Abstract
Objective
Salicornia brachiata, an euhalophyte thriving in saline environments, can be processed into a dried powder (termed bio-salt) from its tender aerial parts. This study aimed to evaluate how three distinct drying methods; freeze-drying (FD), microwave-vacuum drying (MVD) as low temperature drying methods, and heat pump oven drying (HPOD) as a high temperature drying method, affect nutritional composition and bioactivities of S. brachiata. Ultimately, this research aims to identify the most suitable drying approach that retains bioactive and nutritional properties to preserve the health benefits of bio-salt.
Methods
The aerial parts of S. brachiata were washed, air-dried overnight, and then subjected to various drying methods. The proximate composition, colour measurements, mineral composition, and microbial quality of the dried powders were evaluated. The lipid extracts were analyzed using GC-MS and the total phenolic content and antioxidant activity of the resulting powders were assessed.
Results
Among the methods, FD best preserved the plant's color and yielded the highest levels of carbohydrates (42.64%), crude fat (0.88%), and dietary fibers (29.72%). Methanolic extracts of FD powders exhibited the highest phenolic content (43.68 ± 0.04 mg GAE/g DW) and superior antioxidant activity, with IC50 values of 8.72 ± 0.50 µg/mL for the DPPH assay and 19.49 ± 0.76 µg/mL for the ABTS assay, outperforming both MVD and HPOD. The FD extracts contained a higher proportion of unsaturated fatty acids compared to saturated fatty acids. In vitro antibacterial assays revealed that the FD extract at 100 mg/mL demonstrated the strongest inhibition against test bacteria. In an anti-inflammatory assay, FD was identified as the most effective drying method, yielding the lowest IC50 value of 314.78 ± 1.55 µg/mL.
Conclusion
These results suggest that freeze-drying is the most suitable drying method for preserving the bioactive properties and health benefits of bio-salt derived from the aerial parts of S. brachiata.
Introduction
Salicornia presents a remarkable range of nutritional and potential medicinal benefits. Its ability to thrive in saline environments makes it a valuable resource for regions where conventional crops struggle due to high soil salinity. The plant's rich content of proteins, fibers, microelements, and polyunsaturated fatty acids highlights its potential as a functional food. Furthermore, exploring its biological properties such as antioxidant, anti-inflammatory, hypoglycemic, and cytotoxic activities, suggests promising applications in both pharmaceuticals and nutraceuticals. 1
The concept of using dried and powdered tender aerial parts of Salicornia as a natural salt substitute, termed bio-salt (or green salt), not only addresses the dietary need for reducing sodium intake but also taps into sustainable alternatives for traditional table salt production. While Salicornia europaea 2 and Salicornia ramosissima 3 have been extensively studied for their bioactive properties, Salicornia brachiata (Figure 1), a native species of the Indian subcontinent, extending to Myanmar and Sri Lanka, remains largely under explored. However, this species holds significant potential for applications in food, animal feed, nutraceuticals, pharmaceuticals, cosmetics, skincare products, and biofuels, warranting further investigation. The study aims to determine the most effective drying method for preparing bio-salt from S. brachiata while preserving its nutritional composition. Specifically, the objective is to evaluate the phytonutrient content and bioactivities of the plant's tender aerial parts dried using three distinct techniques: freeze-drying (FD), microwave-vacuum drying (MVD), and heat pump oven drying (HPOD). The color, proximate composition, mineral composition, amino acid composition, microbial quality and bioactive properties (antioxidant activity, anti-inflammatory activity and, antibacterial activity) of the dried S. brachiata were assessed to identify which drying technique best preserves these properties.

Salicornia brachiata plants in the salt marshes of Puttalam, Sri Lanka.
Materials and Methods
Sample Collection
The coastal lagoon of Seguwantivu and Vidatamunai in the Puttalam district, Sri Lanka (8° 3’ 21.1” N; 79° 48’ 21.9” E), was chosen as the sampling site due to the high abundance of S. brachiata (Figure 1). Necessary approvals for sampling were obtained from relevant authorities. Plant species identification was performed using standard taxonomic keys and an herbarium specimen was deposited at the National Herbarium, Peradeniya, Sri Lanka, under the accession number R2/RJ1_A. Healthy plants in the vegetative growth phase were randomly selected and harvested, with the aerial parts cut approximately 4 to 5 cm above soil level. These tender aerial parts were transported to the laboratory in April, 2024.
Sample Preparation and Drying
The collected plant samples were thoroughly washed with tap water, followed by Milli-Q water, and air-dried overnight. The plant parts were then manually cut using a food-grade stainless-steel knife, evenly arranged in stainless-steel trays, and subjected to three different drying techniques: FD, MVD, and HPOD. FD samples were dried in a freeze-dryer (Biobase, China) at −40 °C for 72 h. MVD samples were dried in a microwave vacuum dryer (Tianshui Hyuyuan Pharmaceutical Equipment Technology, China) at 50 °C for 4 h, while HPOD samples were dried at 45 °C for 24 h. The drying duration was based on a pre-test conducted where samples were dried until constant weight. All dried samples were ground into a fine powder using a laboratory-scale grinder and sieved through a 0.5 mm sieve. The powdered samples (Figure 2) were stored in a refrigerator (0-4 °C) until further analysis.

Dried powders of S. brachiata; (a) MVD, (b) FD, and (c) HPOD.
Preparation of Extracts
A total of 100 g of powdered sample from each drying method was extracted with 400 mL of methanol using a benchtop orbital shaker (M-TOS-6048F, Australia) at 200 rpm for 24 h at room temperature. The solvent of each extraction was evaporated separately under reduced pressure at 40 °C using a rotary evaporator (IKA, RV 10). The crude extract obtained was used to determine the total phenolic content, total flavonoid content, and condensed tannin content, as well as to conduct in vitro assays for antioxidant, anti-inflammatory, and antibacterial activities.
Determination of Color
The color parameters of the dried samples were assessed using a colorimeter (Chroma Meter CR-410, Konica Minolta) calibrated with a white standard tile in terms of CIELAB coordinates: L*(lightness), a*(greenness-redness), and b*(yellowness-blueness).
Nutritional Characterization
The nutritional profile of each dried plant sample was studied by analyzing the proximate composition, micronutrients, amino acid profile, and fatty acid profile.
Proximate Composition
The moisture, crude fiber, total dietary fiber, ash, crude protein (conversion factor 6.25), and crude fat contents of each sample was determined according to AOAC International (2019) methodologies (934.01, 962.09, 992.16, 930.05, 981.10, 943.01, and 991.36 respectively). 4
The total carbohydrate content was calculated using the formula below.
Mineral Composition
The quantitative determination of Na, Ca, Mg, P, K, Cu, Zn, and Fe was performed using inductively coupled plasma-optical emission spectroscopy (ICP-OES; AOAC 2011.14:2019). 5 The quantitative determination of Cd, As, and Pb was performed by inductively coupled plasma-mass spectroscopy (ICP-MS; AOAC 2013.06: 2013). 4
Amino Acid Composition
Amino acids were determined using high-performance liquid chromatography–ultraviolet spectroscopy (HPLC-UV). 4 The chromatographic separation was performed on Welch Ultisil XB-CN, 250 × 4.6 mm, 5 µm C18 column at a 1 mL/min flow rate. The mobile phase consisted of 100 mmol/L sodium acetate (pH 6.5), acetonitrile (93:7, v/v [solvent A]) and water, acetonitrile (20:80, v/v [solvent B]).
Fatty Acid Composition
Fatty acid profiles were generated from the chloroform (CHCl3) fraction of 1.00 g of powdered samples obtained from each drying method. Fatty acid methyl esters (FAMEs) were prepared 6 and analyzed using gas chromatography/mass spectrometry (GC-MS) with an Agilent 7890B gas chromatograph coupled to a 5977B mass selective detector (Agilent Technologies, Santa Clara, CA, USA) and a 30 m × 0.25 mm i.d. × 0.25 µm DB-5 MS column (J & W Scientific, Folsom, CA, USA) column. Helium was used as the carrier gas at a flow rate of 1 mL/min. The analysis parameters were set as: oven temperature program, 50 °C (5 min), 50 °C to 250 °C (8 °C/min), 250 °C to 290 °C (4 °C/min) for 60 min of a run time, Helium gas flow rate, 37 cm/s; injector and detector temperatures, 290 o C. Samples (1 μL) were injected. EIMS, electron energy, 70 eV; ion source and connecting parts temperature, 250 °C. The acquisition was performed in scanning mode (mass range m/z 35–400 u). Compounds were identified using relative retention times and their mass spectra. The triplicated results were processed using the area percentage obtained from the datasheet of the GC-MS analysis. Each mass spectrum was compared with corresponding reference standard data reported in the mass spectra from NIST 05 in-house libraries.6,7
Microbiological Analysis
The microbiological quality of dried plant samples was determined by enumerating aerobic mesophilic bacteria (ISO 4833-1:2013), 8 Escherichia coli (ISO 7251: 2005), 9 Salmonella spp. (ISO 6579-1: 2017) 10 also yeasts and molds (ISO 21527-1:2008). 11
Chemical Profile of Secondary Metabolites
Total Phenolic, Flavonoid, and Condensed Tannin Content
The total phenolic content (TPC) of the methanol extracts was measured using the Folin-Ciocalteu assay, 12 with gallic acid as the standard. The results were expressed as gallic acid equivalents (GAE). The total flavonoid content (TFC) was determined via a colorimetric assay, 13 which involved the formation of a flavonoid-aluminum complex, with catechin as the standard and results reported as catechin equivalents (CE). The condensed tannin content (CTC) was assessed using the Vanillin/H2SO2 colorimetric method, also with catechin as the standard. 14
Bioactivity Profiles
In Vitro Antioxidant Activity
The DPPH radical scavenging capacity and ABTS assays were performed using methods employed in previous studies. 15 The results of DPPH and ABTS radical scavenging assays were expressed as IC50 values. The ferric-reducing antioxidant power (FRAP) was determined according to the method outlined in a previous study. 16 Butylated hydroxytoluene (BHT) was used as the standard for antioxidant assays.
In vitro Anti-Inflammatory Activity
Human red blood cell (HRBC) assay was performed to evaluate anti-inflammatory activity and IC50 values were calculated. 17
In Vitro Antibacterial Activity
The crude extracts were screened for antibacterial activity against Staphylococcus aureus (ATCC 25923), Bacillus subtilis (MTCC 121), Pseudomonas aeruginosa (ATCC 2785) and Escherichia coli (ATCC 25922) using the agar well diffusion method. The antibacterial activity of the test samples was determined by measuring the diameter of the inhibition zone. 16
Statistical Analysis
All the results were expressed as the mean ± standard deviation (SD) of three independent values. IC50 values were obtained by curve fitting with GraphPad Prism 8 (GraphPad Software, San Diego, CA, USA). Minitab 17 (Minitab Inc., State College, PA, USA) was used to assess the significant (p < 0.05) differences among treatments using one-way ANOVA and Tukey's multiple comparison tests with a 95% confidence level.
Results
Determination of Color
In the present study, dried S. brachiata samples consistently exhibited a range of light green color (Table 1 and Figure 2). This study also analyzed three colorimetric parameters: L*, a*, and b*, then found significant differences (p < 0.05) among treatments. Particularly, the L* parameter indicating lightness, showed that samples treated at lower temperatures (ie, −40 °C) displayed higher brightness compared to those treated at higher temperatures (ie, 50 °C). Furthermore, the greenness parameter (a*) was notably higher in FD samples compared to samples processed through MVD and HPOD. The intensity of the yellow color was observed to be the highest in FD samples, followed by MVD and HPOD samples.
The Proximate Composition, Color Measurements, Mineral Composition and Microbial Quality of S. brachiata Subjected to Different Drying Treatments.

microwave-vacuum drying, ‡freeze-drying, §heat pump oven drying. L* (lightness), a* (green-red), and b* (blue-yellow). The values are the means ± standard deviations for three replicates and those in the same row not sharing the same superscript letter are significantly different from each other (p < 0.05). * Limit of detection µg/g, n. d: not detected.
Nutritional Characterization
Proximate Analysis
The proximate compositions of dried S. brachiata are summarized in Table 1 and depicted in Figure 3. The moisture content was lowest in the HPOD powders, followed by MVD and FD, with significant variation (p < 0.05) observed across the drying methods. Among the three drying methods, HPOD was found to be the most effective in removing moisture.

Effect of different drying method on proximate composition of S. brachiata. Each bar represents mean ± standard deviation of 3 replicates.
The carbohydrate content of dried powders obtained from S. brachiata varied significantly (p < 0.05) depending on the drying method employed. Carbohydrates were the predominant component of the S. brachiata dry mass, followed by ash and crude protein. The carbohydrate content in aerial parts of S. brachiata prepared using different drying methods was significantly different (p < 0.05) from each other, and the FD method showed the highest carbohydrate content.
In terms of protein content, both the FD and MVD samples demonstrated higher crude protein levels compared to the HPOD dried samples. Furthermore, the FD method showed significantly higher crude fat content (p < 0.05) than the HPOD method. A significant treatment effect (p < 0.05) was also observed on both crude fiber and dietary fiber contents, with FD samples yielding significantly higher values for both parameters compared to the other drying methods.
Mineral Composition
Owing to the high ash content of S. brachiata, knowledge of the composition of their mineral fractions is of particular interest. Table 1 shows the mineral profile of S. brachiata samples based on dry weight. Figures 4 and 5 illustrate the effects of different drying methods on the levels of minerals in S. brachiata.

Effect of different drying methods on the level of Sodium (Na), Calcium (Ca), Magnesium (Mg), Phosphorous (P), and Potassium (K) in S. brachiata. MVD - microwave-vacuum drying, FD - freeze-drying, HPOD - heat pump oven drying. Each bar represents mean ± standard deviation of 3 replicates.

Effect of different drying methods on the level of Copper (Cu), Zinc (Zn), Iron (Fe), Arsenic (As), Cadmium (Cd), and Lead (Pb) in S. brachiata. MVD - microwave-vacuum drying, FD - freeze-drying, HPOD - heat pump oven drying. Each bar represents mean ± standard deviation of 3 replicates. * < detection limit (0.1 µg/g).
According to the elemental analysis, Na was the most abundant macro element, followed by P and K. When comparing the Na content among the drying methods, the HPOD method recorded a significantly higher amount of Na compared to the other two drying methods. Interestingly, significant differences in Fe content were observed among the three drying methods. FD samples exhibited the highest Fe content at 206.23 µg/g, which was three and five times higher than that of samples dried using the HPOD and MVD methods respectively. Moreover, the Zn content of FD samples was almost double than that of MVD samples.
Salt content (as NaCl) ranged from 39% to 41% in all drying methods. There were no significant differences in salt content among the three drying methods (Table 1).
In terms of heavy metals, S. brachiata powder contained As and Cd in concentrations ranging from 0.27 to 0.39 µg/g for As and 0.15 to 0.49 µg/g for Cd. However, Pb was undetectable, with concentrations below the detection limit of 0.1 µg/g.
Amino Acid Composition
The study also aimed to assess the amino acid composition in each sample obtained from different drying methods. The results of the quantitative determination of amino acid content are presented in Table 2 and Figure 6. Eight essential amino acids (EAA) - methionine, leucine, lysine, phenylalanine, tyrosine, isoleucine, threonine, and histidine (except tryptophan) - along with nine non-essential amino acids (NEAA) were consistently present in all tested samples. The EAA constituted more than half of the total amino acids (TAA) found in the dried samples, with the EAA/TAA ratio ranging from 0.53 to 0.54. Elevated amounts of threonine, lysine, proline, tyrosine, glycine, alanine, methionine, and cysteine were detected in MVD samples, while the FD samples of S. brachiata reported elevated amounts of glutamic acid, aspartic acid, histidine, and arginine. In addition, S. brachiata, which was subjected to HPOD, reported significantly higher amounts of valine, phenylalanine, isoleucine, and leucine.

Effect of different drying methods on the level of amino acids in S. brachiata. MVD - microwave-vacuum drying, FD - freeze-drying, HPOD - heat pump oven drying. Each bar represents mean value of 3 replicates.
Amino Acid Composition (mg/kg) of S. brachiata Subjected to Different Drying Treatments.

microwave-vacuum drying, ‡freeze-drying, §heat pump oven drying. The values are the means ± standard deviations for three replicates and those in the same row not sharing the same superscript letter are significantly different from each other (p < 0.05). * Tryptophan has not been measured.
Fatty Acid Composition
The oil yields extracted from the tender aerial parts of S. brachiata varied depending on the drying method. The average oil yields from the aerial parts dried using MVD, FD, and HPOD were 0.36%, 0.32%, and 0.35%, respectively. Table 3 presents the fatty acids, and other chemical constituents present in the lipid extract of the aerial parts of S. brachiata prepared using three different drying techniques.
GC-MS Analysis of the Lipid Extract Obtained from Dried Powders of S. brachiata Tender Aerial Parts.
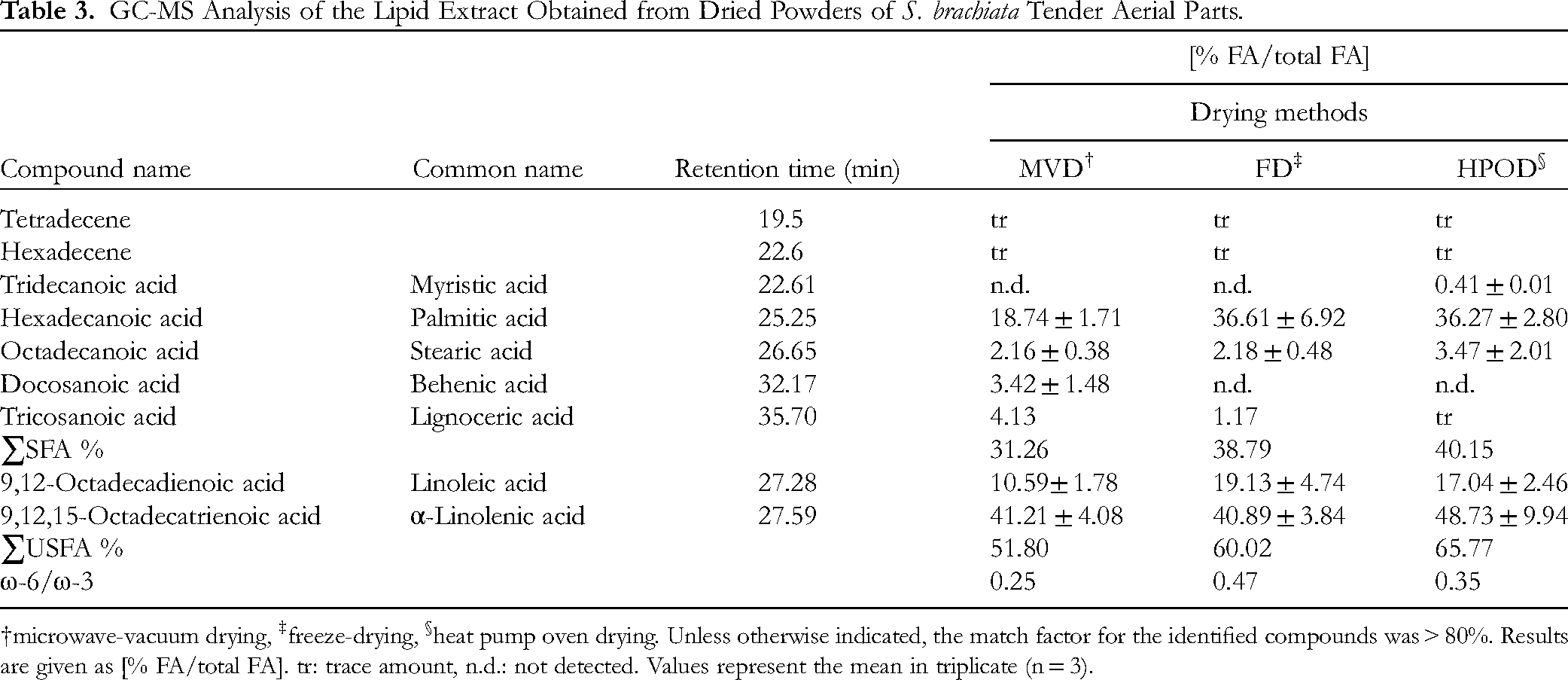
microwave-vacuum drying, ‡freeze-drying, §heat pump oven drying. Unless otherwise indicated, the match factor for the identified compounds was > 80%. Results are given as [% FA/total FA]. tr: trace amount, n.d.: not detected. Values represent the mean in triplicate (n = 3).
The chemical constituents present in the lipid extracts were similar across all three drying methods, though there were differences in the percentages of each constituent. Palmitic acid was identified as the major saturated fatty acid (SFA). Notably, the oils extracted from the aerial parts of S. brachiata were rich in unsaturated fatty acids (USFAs), particularly ω-6 and ω-3 fatty acids. The total USFA content in the FD and HPOD samples was 60% and 66%, respectively. The ω-6 to ω-3 ratios were recorded as 0.25 (MVD), 0.47 (FD), and 0.35 (HPOD), with the FD samples exhibiting the highest ratio.
Microbiological Analysis
In this study, microbiological analysis of dried Salicornia samples revealed significant contamination, with elevated levels (>100 CFU/g) of aerobic mesophilic bacteria as well as yeasts and molds. However, no pathogenic microorganisms, Escherichia coli or Salmonella spp., were detected (Table 1), in accordance with Codex Standard, 18 which mandates the absence of harmful pathogens in food-grade salt.
Chemical Profile of Secondary Metabolites
Total Phenolic, Flavonoid, and Condensed Tannin Content
The effects of the MVD, FD, and HPOD drying method on the retention of TPC, TFC and CTC of S. brachiata were compared on a dry weight basis. The results are presented in Table 4 and Figure 7. Methanol extracts from the FD samples exhibited the highest levels of TPC and TFC, with values of 43.68 mg GAE/g DW and 72.44 mg CE/g DW, respectively. In contrast, the methanol extracts from MVD and HPOD samples showed comparatively lower levels of of TPC and TFC. S. brachiata subjected to three drying methods reported an average CTC of 14.36 mg CE/g DW, with no signifiant differences among the CTC values of the drying methods.
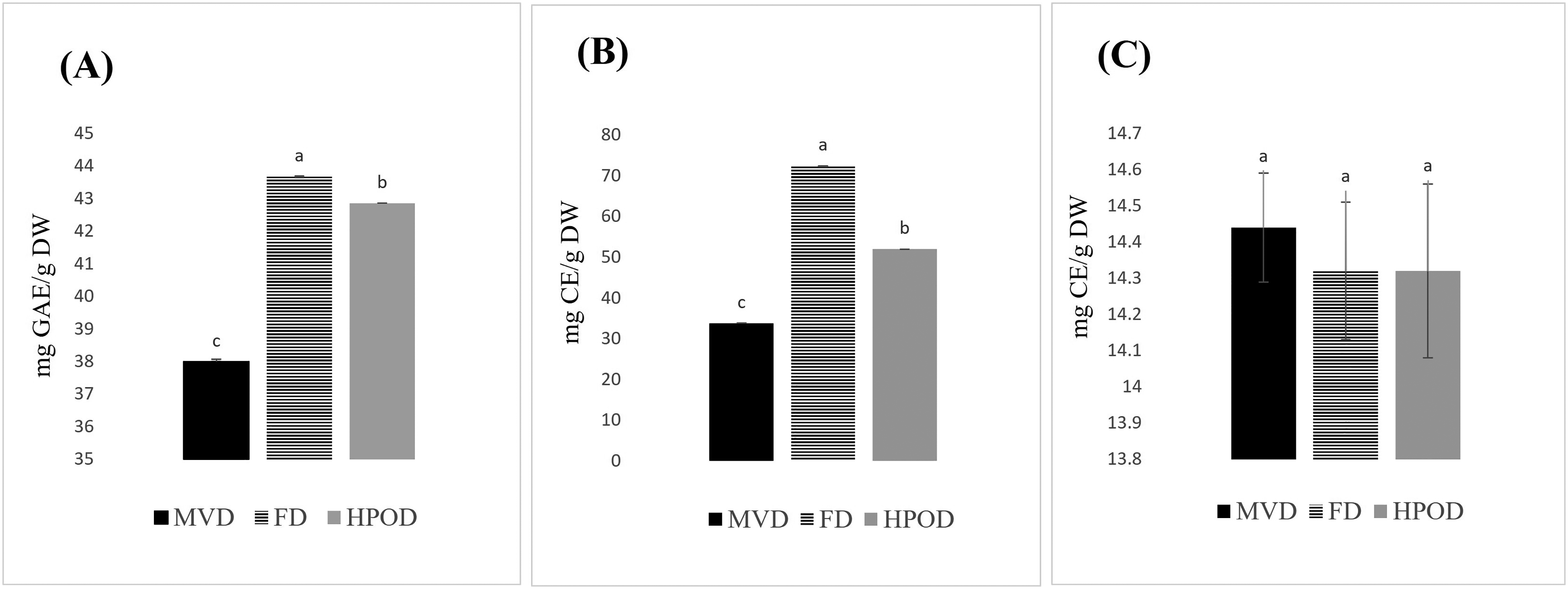
Effect of different drying methods on total phenolic content (A), total flavonoid content (B), and condensed tannin content (C) in extracts of S. brachiata. MVD - microwave-vacuum drying, FD - freeze-drying, HPOD - heat pump oven drying. Each bar represents mean ± standard deviation of 3 replicates.
Total Phenolic Content (TPC), Total Flavonoid Content (TFC), and Condensed Tannin Content (CTC) in Extracts of S. brachiata Obtained Using Three Drying Methods.
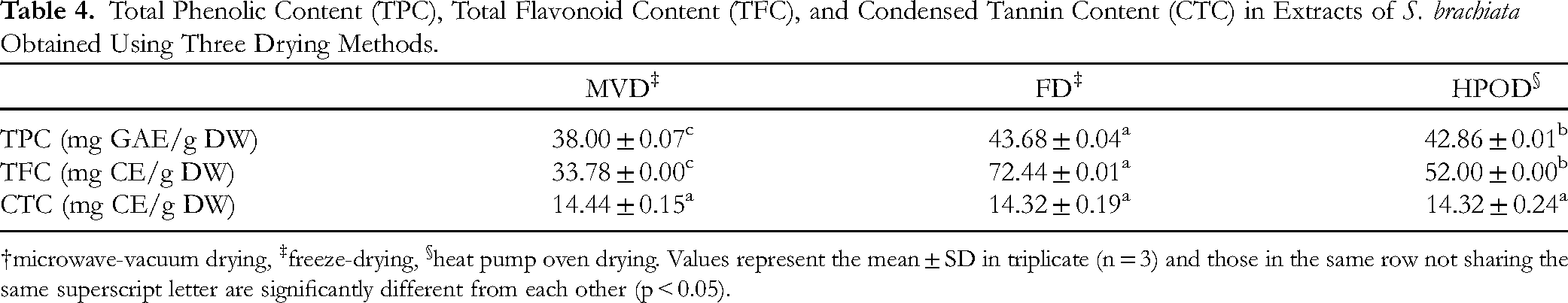
microwave-vacuum drying, ‡freeze-drying, §heat pump oven drying. Values represent the mean ± SD in triplicate (n = 3) and those in the same row not sharing the same superscript letter are significantly different from each other (p < 0.05).
Bioactivity Profiles
In Vitro Antioxidant Activity
The DPPH, ABTS, and FRAP assays were employed to evaluate the radical scavenging capacity of the extracts, with lower IC50 values indicating higher antioxidant activity. Analysis of the DPPH assay reveals that the FD extract exhibited the most robust antioxidant activities, followed by the MVD and HPOD extracts. Furthermore, in the ABTS assay similar results were displayed revealing the highest antioxidant activity in the FD extract with the lowest IC50 value subsequent to the activity given by the MVD and HPOD extracts (Figure 8 and Table 5).

Effect of different drying methods on DPPH (A) and ABTS (B) radical scavenging activity of methanolic extracts of S. brachiata. MVD - microwave-vacuum drying, FD - freeze-drying, HPOD - heat pump oven drying, Standard - Butylated hydroxytoluene (BHT). Each bar represents mean ± standard deviation of 3 replicates.
Anti-Inflammatory Activity and Antioxidant Activity (IC50 Values, µg/mL) of Methanol Extracts of S. brachiata Obtained from MVD, FD, and HPOD.

microwave-vacuum drying, ‡freeze-drying, §heat pump oven drying. BHT was used as the positive control for DPPH and ABTS assays and Aspirin was used for anti-inflammatory assay. Values represent the mean ± SD in triplicate (n = 3). In each column, different letters indicate significant differences.
In the FRAP assay, conducted at a concentration of 0.05 mg/mL, the MVD samples demonstrated the highest reducing power, comparable to the positive control BHT, followed by the FD and HPOD samples (Figure 9A).
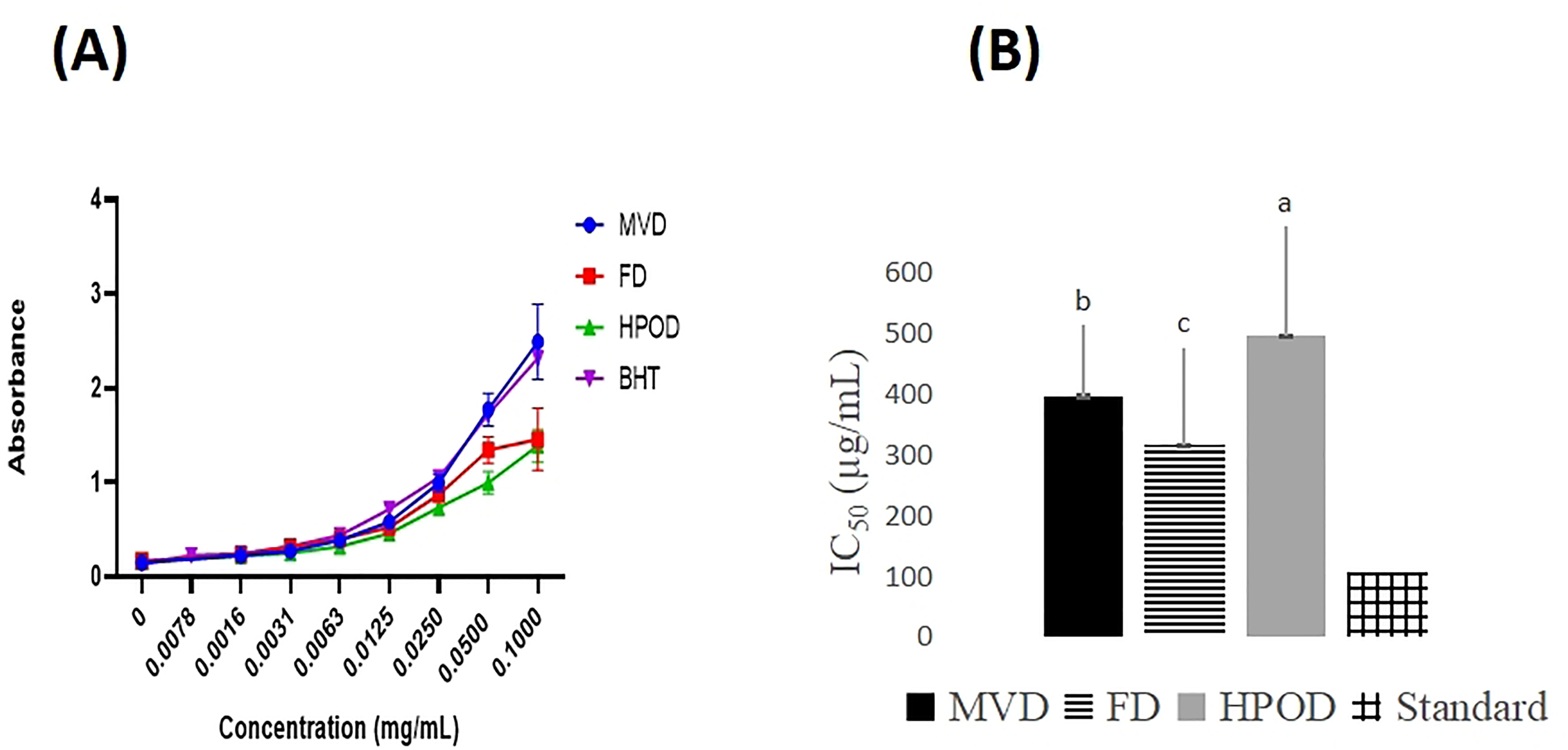
Effect of different drying methods on ferric reducing absorbance power values (A) and anti-inflammatory activity (B) of methanolic extracts of S. brachiata. MVD - microwave-vacuum drying, FD - freeze-drying, HPOD - heat pump oven drying, Standard - Butylated hydroxytoluene (BHT). Each bar represents mean ± standard deviation of 3 replicates.
In Vitro Anti-inflammatory Activity
In the present study, the anti-inflammatory activity of S. brachiata was performed using the heat-induced membrane stabilization method. 17 Table 5 and Figure 9B displays the anti- inflammatory activity of S. brachiata extracts, with aspirin as the standard drug for comparison.
The HRBC test results indicated that extracts from S. brachiata subjected to various drying methods MVD, FD, and HPOD exhibited moderate anti-inflammatory effects compared to aspirin. Among the methods, FD demonstrated the highest activity, surpassing MVD and HPOD.
This suggests that the choice of drying method significantly impacts the bioavailability of the plant's bioactive compounds. Additionally, a recent study reported lower anti-inflammatory activity (IC50 value of 838.67 µg/mL) for the methanolic extract of shade dried S. brachiata (comprising fresh leaves, bark and root) in the HRBC assay. 19
In vitro Antibacterial Activity
Table 6 represents the antibacterial activity of S. brachiata methanolic extracts. Concentration-dependent antimicrobial activity was observed in all samples, with the highest activity recorded in the FD sample. Significant activity was noted against gram-negative bacteria, with E. coli (3.57 ± 0.01 cm) and P. aeruginosa (3.04 ± 0.04 cm), as well as against gram-positive bacteria, including S. aureus (3.05 ± 0.02 cm) and B. subtilis (3.14 ± 0.20 cm). These results suggest that S. brachiata extracts prepared using FD possess antibacterial potential against both gram-positive and gram-negative bacteria, aligning with findings from previous studies. 19
Antibacterial Activity of S. brachiata Extracts Evaluated by Well Diffusion Method.

NS: No sensitivity. Values represent the mean in triplicate (n = 3).
Discussion
A true halophyte, S. brachiata is known for its nutritional and bioactive properties. It is consumed as a food source, particularly in fresh, fermented, cooked, dehydrated, or pickled forms. It has been documented as a rich source of nutrients, especially proteins, vitamins, and unique oligosaccharides. 20 However, these chemical components may be lost or depleted during processing, particularly dehydration. This study evaluated the impact of three drying methods—FD, MVD, and HPOD—on the nutrient composition and bioactive properties of S. brachiata.
The results indicate that, FD is particularly effective in preserving the visual appeal of S. brachiata, retaining its greenness, yellowness, and brightness when compared to HPOD and MVD. As product appearance is critical in food related industries aiming for consumer acceptance, FD offers significant advantages. Food industries aiming to utilize dehydrated S. brachiata would benefit from FD for better preservation of its appearance. Additionally, S. brachiata subjected to all three drying methods reported moisture contents below 7%, which ensures safe storage throughout its shelf life by inhibiting microbial growth and enzymatic activity.
In the proximate analysis, S. brachiata showed higher protein levels than S. ramosissima, but lower than those for S. bigelovii, and S. herbacea.21-25 The presence of higher crude protein content in FD and MVD batches of S. brachiata aligns with previous research, which has demonstrated that FD preserves protein integrity better than high-temperature methods.1,26 High-temperature drying methods, such as HPOD, tend to cause protein denaturation, which reduces protein extractability. Thus, low-impact methods like FD and MVD are recommended for S. brachiata drying, as they minimize protein degradation, making the plant an excellent source of protein for functional foods.
The average crude fiber content of S. brachiata (6% dry weight) was found to be comparable to that of S. bigelovii, but lower than S. ramosissima.23,24 The insoluble fiber in Salicornia offers an opportunity for inclusion in fiber-enriched foods. While the fiber content is moderate, it can still be valuable in food formulations, especially when combined with other high-fiber ingredients.
Halophytes like S. brachiata have a remarkable ability to accumulate both organic and inorganic solutes, making them an intriguing source of dietary minerals. 27 S. brachiata is known to accumulate 30–40% of sodium chloride (NaCl) in its tissues, which aligns with the findings of this study. 28 This makes S. brachiata a promising candidate for bio-salt applications in the food industry, offering a natural alternative to conventional salt. However, the study also found that copper (Cu) levels exceeded the Codex limit for safe consumption in all samples. While other heavy metals like arsenic (As), cadmium (Cd), and lead (Pb) were within acceptable limits, the elevated copper levels pose potential health risks, including kidney damage, neurotoxicity, and gastrointestinal distress. Therefore, assessing the heavy metal content in the soil when cultivating S. brachiata for human consumption is crucial. These findings emphasize the importance of quality control and the adoption of advanced agricultural practices to reduce risks associated with heavy metal contamination.
The seeds of S. brahiata are reported to be rich in sulfur-containing amino acids including cysteine, methionine, and tyrosine. 29 In line with this, the dried aerial parts of this study also showed remarkable levels of these amino acids. However, no consistent relationship was observed between the drying methods and the composition of amino acids.
The lipid extracts S. brachiata aerial parts contained a higher percentage of α-linolenic acid (C18:3) (>40%) compared to the fatty acid composition of S. fruticosa seeds. 30 In all lipid extracts, the percentage of USFAs exceeded that of SFAs. The low ω-6/ω-3 ratio suggests that S. brachiata oil may offer benefits in reducing the risk of cardiovascular diseases, cancer, and autoimmune disorders.
Microbiological analysis is essential for any natural product used as food or food ingredient. Microbiological analysis revealed elevated levels of aerobic mesophilic bacteria and fungi in the samples, suggesting that the harvesting and processing conditions may contribute to microbial contamination. 31 In this study, Salicornia plants were harvested from inland coastal salt marshes and processed without specific sanitizing treatments, other than rinsing with tap water. These findings highlight the importance of implementing stringent sanitation practices during both harvesting and processing stages. To ensure the safety and quality of S. brachiata as a food ingredient, improved hygiene protocols and regular microbiological testing are necessary, particularly in the production of products for human consumption.
Salicornia spp., thrives in extreme environments such as high salinity and low humidity, likely produces robust defense mechanisms, including antioxidant compounds, in response to these stressors. 25 It is evident by the high antioxidant properties of S. brachiata observed in this study.
The results of TPC, TFC, and DPPH and ABTS inhibition indicate that FD is the most effective drying method for preserving antioxidant compounds in S. brachiata. Extracts from FD samples exhibited significantly higher TPC compared to those from MVD and HPOD. This finding aligns with previous studies on S. ramosissima, where freeze-drying also retained higher TPC compared to oven drying. 3 Similarly, studies on S. fruticosa exposed to high oven-drying temperatures (70 °C) showed a significant reduction in polyphenol content, which contributed to the loss of antioxidant properties. 32 The decrease in phenolic compounds with increasing drying temperatures (40-70 °C) has been attributed to both enzymatic and non-enzymatic oxidation reactions.33,34 However, tannin content remained stable across all drying methods, suggesting that tannins in S. brachiata are relatively resistant to degradation, regardless of the drying conditions.
The preservation of bioactive compounds through freeze-drying would enhance the use of S. brachiata in functional foods, dietary supplements, and nutraceutical formulations. Specifically, freeze drying would be crucial in the development of bio salts, where the retention of bioactive compounds is essential for maximizing health benefits.
Conclusion
This study is notable for several reasons and offers significant insights for both the scientific community and the general public. Firstly, while S. europaea, S. ramosissima, and S. herbacea have been extensively studied regarding bioactivities and proximate analysis, comprehensive research on S. brachiata, a halophyte native to the Indian subcontinent, Myanmar, and Sri Lanka, is limited. This study fills this gap by focusing on S. brachiata.
Secondly, while most of the existing studies have utilized air-dried samples, this research, compared the effects of three different drying methods, freeze-drying (FD), microwave-vacuum drying (MVD), and heat pump oven drying (HPOD), on bioactivities and proximate composition. The study demonstrated that S. brachiata is rich in phytochemicals and exhibited significant bioactivities, including antioxidant, anti-inflammatory, and antibacterial properties. Among the drying methods, FD proved the most effective in preserving these beneficial properties. It was also revealed that the lipid extract obtained from the aerial parts of S. brachiata is rich in unsaturated fatty acids.
Thirdly, the study highlights the importance of monitoring soil for heavy metal contamination, as S. brachiata has a notable capacity to accumulate heavy metals, which is crucial when cultivating plants for human consumption. This is the major limitation of this article.
Finally, this paper provides a foundational reference for selecting an effective drying method for preparing bio-salt from the tender aerial parts of S. brachiata on an industrial scale, while maintaining its bioactivities. It serves as both a comprehensive guide and a baseline study for future research and industrial applications.
Footnotes
Acknowledgements
The authors thank A.T. Kannangara for GC/MS analysis, and S.A.A.K. Suriyaarachchi for technical support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable for this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was conducted as part of an industrial partnership between Rajarata University of Sri Lanka and Mega Plantations (Pvt.) Ltd, Sri Lanka. It was supported by the Science and Technology Human Resource Development Project, Ministry of Education, Sri Lanka, and funded by the Asian Development Bank (grant no. CRG/R2/RJ1).

