Abstract
Introduction
Phytopathogens have attracted the attention of global agricultural chemists because of their harmful effects on agricultural production. For example, phytopathogens can parasitize many important crops, thus inducing a variety of crop diseases and causing huge economic losses to global agricultural production every year.1‐3 Compared with other control methods, chemical control has significant advantages, such as high efficiency, low cost, and convenient application. However, due to a large amount of abuse of existing agricultural antimicrobial agents caused by the increasing drug resistance of pathogens, drug control effects, environmental pollution, and other issues, the search for efficient, low-toxicity, and novel chemical structures of fungicides has become a research hotspot in the field of pesticide chemistry.4‐6
Nitrogen-containing heterocyclic compounds have become the research focus for new pesticides because of their diverse structural forms and broad-spectrum biological activities.7,8 Most new heterocyclic compounds have low toxicity to mammals, birds, and fish, but high selectivity to pests and bacteria, which opens up a broad space for pesticide development. Among them, 1,3,4-thiadiazole derivatives are important five-membered heterocyclic compounds, which ‘C-N-S’ structure can chelate with metalions and have a wide range of pharmaceutical and pesticide activities. Commercialized 1,3,4-thiadiazolesinclude the antihypertensive drug tiodazosin, the agricultural fungicide thienone, bismerthiazol, and a novel antibacterial agent of 1,3,4-thiadiazole derivatives containing an amide moiety were designed.9‐11 The active substructure splicing method is an effective method for discovering highly active pesticide molecules, which is helpful in achieving a synergistic effect between different pharmacophores. 12 5-phenyl-1,3,4-thiadiazol-2-amine was used as the starting material. Based on the principle of active substructure splicing and condensation with benzoic acid to form the corresponding amide, phenyl 1,2,3-triazole was introduced into the amide bond through click chemistry, and 20 new thiadiazole-linked compounds were designed and synthesized (Figure 1). Their in vitro inhibitory activities against plant pathogenic fungi were tested using a mycelium growth inhibition assay, and the relationship between the antifungal activity of the target compounds and their molecular structures was preliminarily discussed.

Design thiadiazole derivatives to achieve potential antibacterial activity.
Materials and Methods
General Information
All reagents used for the chemical synthesis were purchased from Xiya Reagent Company. The solvents were distilled before use. The progress of all reactions was monitored by thin layer chromatography (TLC), and the TLC plates were visualized by exposure to ultraviolet light (254 and 365 nm). The products were purified using column chromatography. All NMR spectra (1H-NMR and 13C-NMR) were recorded on a Bruker AV-300 instrument, using CDCl3 as the solvent and TMS as the internal standard. Chemical shifts are expressed in ppm and coupling constants in Hertz. All chemical shifts are given in parts per million relative to that of tetramethysilane. The mass spectra were measured using an HP1100LC instrument (Agilent Technologies, USA).
General Synthetic Procedures
General Procedure for the Synthesis of Compounds A1-A20
A stirring mixture of substrates benzoic acid (8.0 mmol), thiosemicarbazide (729.1 mg, 8.0 mmol) and POCl3 (6 mL) was heated at 80 °C for 2.5 h. After cooling to room temperature, water (16 mL) was added. The reaction mixture was refluxed for 4 h. After cooling, the mixture was basified to pH 8 by dropwise addition of a 40% NaOH solution under stirring. The precipitate was filtered and recrystallized from ethanol to obtain the intermediate compound A as a white solid with a Yield of 81.1%-83%.
General Procedure for the Synthesis of Compounds B1-B20
To a solution of benzoic acid (122.1 mg, 1 mmol) in dichloromethane (DCM, 15 mL) the compounds A1-A20 (2.0 mmol), 1-Hydroxybenzotrizole (HOBt, 70.3 mg, 0.52 mmol), 1-Ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride (EDCI, 210.9 mg, 1.1 mmol), 4-Dimethylaminopyridine (DMAP, 134.4 mg, 1.1 mmol) and triethylamine (111.3 mg, 1.1 mmol) were added. The mixture was then stirred under nitrogen at room temperature for 16-24 h. When TLC showed completion of the reaction, the reaction mixture was poured into water and extracted with DCM. The organic phase was washed with 2% HCl and water and dried over anhydrous Na2SO4. Evaporation of the solvent yielded a residue that was purified by chromatography to yield the amide product. The yields of the purified compounds ranged between 41.0% and 74.5%.
General Procedure for the Synthesis of Compounds C1-C20
To a suspension of K2CO3 (207.3 mg, 1.5 mmol) in acetone (15 mL) was added a solution of appropriate compounds B1-B20 (1 mmol). After the reaction was heated to 60 °C for 30 min, a solution of propargyl bromide (142.8 mg, 1.2 mmol) in acetone (10 mL) was added in a drop wise manner and the mixture was refluxed for 3 h (monitored by TLC). The reaction mixture was poured into water, extracted with DCM, dried over anhydrousNa2SO4 and concentrated under vacuum, and purified by column chromatography to obtain the desired product. Yield: 74%-86%.
General Procedure for the Synthesis of Compounds D1-D20
In a round bottomed flask, compounds C1-C20 (1 mmol) were added to a mixture of CuSO4·5H2O (30 mg, 0.1 mmol), azidobenzene solution (119.1 mg, 1 mmol) and sodium ascorbate (13.3, 0.1 mmol) dissolved in1:1 of t-BuOH and H2O (16 mL) at room temperature. The reaction mixture was stirred for 12 h at room temperature and then poured into water (20 mL). The aqueous layer was extracted thrice with DCM (10 mL). The combined organic layers were concentrated in vacuo. The residue was purified by column chromatography on silica gel to afford desired products D1-D20 in 74%-83.1% yield.
Bioassays
Antifungal activities of the title compounds in vitro was evaluated against five types of fungi (R. solani, B. cinerea, F. graminearum, S. lycopersici, C. lunata) by the typical mycelium growth inhibition method from previously reported method.13‐15 A commercial fungicide (difenoconazole) was used as the positive control. Each compound was dissolved in dimethyl sulfoxide (DMSO) and mixed with sterile molten PDA to obtain a concentration of 50 mg/L for the initial preliminary screening with three replicates. The compounds with good activity were evaluated further by the determination of EC50 values with the concentration gradients of 50, 12.5, 3.13, 0.78 mg/L, which were calculated using log-probit analysis via SPSS v.18.0 software.
The relative inhibition rate of the synthetic compounds was calculated via the following equation:
Statistical Analyses
SPSS 18.0 software (IBM, Chicago Illinois, USA) is used for statistical analysis.
Results
Compound Design
Thiadiazole derivatives exhibit a wide variety of antibacterial activities. 16 Therefore, many antibacterial compound designs have introduced thiadiazole.17‐19 Consulting the literature, it was found that substituted 5-phenyl-1,3,4-thiadiazol-2-amine synthesizes derivatives with good antibacterial activity through condensation with acid to increase the lipophilic region of the phore. Amjad. Qandil et al synthesized 1,3,4-thiadiazole derivatives and tested their antimicrobial activities using the broth macrodilution method. The most potent member was compound 4c (Figure 1), showing selectivity to Gram-positive S. aureus at the minimum inhibitory concentration (MICs) (2 μg/mL). 20 Mali et al designed and synthesized a series of fatty acid-thiadiazole derivatives using a molecular hybridization approach. In vitro antimycobacterial potential was established by screening synthesized compounds against Mycobacterium tuberculosis H37Rv strain. Compound 5a (Figure 1) exhibited a minimum inhibitory concentration of 2.34 μg/mL against M.tb H37Rv. 21 Benzotriazole can significantly enhance the activity of the compound owing to its single structure 22 ; therefore, this article is inspired by the above design to introduce benzotriazole into the thiadiazole amide structure to synthesize D1-D20.
Chemistry
The synthetic outlines of the starting and target compounds are shown in Figure 2. Compound

Synthetic route of target compounds. Reagents and conditions: (Ⅰ) POCl3, reflux, 2.5 h; (Ⅱ) benzoic acid, EDC, DMAP, HOBt, DCM, r.t., 5 h; (Ⅲ) propargyl bromide, acetone, K2CO3; (Ⅳ)Ph-N3, CuSO4·5H2O, sodium ascorbat, H2O:t-BuOH = 1:1(V:V), rt.
Biological Assay
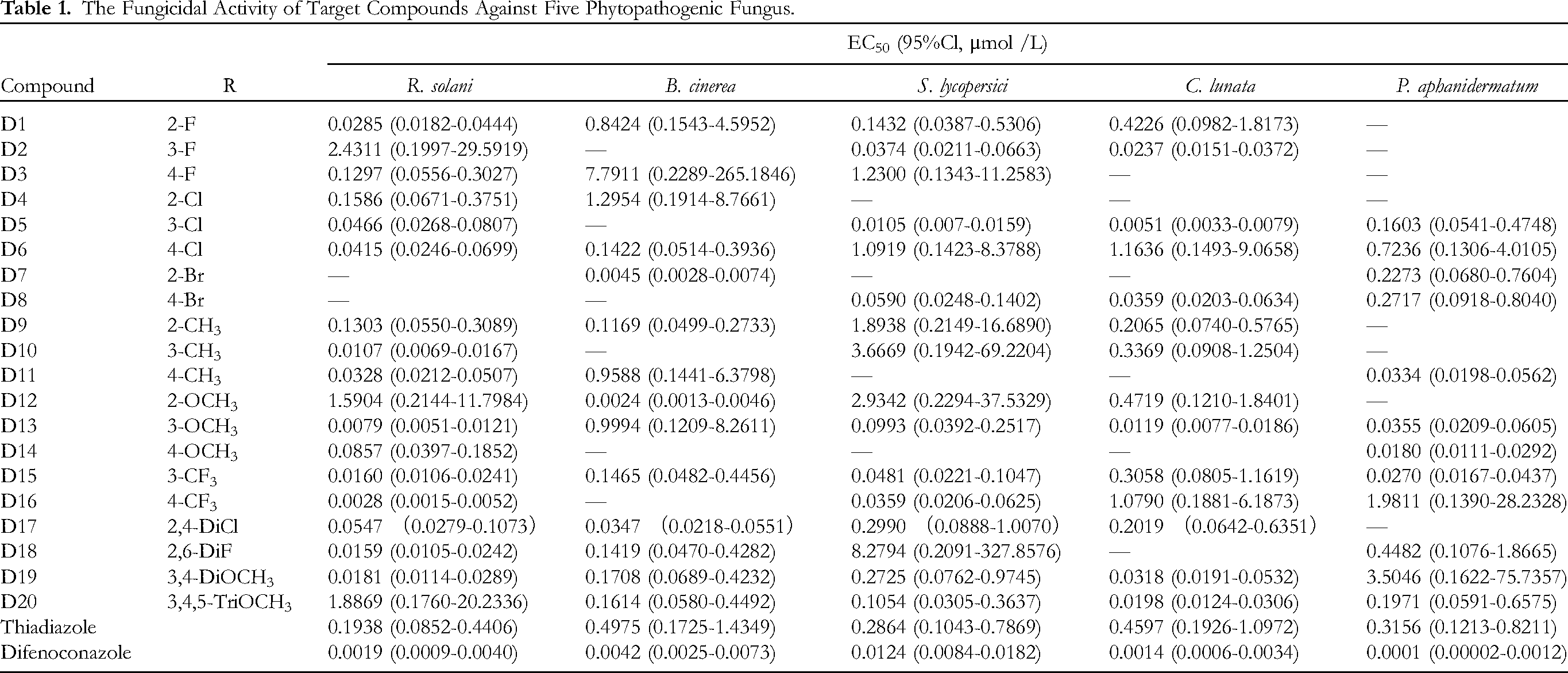
The fungicidal activities of all target compounds against five phytopathogenic fungi (Rhizoctonia solani, Botrytis cinerea, Stemphylium lycopersici, Curvularia lunata, Pythium aphanidermatum) were tested using an in vitro mycelium growth inhibition assay, and the commercial fungicide difenoconazole was used as the positive control. The synthetic compound thiadiazole was also used as a positive control to compare the difference between the pre-and post-structure optimizations. The inhibition of mycelial growth was evaluated by measuring colony diameters in the presence and absence of the tested compounds.
As shown in Table 1, most of the title compounds exhibited significant fungicidal activity; however, there were significant differences among the fungicidal activities of the target compounds. In particular, compounds D16, D13 and D10 showed pronounced fungicidal activities against R. solani with EC50 values of 0.0028, 0.0079 and 0.0107 μmol·L−1 respectively, D12 and D7 against B. cinerea with EC50 values of 0.0024 and 0.0045 μmol·L−1 respectively, D5 against S. lycopersici with EC50 value of 0.0105 μmol L−1, D5 and D13 against C. lunata with EC50 values of 0.0051 and 0.0119 μmol·L−1respectively, D14 against P. aphanidermatum with EC50 value of 0.0180 μmol·L−1. Based on the experimental results, EC50 values of the target compounds were evaluated to confirm their fungicidal effects on the five phytopathogenic fungi.
The Fungicidal Activity of Target Compounds Against Five Phytopathogenic Fungus.
Discussion
In summary, we designed and synthesized a series of derivatives and evaluated their antimicrobial activity against five pathogenic bacteria, R. solani, B. cinerea, S. lycopersici, C. lunata and P. aphanidermatum. As shown in results, the order of activity against R. solani was 3-CF3 > 4-OCH3 > 4-CH3 > 2,6-DiF > 4-CF3 > 2-F. Except for the meta-substitution activity of CF3 and F, which was better than the para-substitution activity, the best activity of other element substitutions was the para-position, and the meta-substitution was better than the para-position. Only the presence of OCH3 and Br substitutions had a good antibacterial effect against B. cinerea. Only the para-Cl substituted compound showed the same activity as the positive control drug against S. lycopersici.
Therefore, in the present study, a preliminary investigation of the potential antifungal activity of thiadiazole and its derivatives was carried out and the relationship between the antimicrobial effect and the moiety was explored, which is of positive significance for the future development of new fungicides that are more effective and environmentally friendly.
Due to various factors, this research is still at a relatively early stage and has many limitations. For example, we focused on the antibacterial aspect of plant pathogens, but although the compounds had antibacterial activity, they did not achieve the good results we expected, so we did not carry out subsequent research on the cellular level of plants and animals for the time being, and if the activity of the series of compounds is further improved in subsequent research, we will carry out cell-related experiments in the subsequent research.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241304621 - Supplemental material for Design, Synthesis and Fungicidal Activity of N-(5-Phenyl-1,3,4-Thiadiazol-2-yl)-N-((1-Phenyl-1H-1,2,3-Triazol-4-yl) Methyl) Benzamide Derivatives
Supplemental material, sj-docx-1-npx-10.1177_1934578X241304621 for Design, Synthesis and Fungicidal Activity of N-(5-Phenyl-1,3,4-Thiadiazol-2-yl)-N-((1-Phenyl-1H-1,2,3-Triazol-4-yl) Methyl) Benzamide Derivatives by Ziyi Kong, Xinyu Zhang, Ming Bian and Danni Fu in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The authors declare that they haveno competing interests.
Ethical Approval
Ethical Approval is not applicable for this article.
Funding
This study was supported by the Inner Mongolia Natural Science Foundation Project (2019BS08016), and Inner Mongolia University for Nationalities Doctoral Research Initiation Fund (No.BS579).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

