Abstract
Parinari is a genus of woody medicinal plants with expansive ethnomedicinal uses. While extensive data are available for several members of this genus, this information is generally scattered. This review therefore crystallizes major reports relating to the ethnomedicinal uses, phytochemistry, and biological activities of this genus for easy accessibility. The rich therapeutic applications of this genus include their use as antimalarial, antidiabetic, anti-inflammatory, antimicrobial, anti-snake venom, and anticancer agents. However, the overarching biological theme for this genus appears to be their application in treating infection and infection-related diseases. These bioactivities have largely been attributed to molecules isolated from this genus and categorized under; kaurene-type diterpenoids, triterpenoids (ursane, oleanane, and lupane types), flavonoids, steroids, and phenolic compounds. While data is available for most parts of the plants, the stem appears to be the most used part. Given the varied medicinal uses of this genus, and yet the scarcity of information on many Parinari species, studies on the ethnomedicine, phytochemistry, and biological activities that fully explore the medicinal potential of this genus for drug discovery are critical. These include the need for comprehensive phytochemical profiling to uncover novel compounds, detailed studies on mechanisms of action, and translational research to validate clinical efficacy. Additionally, comparative ethnobotanical studies, standardization of extraction methods, and sustainable harvesting practices are critical to fully realizing the medicinal potential of Parinari species.
Keywords
Introduction
Parinari (synonymous with maranthes) 1 is a genus of woody plants in the family of Chrysobalanaceae. There are about 51 species of trees and shrubs in this family classified into 17 genera, majority of which are found mostly in the tropics and sub-tropical regions of the world.2–4 They can be identified from the other genera under Chrysobalanaceae by their zygomorphic floral symmetry with a unilaterally attached flowers that have about 6-10 stamens each. Their ovaries are located at the side or opening of a receptacle tube, while the leaves that are lanate on the lower surface have hair-filled stomatal cavities. A parallel but closely spaced secondaries, coupled with a large pair of glands on the leaf petiole and woody fruits, further distinguish these plants. 5 Some of the common Parinari species include; curatellifolia, occidentalis, anamensis, congolana, congensis, costata, campestris, montana, excelsa, robusta, polyandra, floribunda, campestris, sprucei, hypochrysea, and kerstingii.
Humans since prehistoric epochs have relied on medicinal plants as their natural sources of drugs for prevention, management, and treatment of diseases. 6 Predicated on this observation, scientific endeavors in the early days of drug discovery relied heavily on pharmacophores of natural origin. However, technological advancements that started during the last few decades resulted in more efficient synthesis of molecules, thereby accelerating high-throughput screening capabilities for the discovery of hit compounds during synthetic drugs discovery efforts. This makes it seamless to test thousands of samples per day on an array of targets, 7 subsequently dousing the high rising flames of natural products drug discovery. There is however a renaissance in natural products drug discovery in recent times largely due to the expanding pathogen and disease repertoire, prohibitive cost of synthetic drugs, toxicity and unwanted side effects, and high resistance landscape of many pathogens to synthetic drugs. 8
The plethora of compounds present in plants and other natural products sources, and the intricate link between human evolution and biodiversity, make them very important avenues for the discovery of therapeutically important molecules that not only serve as direct antidotes to several human and other zoonotic diseases, but also as precursors for some synthetic drugs.
The success of plant-based natural products drug discovery programs hinges on reliable information on their ethnomedicine, phytochemistry and pharmacology. The use of different plant parts by different cultures and ethnicities and how they are prepared in treating same or different diseases, is perhaps the first glance into the nature of compounds responsible for the observed therapeutic effects, sometimes gleaned only from sensory effects of the extracts. The nature of biodiversity around areas of human habitation undoubtedly are influenced by their use, handed down through folklore from generation to generation. Synthesis of the folkloric uses of plants’ parts by different ethnicities, and the different medicinal application of a particular plant, provides very important information that serve as starting points for scientific investigations. For instance, in Guinea, Parinari excelsa stem bark is used to treat infectious diseases 9 while in some parts of West Africa, same is used as an anthelmintic. 10 On the other hand, it is used as an antiseptic and antimalarial in Tanzania. 11 Such knowledge can be very useful in exploring the medicinal potential of the plant. Therefore, a cross-fertilization of knowledge from different cultures on the genus Parinari could give a comprehensive view of its ethnomedicinal potential that could stimulate drug discovery efforts.
Suffice to say that ethnomedicinal evidence alone is not sufficient as a basis for drug development. Scientific evidence obtained through a series of biological activities using in-vivo and in-vitro models are a necessity. An observed biological activity offers scientific evidence of the efficacy of the plant while a lack of activity questions the folkloric claims. Furthermore, information on the dosage required to elicit a therapeutic response or toxicity is critical for drug development. Biological assays performed on extracts of Parinari species have shown that these plants possess antibacterial,12,13 antimalarial,14,15 cytotoxic, 16 anti-snake venom17,18 and anti-inflammatory 19 activities. They are also used against venereal diseases, toothache, dysentery and diabetes.15,20,21
These observed biological effects are attributed to secondary metabolites produced by these plants. 22 Many of these secondary metabolites are produced in response to stressors caused by biotic factors such as insect infestation, invasion by herbivores, or infection by pathogens. Others are produced in response to abiotic effects such as heat, cold, drought, and climate change. 23 Therefore, in essence, humans are opportunistic beneficiaries of these metabolites. These secondary metabolites such as phenolic compounds, flavonoids, sterols, alkaloids, terpenoids, etc, are widely varied in structure and bioactivity. 24 Therefore, maturity of the plant, the geographical location, the season of harvesting and the part used, influence the nature, type, and quantity of secondary metabolites even in the same species of plants 23 depending on the acute nature of some of the factors already mentioned.
Natural products remain integral to drug discovery efforts with current estimates showing that secondary metabolites sourced from medicinal plants constitute about 30% of the currently available drugs on the market. 25 Among the myriad of compound classes isolated from this genus include kaurene-type diterpenes,15,16 flavonoids, steroids, phenolic compounds. 26 Parinari species are distributed globally but are predominant in tropical Africa and south America as shown in Figure 1. They are not critically endangered, however, habitat loss due to deforestation and land use changes pose significant risks to conservation efforts. According to the IUCN, most Parinari species such as curatellifolia, occidentalis, glabra, congensis, sprucei, congolana, excelsa, hypochrysea, campestris, polyandra and robusta are considered species of least concern with regards to threatened species. 27

Global distribution of Parinari species across South America and Africa. The yellow dots represent Parinari species. Image credit: GBIF.
Methodology
To gather comprehensive information on the ethnomedicine, phytochemistry, and biological activities of the Parinari genus, a systematic review was conducted utilizing a variety of reputable electronic databases, including Google Scholar, JSTOR, ScienceDirect, Elsevier, ResearchGate, Web of Science, PubMed and Scopus. A broad range of keywords was employed during the search process, including “Parinari,” “Ethnomedicine,” “Traditional uses,” “Phytochemistry,” “Isolated compounds,” “Biological activities,” and “Pharmacological activities.” These keywords were strategically selected to encompass all relevant aspects of Parinari species and their various applications. To ensure thorough coverage of the subject, both botanical and synonymous botanical names of Parinari species were included in the search. These names were obtained from well-established online databases, such as The Plant List, JSTOR, Useful Tropical Plants, and Google Scholar, which provided authoritative sources on plant taxonomy and nomenclature. This approach was critical in capturing all potential studies on Parinari species, as the genus may be referred to differently across various regions and in different contexts.
The search process was followed by a careful screening of the gathered studies to ensure that only relevant and high-quality data were included in the review. The studies were evaluated based on their scientific thoroughness, the clarity of their findings, and their contribution to understanding the medicinal, phytochemical, and biological aspects of Parinari. To focus on the most reliable and recent information, preference was given to studies published in the past decade, particularly those published in peer-reviewed journals, books, or academic sources. As part of the selection process, certain inclusion and exclusion criteria were applied. Studies were included if they specifically focused on any aspect of the Parinari genus, including its traditional medicinal uses, isolated bioactive compounds, or biological and pharmacological activities. Only peer-reviewed studies published in English were considered, ensuring the credibility and accessibility of the research. Studies without full-text availability or those published in languages other than English were excluded, as were articles that did not directly pertain to the Parinari genus or its medicinal properties. The data obtained from the selected studies were carefully synthesized and analyzed to create a detailed overview of the Parinari genus, with a particular focus on its ethnomedicinal applications, phytochemical profile, and biological activities. Through this process, the review aims to contribute to the understanding of Parinari species and their potential for future research and drug discovery.
Results
Ethnomedicine
Species of Parinari have several ethnomedicinal uses. These plants have been copiously relied upon in meeting the medical needs of many ethnicities around the tropical and sub-tropical world. A summary of the rich ethnomedicinal use of this genus in the treatment of infections and inflammation is provided in Table 1, while Table 2 provides a summary of uses for other diseases. Interestingly, for most of these plants, the stem bark is the preferred part for the treatment of these diseases in most cultures with roots, leaves, branches and seeds occasionally mentioned.
Parinari species Used Ethnomedicinally for Treating Infection and Inflammation.

Other Ethnomedicinal Uses of Parinari species.

Phytochemistry of Parinari
Phytochemical constituents accumulate in fast dividing cells of plants including leaves, flowers, fruits, seeds, stem, and root bark. Unsurprisingly, root barks and stem barks are among the most popular sources of these bioactive phytochemicals. This is a smart evolutionary move partly because these parts serve as direct barrier to entry by pathogens that constitute a major biotic threat as well as a mediator for many abiotic fluctuations. Various phytochemical studies have been carried out on different parts of Parinari plants. The stem bark remains the most widely studied, consistent with its ethnomedicinal use.
In general, the stem barks of Parinari plants contain alkaloids, saponins, cardiac glycosides, flavonoids, triterpenoids, tannins, phenolic compounds, steroids and reducing sugars.21,37,41–44 The root barks on the other hand contain resins, alkaloids, saponins, cardiac glycosides, flavonoids, triterpenoids, tannins, and anthraquinones,18,45 while the leaves contain alkaloids, saponins, cardiac glycosides, flavonoids, triterpenoids, and tannins. 45 Phytoconstituents of the seeds have been reported as alkaloids, cardiac glycosides, flavonoids, triterpenoids, steroids, phenolic compounds and anthraquinones.46–48 Consistent with other families of plants and evolutionary data on biosynthetic pathways in plants, similar classes of phytochemicals (alkaloids, cardiac glycosides, flavonoids, and triterpenoids) have been found common to the leaves, seeds, stem barks and root barks of this genus. Interestingly, reducing sugars has been reported only in the stem bark, while anthraquinones were reported in the seeds and root barks only. Steroids also appear to be present only in the seeds and stem barks.
These phytochemicals have been extracted using several solvents and solvent mixture including ethanol, hydro-ethanol, methanol, water, 49 hexane,41,48 and ethyl acetate. 48 The solvent most frequently used that guaranteed the extraction of more phytochemicals of this genus in general was hydroethanol21,37,42,46 followed by absolute ethanol41,49 and methanol.43,44,48 This is consistent with the nature of compounds reported and the chemical properties of these solvents including their polarities.
Silica gel-based column chromatography has been the most popular method for the isolation of these compounds from various parts of the plant while sephadex has also been used. Successful isolations of these compounds from these plants have relied on isocratic solvents and their combined mixtures including hexane/dichloromethane/methanol in an increasing order of polarity, 26 chloroform/ methanol, chloroform/ ethyl acetate,50–52 petroleum ether/dichloromethane/ethyl acetate/ethanol, 15 and dichloromethane/ethyl acetate/methanol, 53 all in an increasing order of polarity. Tables 3-8 provide a summary of the various compounds isolated from these plants according to their class.
Kaurene Diterpenes Reported.

Kaurane-Type Diterpenoids
Kauranes are a group of tetracyclic diterpenes composed of a perhydrophenanthrene unit (A, B and C rings). A cyclopentane unit (D ring) is joined to ring C creating a bridge of two carbons between C-8 and C-13 as shown in Figure 2. Kaurane-type diterpenes have been isolated from many plant species belonging to many families including Chrysobalanaceae, Fabaceae, Asteraceae, Euphorbiaceae, Rutaceae, Celastraceae, Lamiaceae, Jungermanniaceae, Annonaceae, Rhizophoraceae Apiaceae and Erythroxylaceae. 58

Carbon skeleton of Kaurane-type diterpene.
The Parinari genus appears to be one of the richest sources of kaurene-type diterpenoids. Bioaccumulation of this class of compounds occurs predominantly in the leaves with about thirty (30) kaurene-type diterpenes isolated and characterized. Details of the structural diversity of this class of compounds is provided below (1-30) and in Table 3. These compounds display an array of biological activities including antifungal 53 and anticancer. 16 The structural diversity of these compounds present in Parinari is generated by functional group transformations on the parent tetracyclic kaurene skeleton such as intramolecular cyclization, degradation, rearrangements, reduction and oxidation. 59


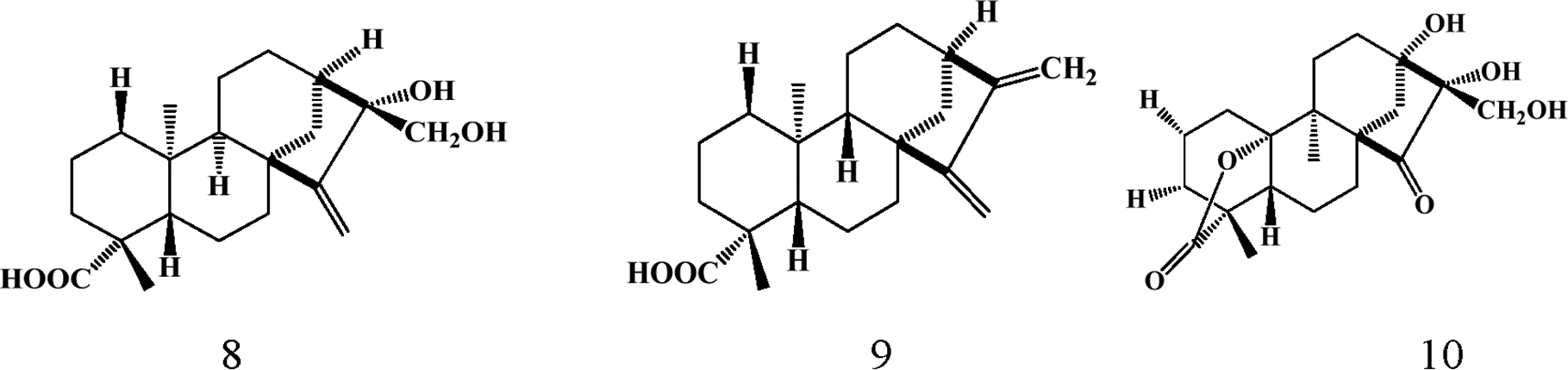

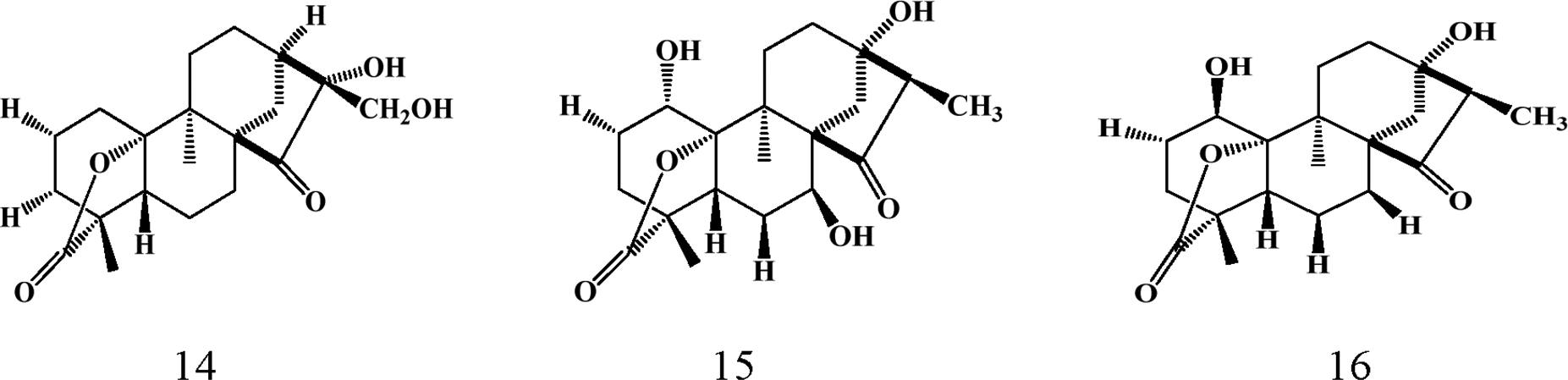







Triterpenoids
Pentacyclic triterpenoids are among the most medicinally useful class of natural products produced by plants for chemical warfare. Unsurprisingly, the Parinari genus is not an exception to this with several triterpenoids identified and characterized. Isolated triterpenes from these plants are mostly of the lupane, ursane and oleanane types as indicated below. Structural details of these compounds are shown below (31-42) and other details in Table 4. Unlike the kaurene-types that bioaccumulate in the leaves, these pentacyclic triterpenoids are mostly found in the stem bark and exhibit various pharmacological effects such as anti-snake venom activity, 17 anticonvulsant and sedative effect. 17





Triterpenoids Reported.

Steroids
The steroidal backbone, also known as cyclopentano-perhydrophenanthrene is a phenanthrene with a cyclopentane ring attached creating a 17-carbons core structure of a four-ring system. They are similar in structure to cholesterol but occur copiously in plants with structures varying in the absence or presence of a double bond or other functional groups attached to the four-ring core. 55 Lanostane and beta-sitosterol displayed below are typical examples of steroids. As shown in Table 5 below, ten steroidal compounds (43-51) such as β-sitosterol and its glycosides and stigmastane and its derivatives have been isolated, mostly from the stem bark. These phytoconstituents have wide pharmacological applications, eg, stigmast-4-en-3-one is reported to exert a hypoglycemic effect 60 and β-Sitosterol is a popular anti-inflammatory agent. 61





Steroids Reported.

Flavonoids
Flavonoids contain a 15-carbon unit in their core structure arranged in the form C6-C3-C6 with two aromatic rings A and B connected by a bridge of propane which either forms a closed pyran ring or remains open (chalcone). 62 Noted for their antioxidant, and anticancer activities as well as their ability to prevent atherosclerosis, 63 Parinari remains a good source of these compounds. Table 6 shows details of characterized flavonoids from this genus. A total of eight (8) flavonoids (structures 52-59) have been isolated from the leaves of Parinari plants with quercetin being the common flavonoid shown below.
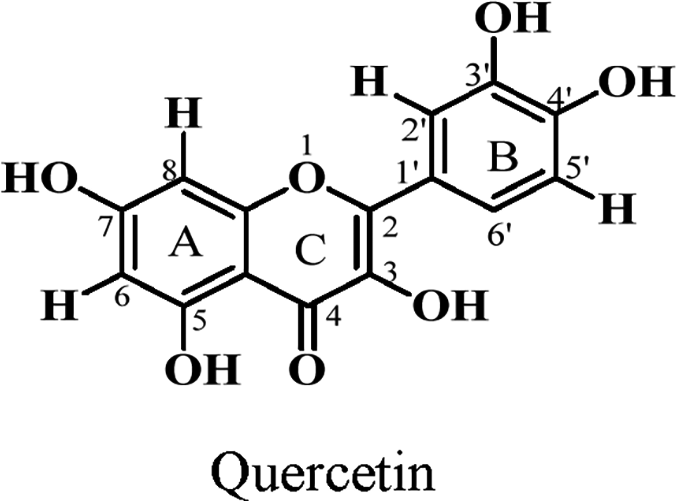


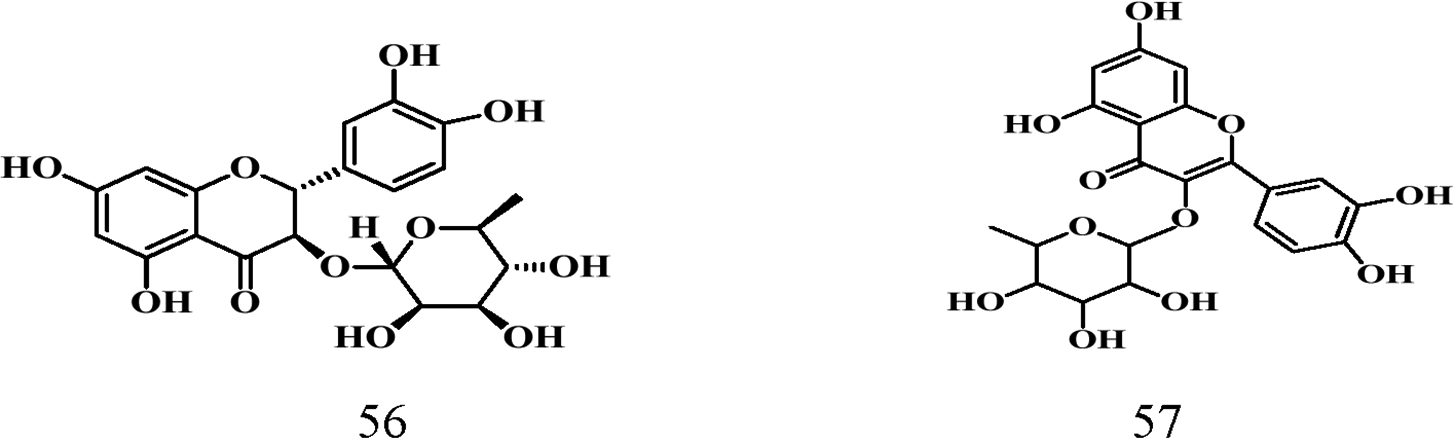
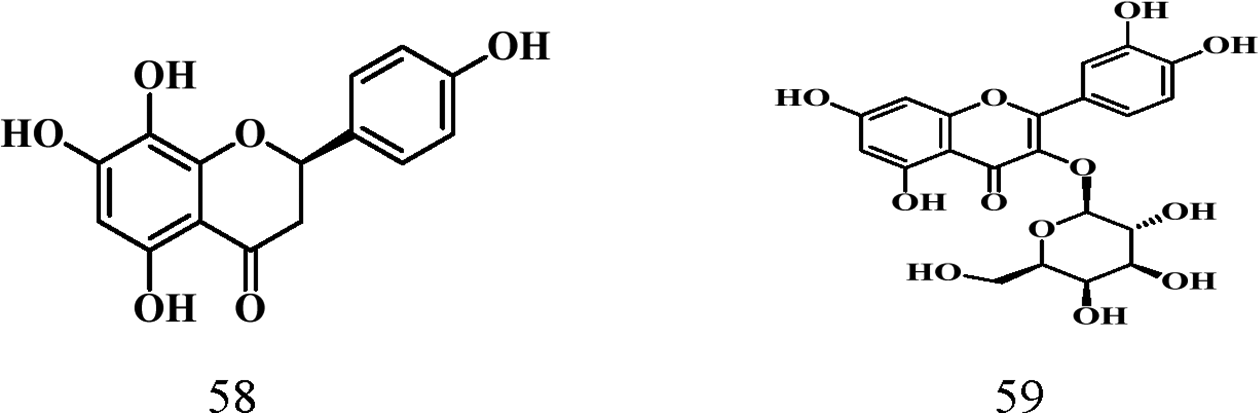
Flavonoids Reported.

Phenolic Compounds
This is an extensive and ubiquitous group of secondary metabolite molecules in plants that possess a wide range of structures and functions. They are made up of water-soluble compounds including phenylpropanoids, quinones, and phenolic acids, as well as water-insoluble compounds such as condensed tannins, cell-wall bound hydroxycinammic acids, and lignins. Phenolic compounds get synthesized during the normal development and growth of the plant as well as when exposed to adverse conditions such as stress and UV radiation. They play a role in preventing degenerative diseases caused by oxidative damage by scavenging free radicals from cell metabolism. 64 They are compounds with hydroxylated aromatic rings in which the hydroxy group is directly attached to the phenyl, or the substituted phenyl, or other aryl group. A typical example of phenolic compounds is benzoic acid shown below. Few phenolic compounds (four) from the root and stem barks have been isolated from Parinari (structures 60-63) details of which are provided in Table 7.
Phenolic Compounds Reported.




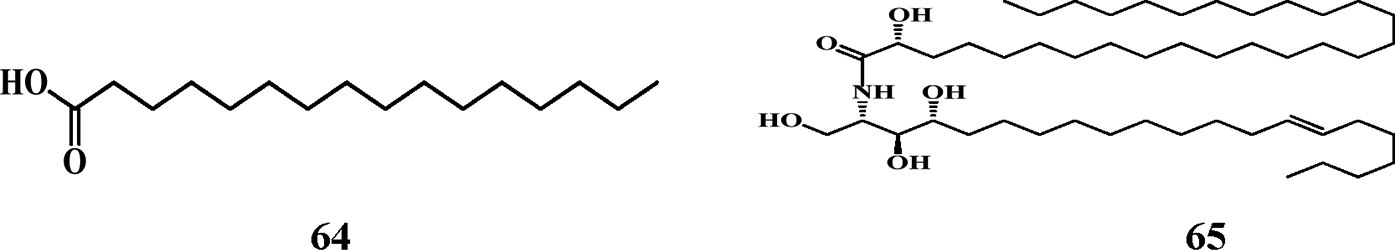
Other Compounds Reported.
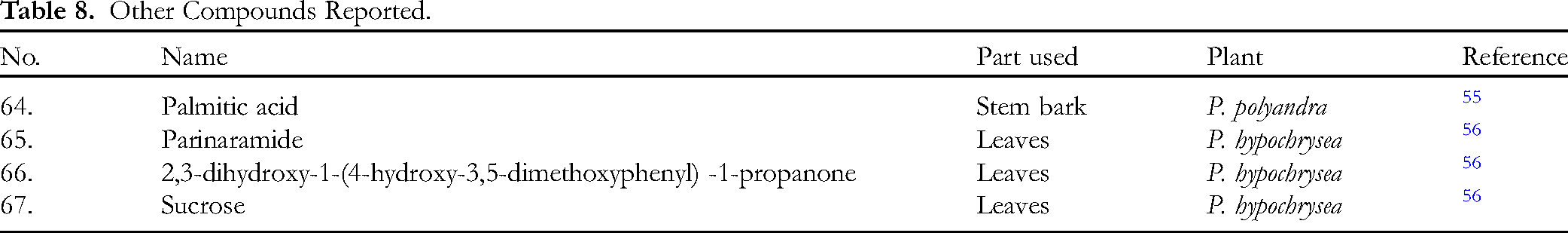
Other Compounds Reported
Other compounds reported include a fatty acid (64), a ceramide or lipid (65), a phenylpropanone (66) and a sugar (67) as shown in Table 8 with their chemical structures below.

Biological Activities
Cytotoxic Activities
Several classes of compounds found in Parinari demonstrate some cytotoxic activity to a range of cell lines consistent with its folkloric use in the treatment of cancer and related diseases. For instance, Kaurane diterpenes isolated from P. curatellifolia root bark and generously abundant in Parinari, exhibited a broad-spectrum cytotoxicity against several cell lines from humans including lung cancer cells, epidermal carcinoma cells, colon cancer cells, breast cancer cells, and prostate cancer cells with ED50 values ranging from 0.3-16.5 µM. The compounds were most active against the epidermal carcinoma cells with ED50 0.6, 0.3 and 0.3 µM for compounds 1-3 respectively. 16 Gororo et al, 51 also reported inhibitory activity of the root bark extract of same plant on xanthine oxidase (an enzyme implicated in oncogenesis) and cytotoxic effect on Jurkat-T cells. They found that DNA from cells treated with extracts of P. curatellifolia showed cleavages suggesting that apoptosis may be the mechanism of cytotoxity. Similarly, 50% ethanolic extracts of P. anamense displayed considerable cytotoxicity against malignant human hepatoma (HepG2) cell line and a normal African green monkey kidney (Vero) cell line with IC50 values of 546 µg/mL and 431 µg/mL respectively with selectivity index (SI) of 0.8. 31
In a cytotoxicity screening of Tanzanian medicinal plants, Kamuhabwa et al, 11 reported that extracts of the stem bark of P. excelsa exhibited antiproliferative activity against human cancer cells of the cervix, colon and skin. The extracts at 100 µg/mL, inhibited the proliferation of 75%-100% of the cervical cell carcinoma, 50%-75% of the colon cell adenocarcinoma and 25%-50% of the skin carcinoma cells.
Antimicrobial Activities
A continued surge in antimicrobial resistance to virtually all drugs on the market has brought into sharp focus the need for new and effective antimicrobial agents with different mechanisms of action and higher barrier to resistance. Parinari quickly comes into focus as a genus with the potential for new antimicrobial agents. In this regard, the ethyl acetate extract of P. curatellifolia root bark and one of its isolated molecules (compound 1) have been reported to exhibit antifungal activities against candida albicans, cryptococcus neoformans, and aspergillus niger. 53 The ethyl acetate extract of the seeds of P. polyandra (Maranthes polyandra) on the other hand demonstrates impressive sensitivity against various microbes such as S. aureus, S. pyogens, S. typhi, S. dysenteriae, C. albicans, C. tropicalis, C. stellatoidea and T. rubrum. 48 Additionally, the ethanolic extract of P. excelsa seeds has also been shown to possess antimicrobial activity, with moderate sensitivity against clinical isolates of S. aureus, E. coli, K. pneumonia and S. typhi. 49 Bhunu et al, 65 reported that extracts of the leaves of P. curatellifolia exhibit strong activity against Mycobacterium tuberculosis. They reported MICs of 6.2 μg/mL for the acetone extract, 12.5 μg/mL for the ethanol extract while the methanol and ethyl acetate extracts recorded 50 μg/mL. Additionally, the ethanol extract, dichloromethane extract and water extracts inhibited biofilm formation using the model organism Mycobacterium smegmatis. This data was corroborated by Mawire et al, 66 who also reported that various extracts of P. curatellifolia demonstrated robust antimicrobial activity against Candida krusei and M. smegmatis with MIC of 25 µg/mL.
Antidiabetic Activity
Hydrolysis of starch and other polysaccharides to simple sugars after meals that lead to postprandial spike in blood glucose level is catalyzed by α-amylase and α-glucosidase. 67 Inhibition of these enzymes thus provides a therapeutic strategy for diabetic patients in reducing blood glucose levels. Anecdotally, Parinari have been used to treat diabetes with some scientific evidence showing that some extract from plants in this genus demonstrate strong activity against these enzymes. In support of the use of these plants for treating diabetes, Srisongkram et al, 42 reported that extract of the twigs of P. anamense inhibited the activity of α-glucosidase with an IC50 of 0.52 μg/mL. The specificity of this activity was confirmed when they showed that this extract had no α-amylase activity. Additionally, aqueous extracts of P. excelsa stem bark, when tested against alloxan-induced diabetic rats, and on overnight (16 h) fasted rats by administering glucose (4 g/kg) 90 min after treatment, showed that the extract at 100 and 300 mg/kg/day for 7 days reversed the hyperglycemia induced by the alloxan. In this experiment, the blood glucose decreased from 3.11 to 0.91 g/L and from 3.60 to 0.85 g/L, respectively for the two doses. Thus, the oral treatment with P. excelsa stem bark aqueous extract at the dose of 100 and 300 mg/kg significantly reduced the blood glucose level at 1/2h. 21 Furthermore, the antidiabetic activity of coconut water extract of P. polyandra seeds, 68 and ethanolic extract of P. curatellifolia stem bark 18 have also been confirmed, consistent with their traditional use for the treatment of diabetes.
Anti-Inflammatory Activity
Inflammation is a protective response to harmful or potentially harmful substances in the body and hence a key component of a functional innate immunity. Prolonged inflammation can however have a deleterious effect on the body because of tissue damage resulting in diseases such as arthritis and hay fever. In this regard, inhibition of inflammation is necessary to prevent excessive tissue loss and reduce pain and redness. The genus Parinari is rich in compounds that exert anti-inflammatory activity. For instance, the methanolic extract of P. polyandra stem bark exhibited anti-nociceptive and anti-inflammatory activities in different in vivo models of laboratory rats and mice. In the acetic acid-induced abdominal constriction test model of rats, the extract was effective at 100 mg/kg dose level, was also effective at 200 mg/kg dose for the hot plate-induced nociceptive model in mice and demonstrated anti-inflammatory activity in the egg albumin-induced inflammation or edema in rats with a dose of 200 mg/kg. 43
P. kerstingii methanolic leaf extract demonstrated significant acute anti-inflammatory effects on egg albumin-induced paw edema, with activity most pronounced at the 100 mg/kg dose at the third hour after induction of edema. The extract was also found to show anti-inflammatory activity by significantly reducing carrageenan-induced paw edema concomitant with a significant reduction in the production of IL-1, TNF-α, COX-2, NF-кB, and PGE2. 33 Also, the stem bark ethanolic extract of M. glabra (P. montana) was found to downregulate the expression of proinflammatory cytokines. 19 Gweshelo et al, 69 reported that the ethanolic extract of P. curatellifolia was a potent inhibitor of glutathione s-transferase (GST), a key enzyme in the biosynthesis of prostaglandins with an IC50 value of 12 μg/mL comparable to that of ethacrynic acid, a standard GST inhibitor, found to be 10 μg/mL, confirming its traditional use in ameliorating pain.
Antimalarial Activities
Malaria continues to be an important public health enigma causing mortality and morbidity to millions of people a year especially in sub-Sahara Africa. Plant secondary metabolites have been very important in the fight against malaria with most of the current drugs for treating malaria derived from plant sources. Parinari genus is endowed with secondary metabolites that have strong antimalarial activity. Three diterpene lactones (compounds 1-3) isolated from both the petroleum ether and dichloromethane extracts of the stem bark of P. capensis showed anti-plasmodial activity against chloroquine-resistant strain of Plasmodium falciparum with IC50 of 0.67, 0.54 and 1.57 μg/mL respectively for compounds 1, 2 and 3. 15 In an in vitro experiment, P. curatellifolia stem bark extracts (ethanolic and n-hexane) inhibited the growth of P. falciparum parasites in a manner that was dose and duration-dependent. The ethanolic extract exhibited a higher parasite inhibition than the hexane extract. 41 The hydroethanolic extract of P. congensis stem bark was found to display moderate activity against chloroquine-sensitive field isolates and chloroquine-resistant strains of P. falciparum respectively. 37 These pieces of scientific evidence lend credence to the ethnomedicinal application of some Parinari plants for malaria treatment.
Anti-Tyrosinase Activities
Tyrosinase is an enzyme that mediates the biosynthesis of melanin in plants and animals and is implicated in skin diseases, including hyperpigmentation. In investigating the cosmetic use of P. anamensis, Werawattanachai et al, 26 tested isolated compounds (43, 46, 54, 57, 58, 59 and 63) from the stem and leaves for tyrosinase inhibitory activity. These compounds inhibited tyrosinase activities with IC50 values that ranged from 0.80-5.38 µM with compound 63 found to be the most active. In this experiment, the IC50 value of kojic acid, the positive control was reported as 0.001 µM. The ability to inhibit tyrosinase implicated in dermatological disorders scientifically supports the application of some species of Parinari for skin disorders.
Discussion and Conclusion
Parinari is distributed across the tropical and subtropical regions of Africa as well as Central and Southern America.3,4 This vast geographical distribution straddles different ethnicities and cultures resulting in myriad and varied ethnomedicinal applications that can be harnessed for drug discovery. Some uses reported include the treatment of snake bites, worm infections, erectile dysfunction, sexually transmitted infections, anemia, skin itches, malaria, fractures, diabetes, hypertension, liver-related diseases, parasitic infections, inflammations, dysentery, stomach aches, and cancer. The ethnomedicinal data gathered revealed that treatment of infections and infection-related diseases was dominant among the many folkloric uses across cultures. Various parts of the plants including the leaves, stem barks, roots, seeds, fruits, twigs, and branches are used but the stem bark appears to be the part most widely used in ethnomedicine.
A review of the phytochemistry of this genus revealed the presence of alkaloids, saponins, tannins, phytosterols, terpenes, flavonoids, cardiac glycosides, anthraquinones, fatty acids, reducing sugars and phenolic compounds in the various parts of the plants. Compounds isolated from various parts of these plants can be broadly categorized into kaurene-type diterpenes, triterpenoids of the ursane, lupane and oleanane types, flavonoids mainly of the quercetin, kaempferol and taxifolin types, steroidal compounds, phenolic compounds, fatty acids, lipids, sugars, and simple organic compounds. Interestingly, while phytochemical screening has identified alkaloids from many Parinari species, no alkaloid has been isolated and characterized from this species to the best of our knowledge.
Column chromatography was reported as the most common method of compound isolation with solvent extraction the commonest method of extracting these metabolites. Solvent systems frequently used for the isolation were reported as CHCl3: MeOH and CH2Cl2: MeOH for normal phase chromatography and MeOH: H2O for reverse phase.14,50,52,54 Kaurane-type diterpenes and triterpenoids are the most abundant secondary metabolites isolated from Parinari mostly from the leaves and stem bark respectively. These diterpenes were found to possess antispasmodic, antidiabetic, antinociceptive, antiallergic, anticancer, and antimicrobial properties 58 while the triterpenoids were found to exert antibacterial, antiviral, 70 anti-inflammatory, anticancer, antioxidant 71 and anti-snake venom activities. 17 Isolated flavonoids however exhibited chemopreventive and chemotherapeutic activities, 72 while the steroids were noted to have anti-inflammatory activities. 73 Consistent with other reports in the literature, the phenolic compounds demonstrated anticancer, antibacterial, antihypertensive and antidiabetic activities. 74 The medicinal importance of these isolated secondary metabolites suggest that there is enormous potential to discover new therapeutics against cancer, inflammation, oxidative stress, nociception, snake venoms as well as hypertension from this genus.
While 67 bioactive compounds have been identified, this represents only a fraction of the potential metabolites present in Parinari species. Plants often produce a wide array of secondary metabolites, some of which may have unique and synergistic therapeutic effects. Advanced analytical techniques such as metabolomics, Liquid chromatography-mass spectrometry (LC-MS), or Nuclear magnetic resonance (NMR) spectroscopy could help identify these uncharacterized molecules. This is crucial for discovering novel compounds that could serve as drug leads. 75 Establishing the mechanism of action is another relevant research gap to consider. Although the biological activities of Parinari have been documented (eg antimalarial, antimicrobial, anticancer), the molecular or cellular pathways these compounds target are often unknown. Understanding how these compounds work at the molecular level (eg, their effect on enzymes, receptors, or signaling pathways) can help optimize their use in medicine and even guide synthetic modifications to enhance efficacy and/or reduce side effects. 76 Most studies on Parinari have been conducted using in vitro (cell-based) or in-vivo (animal-based) models. These provide valuable insights but don’t guarantee the same effects in humans due to differences in physiology, hence clinical relevance remains one critical research gap. To bring Parinari closer to therapeutic use, it is essential to conduct clinical trials that evaluate safety, efficacy, the pharmacokinetics and pharmacodynamics of the various extracts.
Standardization and quality control is another research gap in the literature. Variations in how Parinari plants are harvested and prepared (eg, seasonal and geographical variations, different solvents, plant parts, or methods) can lead to inconsistent results in research and hinder pharmaceutical development. Establishing standardized extraction protocols and ensuring consistent quality in derived products are critical for reproducibility and reliable therapeutic use. Without standardization, it is challenging to develop Parinari-based treatments that meet regulatory approval. 73 Finally, the widespread use of Parinari, especially the stem bark, can lead to overharvesting and threaten the plants’ survival. Bark harvesting often kills the tree, making conservation even more critical. Sustainable harvesting practices and conservation strategies, such as cultivating Parinari or using alternative plant parts (eg, leaves or roots) are essential for ensuring the plant remains available for both traditional and modern medicinal use. 77
Footnotes
Abbreviations
Acknowledgment
The team is grateful to the Centre for Plant Medicine Research and the University of Ghana, Legon for the use of their facilities.
Author's Contribution
All authors contributed to the realization and completion of this work. They also read and approved this manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported with funding from the Building the New Generation of Academics in Africa (Banga-Africa) Project, funded by the Carnegie Cooperation of New York.
