Abstract
Due to the excessive use of antifungal agents, drug resistance and ecological problems are increasing. Some antifungal agents are difficult to degrade and have high toxicity and several side effects. In this study, 15 novel tetrahydrogeranyl quaternary ammonium salts (
Introduction
Plant pathogenic fungi are the main factors causing various plant diseases that result in reduced production in agriculture and forestry.
1
Antifungal agents are prepared from compounds with certain activities that can kill or inhibit the reproduction of bacteria, fungi, and other microorganisms within a certain period of time.2–4 However, the excessive use of antifungal agents will lead to drug resistance and ecological problems. Some highly toxic antifungal agents are difficult to degrade and have serious side effects.
5
It is very important to develop new plant antifungal agents with low toxicity and high efficiency. Citral is the main component of
It is generally recognized that a new generation of antifungal agents with environmental friendliness and high efficiency can hopefully be synthesized from some natural products. Rastija et al
9
studied a series of coumarin derivatives that were effective against
Results
Synthesis
The synthetic routes of tertiary amines and quaternary ammonium salts are shown in Figures 1 and 2. The tertiary amines (compounds
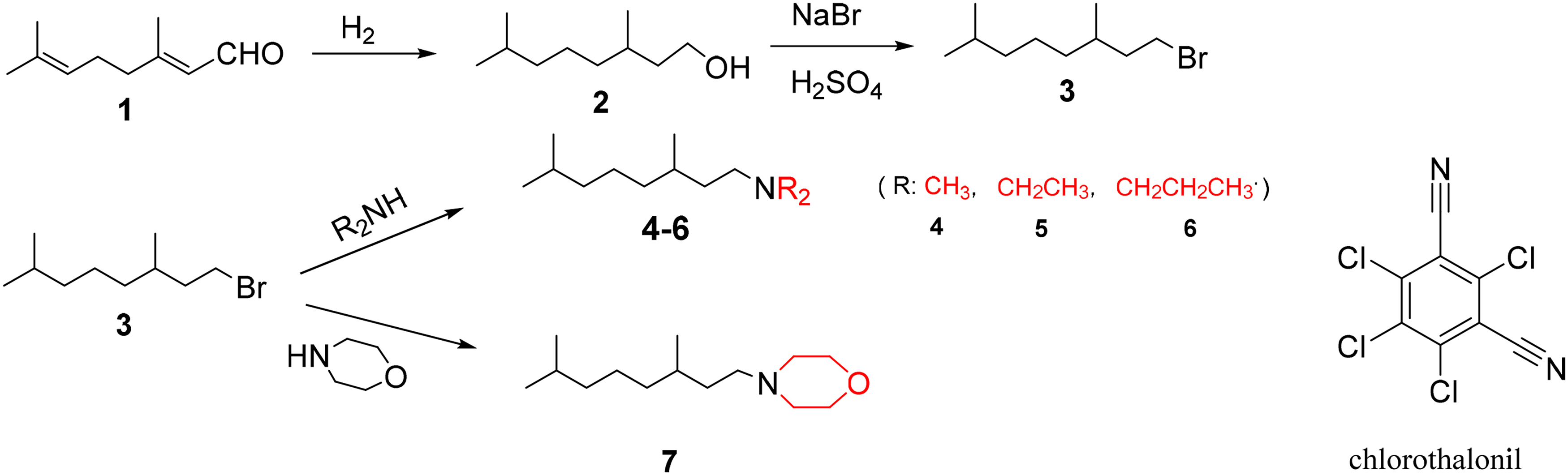
Synthetic route of tetrahydrogeranyl tertiary amines and the structure of chlorothalonil.

Synthetic route of tetrahydrogeranyl quaternary ammonium salts.
The molecular structures of the tetrahydrogeranyl quaternary ammonium salts were characterized by Fourier transform infrared (FTIR), proton nuclear magnetic resonance (1H NMR), and carbon-13 nuclear magnetic resonance (13C NMR) spectroscopy and mass spectrometry (MS), and the results showed that the target derivatives 8
Taking compound
Biological Activity
The inhibition rates of 15 tetrahydrogeranyl quaternary ammonium salts for the growth of 10 phytopathogenic fungi are shown in Table 1.
Inhibition Rates of Compounds for the Mycelium Growth of Plant Pathogenic Fungi.

At a concentration of 0.25 mg/mL, the inhibition rates of compound
From the structural differences of these compounds, it can be seen that compounds
Discussion
Citral is a natural plant resource with a unique active structure, and it was found that the biological activities of citral derivatives were higher than those of citral. For example, Zhou et al
20
studied the minimum inhibitory concentration and minimum fungicidal concentration of citral against
In this study, 15 tetrahydrogeranyl quaternary ammonium salt derivatives were synthesized from citral. The antifungal activity of the synthesized derivatives against 10 plant pathogens that cause significant damage to crops was tested in this study.
In addition, some relationships between the structure of compounds and the antifungal rates were found. For
Materials and Methods
General
Methyl bromide, ethyl bromide,
Plant pathogenic fungi used in the study were preserved and provided by the Administration on Forest Ecosystem Protection and Restoration of Poyang Lake Watershed:
FTIR spectra of the compounds were recorded on a Nicolet IS10 FTIR spectrometer, and 1H NMR and 13C NMR spectra on a Bruker AVANCE 400 NMR spectrometer (Bruker) using deuterium oxide (D2O) as a solvent and trimethylsilane as the internal standard. Electrospray ionization MS was conducted on a Bruker Amazon SL mass spectrometer, and electron impact (EI)-MS on a Bruker Amazon SL mass spectrometer (Bruker). The purity of the compounds was detected by liquid chromatography (Agilent Technologies Singapore International Pte., Ltd). Melting points were determined in a WRS-2 melting point apparatus (Shanghai Precision & Scientific Instrument Co., Ltd) and were uncorrected, and a GHP-250 Intelligent incubator (Shanghai Sanfa Scientific Instrument Co., Ltd) was selected to culture strains during the experiment.
Quaternary Ammonium Salt Synthesis
Tertiary amine
The characterization data of the compounds are shown in Table 2.
Structural Analysis of 15 Quaternary Ammonium Salts.

Abbreviations: m.p., melting point; IR, infrared. EI-MS, electron impact-mass spectrometry.
Biological Activity Evaluation
The inhibitory activities of 15 new compounds against 10 plant pathogenic fungi were determined by the mycelium rate method. The compound was quantitatively dissolved in sterile water and added to potato dextrose agar (PDA) medium to a final quaternary ammonium salts concentration of 0.25 mg/mL. PDA medium with no compound was used as a negative control, and chlorothalonil was used as a positive control. All prepared PDA medium was poured into culture dishes (9 cm in diameter). After cooling and solidification, the PDA medium was inoculated with fungal strains (0.5 cm). Each strain was cultured in 3 repeated dishes. After inoculation, mycelium was cultured in a constant temperature incubator (27 °C) for several days. When the mycelium of the negative control group grew to ∼7 cm, the diameter of the fungus cake was measured by the crossing method. The antifungal rate was calculated by equations (1) and (2):

Fifteen tetrahydrogeranyl quaternary ammonium salt derivatives were synthesized from citral in this study, and their structures were characterized by FTIR, MS, 1H NMR, and 13C NMR. The antifungal activities of tetrahydrogeranyl quaternary ammonium salt derivatives against 10 plant pathogenic fungi were determined by the mycelium growth rate method, and the test results showed that all compounds had certain inhibitory activities against the tested strains. The structure–activity relationship analysis showed that the
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221078452 - Supplemental material for Synthesis and Antifungal Activity of Novel Tetrahydrogeranyl Quaternary Ammonium Salts
Supplemental material, sj-docx-1-npx-10.1177_1934578X221078452 for Synthesis and Antifungal Activity of Novel Tetrahydrogeranyl Quaternary Ammonium Salts by Yun Peng, Jiayu Chang, Zhuangquan Xiao, Jiazong Huang, Ting Xu, Shangxing Chen, Guorong Fan, Shengliao Liao, Zongde Wang and Hai Luo in Natural Product Communications
Footnotes
Acknowledgments
The authors thank Ting Xu and Jiazong Huang for their help.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Jiangxi Province Academic and Technical Leaders Training Program (grant numbers 20204BCJ22022, 20204BCJL23045, YC2020-S235, 31960295, and jxsq2019201016).
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
