancer properties. To verify the native occurrence of these species, each plant was cross-checked against the Herbarium plant database of National Herbarium and Plant Laboratories (KATH). Subsequently, the articles featuring plant species that demonstrated anticancer activity and were confirmed to exist in Nepal were thoroughly examined with specific focus on anticancer phytochemicals reported and documenting the methodologies for their screening.
Results and Discussion
Anticancer Medicinal Plants of Nepal
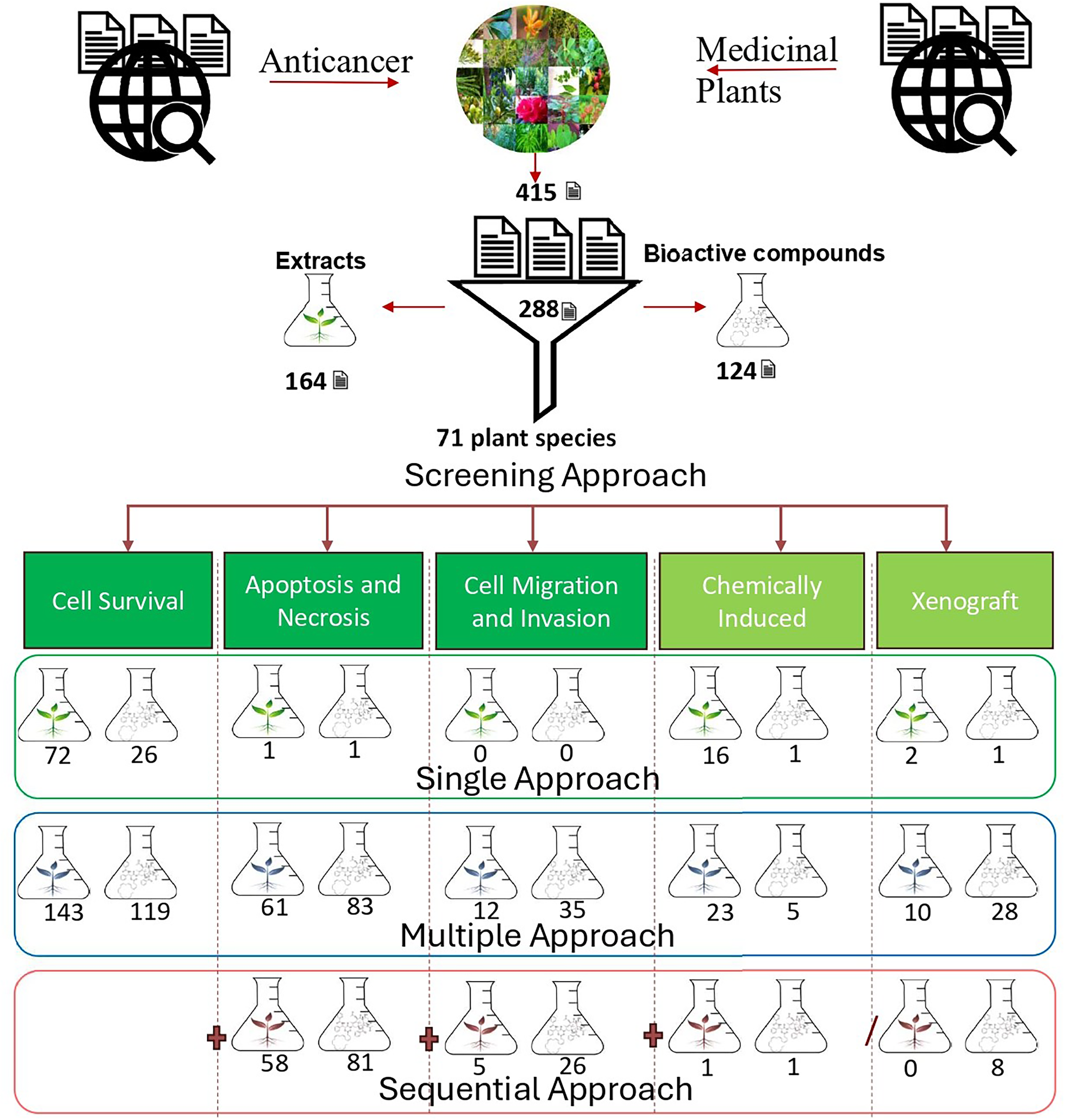
There has been a growing interest in exploring the potential anticancer properties of medicinal plants. The rich botanical resources of Nepal present a unique opportunity to explore the hidden potential in the ongoing research against cancer. 12 In this study “Google Scholar” and “PubMed” were searched for full-text original research articles published from 2003 to 2023 which resulted in 415 articles and associated medicinal plants. Next, the presence of the plants in Nepal was verified using the National Herbarium and Plant Laboratories (KATH) Plant Database of Nepal. This yielded 288 articles on 71 plant species with anticancer properties. The list of plants is provided in Table 1. Among the total 288 papers, 164 (56.95%) used plant extracts, and 124 (43.05%) used isolated bioactive compounds. The distribution suggests nearly equal exploration of both crude extracts and purified compounds, reflecting the interest in identifying anticancer agents from natural sources.
Medicinal Plants Found in Nepal with Anticancer Bioactive Compounds and Screening Methods of Anticancer.

Screening method used
Active Phytochemicals in Nepalese Medicinal Plants
The phytochemicals that have demonstrated their therapeutic effect against cancer include alkaloids, polyphenols, saponins, phytosterols, terpenoids, flavonoids, and polysaccharides. 13 The 71 plants listed in Table 1 contained 84 bioactive compounds, namely 7 alkaloids, 10 polyphenols, 1 saponins, 5 phytosterols, 29 terpenoids, 11 flavonoids, 3 polysaccharides, and 18 other miscellaneous compounds (Table 1) that showed anticancer activities. Among these, some of the compounds that reached clinical trial phase 3 includes Artemisinin, derived from the Artemisia genus,14,15 Berberine isolated from Berberis vulgaris,16,17 Curcumin, a polyphenolic compound derived from Curcuma longa,18,19 taxol extracted from Taxus wallichiana,20,21 Cannabidiol, reserpine, and withaferin-A like compounds.
Screening of Anticancer Compounds in Plants
The process of identifying potential anticancer compounds from medicinal plants demands a systematic approach and relies heavily on appropriate screening methods. It typically begins with the collection of plant material, followed by an extraction process using suitable solvents to obtain crude extracts. Subsequently, fractionation techniques are employed to isolate specific compound(s) from these crude extracts. The fractions or isolated compounds are then evaluated for their anticancer properties. 295 While clinical trials are the ultimate test, they are often hindered due to ethical, medical, financial constraints, and the limited number of eligible patients. Hence, preliminary screening methods that are more accessible play a fundamental role. This method includes both in vitro and in vivo techniques. However, the evaluation of anticancer compounds using the techniques necessitates selecting suitable cancer cell lines to determine the effectiveness of these substances. 296
In this study, it was observed that cancer cell lines were used in 271 (94.09%) papers for screening and evaluating anticancer compounds. Out of these, 157 (54.51%) papers used multiple cell lines, while the remaining 114 (39.58%) used a single cell line in their studies. Similarly, 95.6% of the cell lines used were human cell lines, 3.58% were murine cell lines, and 0.75% were from other animal sources. Figure 1 represents the types of human cell lines used for this purpose. Among these, breast cancer cell lines were the most commonly used, accounting for 24.21% of the studies. This could be attributed to breast cancer being the second most common cause of cancer-related death, 297 causing it to be one of the most commonly studied cell lines for investigating compounds with potential anticancer properties. Other frequently used cell lines included colon, cervical, lung, liver, prostate, and leukaemia cancer cell lines, representing 13.78%, 9.45%, 9.25%, 8.07%, 7.68%, and 5.51%, respectively. Additionally, other human cell lines such as melanoma, ovary, and other parts of the human body were also used but less frequently, collectively representing 22.05% of the studies. This diversity reflects the wide range of cancer types being studied. Surprisingly, only a small proportion of research evaluated the anticancer effects on normal cells, highlighting a gap in understanding the selectivity of these active compounds and their potential impact on normal cell physiology.

Cell lines used in screening and evaluation of anticancer compounds.
In Vitro Approaches
In vitro methods are utilized to evaluate various aspects such as cell viability, proliferation, apoptosis assessment, cell cycle progression, and cell migration and invasion for the assessment of anticancer agents.298,299 These methods play a crucial role in the initial stages of anticancer drug development by identifying promising compounds. This significance is underscored by the substantial number of papers, 268 (93.06%), that employed in vitro methods for screening and evaluating anticancer compounds. Among these, 224 (77.78%) papers exclusively utilized in vitro methods, highlighting their essential role in the identification of promising compounds (Figure 2).
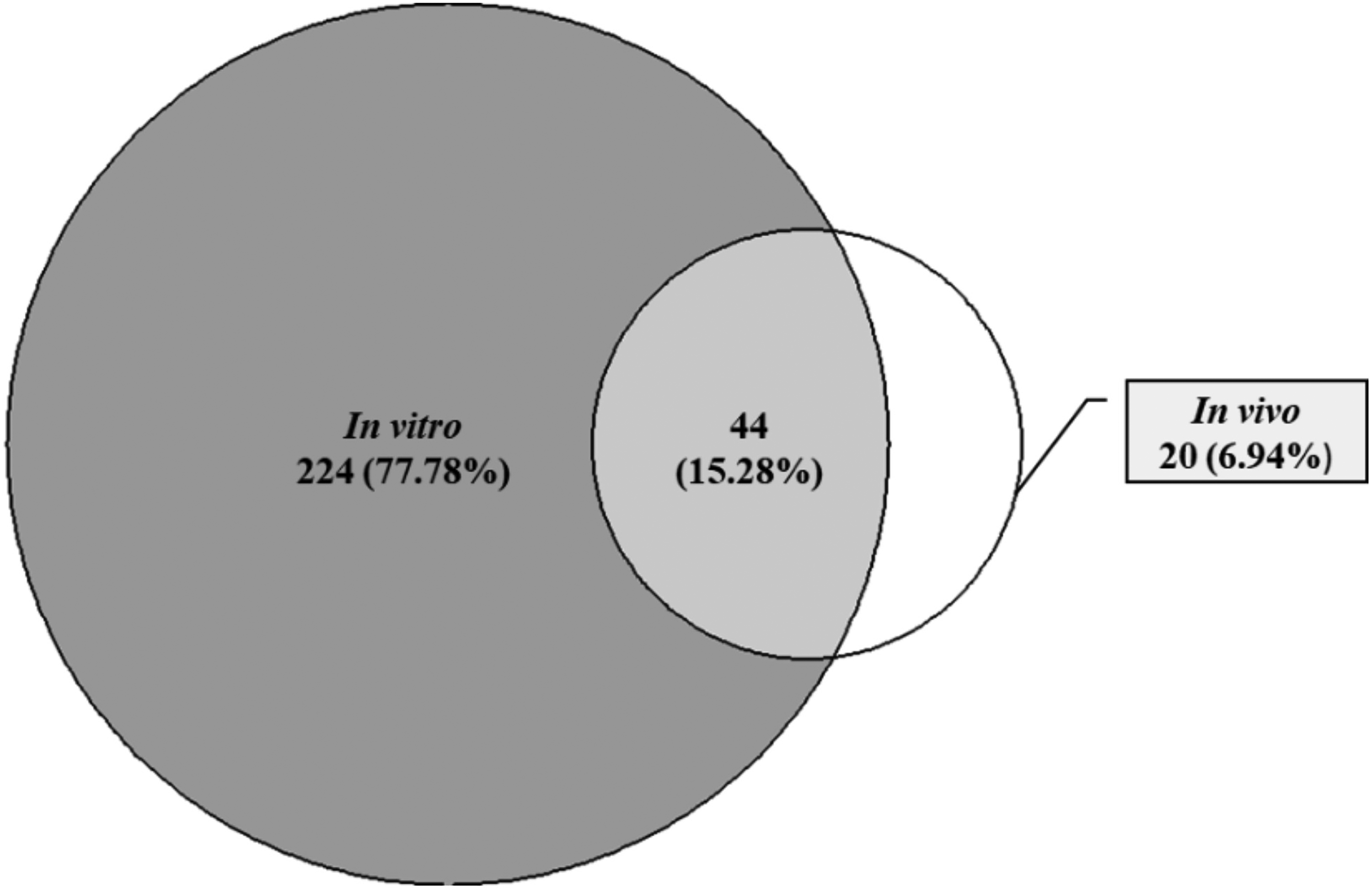
Approaches used in screening and evaluation of anticancer compounds.
In Vitro Cell Survival Assay
Cell survival assays, including cell proliferation, viability, and toxicity assays, serve as an initial assessment, providing insight into the overall health and survival of cells following exposure to various compounds. One widely used method for assessing cell viability, proliferation, and toxicity is the MTT assay. This method measures cellular metabolic activity by introducing tetrazolium salt, MTT (3-(4, 5-dimethylthiazol-3-yl)-2, 5-diphenyl tetrazolium bromide), which is converted into a colored formazan product by viable cells. 22 Similarly, the Alamar Blue assay offers insights into cellular metabolic activity, growth characteristics, and safety profiles. In the broader spectrum of in vitro assays, additional methods such as the XTT, 23 MTS, 41 Trypan Blue Exclusion, Presto Blue assay, 91 SRB (Sulforhodamine B) assay, WST-8 assay, Bromodeoxyuridine (BrdU) incorporation, Anchorage-independent Colony Formation assay, Clonogenic assay, LDH release assay, and cell proliferation by RTCA provide diverse approaches for evaluating proliferation, cytotoxicity, and growth characteristics are used.95,112,173 These assays are known for their simplicity, reliability, and compatibility with high-throughput screening, playing a collective role in determining the effects of compounds on cells, thus making them integral to biomedical research. For instance, cytotoxicity and the selectivity of Annona muricata and Terminalia bellirica extracts were studied by the MTT assay on both cancerous cell lines, and normal cell lines. It was observed that the extracts acted in dose dependent manner in cancerous cells while having minimal effects on normal cells, hence giving very promising selectivity.300,301,266
It was observed that a significant proportion of the studied research papers, 262 (90.97%), used cell survival assays along with other in-vivo and in-vitro methods, including apoptosis, invasion, cell migration, and invasion for screening and evaluating anticancer compounds, as illustrated in Figure 3. Out of these, 119 (41.32%) papers utilized bioactive compounds isolated from plants, and 143 (49.65%) used plant extracts in their studies. Additionally, 98 (34%) papers employed only cell survival assay methods for screening and evaluating anticancer compounds, with 26 (9%) using isolated compounds and 72 (25%) studying plant extracts. This demonstrates that in the continuum of in vitro screening for potential anticancer agents, viability, proliferation, and cytotoxicity assays are equally crucial, offering deeper insights into the impact of these compounds on the growth and division characteristics of cancer cells, as well as their safety profiles.

Distribution of papers by cell survival method and cell survival along with other methods for screening and evaluation of anticancer compounds of plant extracts and plant isolated compounds.
In Vitro Apoptosis Assays
Apoptosis and necrosis assays are pivotal for assessing the potential anticancer properties of medicinal plants, shedding light on their impact on cell death pathways. A plethora of techniques are harnessed to discern apoptosis and necrosis, each offering unique insights into the mechanisms at play.302,303 Flow cytometry, a robust tool in this context, scrutinizes alterations in cellular morphology and DNA content, enabling the discrimination of early and late-stage apoptosis and necrotic cells. This is achieved through the use of fluorescent dyes such as Annexin V, Propidium Iodide, Acridine orange/Ethidium bromide, Hoechst 33342 binding to DNA or phosphatidylserine exposed in the outer membrane of apoptotic cells.25,36,63 Concurrently, DNA fragmentation assays, such as the TUNEL (Terminal deoxynucleotidyl transferase dUTP Nick End Labelling) assay, identify DNA breaks characteristic of apoptosis. Labelled nucleotides are incorporated into fragmented DNA ends, allowing for quantification via fluorescence or immunohistochemistry. 16
Annexin V-FTIC staining and Propidium Iodide (PI) staining are also common techniques for apoptosis detection. Annexin V, a protein with a high affinity for phosphatidylserine, is labelled with a fluorochrome (eg, FTIC) and binds to phosphatidylserine on the outer membrane of apoptotic cells. 50 In parallel, PI staining is widely employed for assessing apoptosis in various experimental models. Nowadays, the Annexin V, Yo-Pro-1, and PI methods are often combined for comprehensive apoptosis detection. 26 Additionally, these methods provide valuable information about alterations in cell cycle distribution, highlighting the interconnectedness between apoptosis and cell cycle regulation. Furthermore, researchers may explore Caspase activity assays, DAPI staining, measurement of Mitochondrial membrane potential (MMP), intracellular ROS analysis, immunohistochemistry, and RNA expression analysis, all of which offer multifaceted approaches to delve into the intricate dynamics of cell death pathways. The induction of apoptosis as represented by caspase activity and changes in the expression of genes such as BAX and BCL2 represents a key indicator of anticancer agents. In one study, in vitro apoptosis assay revealed that the mechanism of action for anticancer activity from extracts of Annona muricata is the upregulation of proapoptotic genes like BAX and caspase-3/7 and downregulation of anti-apoptotic genes such as BCL2. 301 These assays collectively provide indispensable tools for probing the anticancer potential of medicinal plants and their ability to induce programmed cell death. 304
When examining studies that integrate apoptosis assessment with other screening phases (Figure 4), a total of 144 (50.0%) articles were identified as performing such evaluations. Among these studies, 61 (21.18%) papers utilized plant extracts, while 83 (28.82%) used isolated compounds. Furthermore, 139 (48.26%) papers performed apoptosis after cell survival studies, of which 58 (20.14%) studies were based on plant extracts, while 81 (28.13%) studies used plant-isolated compounds. This distribution highlights the consistent exploration of apoptosis in conjunction with other cellular processes, emphasizing its integral role in elucidating the mechanisms underlying the cytotoxic effects of medicinal plants. Interestingly, only 2 (0.69%) papers, 1 for plant extract and 1 for plant isolated compounds performed only the apoptosis method for screening and evaluating anticancer compounds.

Distribution of papers on apoptosis, apoptosis along with cell survival, and apoptosis along with other methods for screening and evaluating anticancer compounds from plant extracts and isolated compounds.
In Vitro Cell Migration and Invasion Assays
In the quest to uncover the anticancer properties of medicinal plants, an essential avenue of investigation involves the utilization of migration and invasion assays to scrutinize their impact on the movement of cancer cells. Several methodologies, including the Boyden Chamber assay, encompass transwell migration assays and scratch wound healing assays. 84 The Boyden Chamber assay, a versatile term encompassing a range of migration assay variations within a transwell system, allows us to delve into the cell invasion potential. It introduces an additional layer, such as the basement membrane matrix or Matrigel, faithfully replicating the extracellular matrix encountered by cells during their invasive journey. 77 By incorporating medicinal plant extracts or compounds into these assays, we gain the means to assess their influence on cell invasiveness.
The transwell migration assay utilizes a porous membrane embedded in a transwell insert, a well-established method in the scientific community. This assay plays a crucial role in quantifying the effects of medicinal plant extracts by observing cell migration towards chemoattractants. On a simpler note, the scratch wound healing technique, a straightforward yet effective method, assesses both cell movement and wound closure. Its widespread adoption underscores its effectiveness in studying wound repair dynamics in response to medicinal plant extracts or compounds. 305 These assays collectively serve as essential tools in uncovering how medicinal plants impact the migration, invasion, and wound healing of cancer cells. Their frequent use and popularity underscore their vital role in enhancing our understanding of potential anticancer agents derived from the diverse world of medicinal plants.
An account of studies conducted on cell migration is presented in Figure 5. It reveals that a total of 47 (16.32%) papers utilized cell migration in conjunction with other methods for screening and evaluating anticancer compounds. Among these, 12 (4.16%) studies employed plant extracts, while 35 (12.15%) studies focused on plant isolated compounds. Additionally, 31 (10.76%) studies combined cell migration and invasion assays with cell survival and apoptosis studies, with 5 (1.74%) studies using plant extracts and 26 (9.30%) studies centered on plant isolated compounds. This sequential integration enables researchers to investigate the interconnectedness among various cellular processes and their collective impact on cancer progression.
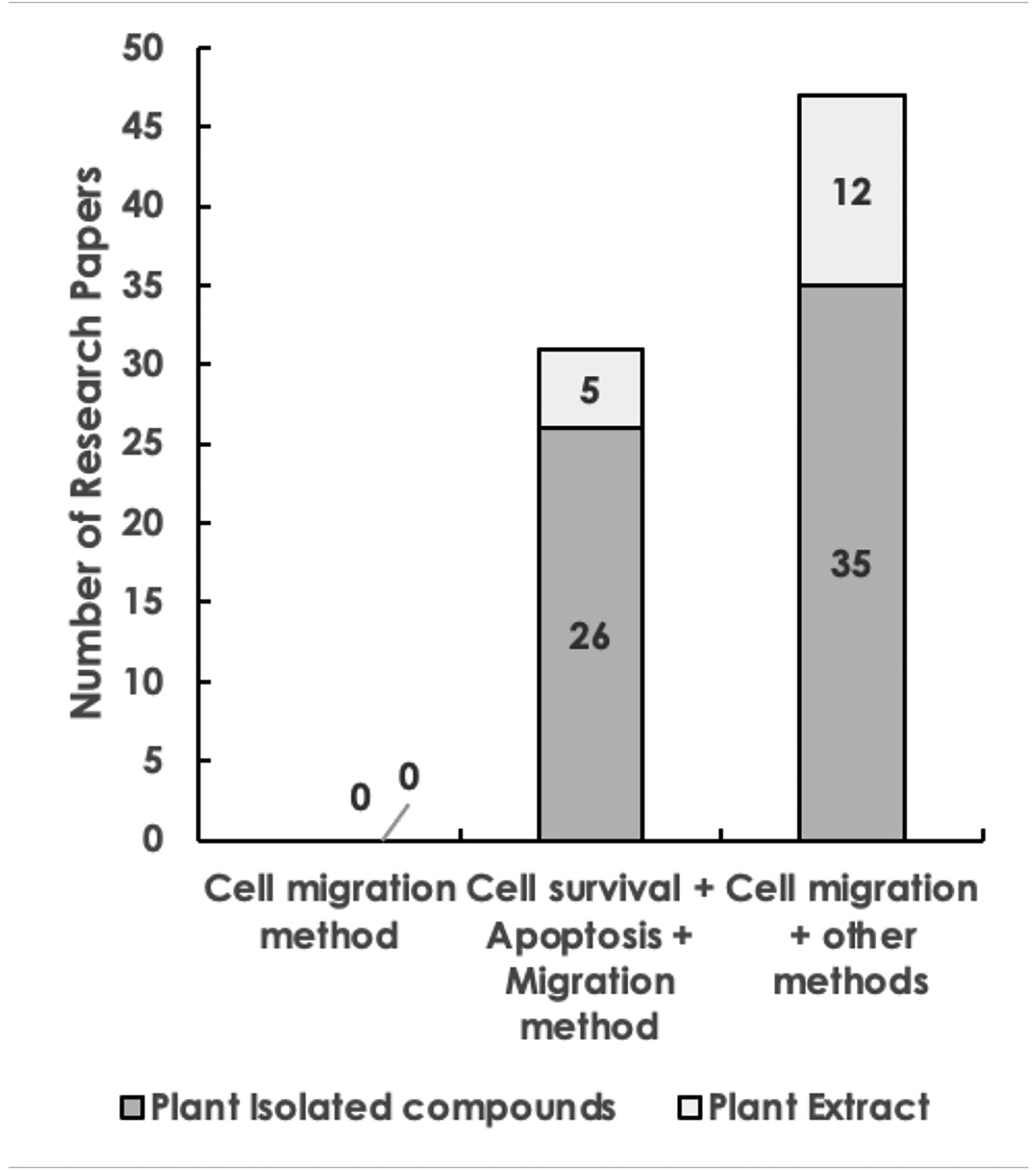
Distribution of papers by cell migration, cell migration along with cell survival and apoptosis, and cell migration along with other methods for screening and evaluation of anticancer compounds of plant extracts and plant isolated compounds.
In Vivo Approaches
In vivo studies delve into the intricate mechanisms underpinning the actions of anticancer agents, offering valuable insights into their real-world performance within living organisms. The in vivo experiments provide a crucial bridge between the laboratory and clinical applications. 306 This study revealed that 64 (22.22%) of the articles studied used both in vitro and in vivo methods. Out of these, 20 (6.94%) of the papers exclusively utilized in vivo methods, such as tumor xenograft and chemically induced animal models. These approaches, often employing animal models like mice or rats, facilitate an exploration of how plant-derived compounds interact with the body's intricate systems and their impact on cancer progression.307,308
In Vivo Chemically Induced Tumor Models
The administration of specific chemicals or carcinogens to animals, often rodents are involved in chemically induced tumor models, to induce the development of tumors.309,310 These models allow researchers to study the effects of medicinal plant extracts or compounds in the context of cancer prevention and treatment. 311 For instance, plants like Azadirachta indica, Rauwolfia serpentina, Swertia chirayita, Terminalia chebula, Tinospora cordifolia, Berginia ciliata, Cannabis sativa, Cuscuta reflexa, Cyanodon dactylon, Podophyllum hexandrum, Rubia cordifolia, and Zanthoxylum acanthopodium have been studied using chemically induced animal models, showcasing their potential in inhibiting tumor formation. The process involves carefully controlled exposures to carcinogens such as 7,12-dimethylbenz[a]anthracene (DMBA), diethylnitrosamine (DEN), 1,2-dimethylhydrazine (DMH), and carbon tetrachloride (CCl4) as potent carcinogen, with DMBA often employed, 254 leading to the formation of tumors in specific organs or tissues. The use of polycyclic aromatic hydrocarbons in these controlled experiments, highlighting their role in initiating tumorigenesis.94,233 An account on studies performed on chemical induced model is given in Figure 6. It shows that 28 (9.72%) papers integrated chemically induced animal models with any other screening methods, with 23 (7.99%) of studies utilizing plant extracts and 5 (1.74%) using plant isolated compounds. Interestingly, only two papers, one for each plant extract and plant isolated compounds used chemically induced animal model along with other three in vitro methods. Moreover, 17(5.90%) papers dedicated solely to chemically induced animal models of which 16 (5.55%) studies utilized plant extracts, while 1(0.35%) study used plant isolated compounds. These results emphasize the value of this animal models to evaluate medicinal plants’ potential as anticancer agents. These models are particularly useful for investigating the potential of medicinal plants in cancer prevention by assessing their ability to inhibit or delay tumor formation. 312

Distribution of papers by chemically induced animal model, chemically induced animal model along with cell migration, cell survival and apoptosis, and chemically induced animal model along with other methods for screening and evaluation of anticancer compounds of plant extracts and plant isolated compounds.
In Vivo Tumor Xenograft Models
Tumor xenograft models involve the transplantation of tumor cells or tissues from one species into immune-deficient individuals of another species. 313 This approach replicates the behaviour of human cancer cells within a living organism, providing insights into the effectiveness of potential anticancer agents. In this model, animals like mice and rats bearing human tumor xenografts are treated with medicinal plant extracts or compounds, and any changes in tumor size and volume are monitored over a specified period. A significant reduction in tumor growth compared to control groups indicates the potential anticancer activity of the tested medicinal plants. Furthermore, histopathological analysis is carried out to study the structural and cellular changes within tumor tissues following treatment. 314 These analyses help in understanding the mechanisms by which medicinal plant compounds affect tumor growth.
Our findings indicate that 38 (13.19%) of the total papers utilized tumor xenograft models. Among these, 10 (3.47%) papers were based on plant extracts, and 28 (9.72%) papers were based on plant-isolated compounds (Figure 7). Additionally, 8 (2.78%) papers were based on plant-isolated compounds that integrated xenograft mice models with cell migration and invasion assays, as well as cell survival and apoptosis studies. Moreover, 3 (1.04%) papers utilized only tumor xenograft models for screening anticancer agents, with 2 (0.69%) papers using plant extracts and 1 (0.35%) paper using plant-isolated compounds.

Distribution of papers by tumor xenograft animal model, tumor xenograft animal model along with cell migration, cell survival, and apoptosis, and tumor xenograft model along with other methods for screening and evaluation of anticancer compounds of plant extracts and plant isolated compounds.
Although the preclinical findings expose the anticancer potential of Nepalese plants, their translational application involves rigorous clinical trials. Accomplishments from preclinical studies to clinical relevance will involve not only efficacy but also safety profiling through toxicological studies and the understanding of mechanisms of action. A number of bioactive compounds found in Nepalese medicinal plants exhibits anticancer effects via a variety of mechanisms. Catharanthus roseus's vinca alkaloids inhibits tubulin polymerization, which prevents cancerous cells from proliferating. In breast cancer models, curcumin from Curcuma longa demonstrated to trigger apoptosis and prevent metastasis, and when administered as Meriva (curcumin phytosomes) in a controlled study, shown effectiveness in colorectal and pancreatic cancer. 5 Furthermore, one of the most popular plant-derived chemotherapeutic agents is still taxol from Taxus wallichiana, which stops cell division in a variety of cancers by stabilizing microtubules.315,316 Moreover, the advancement of compounds such as berberine, curcumin, and paclitaxel into clinical trials demonstrates how traditional knowledge coupled with efficient scientific validation can narrow the gap between ethnobotanical practices and modern medicine. Such phytochemicals, which have reached clinical trials, serve to provide a framework for further development of several promising phytochemicals. Integration of traditional knowledge with modern pharmacology may speed up the development of plant-derived anticancer drugs.
Conclusion
This study analyzed 288 articles on 71 Nepalese plant species with anticancer properties, highlighting approaches to evaluate their potential. Methanolic extract was found to be predominantly utilized, with leaves being the most commonly studied plant part. Many studies were limited to in vitro approaches, while other studies mainly focused on isolated compounds and were broadly evaluated through both invitro and in vivo approaches. (Figure 8) Combining in vitro and in vivo methods, with multiple cancer cell lines, provided a holistic evaluation of anticancer potential of medicinal plants. Despite the benefits, mandatory toxicological assessments are crucial to identify their potential adverse effects. In conclusion, the combination of in vitro and in vivo screening methods presents a significant approach to identifying promising candidates for further development as anticancer agents.
In vitro (dark green) and In vivo (light green) screening methods of phytochemicals with anticancer activity from Nepalese plants.
Footnotes
Acknowledgements
Everyone who made substantial contributions to this study has been acknowledged and listed.
Credit Authorship Contribution Statement
Anup Luitel; Data collection, Manuscript preparation, Lalit Mohan Pant; Data collection, Manuscript preparation; Niru Thapa; Manuscript preparation, Data analysis, Manika Shrestha; Manuscript preparation, Data analysis, Anup Adhikari; Data collection, Ashwinee Kumar Shrestha; Supervision, Data analysis, Ronak Shrestha; Data analysis, Janardan Lamichhane; Conceptualization of Topic, Supervision, Rajan Shrestha; Supervision, Conceptualization of Topic, Data analysis and Data review.
Data Availability Statement
All data analysed during this study are included in this published article and its Supplemental materials.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
