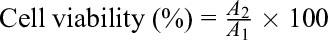Abstract
The effects of drying methods on the contents of four nonvolatile and five volatile components and the immunoregulatory activities of four components in Osmamthus fragrans flowers were investigated. In general, microwaving preserved more nonvolatile components than the other methods, while the sun or shade method preserved more volatile components. Nonvolatile components such as salidroside and acteoside and volatile ingredients such as linalool and linalool oxide exhibited better immunoregulatory activity than the other ingredients. Taken together, O. fragrans flowers dried by microwaving resulted in the best immunoregulatory activity. This study provides evidence for the optimal drying method for O. fragrans flowers as food and medicine.
Osmamthus fragrans (Thunb.) Lour., family Oleaceae, 1 is not only a famous aromatic and ornamental plant, but also an excellent edible and medicinal species. 2 Its flowers can be made into scented tea, wine, and cake. 3,4 In addition, these can be used to treat stomach, liver, and kidney diseases. 5
The utilization of O. fragrans flowers in the treatment of diseases is based on their bioactive constituents, which consist of nonvolatile and volatile components. The nonvolatile compounds mainly include phenylpropanoids, flavonoids, terpenoids, and lignans. 6 In O. fragrans flowers, the phenylpropanoids mainly exist in the form of either p-hydroxyphenylethanol esters or glycosides. Eleven phenylpropanoids, such as acteoside, salidroside, and isoacteoside, have been identified in the aqueous extract of O. fragrans flowers. 7 Phenylpropanoids have antioxidant, anti-melanogenesis, anti-hypoxia, anti-inflammatory, anti-aging, and other biological activities. 8 -12 Six flavonoids were isolated from the ethanol extract of O. fragrans flowers, namely, quercetin, rutin, genistin, kaempferol, isorhamnetin, and naringin, which have different anti-inflammatory, DPPH-free radical scavenging, and anti-proliferation activities. 13 A secoiridoid glycoside (8E-ligstroside) and a coumaric acid analog with a monoterpene moiety (floraosmanol A) inhibit the activity of beta-secretase and nitric oxide (NO) production in lipopolysaccharide-activated RAW264.7 macrophages, respectively. 14,15 Lee and collaborators obtained seven lignans from O. fragrans flowers, of which phillygenin has a great effect on protecting liver and preventing NO production. 16,17 In addition, nine polyphenols with antioxidant properties and 8 monosaccharides were isolated and identified in ethanol extracts of O. fragrans flowers. 18,19 The volatile components typically include terpenes, aldehydes, esters, ketones, and alcohols. 20 Forty-one volatile organic components were identified from 29 cultivars of O. fragrans groups, and the major volatile compounds were alpha-ionone, beta-ionone, linalool, linalool oxide, and geraniol. 21 A total of 84 volatiles were detected by GC-MS analysis, and among these, terpenoids, ketones, and esters were predominant in the SPME extracts, while aromatics and alcohols were abundant in SDE extracts. 22 Eugenol and geraniol were screened as the main active ingredients with potential neuroprotective and anti-tumor effects. 23
Drying is necessary as a means of removing moisture in fresh products to ensure their enzymatic and microbiological stability. 24 Different drying methods such as hot air-drying, microwave drying, infrared drying, freeze-drying, and vacuum oven drying have their own unique characteristics and different effects on the chemical components and bioactivities of fresh products. Microwave drying and halogen lamp-microwave drying performed significantly better than traditional hot-air drying in enhancing the quality of black tea, especially the volatile and taste compounds. 25 Freeze-drying results in better physicochemical properties and antioxidant activities of mulberry leaf polysaccharides in comparison with air-drying and hot air-drying. 26 Also, freeze-drying produced the best dried jujube fruits and bitter gourd slice chemical composition, antioxidant capacity, and sensory quality. 27,28 Generally speaking, lower drying temperatures are more conducive to the preservation of volatile components. 29 Oven and vacuum oven-drying at 35 °C were recommended for Thymus daenensis as these generated the highest quantity of essential oils of the appropriate quality. 30
Previous studies have reported that harvest stages, flower tissues and organs, cultivated varieties, extraction methods, and preservation methods all had a great impact on the volatile components of O. fragrans flowers. 31 -38 Nevertheless, our current understanding of the effects of different drying methods on chemical components in O. fragrans flowers is limited. Also, to the best of our knowledge, no investigations on the immunomodulatory activities of chemical components in O. fragrans flowers have been conducted to date. Therefore, the aim of this research was to determine the optimal drying method for O. fragrans flowers from three O. fragrans groups using the contents of nine components as indices. Another goal of the present study was to determine the cell viability and phagocytic activities of RAW 264.7 cells to assess the immunoregulatory activity of O. fragrans.
Materials and Methods
Chemicals and Reagents
Acetonitrile and methanol (HPLC-grade) were purchased from Merck (Darmstadt, Germany). Standard products such as salidroside, acteoside, oleanolic acid, and ursolic acid were obtained from the National Institutes for Food and Drug Control (Beijing, China). L (-)−2-Octanol (as internal standard), linalool, linalool oxide, geraniol, α-ionone, and β-ionone were purchased from Shanghai Yuanye Biotechnology Co., Ltd. (Shanghai, China). Light petroleum, boiling range of 80 to 120 °C, was purchased from J&K Chemical, Ltd. (Beijing, China). Water of HPLC grade was purified by a reverse osmosis system (Millipore, Ireland). All the reagents of HPLC grade were filtered across a 0.45 µm membrane before the experiment. Dimethyl sulfoxide (DMSO), 3-(4,5-dimethylthiazol-2-y1)−2,5-diphenyltetrazolium bromide (MTT), trypan, concanavalin A (Con A), penicillin G, and streptomycin sulfate were obtained from Sigma Chemical Co. (St. Louis, MO, USA), and Gibco 1640 medium and fetal bovine serum (FBS) from Gibco Invitrogen Corp. (San Diego, CA, USA). Other reagents and chemicals were of analytical grade.
Plant Material
Fresh O. fragrans flowers were harvested from 3 different O. fragrans groups in our campus (32°33’ N and 117°1’ E), which showed flower colors of almost orange, silver, and gold. These were identified by Dr Li Feng from Shandong University of Traditional Chinese Medicine separately as O. fragrans var. aurantiacus (Batch number: 20191006‐1), O. fragrans var. latifolius (Batch number: 20191006‐2), and O. fragrans var. thunbergii (Batch number: 20191006‐3). For convenience, these three groups will hereafter be called “Aurantiacus,” “Latifolius,” and “Thunbergii.” After drying, the flowers were stored in a glass dryer filled with desiccant silica gel. They were analyzed within a month after drying.
Drying Methods
Shade drying (SHD), sun drying (SUD), quick-lime drying (QLD), oven drying (OD), and microwave drying (MD) of fresh flowers from three O. fragrans groups were utilized. SHD and SUD were separately conducted either indoors or in the sun. QLD was performed by placing fresh flowers in the upper glass dryer, while the lower layer was filled with quick lime. OD was operated separately at 40 °C, 50 °C, 60 °C, and 70 °C in a drying oven (DHG-9070B, Shanghai Pei for Experimental Instrument Co., Shanghai, China). MD was performed separately at medium-low power (MLP), medium power (MP), medium-high power (MHP), and high power (HP) in a microwave oven (M1-L213B, Midea Group Co., Foshan, China). All dried O. fragrans flowers were dried until constant weight.
Sample Extraction and Preparation
The dried flowers from three O. fragrans groups were extracted and prepared for HPLC analysis to identify four kinds of non-volatile components. Briefly, 0.1 g of the dried O. fragrans flowers was measured precisely and placed into a 50 ml conical flask with a stopper. Then, 10 ml of methanol was added to the flask, and extraction was performed for 30 minutes under 100 w power and 40 kHz frequency at room temperature using an ultrasonic cleaner (KQ-100 DE, Kunshan Ultrasound Instrument Co., Suzhou, China). The lost weight was compensated by methanol after being cooled to room temperature, and the extracts were passed through a double-layer filter paper. The continuous filtrates above were collected and stored at 4 °C for the subsequent content determination of the four kinds of non-volatile components.
The dried flowers from three O. fragrans groups were extracted and prepared for GC analysis to identify five kinds of volatile components. Briefly, 10 g of the dried O. fragrans flowers was measured precisely and placed into a 500 ml round-bottom flask. Then, 300 ml of deionized water was added to the flask. Using a Clevenger apparatus, extraction was performed for 4 hours. Then, 2 ml of L (-)−2-octanol light petroleum solutions with a boiling range of 80 to 120 °C was added to the distilled extracts. The above liquids were thoroughly mixed, and the supernatants were separated and stored at 4 °C for subsequent identification of volatile components. All experiments were performed in triplicate.
HPLC
Four kinds of non-volatile components were analyzed by HPLC using an E2695 LC instrument (Waters Instruments Co., MA, USA) with an online degasser, a quaternary pump, a 2489 ultraviolet-visible detector (UVD), and an autosampler. Approximately 10 µL of the HPLC samples was analyzed with a Symmetry C-18 analytical column (250 mm × 4.6 mm, 5 µm, Waters Corp., USA) using a linear gradient with water-phosphoric acid (100:0.05, v/v) as solvent A and acetonitrile as solvent B. The gradient program was as follows: 10% B in 0‐8 minutes, 10% to 30% B in 8‐22 minutes, 30% to 85% B in 23‐36 minutes, and 85% to 10% B in 37‐66 minutes. The velocity of flow was 1.0 mL min-1, and the column temperature was kept at 30 °C. Detection was performed at a wavelength of 220 nm in 0‐25 minutes and 210 nm in 26‐66 minutes.
Gas Chromatography (GC)
Five kinds of volatile components of the GC samples were analyzed using a GC-4000A GC instrument (East & West Analytical Instruments Co., Beijing, China) with a nitrogen canister, a flame ionization detector (FID), a hydrogen generator, an air pump, and a 1 µL injection needle. Approximately 1 µL of sample was injected into the GC instrument fitted with an AE.SE-54 nonpolar analytical column (30 m × 0.32 mm, Lanzhou Atech Technologies Co., Lanzhou, China) and with high purity nitrogen gas as carrier using a temperature gradient program. The gradient program was as follows: 50 °C for 2 minutes, 50 to 160 °C at the rate of 2 °C min-1, 160 to 250°C at the rate of 22.5 °C min-1, and 250 °C for 12 minutes. The temperature of the gasification chamber and flame ionization detector was set at 250 °C. The pressure of nitrogen, air, and hydrogen were 0.4, 0.2, and 0.09 MPa, respectively.
Assessment of Cell Viability
The cytotoxicity of the relevant chemical components of O. fragrans flowers, using various concentrations, was evaluated using an MTT test, with some modifications.
39
Approximately 1
where A1 is the absorbance value of the blank control group, and A2 is the absorbance value of the treatment group.
Phagocytosis Assay
The phagocytic activities of RAW 264.7 cells treated with various concentrations of relevant chemical components of O. fragrans flowers were examined by neutral red uptake assay.
40
RAW 264.7 cells were seeded at a density of 2.7
where B1 is the absorbance value of the blank control group, and B2 is the absorbance value of the treatment group.
Statistical Analysis
All data were reported as the mean ± standard deviation of 3 samples. Statistical analysis was performed with SPSS 19.0. Significance differences were tested using ANOVA at a significant level (P < 0.05).
Results
Yields and drying time of dried O. fragrans flowers with different drying methods.
To avoid moisture interference with the chemical composition content, the yields of dried O. fragrans flowers with different drying methods were calculated and compared (Table 1). The yields were calculated using the following equation:
where C1 is the weight of 100 g fresh flowers, and C2 is the weight of 100 g fresh flowers after drying.
Yields and Drying Time of Dried O. Fragrans Flowers Using Various Drying Methods.

Abbreviations: HP, high power; MD, microwave drying; MHP, medium-high power; MLP, medium-low power; MP, medium power; OD, oven-drying; QLD, quick-lime drying; SHD, Shade drying; SUD, sun drying.
Data are presented as the mean ± standard deviations (n = 3).
The different drying methods had no significant effect on dried flower yield (P > 0.05). Also, drying via MD and OD utilized less than 15 minutes and 6 hours, respectively, while SHD, SUD, and QLD spent no less than 3 days. The advantages and disadvantages of these drying methods have also been previously reported. 41
Effect of Different Drying Methods on Nonvolatile Components of O. Fragrans Flowers
The drying process may be accompanied by changes in chemical composition. The peaks of salidroside, acteoside oleanolic acid, and ursolic acid could be well separated from adjacent peaks and, therefore, the analytical conditions could be used to identify the four components.
The amount of ursolic acid in Aurantiacus, Latifolius, and Thunbergii flowers was apparently below the detection level of our quantification method (Tables 2 -4). Also, oleanolic acid was not quantified in Aurantiacus and Thunbergii flowers, but was in Latifolius flowers. Table 3 shows that the highest content of oleanolic acid was about 1.78% (g/100 g) in Latifolius flowers dried by SUD. In addition, oleanolic acid also existed in the Latifolius flowers dried by MD, and their contents initially increased and then decreased with increasing power.
Effects of Drying Methods on the Nonvolatile and Volatile Components of Auranticacus Flowers.

Abbreviations: —, means that the component cannot be detected; HP, high power; MD, microwave drying; MHP, medium-high power; MLP, medium-low power; MP, medium power; OD, oven drying; QLD, quick-lime drying; SHD, Shade drying; SUD, sun drying.
Data are presented as mean ± standard deviations (n = 3).
Effects of Drying Methods on the Nonvolatile and Volatile Components of Latifolius Flowers.

Abbreviations: —, means that the component cannot be detected; HP, high power; MD, microwave drying; MHP, medium-high power; MLP, medium-low power; MP, medium power; OD, oven drying; QLD, quick-lime drying; SHD, Shade drying; SUD, sun drying.
Data are presented as mean ± standard deviations (n = 3).
Effects of Drying Methods on the Nonvolatile and Volatile Components of Thunbergii Flowers.

Abbreviations: —, means that the component cannot be detected; HP, high power; MD, microwave drying; MHP, medium-high power; MLP, medium-low power; MP, medium power; OD, oven drying; QLD, quick-lime drying; SHD, Shade drying; SUD, sun drying.
Data are presented as mean ± standard deviations (n = 3).
The nonvolatile components in O. fragrans flowers are presented in Tables 2 -4. Specifically, the highest contents of acteoside were 2.31%, 1.90%, and 2.24% (g/100 g) in Aurantiacus, Latifolius, and Thunbergii flowers, respectively. Similarly, the highest contents of salidroside were 0.87%, 0.19%, and 0.24% (g/100 g) in Aurantiacus, Latifolius, and Thunbergii flowers, respectively. The highest contents of oleanolic acid, acteoside, and salidroside all appeared in O. fragrans flowers dried with the MD method from the three O. fragrans groups. Taken together, the contents of these three nonvolatile components could be increased by either increasing the temperature (OD) or power (MD). In summary, high temperature such as in OD (60 to 70 °C) and local high temperatures such as MD (MP-HP) are beneficial to the preservation of nonvolatile components.
Effect of Drying Methods on the Volatile Components of O. Fragrans Flowers
Linalool oxide, linalool, geraniol, α-ionone, β-ionone, and δ-decanolide were the most extensively depleted components after natural drying of O. fragrans flowers. 36 Therefore, these were selected as the typical constituents for evaluating the effects of different drying methods on the volatile components. GC was used to determine the contents of these compounds, the peaks of which could be separated from the adjacent ones, and utilized in their assay.
There are several findings on the volatile components of O. fragrans flowers (Tables 2 -4). Specifically, the highest contents of linalool oxide and linalool were 27.93% and 16.64% (mg/100 g) in Aurantiacus flowers, respectively, and 57.77% and 31.87% (mg/100 g) in Thunbergii flowers, respectively. The highest values were observed in Aurantiacus and Thunbergii flowers dried using the OD (40 °C) method. The highest content of linalool oxide was 37.31% (mg/100 g) in Latifolius flowers subjected to the QLD method, while the highest content of linalool was 10.36% (mg/100 g) in Latifolius flowers that underwent the SHD method. The highest contents of geraniol were 17.35%, 29.55%, and 89.93% (mg/100 g) in Aurantiacus flowers dried using the OD (40 °C) method and in Latifolius and Thunbergii flowers with the SHD method, respectively. The highest contents of α-ionone and β-ionone were 94.75% and 2.05% (mg/100 g) in Aurantiacus flowers dried using the SUD method, 72.63% and 1.51% (mg/100 g) in Latifolius flowers dried using the SUD method, and 18.27% and 28.59% (mg/100 g) in Thunbergii flowers dried using the QLD method. Relatively speaking, the contents of linalool oxide, linalool, and geraniol were high in the O. fragrans groups. In addition, the contents of α-ionone were high in Aurantiacus and Latifolius flowers, while that of β-ionone was high in Thunbergii flowers. The high temperature used in OD (50 to 70 °C) and local high temperature such as MD were not beneficial to the preservation of volatile components. In contrast, low temperature such as in OD (40 °C), SHD, SUD, and QLD were beneficial in this respect.
Effect of Chemical Components of O. Fragrans Flowers on Cell Viability of RAW264.7 Cells
The MTT test was used to determine the number of viable cells and cell viability by assessing the activity of mitochondrial succinate dehydrogenase of live cells. The MTT colorimetric method has a very wide range of applications in cell proliferation and differentiation and is related to immunological experiments in cell metabolism. Figure 1 shows that the survival rates of RAW 264.7 cells were basically above 90% after the disposition of salidroside, acteoside, linalool oxide, and linalool within the concentration range of 3.125‐50 µg·mL-1. However, the survival rates of the cells decreased to less than 50% after the disposition of geraniol, α-ionone, and β-ionone in the concentration range of 50‐100 µg·mL-1. As these three compounds showed significant cytotoxic effects, they were not selected for the next phagocytosis assay.

Effect of some relevant chemical components of O. fragrans flowers on cell viability of RAW 264.7 cells.
Effect of relevant chemical components of O. fragrans flowers on the phagocytic activity of RAW 264.7 cells
The phagocytosis of macrophages is the first step in an immune response, and increasing phagocytic activity is characteristic of activated macrophages. 42 Thus, the phagocytic activities of RAW 264.7 cells treated with various concentrations of relevant chemical components were investigated by a neutral red uptake assay. When the tissues and organs undergo pathological changes, macrophages can eliminate antigens through intracellular cytotoxicity. Phagocytosis is a key indicator of evaluating macrophage activities, and activated macrophages help initiate specific defense mechanisms by recruiting other immune cells and play a critical role in the immune system. 42 Salidroside and acteoside (3.125, 100 µg·mL-1) could increase the phagocytosis of RAW 264.7 cells compared with the control cells (Figure 2A). Linalool oxide could also increase the phagocytosis at 3.125‐6.25 µg·mL-1, but not at 12.5‐100 µg·mL-1. Linalool did not increase the rate of phagocytosis. Therefore, salidroside and acteoside had more abilities of enhancing the phagocytic activity of RAW 264.7 cells than linalool oxide and linalool.

Effect of relevant chemical components of O. fragrans flowers on the phagocytic activity of RAW 264.7 cells.
Phagocytosis of pathogens is an important function of macrophages, which participate not only in activating the adaptive immune system, but also in immune regulation. 42 The phagocytic rate was significantly suppressed (P < 0.05) when the cells were exposed to LPS after pretreatment with salidroside, acteoside, linalool oxide, and linalool at 3.125‐100 µg·mL-1 (Figure 2B). The phagocytic rates (P < 0.05) in LPS-stimulated RAW 264.7 cells through treating with salidroside, acteoside, linalool oxide, and linalool at 3.125‐100 µg·mL-1 were lower than those (155.1 ± 5.6%) of LPS-stimulated RAW 264.7 cells. However, linalool oxide and linalool had obvious advantages over salidroside and acteoside in reducing the phagocytic activity of LPS-stimulated RAW 264.7 cells.
Effects of the Drying Method for O. Fragrans Flowers on Cell Viability and Phagocytic Activity
Based on the earlier results, geraniol, α-ionone, and β-ionone induced poor cell proliferation and viability. Salidroside and acteoside possessed notable effects on enhancing the phagocytic activity of natural RAW 264.7 cells. Salidroside, acteoside, linalool oxide, and linalool all had remarkable advantages in reducing the phagocytic activity of LPS-stimulated RAW 264.7 cells. Therefore, salidroside and acteoside exhibited two-way immunoregulation of phagocytic cells. However, the contents of linalool oxide and linalool, only in the order of mg, were lower than salidroside and acteoside, in the order of g per 100 g of fresh O. fragrans flowers. Therefore, salidroside and acteoside played the main part in regulating the activities of phagocytic cells in the immune system. The total content of salidroside and acteoside in “Aurantiacus,” “Latifolius,” and “Thunbergii” was the highest with the MD method in HF. Therefore, the rapid drying method represented by microwave drying maintained the medicinal properties of O. fragrans flowers, and thus should be advocated. In contrast, the unique fragrance of O. fragrans flowers, which is important in the food industry, is retained by traditional drying methods such as shade drying and lime drying.
Discussion
Calculating dried flower yields is employed in determining water content and other chemical components in bitter gourd slices of different groups and subjected to various drying methods. 28 MD had the highest drying efficiency among the 5 studied drying methods. The local high temperature that is involved in MD had the more rapid and greater function of inactivating a large amount of enzymes and preserving glycosides than OD. 43,44 SHD, SUD, QLD, and OD at low temperature and MD at low power retained enzyme activity and caused the hydrolysis of salidroside. Thus, enzyme content and activity play an important role in the nonvolatile components of fresh O. fragrans flowers. In addition, most of the volatile components of O. fragrans flowers have a low boiling point. 45 Therefore, temperature in the drying process of O. fragrans flowers is the dominant factor affecting the content of volatile components, which has been confirmed in the present research. The contents of volatile components are also influenced by product characteristics such as physical structure (density and porosity), plant species, and their localization in the plant. 29,41 Drying methods also play an important role in the preservation of nonvolatile components. 46
Immunomodulation is a process of adapting the immune system to obtain the optimal response via either stimulation or suppression of the immune reaction. 47 Immunotherapy has already been applied to cancer therapy. 48 O. fragrans flowers, as a natural medicine and food, should be comprehensively investigated. RAW 264.7 macrophages are often used to evaluate the immunomodulatory activity of chemical substances. The cytotoxic effects of salidroside, acteoside, linalool oxide, linalool, geraniol, α-ionone, and β-ionone were determined using the MTT assay to ensure the safety of these ingredients. Phagocytosis of RAW 264.7 macrophages was determined using the neutral red phagocytosis experiment to assess their immunomodulatory activities.
Conclusions
O. fragrans flowers from three O. fragrans groups, namely, “Aurantiacus,” “Latifolius,” and “Thunbergii” were dried using 5 different methods. The non-volatile and volatile components such as salidroside, acteoside, linalool oxide, and linalool were determined. The different characteristics appeared in the effects of drying methods on the non-volatile and volatile components of various O. fragrans flowers. The effects of the former quantitative components on the cell viability and phagocytic activity of normal and LPS-stimulated RAW 264.7 cells were investigated. Four components,salidroside, acteoside, linalool oxide, and linalool, all showed good immunoregulatory effects. Considering their different contents, the best drying method was dependent on the total salidroside and acteoside content. Microwave drying and oven drying at high temperature were beneficial for the preservation of the bioactive nonvolatile components for enzymes inhibition and protection of glycosides. Other drying methods, such as shade drying, sun-drying, quick-lime drying, and oven-drying at low temperature were beneficial for the preservation of fragrant volatile components. This research provides evidence for the selection of the appropriate drying method for O. fragrans flowers as food and medicine.
Footnotes
Acknowledgments
The authors thank Dr Li Feng from Shandong University of Traditional Chinese Medicine for the support of identification of O. fragrans flowers, and Dr Liao Yonghong from Peking Union Medical College for proof-reading our manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Anhui University Natural Science Research Projects of China under Grant (KJ2019A0097).