Abstract
Objectives
Amyloid fibrils are misfolded proteins that self-assemble to form insoluble, elongated, β-sheet structures. These fibrils are linked with the progression of neurodegenerative diseases like Alzheimer's Disease and metabolic disorders such as Type II diabetes. A range of molecules have shown the ability to impede the formation of these fibrils, often by acting on the monomeric and oligomeric forms of the protein. Our work focuses on the inhibitory effect of curcumin, gallic acid and mangiferin on the formation of amyloid fibrils.
Materials and methods
Bovine insulin was used as a model system for amyloid formation by lowering the pH at 60 °C, and several different small molecules were used to test inhibition. Samples were analyzed using Atomic Force Microscopy (AFM) in a liquid environment, as well as Congo Red and Thioflavin T assays.
Results
We have been able to observe significant inhibition of amyloid fibril formed from insulin by these compounds as well as observe beginning stages and the morphology of fibrils. Mangiferin, gallic acid, and curcumin were all shown to slow down fibril formation, though by 5 days fibrils have started to form in the gallic acid solution. After 7 days, solutions containing curcumin or mangiferin still show no strong evidence of structured fibrils.
Conclusion
Gallic acid, curcumin and mangiferin are all molecules that have inhibitory effects on amyloid formation using insulin as a model system, with gallic acid being somewhat less effective than the other two. The AFM technique used to image fibrils was performed in an environment that more closely resembles in vivo conditions than air dried samples.
Introduction
Proper folding is an integral part in helping to determine the properties of proteins, as the shape ultimately determines the function. This folding process can also be detrimental to the individual if it occurs incorrectly. Many different diseases are associated with the misfolding of a protein and the formation of well-ordered aggregates known as amyloid fibrils, including Alzheimer's, Parkinson's and Type 2 diabetes.1–8 These aggregates have a high level of ß-sheet structure regardless of the secondary structure of the natively folded protein. The fibrils are protease-resistant with a compact core and strong intermolecular forces that lead to a high level of stability and strength.9–11 The fibrils have been postulated to be formed through the hierarchical assembly model (HAM), which suggests that amyloid formation begins with proteins undergoing a conformation change and forming a partially folded structure. These misfolded monomers assemble to form oligomers. The oligomers are soluble and rich in ß-sheet structure and exposed hydrophobic residues, which then grow and become rod-like protofilaments which elongate with the addition of additional monomeric intermediates. These then intertwine in order to form protofibrils and then assemble to insoluble mature fibrils, which eventually leads to the formation of plaques.12–18 It was previously thought that these fibril accumulation-related diseases could be treated by reversing the formation of these large amyloid structures, yet the energetically favorable state of the fibrils creates a challenging problem. There is also debate in the literature about which is the more toxic form, the fully mature fibrils, the protofibrils, or the smaller oligomers.19–22 Subsequently, one focus of treatment has prioritized towards the prevention of the incorrect folding process through the mediation of certain compounds, including polyphenolic acids.
Amyloidogenic proteins can fall into one or more of four categories: (1) Naturally unstructured proteins that have a disposition towards conformational change, (2) Proteins that are partially unfolded due to environmental conditions, (3) Proteins with one or more mutations that lead to an unfavorable structure, and (4) formation of a fragment due to proteolysis of a larger precursor. Regardless of their categorization, amyloidogenic proteins share some common structural features. These features include formation of the same structural motif, large quantity of exposed hydrophobic residues, and the lack of a localized or globalized high net charge. 23 The high level of ß-sheet structure can have the ß strands oriented either in a perpendicular manner against the fibril axis or orientated parallel to the fibril axis. With this highly organized and tightly packed ß-sheet structure, the amyloid fibrils become resistant to enzymatic digestion, which contributes to the insoluble nature of amyloid plaque deposits,24,25 though studies are focusing on ways the body can break down amyloid fibrils through proteolytic methods.26–28
Natural polyphenols are powerful compounds that have traditionally been used in the treatment of various diseases and are found in a variety of easily accessible sources such as plants, fruits, vegetables, and teas. 29 The structural patterns of polyphenols contribute to this therapeutic ability. Polyphenols contain aromatic rings with hydroxyl groups, which are thought to play an important role in blocking the self-assembly process of maturing amyloid fibrils through hydrophobic interactions. 30 This action may also contribute to their ability to act as antioxidant, anti-inflammatory, and neuro-protective agents. Polyphenols are of high interest in Alzheimer's Disease research because they have shown to play an inhibitory role in the formation and maturation of fibrils. Several studies suggest that polyphenols are able to inhibit amyloid fibril formation, disaggregation, or destabilization of preformed amyloid fibrils.31–37 A variety of polyphenols and flavonoids have been identified as possible candidates to prevent and delay the progression of these types of diseases. One such compound is gallic acid.38–43
Gallic acid is a naturally occurring polyphenol that is found in a variety of green plant tissues including green tea leaves and grape seed oil. It has been found that gallic acid has the ability to lessen the extent of Amyloid beta peptide (Aß) fibril formation and aggregation from insulin in a dose-dependent fashion. 44 Curcumin, a major constituent in turmeric, is another example of a molecule that exhibits neuroprotective characteristics against amyloid fibril formations and can act to disaggregate pre-formed fibrils and reduces disruption of membrane models.45–53 Curcumin is a diarylheptanoid which contains two aromatic rings separated by a chain of seven carbon atoms. Due to its lipophilic quality, it has the ability to cross the blood-brain barrier. 23 Curcumin also can bind to Aß plaques as observed in mice that were either orally fed or injected with the compound. 54 Exposure to a higher concentration of curcumin has exhibited a higher level of inhibition of Aß fibril aggregates compared to a lower concentration and blocked the formation of Aß oligomers. 55
The ability of such natural compounds to block the progression of amyloid fibril formation at different stages of maturation provides insight into the precise steps of amyloid fibril formation. Amyloid fibril formation involves a progressive maturation process containing hierarchical stages beginning with oligomers with a high level of ß-sheet structures, followed by protofilaments and, ultimately, mature amyloid fibrils.56–62 While a basic understanding of amyloid maturation exists, specific details on the nature of each maturing stage are not completely understood. The levels of toxicity of the various stages also remains unknown, with studies showing that the oligomeric form may play a larger role in disease states than originally thought.19–21
The exact mechanism of amyloid fibril toxicity is still under investigation, but it has been heavily focused on fibril / lipid membrane interactions.63–67 Small molecules have also been shown to inhibit amyloid formation in the presence of lipids. 68 Polyphenols have also been used to look at the crowded conditions69,70 and amyloid formation in general.71–74 One possible drawback to the use of polyphenols as inhibitors of amyloid formation in patients is the increased inflammation response found in some insulin preparations with certain phenols, highlighting the need for the use of non-toxic inhibitors.75,76 In addition to further testing the inhibition ability of gallic acid and curcumin, we will also focus on mangiferin, a polyphenol found in mango fruit. Mangiferin has exhibited an abundant number of therapeutic abilities including, antioxidant, anti-inflammatory, antimicrobial, and anti-cancer with potential to reduce oxidative stress. It also has the ability to cross the blood-brain barrier, adding to its potential as a therapeutic aid.77–86 Our study will explore the capabilities mangiferin by observing its potential ability to interact with insulin in various forms and disrupt the progression of amyloid formation.
Our study uses atomic force microscopy (AFM) to further investigate the specific inhibitory role different polyphenols play in the maturation process of amyloid protofibrils and fibrils. We also tested if the acidic nature of gallic acid was responsible for the ability of the compound to lower the rate of amyloid formation. Our AFM observations were completed in a liquid environment using a modified form of the Peak Force Tapping mode as opposed to those done with air-dried samples.87–93 This allowed the fibril growth to be observed and measured in an environment that closer mimic in vivo conditions. Both Congo red absorbance and Thioflavin T fluorescence were also used to verify the presence of amyloid fibrils in the bulk samples.94–102
Materials and Methods
Materials
All materials (Bovine insulin from pancreas, gallic acid, citric acid, ascorbic acid, curcumin, mangiferin, and dyes) were obtained from Sigma-Aldrich (St. Louis, MO USA) unless otherwise noted.
Insulin Fibril Formation
Insulin solutions of 2 mg/mL were made in an Eppendorf tube with 0.1 mL of 0.1 M HCl and 0.9 mL DI H2O for samples of insulin at a pH of 2. Appropriate volumes of DI H2O and stock solutions of insulin and of molecules of interest were mixed to produce 1:1, 1:2, 1:5, and 1:10 M ratios of insulin/small molecule complexes. The final concentration of insulin was always prepared as 2 mg/mL. Samples were vortexed for 30 s, then incubated in a dry bath at 60 °C without agitation or stirring for set experimental times, and were spun down for 2 min in a centrifuge before use.
Solution Preparation
Curcumin stock solutions were prepared at a concentration of 20 µM in aqueous solution using a heated water treatment, 103 mangiferin stock solutions to 0.1 mM and gallic acid stock solutions to 40 mM, which were then used to create working solutions. Citric and ascorbic acid solutions were made to 200 mM (pH = 2.6 and ionic strength of 0.0025, and pH = 3.0 and ionic strength of 0.001 respectively).
Congo Red UV-Vis Measurements
A Congo red solution was made by combining 0.011 g of Congo red, 0.015 g K2HPO4, 1.46 g NaCl with a pH of 7.4, and a 10% ethanol/water solution up to a volume of 1L. Congo red stock solutions were made before each run.100 µL of Congo red solution was added to a cuvette with 1000 µL of 50 mM KCl/NaCl solution, 100 µL of sample (or 0.1 M HCl), and 100 µL of 0.1 M NaOH to neutralize the acidic solution. Samples were then incubated for 30 min at room temperature, and then analyzed under UV-Vis with a Vernier SpectroVis Plus Spectrophotometer then analyzed with Logger Pro Insulin samples were heated for specified times and then frozen until ready for use.
To see the effects of varying amounts of curcumin or mangiferin, the same Congo red solutions were made to a volume of 1600 µL. Then a total of 400 µL protein: curcumin solution was added with varying ratios, incubated for 30 min, and analyzed the same as above.
Thioflavin T Fluorescence Measurements
8 mg of Thioflavin T was added to 10 mL of a 10 mM phosphate and 150 mM NaCl solution at a pH of 7, which was then filtered through a 0.2 µM filter. Fresh stock solutions were made before each experimental run. 1 mL of this stock was added to 50 mL of the phosphate buffer to give a working solution (with the concentration of thioflavin T measured at 412 nm using an extinction coefficient of 36 mM−1 cm−1). 2 mL of this solution was added to the cuvette, and 20 µL of either unheated 2 mg/ml insulin or the heated samples were pipetted in and mixed for 1 min. Samples were then run on an Agilent Cary Eclipse Fluorescence spectrometer with an excitation wavelength of 440 nm. Samples were also monitored at 482 nm for 1 min to confirm the fluorescence change.
AFM Measurements
All AFM images of insulin alone and ratios of insulin/ small molecule complexes after incubation at 60 °C were captured using a Bruker Dimension Icon Atomic Force Microscope. Samples were placed on freshly cleaved mica (which was cleaved before each experiment using scotch tape) which was then mounted on a metal microscope disk using cyanoacrylate glue unless otherwise noted. The device used a Dimension Fluid Cantilever DTFML-DD High Efficiency Direct Drive Fluid Cell Holder which held the tips. ScanAsyst Fluid + Silicon-Nitride tips were used with a frequency of 150 kHz, a tip radius of 2 nm, and a spring constant that varied between 0.8 N/m and 2.3 N/m which was calibrated with each tip using a thermal tune function.
For measurements in the liquid phase: Focus was first performed in air, then fluid was added to the fluid holder and refocused. A 2.0 µL aliquot of experimental solution was added to 40.0 µL of a 100 mM NaCl solution on a freshly cleaved mica slide, and then the Scan Head was lowered and brought into contact with the liquid sample. The images were taken using the ScanAsyst Fluid experiments (which is a modified version of the Peak Force Tapping mode). Images were taken under ambient conditions at a scan frequency of 0.977 Hz at 512 samples/line, a peak force amplitude of 100 nm, a Peak Force feedback force that varied between 0.6 nN and 1.1 nN, and a peak force frequency of 2 kHz. Samples were then analyzed by the NanoScope Analysis package. While there was some variation within each sample, the fibrils were consistent in their properties within each run. All experiments were run on three separate occasions each with a unique sample, with a minimum of four separate scans per sample.
Results
Fibril Formation Can Be Slowed or Halted by Polyphenolic Acids
Bovine insulin was dissolved into the buffer to a concentration of 2 mg/mL, and samples were heated to 60 °C prior to AFM imaging. Figure 1a shows formation of amyloid fibrils by 24 h, showing diameters of 10–20 nm. By the time 2 days had elapsed (Figure 1c), amyloid fibrils are present to a greater degree. After 5 days (Figure 1e) fibrils are still stable. There was a diversity of structures found in these images, with rope chain-like structures found in multiple fibrils. Figure 1b, 1d, and 1f are zoomed in images of the fibrils showing the detailed structures found in the fibrils. These structures are similar to those found in the literature using different proteins in both a liquid environment and fibrils dried in air and seems to indicate that the fibrils are building up from oligomers.104–107

Amyloid fibrils from insulin at 2 mg/mL concentration.
To verify the presence of amyloid fibrils in solution, both a spectroscopic Congo red and Thioflavin T assay were run. Figure 2a shows a shift in the absorbance max and Figure 2b shows a large increase in the fluorescence when insulin fibrils were added to the solutions, which is associated with binding of amyloid fibrils to the dyes.96,108,109

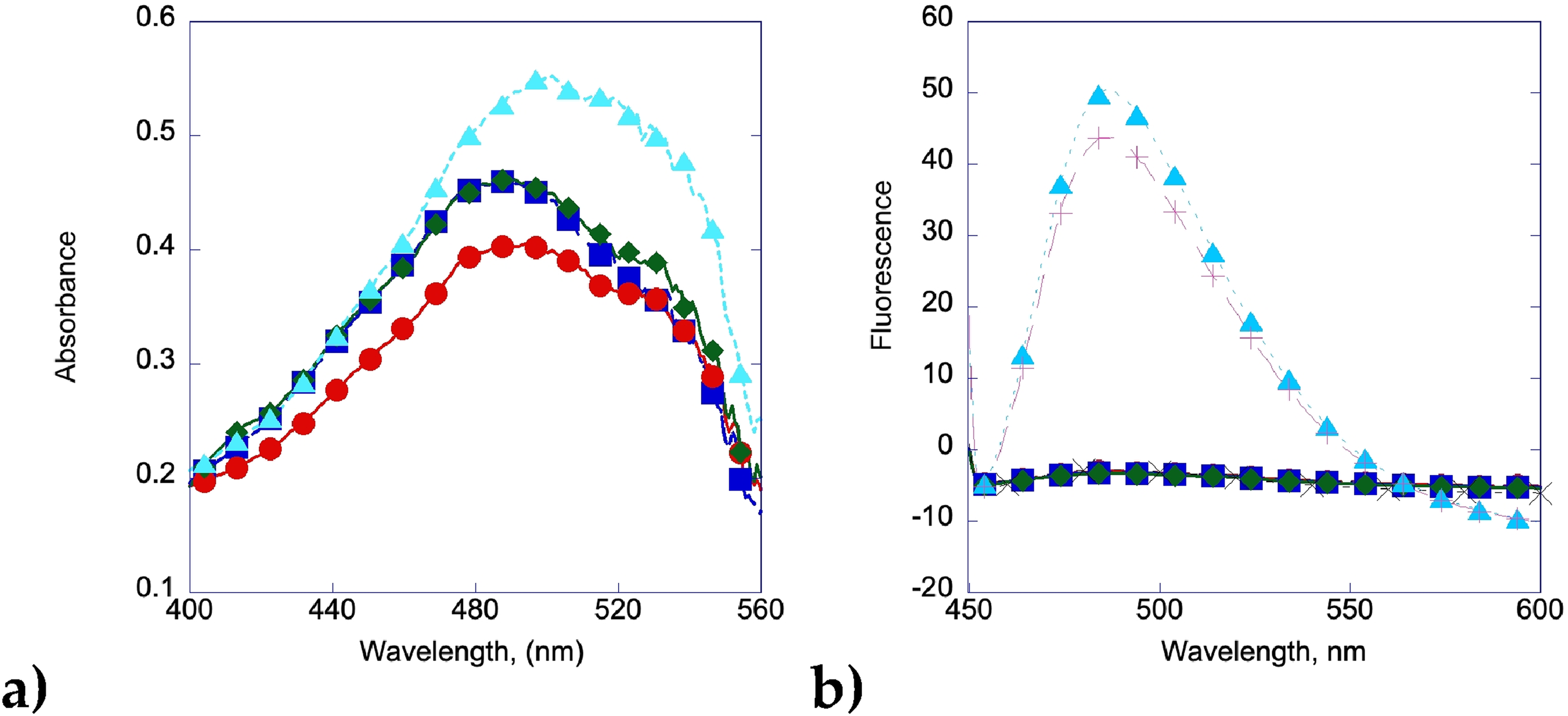
When gallic acid is added in a 2:1 concentration to the insulin, the ability to form amyloid fibrils is significantly lowered. After 1 day (Figure 3a), there are no amyloid fibrils present on the mica surface. Figure 3b shows the beginning of aggregates growing into larger structures starting around three days after incubation began, with Figure 3c showing the beginning of fibrils after 5 days of constant heating. The characteristic shift of the maximum wavelength of Congo red in solution is found by the end of 7 days, but not before, although there is some shifting at the 4 day mark at longer wavelengths (Figure 4a). This is also seen in the Thioflavin T assay, where there is no significant change in fluorescence in the first few days, but by day 6 the fluorescence starts to increase significantly, indicating the eventual formation of amyloid fibrils (Figure 4b).

Amyloid fibrils from insulin incubated with gallic acid at 1:2 M ratio.
Curcumin shows strong inhibition of insulin fibril samples when made under the previous conditions. Figure 5a shows no activity or protein on the mica surface. Figure 5b shows no apparent pattern with scattered aggregates after 2 days of heating at 60 °C. Figure 5c shows scattered aggregates after 7 days but no amyloid fibrils found via AFM.

Amyloid fibrils from insulin incubated with curcumin at 1:2 M ratio. (
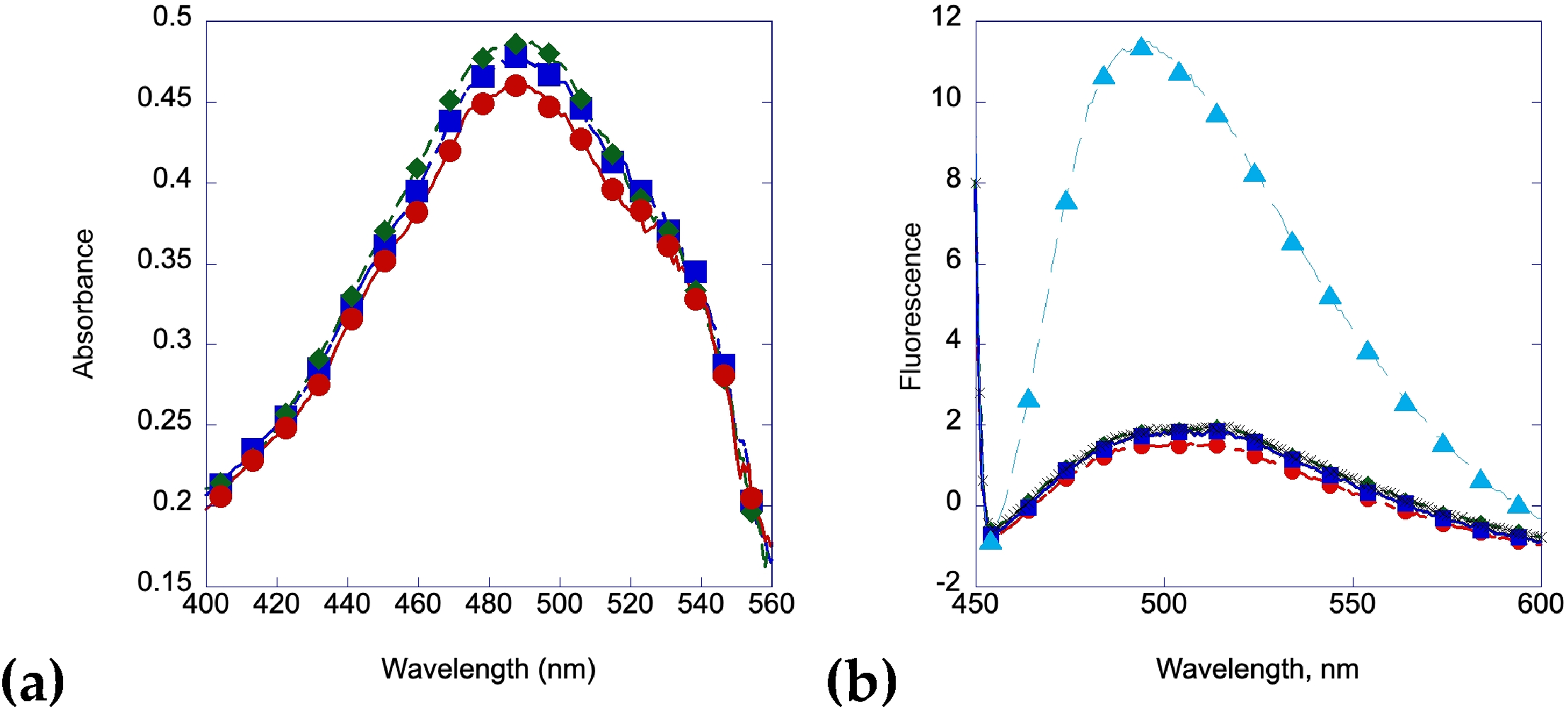
Congo red was used to verify that there were no amyloid fibrils found in the solution. The UV spectrum shifts to the right due to curcumin's absorption and potential interaction with Congo red, but the spectrum remains consistent even after 7 days of heating, indicating that no amyloid fibrils are present in the solution after 1 week (Figure 6a). The Thioflavin T assay shows a similar trend, with no increase in fluorescence after 7 days. Curcumin has been shown to interfere with the fluorescence of Thioflavin T but not Congo Red in certain circumstances, 110 but we were able to use a low enough concentration of curcumin that no effect was observed between a Thioflavin T solution with 20 µL of insulin and with 20 µl of an insulin/curcumin solution (Figure 6b).

When mangiferin is made at a similar concentration and incubated with insulin at 2 mg/mL at 60 °C, a similar inhibition of amyloid fibril formation is observed. Figure 7a shows no activity or protein on the mica surface after 1 day. Figure 7b shows aggregation but no amyloid formation after two days of heating. Figure 7c shows a similar trend after 7 days of heating at 60 °C. These results are corroborated by the Congo red and Thioflavin T binding assays shown in Figure 8a and 8b.

Amyloid fibrils from insulin incubated with mangiferin at 1:2 M ratio. (
Thioflavin T data was also collected at 482 nm. Both mangiferin and curcumin show a strong inhibition of insulin amyloid formation with very little change in fluorescence even after seven days, whereas insulin by itself shows a large increase within 24 h (Figure 9). Gallic acid shows good inhibition, but shows signs of amyloid formation after 5 days, with a large increase in fluorescence by day 6, consistent with the formation of amyloid fibrils also shown in the Congo red and AFM data.

(
Protofibril Growth is Time Dependent and Follows an Ordered Process
Amyloid fibril formation was observed at 21 h (Figure 10 a,b) and 24 h (c,d) of insulin samples. After 21 h, samples were imaged and globule formations were found in various formations, from a line of aggregates (Figure 10a) to a fishnet-like pattern. No fibril structures were observed during this time for these samples.

Early stages of amyloid formation.
At 24 h, samples were imaged and globule formations were found strung together like beads on a string. Figure 9d shows two globules connected by a single thread. The globules measure to a maximum average height of 45 nm with a width of about 100 nm.
At 96 h, fully formed fibrils were found (Figure 11 a,c,e). The fibrils show a left-handed twist with a periodicity of 50–60 nm (Figure 11b) and are approximately 15–35 nm wide.

Amyloid fibril formation from insulin at 4 day incubation, a-c along with height measurements. (
Acidification of the Insulin Samples Using Different Acids
Samples were also incubated with both citric acid (pH = 2.6) and ascorbic acid (pH = 3.0) separately. After incubating the 2 mg/mL insulin solution with 200 mM citric acid (Figure 12a) or ascorbic acid (Figure 12b) at 60 °C prior to imaging, it was found that both had little effect on the inhibition of fibril formation. The fibrils were found to have both a similar shape and structure to the insulin fibrils without any citric acid, though often showed a fibril that appeared to have three sub-units lying side-to-side, each about 15–20 nm wide (Figure 12c).

(
Inhibitory Effects of Curcumin and Mangiferin at Various Concentrations
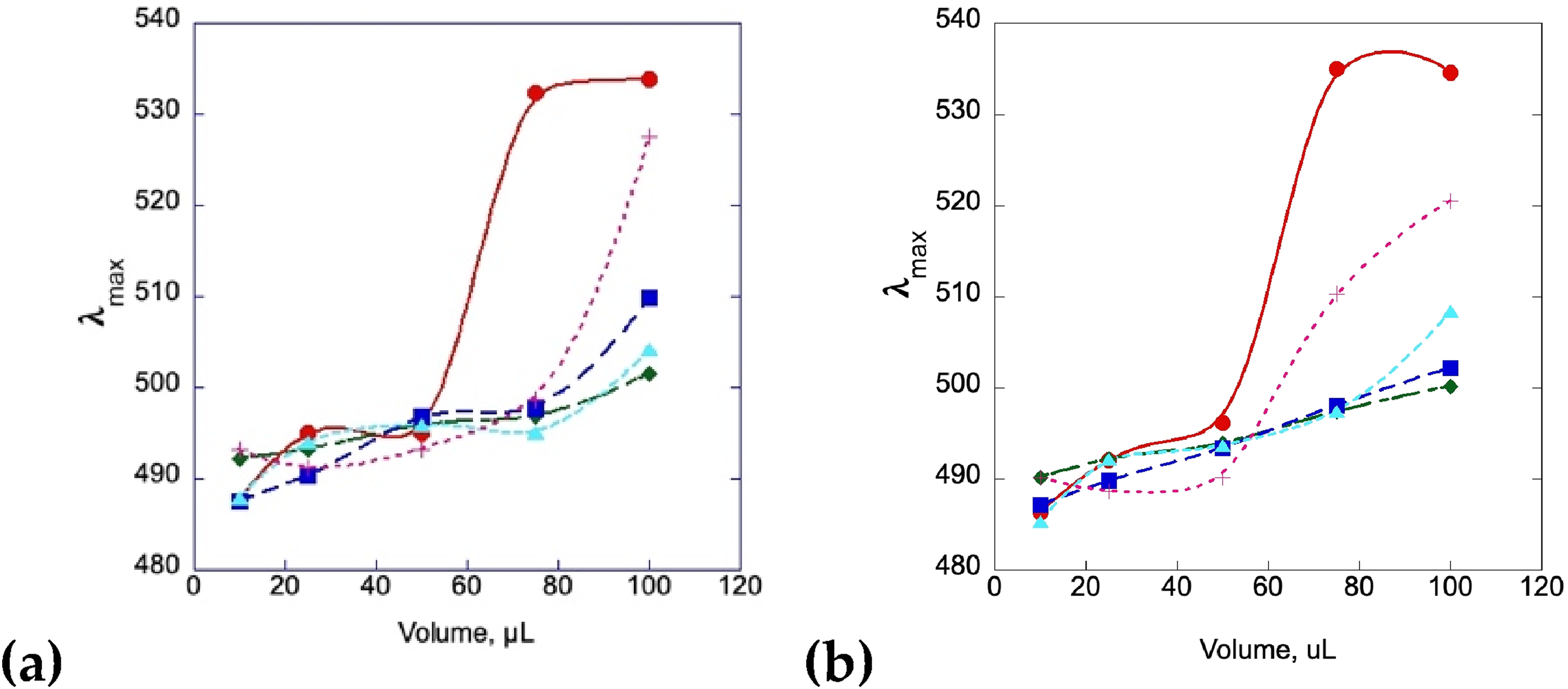
Insulin, insulin/curcumin, and insulin/mangiferin samples were also added to Congo red solutions and monitored by UV-Vis to look for the inhibitory effects on fibril formation at different concentrations of curcumin. Figure 13a shows a shift in the absorbance max of a solution of a 2 mg/mL insulin solution compared to an insulin at the same concentration incubated with curcumin at various concentrations, while 13b shows the same experimental setup with mangiferin. The data in both cases shows a shift in the maximum absorbance with the insulin by itself, which is associated with the binding of amyloid fibrils to the Congo red dye.47–49 The addition of the curcumin and the mangiferin solutions at high concentrations halts the absorbance shift from occurring, indicating that fibrils are not present in these solutions, which is also seen in the AFM data. At very low concentrations of curcumin and mangiferin (0.43 µM), fibrils may be present after 2 days, but this requires a much larger amount of stock to be added to the Congo red in order to be observed.
Discussion
Protein folding is a natural biochemical process that plays an integral role in the maintenance of a variety of biological processes. Nonetheless, the process of protein folding can be filled with a variety of malfunctions, which can sometimes be self-corrected by the body and, other times, lead to amyloidosis. By imaging the fibrils in a liquid environment instead of the more traditional dry scan, we were able to observe the fibrils in a fully hydrated form and visualize them in the environment that they normally grow in. Using this methodology, our investigation further included studying natural small molecules and their ability to disrupt and delay amyloid fibril maturation. Our main conclusions are (1) amyloid fibrils have a distinct ß-sheet and helical conformation, (2) the maturation pattern follows the well-discussed HAM in addition to having bead-like and net-like structural organizations in the very early stages of maturation, and (3) the presence of different small molecules disrupts the maturation to various degrees, depending on the type of molecules present, both polyphenols and diarylheptanoids. The presence of amyloid fibrils was also verified using traditional Congo red and Thioflavin T spectroscopic techniques, although no kinetic measurements were made, limiting the measurements of monomeric and oligomeric interactions. The insulin solutions were heated and then frozen at the appropriate times so that all spectroscopic assays would be run at the same time under the same conditions. Although a freeze-thaw cycle has been shown to promote amyloid formation in antifreeze proteins,111,112 the presence of amyloid fibrils in some samples but not all implies that the freezing of the samples did not promote or deter fibril formation during storage or thawing. It is also important to note that polyphenols are known to auto-oxidize which can lead to a quenching of the signal and erroneous interpretations of data.113,114 By employing fresh stocks and multiple techniques, including those that do not depend on spectroscopy, this potential problem can be minimized. The oxidation process may have benefits in certain cases, as the amyloid inhibition properties of curcumin may be increased by the presence of autooxidation products. 115
Multiple amyloid fibril deposits form between the 24 h and 5-day maturation period as shown in Figure 1. The helical conformation had become highly visible after 24 h of maturation and continues as the fibrils form (Figure 1 and Figure 11). The mature amyloid fibril consists of two chains that are arranged in a helical shape. A closer imaging of the helical formation of the two peptide chains is presented in Figure 10 where one chain overlaps with the second. Figure 11a presents 2 fibrils with prominent twists in their structures. This is observed through the height variation within a single fibril. A repeated pattern of short segments of raised heights followed by segments with shortened heights can be observed. The height variation caused by the helical structure is more clearly visualized in the zoomed images of these two fibrils. The helical twist causes the fibril to have two very distinct heights: a raised area as visualized with the bright yellow/tan color and a lowered area (presented with the darkened color) which correspond to a height difference of 6 nm and 35 nm wide.
Our investigation suggests that the first signs of structured interaction between the fibrils occur within the first 24 h of incubation. At 21 h the first signs of organization are seen (Figures 10c and d). A distinct differentiation can be seen between the positioning of the amyloid fragment and blank mica in Figure 10a, which hints at the interaction and organization method of amyloidogenic proteins. The first structured interaction between fibrils forms a net-like structure as presented in Figures 10b and c. Closer imaging of these early fibrils revealed distinct beads or rings connected by a single strand, where two bulged beads are connected by a fainter line with helical interactions between these early structures (Figure 10d). This observation may help describe the HAM of amyloid maturation and the building up of the fibril from ring or bead-shaped oligomers. 116
A focus of study among many investigations is the ability to disrupt and inhibit the above-described maturation processes of amyloid fibrils with the aid of natural polyphenols. The polyphenols used in this study were gallic acid and mangiferin which both are acidic compounds and have a ring as a part of their structure, and curcumin which is a diarylheptanoid. To test to see if the inhibition of fibril grown was due to the presence of an acidic organic molecule or to a molecule that contained a ring structure, we incubated insulin in solutions containing either ascorbic acid or citric acid. Citric acid was added purely to test the effect of an acidic molecule on fibril formation, and it was found that there was no change in fibril formation compared to that of the insulin control. The ascorbic acid was used to test the effect of low pH environment as well as a ring structure and it was found that this also did not have any inhibition of fibril formation. A variety of studies have analyzed the effects of using compounds like gallic acid and curcumin and has shown the ability to slow down the progression of fibril formation.33,43–45,117–120 Our study similarly revealed that gallic acid, curcumin, and mangiferin all have the ability to inhibit the formation of fibril formation, although to varying amounts. It can be seen in Figure 3b that amyloid formation after 2 days incubation with gallic acid has less fibril formation than our insulin control after a 2-day incubation, and at the 5-day incubation mark the fibrils have a width of 10–30 nm. This shows that gallic acid has the ability to delay the formation of these amyloid fibrils for a period of time which is shorter than for mangiferin or for curcumin.
Curcumin has also been previously studied for its ability to act as an amyloid inhibitor. One study found that curcumin has the potential of inhibiting the formation of amyloid beta oligomers and fibrils. 54 Our study captured AFM images supporting similar findings. Figure 5 shows that the insulin solution exposed to curcumin displayed a delayed maturation period, holding the process at the oligomer stage, which is depicted by the scattered circular congregation of aggregates throughout the mica. The protein appears to stay in this form through the 7-day period, only slightly increasing in number of aggregates since the initial 24 h scan. This is also demonstration from the spectroscopic assays, as there is little shift in the maximum wavelength of a Congo red/Curcumin solution between 1 h and 7 days, and almost no increase or shift in the fluorescence of Thioflavin T. Furthermore, curcumin clearly expresses its inhibitory nature when compared to insulin alone that shows progression of insulin to an amyloid structure just after 24 h. In addition to its inhibitory potential, curcumin also has shown to have the ability to cross the blood-brain barrier. Mangiferin also shows a similar ability to inhibit fibril formation, giving another avenue to elucidate the mode of action of inhibition of small molecules on amyloid formation. This finding further strengthens the two molecule's therapeutic potential for use with amyloidogenic diseases, as well as the power of the imaging technique. For all three of these molecules, the exact method of interaction with the fibrils and the origin of the anti-amyloidogenic ability remains open for debate. These molecules could stabilize a partially unfolded monomer, or make association of the monomers into oligomers less energetically unfavorable. It's also possible that the presence of these molecules interacts with larger oligomers or protofibrils and shifts the aggregation pathway to a process less favorable to full length amyloid fibrils. Further investigation of this ability could lead to a more complete understanding of their method of action and lead to better therapeutic strategies.
Conclusion
In this study we have investigated the ability of three different molecules (mangiferin, curcumin, and gallic acid) to slow down and disrupt amyloid formation using insulin as our model system. A combination of AFM in the liquid phase and spectroscopy was used to monitor these changes, keeping the fibrils in an aqueous environment during the experiments. Gallic acid, while successful at slowing down amyloid formation, allowed fibrils to form before the end of the seventh day, whereas the other two molecules inhibited amyloid formation for the entire run of the experiments.
Footnotes
Acknowledgements
The authors are grateful to the Siena College School of Science for its support in this research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Siena College Center for Undergraduate Research and Creative Activity to Daniel F. Moriarty.
