Abstract
Objectives
This study aimed to investigate the essential oil extracted from the aerial parts of Teucrium viscidum Blume, a perennial herb belonging to the Lamiaceae family, collected in Vietnam. The focus was on determining the essential oil's chemical composition and evaluating its antimicrobial and mosquito larvicidal activities.
Methods
The essential oil of T. viscidum was analyzed using GC–FID/MS to identify its chemical constituents. The antimicrobial activity was assessed against seven microorganisms using the broth microdilution method to determine minimum inhibitory concentration (MIC) and half-maximal inhibitory concentration (IC50) values. The larvicidal activity was evaluated against Culex quinquefasciatus larvae, with lethal concentration (LC50 and LC90) values calculated after 24 and 48 h of exposure.
Results
Chemical analysis revealed that the essential oil of T. viscidum was predominantly composed of sesquiterpene hydrocarbons, with (E)-caryophyllene (47.91%) and germacrene D (19.17%) as the major constituents. The essential oil exhibited significant antimicrobial activity against three Gram-positive bacteria (Enterococcus faecalis, Staphylococcus aureus, and Bacillus cereus), one Gram-negative bacterium (Pseudomonas aeruginosa), and one yeast (Candida albicans), with MIC values ranging from 32 to 64 μg/mL and IC50 values ranging from 9.56 to 24.07 μg/mL. The mosquito larvicidal activity of the essential oil showed strong efficacy against C. quinquefasciatus larvae, with LC50 values of 23.07 μg/mL after 24 h and 19.67 μg/mL after 48 h, and LC90 values of 32.90 μg/mL at 24 h and 29.99 μg/mL at 48 h, indicating increased effectiveness with prolonged exposure.
Conclusion
This study provides a comprehensive analysis of the chemical composition and biological activities of T. viscidum essential oil. The findings highlight its potent antimicrobial and mosquito larvicidal properties, suggesting it as a promising natural alternative to synthetic antimicrobial agents and insecticides. Further research is warranted to explore its mechanisms of action and potential applications in pharmaceuticals and pest control.
Keywords
Introduction
Essential oils, derived from various plant parts such as leaves, flowers, bark, and roots, are complex mixtures of volatile organic compounds with long-recognized bioactivities. 1 These natural oils are widely used across multiple industries, including pharmaceuticals, cosmetics, and agriculture, due to their distinctive aromatic properties and biological activity. 1 Essential oils have garnered particular attention for their antimicrobial, antioxidant, anti-inflammatory, and larvicidal properties, positioning them as natural alternatives to synthetic chemicals.1,2
One of the most well-documented properties of essential oils is their antimicrobial activity, demonstrated against a broad range of microorganisms, including bacteria, fungi, and viruses. 3 This antimicrobial action is primarily attributed to their chemical composition, which includes terpenes, phenolics, aldehydes, and ketones—compounds known to act synergistically to enhance the oil's effectiveness. Given the rising prevalence of antibiotic-resistant bacteria, there has been an intensified search for alternative or adjunct therapies, with essential oils emerging as promising candidates. 4 Their multi-targeted mode of action within microbial cells reduces the likelihood of resistance development, making them valuable in combating multidrug-resistant pathogens. Additionally, the natural origin of essential oils aligns with the growing consumer preference for green and sustainable products, further increasing their appeal across various industries. 5
Beyond their antimicrobial potential, essential oils also exhibit mosquito larvicidal activity, which has drawn attention for eco-friendly vector control strategies. 6 Mosquito-borne diseases, such as malaria, dengue fever, and Zika virus, are significant global public health concerns. Among the mosquito species of concern, Culex quinquefasciatus stands out as a particularly harmful vector. 7 Commonly known as the southern house mosquito, C. quinquefasciatus is responsible for transmitting diseases such as lymphatic filariasis, West Nile virus, and Japanese encephalitis, posing serious public health risks, particularly in tropical and subtropical regions.7,8 These diseases significantly impact the health and well-being of affected populations, leading to long-term disabilities, reduced productivity, and increased healthcare costs. In agricultural communities, outbreaks of mosquito-borne diseases can also disrupt farming activities, causing economic losses due to illness in the workforce and reduced crop productivity. 8 Furthermore, the uncontrolled proliferation of mosquito larvae in stagnant water sources, such as irrigation systems and ponds, exacerbates the problem by creating ideal breeding grounds for these pests. 8
Effective control of C. quinquefasciatus larvae is essential not only for reducing the incidence of these debilitating diseases but also for mitigating their broader environmental and economic impacts. However, the extensive use of chemical insecticides has led to the development of resistance in mosquito populations, alongside concerns about environmental contamination and harm to non-target species. 9 This highlights the need for alternative, eco-friendly larval control approaches. Essential oils, with their ability to target the early developmental stages of mosquitoes, present a promising solution. 10 Their biodegradability and reduced environmental impact, compared to synthetic insecticides, are notable advantages, and their low toxicity to non-target organisms, including humans, makes them suitable for use in residential areas and sensitive ecosystems. 10
The genus Teucrium, belonging to the Lamiaceae family, comprises approximately 434 species distributed mainly in temperate regions of Europe, North Africa, and Asia. 11 Known for their aromatic properties, these plants have been used in traditional medicine for centuries. Species within the Teucrium genus are typically perennial herbs or shrubs, characterized by branches that are square in cross-section, leaves arranged in opposite pairs, and flowers arranged in thyrses, with mostly white to cream-colored, lobed petals. 11 The diverse morphology and adaptability of Teucrium species allow them to thrive in various habitats, from arid deserts to coastal regions. Pharmacologically, Teucrium species are recognized for a range of therapeutic properties, including anti-inflammatory, antipyretic, diuretic, and hepatoprotective activities. 11 These medicinal attributes are largely attributed to the complex chemical composition of the plants, particularly their flavonoids, iridoids, diterpenes, and essential oils.12,13 The bioactive compounds isolated from Teucrium species have been the subject of numerous studies, supporting their potential use in treating various ailments.12,13
The essential oils of Teucrium species, in particular, have attracted attention due to their potent biological activities. 14 These oils contain a diverse array of terpenoids, which contribute to their antimicrobial, antioxidant, anti-inflammatory, and insecticidal properties. 14 Research into the essential oils of Teucrium species has revealed significant variability in their chemical composition, which depends on factors such as geographic location, climate, and extraction method.15–19 This chemical diversity is believed to be a key factor in the broad spectrum of bioactivities observed in these oils.
Among the species of the Teucrium genus, Teucrium viscidum Blume stands out due to its distinctive characteristics and notable bioactivities. T. viscidum is a perennial herb predominantly found in temperate regions of Asia, especially in China, Korea, and Japan. 20 It has been used in traditional medicine to treat ailments such as hemoptysis, hematemesis, pulmonary abscesses, traumatic injuries, and bites from rabies-infected dogs or venomous snakes.21,22 Recent scientific studies have validated some of these traditional uses, particularly highlighting the plant's anti-inflammatory, cytotoxic, and antihyperglycemic properties.21,23,24 Bioactive compounds such as diterpenoids, triterpenoids, steroids, and lignans in T. viscidum are thought to underlie its therapeutic potential.20,22–26 Despite these findings, the chemical composition and biological activities of T. viscidum essential oil have not yet been studied. In this study, we analyzed the chemical composition of the essential oil from T. viscidum and evaluated its antimicrobial and larvicidal activities. The specific objectives were: (1) to analyze the chemical composition of the essential oil from the aerial parts of T. viscidum collected in Vietnam; (2) to evaluate the antimicrobial activity of the essential oil against seven different strains of microorganisms using the broth microdilution method; and (3) to test the mosquito larvicidal activity of the essential oil against C. quinquefasciatus larvae. By exploring the biological properties of T. viscidum essential oil, this study contributes to the growing body of research on natural products with potential applications in medicine and vector control.
Materials and Methods
Plant Material
The aerial parts of T. viscidum were collected in August 2023 during a field expedition in Pu Luong Nature Reserve, located in Thanh Hoa province, Vietnam. The plant specimen was authenticated and identified by Assoc. Prof. Dr Le Thi Huong, an expert from Vinh University, Vietnam. Following identification, a voucher specimen was deposited in the university's herbarium under the code LTH33 for future reference. The collected aerial parts were promptly transported to the laboratory, where the extraction process was carried out without delay. This careful handling was crucial in preserving the bioactive compounds present in the plant material.
Essential Oil Extraction
A total of 6 kg of aerial parts of T. viscidum was collected, with 2 kg used for each extraction process. The extraction was repeated three times. Essential oil extraction was performed using the hydrodistillation method in a Clevenger-type apparatus with distilled water, following the procedures outlined in the Vietnamese Pharmacopoeia 27 and established studies.28,29 The process lasted 4 h under normal atmospheric pressure. After extraction, the essential oil was dried using anhydrous sodium sulfate to remove residual moisture. The oil was then transferred into sterilized glass vials and stored at 4 °C, with a maximum storage time of one month before chemical composition analysis to preserve its integrity.
Essential Oil Analysis
The essential oil sample of T. viscidum was analyzed using both gas chromatography–flame ionization detection (GC–FID) and gas chromatography–mass spectrometry (GC–MS) techniques, following previously described studies.29,30 For the GC–FID analysis, an Agilent Technologies HP 7890A Plus gas chromatograph equipped with a flame ionization detector (FID) and an HP-5 ms capillary column (30 m length × 0.25 mm internal diameter, 0.25 μm film thickness) was utilized. Hydrogen was employed as the carrier gas at a constant flow rate of 1 mL/min. The injector temperature was set to 250 °C, while the detector temperature was maintained at 260 °C. The column temperature program began at 60 °C (held for 2 min), followed by an increase at a rate of 4 °C/min until reaching 220 °C, where it was held for 10 min. A split ratio of 10:1 was applied during the injection of 1.0 μL of the sample. The inlet pressure was maintained at 6.1 kPa to ensure stable chromatographic performance. The percentage composition of the essential oil components was determined by normalizing the peak areas, without the use of corrective factors.
For the GC–MS analysis, the same chromatographic conditions and equipment were employed, with an HP 5973 mass selective detector (MSD) integrated into the system. Helium was used as the carrier gas at a flow rate of 1 mL/min. The mass spectrometer operated with an electron ionization voltage of 70 eV and an emission current of 40 mA. The mass spectrometer scanned a mass range from 35 to 350 atomic mass units (amu) at a rate of 1 scan per second. Compound identification was conducted through a combination of co-injection with authentic standards (Sigma-Aldrich, USA), comparison of retention indices (RI), and detailed mass spectral analysis.31,32
Antimicrobial Assay
The antimicrobial activity of the essential oil extracted from T. viscidum was evaluated against a range of microorganisms, including three strains of Gram-positive bacteria (Enterococcus faecalis ATCC 299212, Staphylococcus aureus ATCC 25923, Bacillus cereus ATCC 14579), three strains of Gram-negative bacteria (Escherichia coli ATCC 25922, Pseudomonas aeruginosa ATCC 27853, Salmonella enterica ATCC 13076), and one strain of yeast (Candida albicans ATCC 10231). These microbial strains were obtained from the American Type Culture Collection (ATCC, Manassas, USA). The minimum inhibitory concentration (MIC) and half-maximal inhibitory concentration (IC50) values of the essential oil were determined using the broth microdilution method, as outlined in previous studies.30,33
In the experiments, Mueller-Hinton Agar (MHA) was used as the medium for bacterial testing, while Sabouraud Agar (SA) was employed for fungal assays. The essential oil solutions were first prepared in dimethylsulfoxide (DMSO) and then further diluted in sterile distilled water within micro-test tubes. These diluted solutions were subsequently transferred to 96-well microtiter plates for testing. Prior to testing, bacterial cultures were standardized to a concentration of 5 × 105 CFU/mL in Mueller-Hinton broth, while fungal cultures were adjusted to 1 × 103 CFU/mL in Sabouraud dextrose broth. Sterile distilled water was used as the negative control, while streptomycin and cycloheximide served as the positive controls for antibacterial and antifungal activities, respectively. The plates were incubated for 24 h at 37 °C for bacteria and 30 °C for fungi to facilitate microbial growth. After incubation, microbial growth was assessed by measuring the optical density at 600 nm using a Spectramax 190 microplate reader. The MIC value was defined as the lowest concentration of the essential oil that completely inhibited visible microbial growth.
Additionally, IC50 values were calculated to quantify the degree of microbial growth inhibition. This was achieved by assessing the percentage of growth inhibition through turbidity measurements using an EPOCH2C spectrophotometer and Rawdata computer software. The IC50 value represents the concentration of the essential oil required to achieve 50% inhibition of microbial growth, as indicated by a 50% reduction in turbidity.
Mosquito Larvicidal Assay
The mosquito larvicidal activity of the essential oil extracted from T. viscidum was assessed against C. quinquefasciatus larvae, following established protocols.30,33 Third instar larvae were collected and reared in the laboratory of Duy Tan University, Vietnam. For the bioassay, the essential oil was first dissolved in DMSO to prepare a 1% stock solution. Serial dilutions of this stock solution were then prepared to obtain final concentrations of 3.125, 6.25, 12.5, 25, 50, and 100 µg/mL. Twenty larvae were introduced into 300 mL beakers containing the different concentrations of the essential oil. The experiments were conducted at room temperature (25 °C), with four replicates for each concentration. Permethrin was used as a positive control to evaluate the effectiveness of the essential oil, while DMSO served as the negative control. Larval mortality was recorded at two time points: 24 h and 48 h post-exposure. The lethal concentration values (LC50 and LC90) and their 95% confidence intervals were calculated using log-probit analysis.
Statistical Analysis
All experiments in this study were conducted in triplicate to ensure the reliability and reproducibility of the results. The IC50 values for antimicrobial assay were determined using a non-linear regression model to fit the dose-response curve. The LC50 and LC90 values for the larvicidal assay were calculated using log-probit regression analysis with 95% confidence intervals. All statistical analyses were performed using Minitab 19.2020.1 (State College, PA, USA) and GraphPad Prism 9.5.1.733 software (GraphPad Software Inc., San Diego, CA, USA).
Results and Discussion
Chemical Composition of Teucrium viscidum Essential Oil
The hydrodistillation of T. viscidum yielded an essential oil with a yield of 0.23% ± 0.01 (v/w), characterized by a rich and complex chemical profile. GC–FID and GC–MS analyses identified 31 compounds, accounting for 97.20% of the total oil composition (Table 1). The oil was predominantly composed of sesquiterpene hydrocarbons, constituting 91.01% of the total composition. The major constituents in this class were (E)-caryophyllene (47.91%), germacrene D (19.17%), and β-bourbonene (4.31%). Oxygenated sesquiterpenes contributed 3.73%, with caryophyllene oxide (2.20%) as the most abundant compound. Monoterpene hydrocarbons were present in minor quantities, comprising only 2.46% of the total composition.
Chemical Composition of the Essential Oil from Teucrium viscidum.
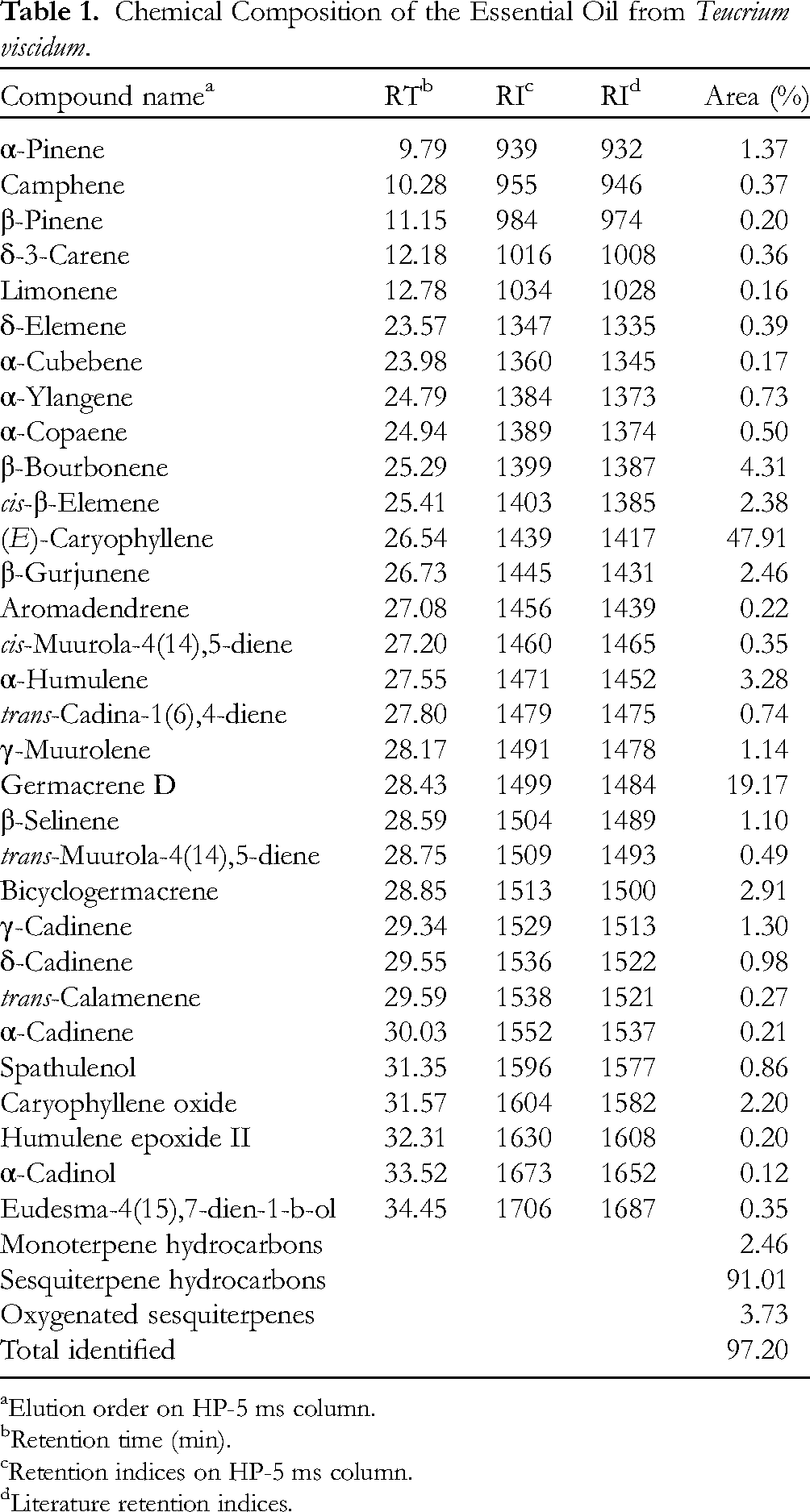
Elution order on HP-5 ms column.
Retention time (min).
Retention indices on HP-5 ms column.
Literature retention indices.
This study presents the first detailed analysis of the chemical composition of T. viscidum essential oil, limiting direct comparisons with previously analyzed samples of the same species. However, comparisons with other Teucrium species reveal several trends and differences. Sesquiterpenes are consistently reported as dominant across the genus, though their specific composition and relative abundance vary. For example, T. marum is characterized by high levels of isocaryophyllene (20.24%) and β-bisabolene (14.73%), 34 while β-caryophyllene is the predominant component in T. arduini (24.5%) and T. chamaedrys (26.9%). 35 This suggests a general trend of sesquiterpene dominance across the genus.
In contrast, T. montanum exhibits a more diverse sesquiterpene profile, including δ-cadinene (17.19%), β-selinene (8.16%), and α-calacorene (4.97%), along with unique compounds such as 1,6-dimethyl-4-(1-methylethyl)-naphthalene (4.91%). 36 Similarly, T. brevifolium demonstrates significant sesquiterpene content (55.5%), with notable constituents including spathulenol (9.0%) and δ-cadinene (4.2%). 37 Other species, such as T. flavum and T. montbretii, also show high sesquiterpene proportions (54.5% and 50.1%, respectively), though with different major components such as caryophyllene (12.2%) and carvacrol (13.9%). 37 In T. polium, sesquiterpenes account for 55.8% of the oil, with carvacrol (10.1%) and caryophyllene (9.8%) being key constituents. 37 T. botrys is notable for its β-caryophyllene (20.4%) and (E)-β-farnesene (17.7%), highlighting variability within sesquiterpene-rich profiles. 35 Lastly, T. scordium presents a terpene profile dominated by α-pinene (17.7%) and β-pinene (10.0%), distinguishing it from other species in the genus. 35
Overall, while sesquiterpenes typically dominate in the Teucrium genus, each species exhibits a unique chemical profile, reflecting the genus's diversity. The high concentration of sesquiterpene hydrocarbons and the low presence of oxygenated compounds in T. viscidum essential oil may have implications for its biological activities, such as antimicrobial, anti-inflammatory, and antioxidant properties. The absence of certain monoterpenoids, commonly found in other essential oils, may also influence its olfactory properties and potential applications in medicinal and industrial contexts. Variations in chemical composition among different Teucrium species and within T. viscidum itself can be attributed to factors such as genetic diversity, environmental conditions, and the specific plant organs used for oil extraction.38,39
While this study offers important insights into the chemical profile of T. viscidum, several limitations must be acknowledged. First, the analysis is confined to essential oil extracted from the aerial parts of the plant; the chemical composition may differ if oils were obtained from other plant organs, such as roots or flowers. Additionally, seasonal variations, harvesting time, and environmental conditions (eg, soil type, altitude, and climate) were not addressed, all of which could significantly impact the oil's composition. Future research should investigate these factors in detail, as they are critical for ensuring consistency in the oil's chemical profile and its subsequent bioactivities. Lastly, the extraction method employed—hydrodistillation in this case—may affect both the yield and chemical composition. Other extraction techniques, such as steam distillation or solvent extraction, might yield oils with different proportions of bioactive compounds, potentially enhancing or reducing their efficacy in various applications.
Antimicrobial Activity of Teucrium viscidum Essential Oil
The antimicrobial effects of T. viscidum essential oil were assessed against seven microorganisms, including three Gram-positive bacteria, three Gram-negative bacteria, and one yeast. The potency of the essential oil was quantitatively evaluated using MIC and IC50 values, as presented in Table 2. The essential oil exhibited significant activity against all three Gram-positive bacteria (E. faecalis, S. aureus, and B. cereus), one Gram-negative bacterium (P. aeruginosa), and one yeast (C. albicans), with MIC values ranging from 32 to 64 μg/mL and IC50 values ranging from 9.56 to 24.07 μg/mL. Specifically, the essential oil showed strong activity against S. aureus, B. cereus, P. aeruginosa, and C. albicans, with an MIC of 32 μg/mL and IC50 values of 9.56, 10.22, 10.54, and 14.89 μg/mL, respectively. The oil also displayed activity against E. faecalis, with an MIC of 64 μg/mL and an IC50 of 24.07 μg/mL. However, the essential oil did not exhibit activity against the Gram-negative bacteria E. coli and S. enterica, suggesting selective antimicrobial action. This selectivity is consistent with findings for essential oils from other species in the Teucrium genus.34,36,40,41
Antimicrobial Activity of the Essential Oil from Teucrium viscidum.

Minimum inhibitory concentration (µg/mL).
Half-maximal inhibitory concentration (µg/mL).
The positive controls for bacteria and yeast were streptomycin and cycloheximide, respectively.
The positive control, streptomycin, used against both Gram-positive and Gram-negative bacteria, exhibited an MIC of 32 μg/mL against E. faecalis, S. aureus, and B. cereus, and an MIC of 128 μg/mL against P. aeruginosa and S. enterica. Streptomycin showed a higher MIC of 256 μg/mL against E. coli. Cycloheximide, the standard antifungal agent, demonstrated activity against C. albicans with an MIC of 32 μg/mL. Notably, the antimicrobial activity of the essential oil was comparable to or, in some cases, superior to that of the positive controls. Previous studies have established that essential oils exhibit strong antimicrobial effects when MIC values are below 100 μg/mL.42,43 Based on this criterion, T. viscidum essential oil in this study demonstrated strong antimicrobial activity.
The observed antimicrobial activity of T. viscidum essential oil can be attributed to its chemical composition, particularly the presence of bioactive sesquiterpenes. Compounds such as (E)-caryophyllene, germacrene D, and caryophyllene oxide, which are abundant in this oil, have been reported to possess significant antimicrobial properties.44–48 These compounds may disrupt microbial cell membranes, inhibit essential microbial enzymes, or interfere with microbial processes, resulting in growth inhibition.5,49 The substantial presence of sesquiterpene hydrocarbons suggests a broad-spectrum antimicrobial effect, as these compounds have demonstrated potent activity against both Gram-positive and Gram-negative bacteria, as well as fungi. 50 The observed antimicrobial effects are likely influenced by synergistic interactions between the major sesquiterpenes and the oil's minor components. Though present in smaller quantities, minor compounds may enhance the efficacy of the primary bioactive sesquiterpenes, leading to more effective disruption of microbial cell structures or inhibition of microbial enzymes. 51 However, the lack of activity against Gram-negative bacteria, such as E. coli and S. enterica, may be due to the presence of an outer membrane in these bacteria, which acts as a barrier to hydrophobic compounds like essential oils. Differences in cell wall structure and composition between Gram-positive and Gram-negative bacteria that affect their susceptibility to antimicrobial drugs have been previously reported.52–54
While compounds such as (E)-caryophyllene and germacrene D are known to disrupt microbial membranes or inhibit enzymatic activity,44–48 the precise mechanisms by which the oil inhibits microbial growth remain to be fully elucidated. Further research is needed to identify the specific targets of these compounds within microbial cells. Additionally, investigating potential synergistic interactions between the essential oil and conventional antimicrobial agents could provide valuable insights into its use as an adjunct treatment, particularly for infections caused by antibiotic-resistant strains. However, before the essential oil can be considered for clinical application, more comprehensive studies on its toxicity and safety profile in humans and animals are essential.
Mosquito Larvicidal Activity of Teucrium viscidum Essential Oil
The insecticidal properties of essential oils have been extensively studied and shown to be effective against various insect pests, including mosquitoes.55,56 In this study, the larvicidal potential of T. viscidum essential oil was evaluated against C. quinquefasciatus larvae, with results presented in Table 3. After 24 h of exposure, the essential oil exhibited significant larvicidal activity, with an LC50 value of 23.07 μg/mL and an LC90 value of 32.90 μg/mL. Prolonged exposure to 48 h resulted in enhanced larvicidal effectiveness, with a reduced LC50 of 19.67 μg/mL and an LC90 of 29.99 μg/mL. These results are consistent with the larvicidal activity observed in essential oils from other species in the Teucrium genus.57,58
Mosquito Larvicidal Activity of the Essential Oil from Teucrium viscidum against Culex quinquefasciatus (μg/mL).

The observed larvicidal activity of T. viscidum essential oil can be attributed to its chemical composition, particularly the presence of bioactive sesquiterpenes. These compounds are known for their insecticidal properties, including disruption of insect nervous systems and interference with physiological processes.59,60 Sesquiterpenes such as (E)-caryophyllene and germacrene D, both present in significant quantities in T. viscidum essential oil, have demonstrated larvicidal activity against various mosquito species.61,62 Oxygenated compounds like caryophyllene oxide further contribute to the oil's overall larvicidal efficacy. 63 The effectiveness of T. viscidum essential oil is also enhanced by synergistic interactions between its major and minor components. 64 While the main constituents, such as (E)-caryophyllene and germacrene D, play a crucial role in the oil's insecticidal properties, the secondary constituents, though present in smaller quantities, can significantly amplify the oil's overall efficacy. These secondary compounds, while not as potent individually, can enhance the activity of the primary components, leading to a more powerful combined effect.60,64
Permethrin, a well-established synthetic insecticide, exhibited an LC50 of 0.00638 μg/mL and an LC90 of 0.0232 μg/mL after 24 h of exposure, values much lower than those for T. viscidum essential oil (LC50 of 23.07 μg/mL and LC90 of 32.90 μg/mL). This highlights the substantially higher potency of permethrin in killing C. quinquefasciatus larvae over the same exposure period. However, while permethrin is more potent, it is also associated with several environmental and health concerns, including insecticide resistance in mosquito populations, potential toxicity to non-target organisms (including beneficial insects and aquatic life), and environmental persistence. Over time, these factors can diminish its long-term effectiveness and sustainability as a mosquito control strategy.
In contrast, T. viscidum essential oil, although less potent than permethrin, offers certain ecological advantages. Essential oils are biodegradable, have a lower environmental impact, and are generally considered safer for humans and non-target species. 10 Their mode of action—often involving multiple biochemical pathways—also reduces the likelihood of resistance development in mosquito populations. The increased efficacy of T. viscidum essential oil with prolonged exposure (as shown by the reduced LC50 and LC90 values after 48 h) suggests that it could be effectively used in longer-term applications, potentially in combination with other eco-friendly pest management strategies.
Compared with other plant-based essential oils, the LC50 values obtained for T. viscidum essential oil fall within the effective range for mosquito larvicides.60,64 This study represents the first investigation into the larvicidal activity of T. viscidum essential oil, making direct comparisons with other studies on the same species challenging. However, previous research has shown that essential oils with LC50 values below 100 μg/mL exhibit significant larvicidal potential.60,64 Based on this criterion, T. viscidum essential oil demonstrates considerable larvicidal activity against C. quinquefasciatus larvae.
The observed increase in efficacy from 24 to 48 h underscores the importance of exposure duration in larvicidal assessments. Extended exposure allows the bioactive compounds in the essential oil to exert a more profound impact, potentially leading to prolonged physiological disruption and increased mortality. 65 This time-dependent effect suggests that optimizing application strategies to consider exposure duration could enhance larvicidal outcomes. Additionally, variation in the susceptibility of C. quinquefasciatus larvae to the oil's active compounds may be influenced by species-specific factors, such as metabolic rate and detoxification mechanisms.60,64 These physiological and biochemical differences can affect how larvae absorb and respond to the essential oil, highlighting the need to consider species-specific responses when developing effective larvicidal treatments. 66
However, there are limitations to the use of essential oils as larvicides. Environmental factors such as UV light exposure, temperature, and humidity can degrade the active compounds in essential oils, reducing their efficacy in field applications. Additionally, the slow-acting nature of essential oils, as demonstrated by the improved efficacy over 48 h, may be less practical in situations requiring rapid control of mosquito populations. Further research is needed to optimize formulations of the essential oil to enhance its stability and prolong its larvicidal activity. Combining T. viscidum essential oil with other botanical or synthetic agents could improve its potency and durability, making it a more viable option for mosquito control programs.
Conclusions
In conclusion, T. viscidum essential oil emerges as a potent natural product with significant potential in healthcare and vector control. Its demonstrated antimicrobial and larvicidal activities, combined with its environmentally friendly profile, make it a valuable alternative to synthetic chemicals. The diverse chemical composition of the oil, particularly its bioactive sesquiterpene compounds, likely plays a key role in its efficacy. Future research should focus on developing essential oil-based formulations for practical applications in medicine and pest control. Investigating the oil's mechanisms of action and exploring synergistic blends with other natural or synthetic agents could enhance its effectiveness. Additionally, comprehensive studies on its safety profile and long-term environmental impact will be crucial for its integration into sustainable disease control strategies. By exploring these avenues, T. viscidum essential oil could play an important role in addressing global challenges such as antibiotic resistance and insecticide-resistant mosquito populations.
Footnotes
Acknowledgments
The authors would like to thank the editor and anonymous reviewers for their thoughtful comments and efforts toward improving our manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
