Abstract
Introduction
Benzothiazole skeleton is an important heterocyclic nuclei of many natural and synthetic active compounds. 1 Benzothiazole has been identified as a volatile constituent from several kinds of microorganisms including fungi Aspergillus clavatus, 2 Polyporus frondosus, ligninolytic basidiomycetes Armillaria astoyae, 3 and bacteria Pseudomonas chlororaphis. 4 It is demonstrated that this compound is used as a fumigant to control fungi Sclerotinia sclerotiorum, 4 nematode Ditylenchus destructor, 5 and insect Tribolium castaneum, 6 Bradysia odoriphaga 7 in nature. In fact, benzothiazole analogues have been found to exhibit a wide range of pharmacological actions including anticancer, antibacterial, antifungal, antiviral, anthelmintic, anti-inflammatory, analgesic, anticonvulsant, antidiabetic, anti-oxidant and neuroprotective properties. 8 Most of the reported antibacterial and antifungal benzothiazole compounds are 2-substituented derivatives, in which 2-N and 2-S substituents are notable. They displayed favourable antibacterial activities against Staphylococcus aureus, Escherichia coli, and antifungal activities against Candida albicans, Candida parapsilosis, Candida tropicalis. Research sugests that the antibacterial target of the benzothiazines is dihydroorotase, 9 while the antifungal target is lanosterol 14a-demethylase (CYP51). 10 Some benzothiazole derivatives have been developed as commercial fungicides and bactericides (Benthiazole, Benthiavalicarb-isopropyl), or herbicides (Benzthiazuron, Mefenacet, Figure 1).

Natural or synthesized active benzothiazole and acylthiourea compounds.
Acylthiourea is a valuble structure unit of fungicides. Such as thiophenate and thiophanate-methyl have been using as agricultural fungicide since 1960s. In plants, thiophenate and thiophanate-methyl are first converted to ethyl carbendazim and carbendazim respectively, which could affect the division of fungal cells, damage the germ tubes and thereby kill the fungi.
11
Though being used for several decades, these fungicides are still widely used today due to their broad-spectrum and highly effective. Acylthiourea compounds also exist in nature source. For example compound ethyl 4-(o-nitrophenyl)-3-thio-allophanate (Figure 1) was once identified from the resistant pyricularia oryzae Cav. Rive variety, which could strongly inhibit the growth of microorganism Agrobacterium tumefaciens and suppress the gene expression of virulence region.
12
Further research found that benzothiazole thio-allophanate exhibited strong inhibition on the growth of agrobacterium.
13
Subsequently, some 5-substituted (-NO2, -NH2 or Br) benzothiazol-2-yl acylthiourea compounds are found to have antimicrobial activities, in which compounds
As an ongoing work to search for new agricultural fungicidal compounds, we focused our interest in benzothiazole and acylthiourea moiety. To understand how the electrostatic effect of substitutes on benzothiazole ring affect the fungicidal activity of benzothiazole acylthiourea, we would plan to design and synthesize a series of target compounds with -NO2 or -OCH3 at different site of benzothiazole ring and evaluate their antifungal activities. Using o-, m-, or p-nitrophenylamine as starting material, 4-, 5- and 6-NO2 benzothiazol-2-yl arylacylthiourea were successfully obtained accordingly by our synthetic scheme. But only 6-OCH3 benzothiazol-2-yl carboxamides instead of arylacylthiourea were produced from p-anisidine no matter with alkyl or aryl chlorides by the same synthetic method.
Since 6-OCH3 benzothiazol-2-yl carboxamides have similar core structure as commercial fungicide Carbendazim, the antifungal activities of these compounds should be screened. Aditionally, some 6-substituted benzothiazol-2-yl cyclohexanecarboxamides and cyclopropanecarboxamides are reported to exhibit antimicrobial activities,15,16 in which 6-MeO benzothiazole derivative
In summary, the NO2 substituted benzothiazole arylacylthiourea and 6-OCH3 benzothiazole carboxamides were synthesized and their in vitro antifungal activities against 2 kinds of phytopathogens fungi were screened. The in vivo antifungal activities, mycelium morphology and permeability of cell membrane of the candidate compounds on B. cinerea were also investigated.
Materials and Methods
Instruments and Chemicals
All of the chemicals were purchased from commercial suppliers and used without further purification. Reactions were monitored by TLC and visualized under UV light at 254 nm. Melting points (uncorrected) of compounds were measured using an X-4B micro melting point instrument (Yidian Physical Optical Instrument Co. Ltd Shanghai, China). The 1H and 13C NMR spectra were displayed on a BRUKER AVANCE III HD 500 MHz (Bruker, Switzerland) using deuterated chloroform (DCCl3) or deuterated dimethyl sulfoxide (DMSO-d6) as solvent. High-resolution mass spectra (ESI TOF (+)) were recorded on the Solari X70FT-ICR-MS (Bruker, Germany). The x-ray diffraction was measured by Bruker D8 QUEST-X (Bruker, Germany).
General Procedure for Synthesis of Intermediates 1 -4
Substituted 2-amino-benzothiazoles (
General Procedure for Synthesis of Title Compounds A1 -A12 and B1 -B12
Target compounds
N-(6-Methoxybenzo[d]Thiazol-2-yl)Butyramide (A1 )
Yellow solid; yield 72.3%; mp: 174-178 °C; 1H NMR (500 MHz, CDCl3) δ 7.57 (d, J = 8.9 Hz, 1H), 7.23 (d, J = 2.5 Hz, 1H), 7.00 (dd, J1 = 8.9 Hz, J2 = 2.5 Hz, 1H), 3.81 (s, 3H), 2.46 (t, J = 7.5 Hz, 2H), 1.75-1.67 (m, 2H), 0.91 (t, J = 7.4 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ 171.85, 158.17, 156.97, 141.11, 132.84, 120.65, 115.49, 104.50, 55.88, 38.27, 18.54, 13.62. HRMS (ESI): calcd for C12H15N2O2S ([M + H]+), 251.0848, found, 251.0850.
N-((6-Nitrobenzo[d]Thiazol-2-yl)Carbamothioyl)-3-(Trifluoromethyl)Benzamide (B1 )
Yellow solid; yield 73.2%; mp: 196-198 °C; 1H NMR (500 MHz, CDCl3) δ 11.31 (s, 1H), 8.82 (s, 1H), 8.33-8.23 (m, 3H), 7.92 (d, J = 7.7 Hz, 1H), 7.72 (t, J = 7.8 Hz, 1H), 7.61 (d, J = 8.9 Hz, 1H); 13C NMR (125 MHz, CDCl3) δ 164.32, 163.31, 152.24, 144.32, 132.41, 132.06, 131.97(q, J = 32.7 Hz), 131.14, 130.28 (q, J = 3.6 Hz), 130.07, 128.39 (q, J = 230.1 Hz), 124.91 (q, J = 4.1 Hz), 124.37, 122.19, 120.74, 118.37. HRMS (ESI): calcd for C16H10F3N4O3S2 ([M + H]+), 427.0140, found, 427.0141.
The data of the other compounds and their NMR and HRMS spectral details can be found in the Supplemental Material.
Bioassay
Mycelium growth rate method was used to evaluate the in vitro antifungal activities of title compounds against 2 kinds of vital plant pathogens V. mali and B. cinerea.
19
Each treatment was conducted in triplicate. And commercial fungicide Hymexazol was used as a reference drug. The in vivo curative and protective effects of compounds
Statistical Processing
Values are expressed as mean ± standard error of the mean. Analyses were performed using Excel Statistics. Toxicity regression equations with a correlation coefficient and effective concentration that inhibited mycelium growth by 50% (EC50) were expressed as the mean of values obtained using toxicity regression equation software. 23
Results
In reference to Arpana's method,
18
we used less toxic toluene instead of benzene in the second and 3ird step of the Scheme to prepare the products (follow Scheme 1, series

The X-ray diffraction structure of target compound
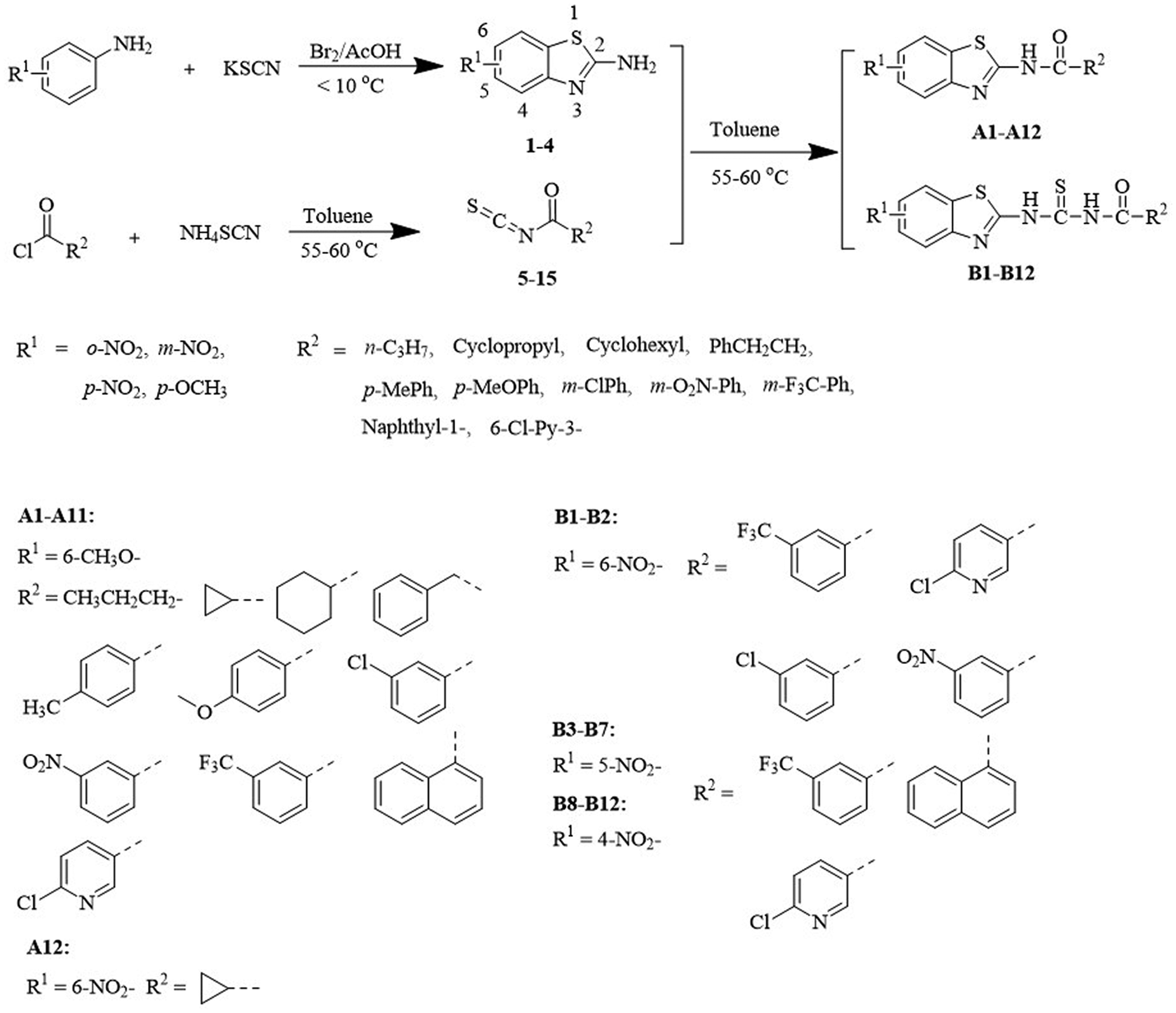
Synthesis of target compounds
The inhibitory rates of target compounds against the mycelium growth of V. mali and B. cinerea were listed in Table 1. In series
In Vitro Antifungal Activities of the Target Compounds (100 μg/mL; Inhibition Rate, %).

To understand the potential of the high active compounds screened above, the toxic regression equation and EC50 of
EC50 of Target Compounds Against V. mali and B. cinerea.

95% confidence limit.
The most active and easily obtained compounds

Protective effect of compound
Curative and Protective Effects of Compounds

When the mycelium of B. cinerea cultivated with compound

The effect of compound
To understand the effect of compound

Effect of
Discussion
For benzothiazol-2-yl carboxamides (series
It is reported, for the 6-substituted 2-aminobenzothiazole derivatives, the electron-withdrawing groups (Cl, F and CF3) at the 6-position of the benzothiazole moiety were better than small electron-donating groups (CH3O, C2H5O) against all Candida spp. tested.
10
Our above conclusion is agree with this result. However, more 6-NO2 benzothiazol-2-yl carboxamides should be designed and evaluated for their antifungal activities to illustrate the SAR of series
In NO2 substituted benzothiazol-2-yl acylthioureas (series
It is reported that benzothiazole fungicide Benthiavalicarb-isopropyl could inhibit mycelia growth, zoosporangia and cystospore germination, and also inhibit the sporulation of Phytophthora infestans at a very low concentration. It is presumed that benthiavalicarb-isopropyl inhibits the fibrillization of cellulose, which is involved in the biosynthesis of the cell wall. 24 As mentioned above, acylthiourea fungicide Thiophenate mainly affect the division of fungal cells, damaging the germ tubes and thereby killing the fungi. 11 However, there is no report about the mechanism of the hybird compound benzothiazole acylthiourea. Our preliminary research found that our target compound did affect the mycelial cell wall and cell membrane of B. cinerea. But whether our compound inhibits the fibrillization of cellulose, there need more research to verify. And we still do not know whether the selected compound affect the division of fungal cells.
Conclusions
In conclusion, target compounds
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241311738 - Supplemental material for Synthesis, Characterization and Antifungal Activities of Amide and Acylthiourea of Substituted 2-Amino-1,3-Benzothiazole
Supplemental material, sj-docx-1-npx-10.1177_1934578X241311738 for Synthesis, Characterization and Antifungal Activities of Amide and Acylthiourea of Substituted 2-Amino-1,3-Benzothiazole by Junping Wang, Chuanping Wang, Hongmei Wang, Yan Wei, Jin Lin and Shuanghong Hao in Natural Product Communications
Footnotes
Acknowledgments
The authors are indebted to the National Natural Science Foundation of China (No. 31471808), the Natural Science
Foundation of Shandong Province (No. ZR2021MC022), and Investigation and Control of Forestry Pests (No. 1106309) for financial support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Shandong Province, National Natural Science Foundation of China, Investigation and Control of Forestry Pests, (grant number ZR2021MC022, 31471808, 1106309).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
