Abstract
Keywords
Introduction
Inflammation, a fundamental response to injury or infection, is crucial for maintaining bodily homeostasis. 1 However, its dysregulation or chronic manifestation can lead to the development of various debilitating diseases, including cardiovascular disorders, cancer, and neurodegenerative illnesses.1-4 Understanding the intricate molecular mechanisms governing inflammation is essential for devising targeted therapeutic interventions.
Within the complex network of inflammatory responses, lipopolysaccharide (LPS), a potent inducer derived from the outer membrane of Gram-negative bacteria, serves as a key trigger for inflammation. 5 During LPS-induced inflammation, adenosine monophosphate-activated protein kinase (AMPK) emerges as a critical regulator, acting as a cellular energy sensor with the potential to modulate inflammation by regulating cellular metabolism and suppressing pro-inflammatory signals.6-8 However, the precise mechanisms through which AMPK influences the inflammatory response during LPS stimulation require further elucidation. Janus kinase 2 (JAK2) and signal transducer and activator of transcription 3 (STAT3) are other crucial players in LPS-induced inflammation,9,10 known for their involvement in cytokine signaling and their influence on the inflammatory response. 10 The potential interplay and crosstalk among AMPK, JAK2, and STAT3 hint at the existence of a signaling pathway that could hold therapeutic significance. 11 Therefore, the AMPK/JAK2/STAT3 signaling pathway presents itself as a promising target for therapeutic interventions in inflammation, necessitating a deeper exploration of its intricacies. 12
Glabridin (GBD, Figure 1A), a natural flavonoid isolated from licorice root, has garnered attention for its anti-inflammatory and antioxidant properties. 13 Previous studies have reported its ability to inhibit the production of pro-inflammatory cytokines, such as tumor necrosis factor (TNF)-α, interleukin (IL)-6, and IL-1β, as well as nitric oxide (NO), in various cell types.13-15 Some studies have explored some aspects of the STAT3 or AMPK signaling pathways modulated by GBD.16-18 While previous studies have examined individual aspects of GBD's effects on inflammatory pathways, the potential interplay between AMPK, JAK2, and STAT3 signaling in response to GBD treatment, particularly in LPS-induced macrophage inflammation, remains largely unexplored. To address this gap, we have methodically investigated the relationship between these keys signaling pathways in the context of GBD's anti-inflammatory effects on LPS-stimulated macrophages. By focusing on these specific pathways, this study seeks to expand our knowledge of GBD's molecular mechanisms and provide new insights into how GBD exert their anti-inflammatory effects through multiple signaling pathways, potentially offering new directions for therapeutic strategies in inflammatory diseases.

Effects of Glabridin (GBD) on cell viability and morphological changes in lipopolysaccharide (LPS)-stimulated RAW264.7 cells. (A) Structural representation of GBD. (B,C) RAW264.7 cells were treated with either 0.1% DMSO or pre-treated with GBD (1-10 μM), followed by exposure to LPS (1 μg/mL) or not for 24 h. Cell viability was assessed according to the methods described in the Materials and Methods section. (D) Cellular morphology was examined using optical microscopy at 200x magnification. Scale bar: 25 μm. (E) The activation index percentage illustrates the proportion of cells displaying activated morphology relative to the total cell count. The data is presented as means ± SEM (
Results
Effects of GBD on Cytotoxicity and Morphology in RAW 264.7 Cells
Cytotoxicity assessments using the MTT assay demonstrated that GBD concentrations ranging from 1 to 10 μM did not induce cytotoxic effects in RAW264.7 cells (Figure 1B). Subsequently, when examining the impact of GBD on LPS-induced cytotoxicity in RAW264.7 cells (Figure 1C), the MTT assay revealed that GBD did not exacerbate cytotoxicity under LPS stimulation.
Microscopic analysis of cellular morphology (Figure 1D) provided visual insights into the effects of GBD on RAW264.7 cells. In the control group, DMSO + LPS group, and 10 μM GBD group showed similar cellular morphology, with no notable changes observed. However, under LPS stimulation, a distinct activation-induced morphological change was observed. Specifically, cells transformed from a round shape to a more flattened and irregular form, with increased intercellular spaces and the formation of cellular protrusions. These changes are indicative of cellular activation. Remarkably, co-treatment with 10 μM GBD and LPS mitigated this activation, restoring cellular morphology to a state comparable to the control group, where cells maintained their round shape, remained closely packed, and showed no obvious protrusions. Furthermore, the 10 μM GBD group exhibited cellular morphology similar to the control group, suggesting that GBD alone does not induce significant morphology alterations. The activation index, derived from the morphological observations (Figure 1E), substantiated these findings. The control group displayed a baseline activation index of 4.7 ± 0.8%. The DMSO + LPS group showed a significant increase in activation index to 37.1 ± 3.9% (
GBD Regulated LPS-Induced Inflammation Cytokines in RAW264.7 Cells
Evaluation of NO production, as determined by the Griess assay, revealed a significant increase in the DMSO + LPS group, serving as our positive control. However, an inhibitory effect only in the 10 μM GBD with LPS group (

Effects of GBD on nitric oxide (NO) production, protein expression of inducible nitric oxide synthase (iNOS) expression, interleukin-1 beta (IL-1β), and tumor necrosis factor-alpha (TNF-α) in LPS-stimulated RAW264.7 cells, with additional comparison of no inhibition by GBD and L-NAME. (A) RAW264.7 cells were pre-treated with GBD (1-10 μM) and subsequently stimulated with LPS for 24 h. Griess reagent was used to measure NO levels; (B–D) Pre-treatment of RAW264.7 cells with GBD (5 and 10 μM) followed by LPS stimulation for 24 h. Evaluation of (B) iNOS, (C) IL-1β, and (D) TNF-α protein expression was conducted as outlined in the Methods section. (E) Comparative effects of GBD and L-NAME on LPS-induced NO production. Cells were pretreated with DMSO, GBD (10 μM), or L-NAME (20 μM) for 30 min, followed by LPS stimulation for 24 h. NO levels were measured as described in (A). The data is presented as means ± SEM (
Subsequent experiments using both 5 μM and 10 μM GBD further dissected the impact on inflammatory mediators. In terms of iNOS protein expression (Figure 2B), the DMSO + LPS group showed a marked increase (
Figure 2C depicts the impact of GBD on IL-1β protein expression. The DMSO + LPS group showed elevated levels of IL-1β, whereas both 5 μM and 10 μM GBD significantly inhibited IL-1β protein expression (
In addition, L-NAME (8.7 ± 1.4 μM,
Modulation of JAK2/STAT3 Signaling and AMPK by GBD Alters LPS-Induced Inflammation in RAW264.7 Cells
Western blot results (Figure 3A and B) indicated a significant increase in phosphorylated JAK2 and STAT3 upon LPS stimulation, indicative of pathway activation. Co-treatment with GBD demonstrated a dose-dependent suppression of JAK2 and STAT3 phosphorylation, suggesting a robust inhibitory effect on the JAK2/STAT3 signaling axis. Examining AMPK expression through Western blot analyses (Figure 3C), a significant reduction in AMPK phosphorylation levels was observed in LPS-induced macrophages (

GBD's impact on Janus kinase 2 (JAK2), signal transducer and activator of transcription 3 (STAT3), and adenosine monophosphate-activated protein kinase (AMPK) phosphorylation in RAW264.7 cells. Cells treated with 0.1% DMSO or GBD (5 and 10 μM), followed by LPS stimulation 3 h. Immunoblotting assessed phosphorylation levels of (A) JAK2, (B) STAT3, and (C) AMPK. Data: means ± SEM (
Impact of JAK2 Inhibitor AG490 and AMPK Inhibitor Compound C on Inflammatory Responses in LPS-Induced RAW264.7 Cells
The roles of JAK2 and AMPK signaling pathways in regulating inflammatory responses were investigated by examining the effects of their specific inhibitors on the inflammatory markers in LPS-induced RAW264.7 cells. AG490, a JAK2 inhibitor, and Compound C (CC), an AMPK inhibitor, were utilized to assess the involvement of these markers. Figure 4A illustrates the dose-dependent inhibitory effect of AG490 (10-80 μM) on LPS-induced NO production, indicating a significant reduction in NO levels. Similarly, Figure 4B demonstrates the inhibitory effects of CC (1-10 μM) on LPS-induced NO production. The concentration-dependent response suggests that CC effectively suppresses NO production in RAW264.7 cells under LPS stimulation.
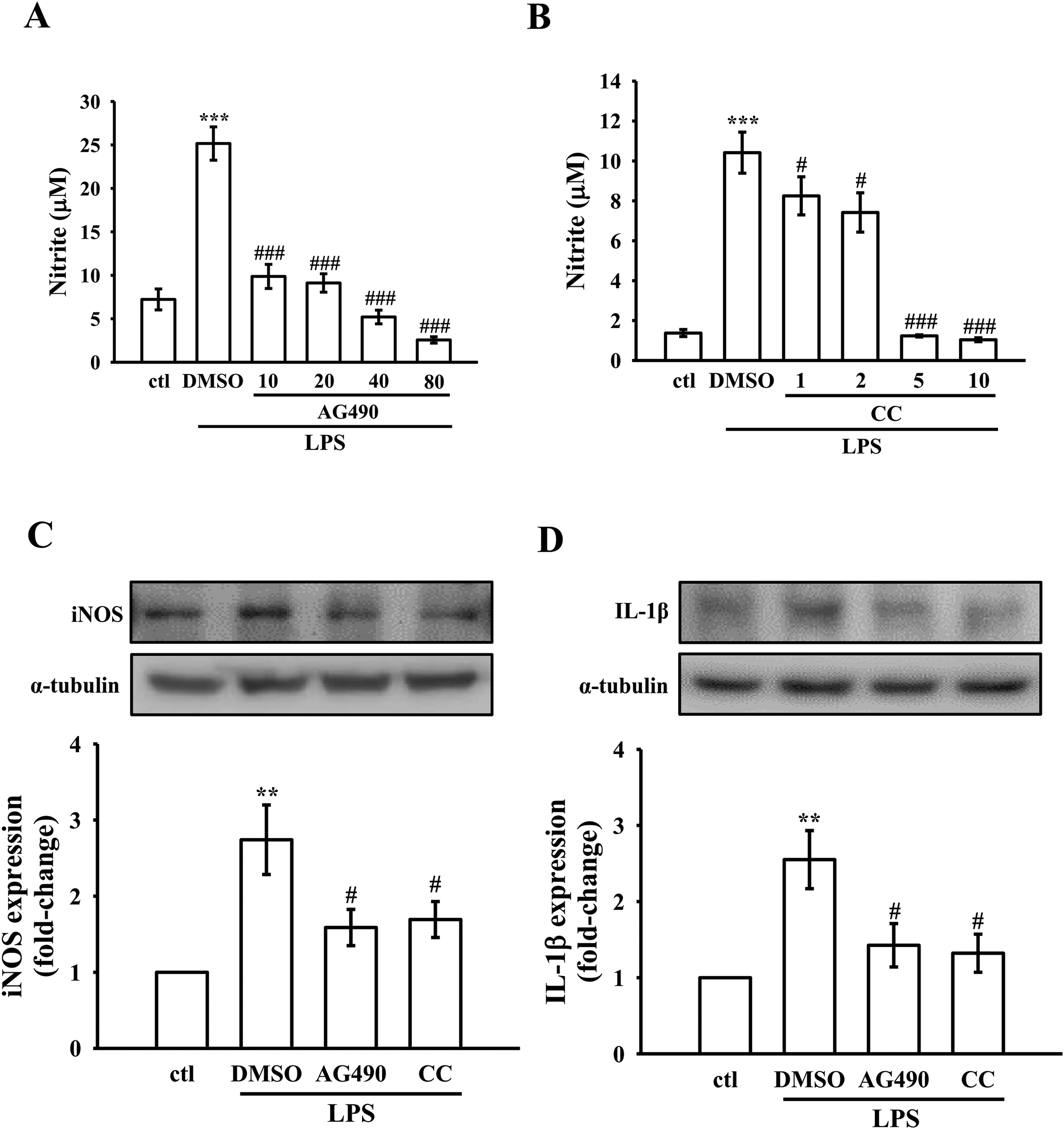
Effects of inhibitors on NO production and the protein expression of iNOS expression, and IL-1β in LPS-stimulated RAW264.7 cells. (A-B) RAW 264.7 cells were pre-treated with inhibitors (AG490 and compound c (CC)) and subsequently stimulated with LPS (1 μg/mL) for 24 h. Griess reagent was used to measure NO levels; (C–D) Pre-treatment of RAW264.7 cells with 0.1% DMSO or inhibitors, followed by LPS stimulation for 24 h. Evaluation of (C) iNOS and (D) IL-1β protein expression was conducted as outlined in the Methods section. The data are presented as means ± SEM (
At the protein expression level, Figure 4C and D provide insights into the impact of AG490 and CC on specific inflammatory markers. In Figure 4C, both 10 μM AG490 and 1 μM CC exhibit inhibitory effects on LPS-induced iNOS protein expression, suggesting a regulatory role in modulating iNOS. Figure 4D delves into the modulation of IL-1β protein expression under LPS-induced conditions. Both 10 μM AG490 and 1 μM CC showcased inhibitory effects on LPS-induced IL-1β protein expression. These findings underscore the anti-inflammatory potential of JAK2 and AMPK in mitigating LPS-induced inflammatory responses in RAW264.7 cells.
Inhibition of Signaling in LPS-Induced RAW264.7 Cells by AG490 and CC
To elucidate the relationship between JAK2/STAT3 and AMPK pathway in LPS-induced RAW264.7 cells, specific inhibitors AG490 and CC were employed. Western blot results (Figure 5A-C) demonstrated that AG490, a JAK2 inhibitor, effectively suppressed the phosphorylation of JAK2 and STAT3, without affecting AMPK phosphorylation. In contrast, CC, an AMPK inhibitor, reduced the phosphorylation levels of JAK2, STAT3, and AMPK (Figure 5A-C). These results suggest that while AMPK may influence JAK2/STAT3 signaling, the inhibition of JAK2/STAT3 does not appear to affect AMPK phosphorylation, indicating a potentially unidirectional relationship between these pathways in this context.

Inhibitors’ influence on the phosphorylation of JAK2, STAT3, and AMPK in RAW 264.7 cells. (A) RAW264.7 cells were treated with either 0.1% DMSO or inhibitors, followed by stimulation with LPS for 3 h. Immunoblotting was employed to assess the phosphorylation levels of (A) JAK2, (B) STAT3, and (C) AMPK. The data is presented as means ± SEM (
Discussion
Licorice root, renowned in traditional medicine for its extensive pharmacological properties, has a rich history of use spanning centuries across Asia and Europe.13,14 Within licorice extract, GBD, a key component of the hydrophobic fractions, stands out for its exceptional medicinal attributes. The results of this study demonstrated that GBD's potent anti-inflammatory activity in LPS-induced macrophages, potentially achieved through the modulation of the AMPK/JAK2/STAT3 signaling pathway (Figure 6).

Schematic representation of the anti-inflammatory mechanism of GBD
Notably, our investigation revealed a nuanced dose-dependent effect of GBD on macrophage function and inflammation. The effective mitigation of inflammation was observed at GBD concentrations of 10 µM. At this optimal concentration, GBD demonstrates significant immunomodulatory effects by suppressing the activated phenotype induced by inflammatory stimuli without compromising macrophage viability. This finding is crucial as it suggests a favorable therapeutic window for GBD, highlighting its potential as a promising candidate for innovative anti-inflammatory applications in clinical settings.
Inflammatory cytokines, including iNOS, TNF-α, and IL-1β, play pivotal roles in the inflammatory response. Our study demonstrates that GBD significantly reduced IL-1β levels and NO production, while also inhibiting iNOS expression. However, it's noteworthy that GBD had no significant effect on TNF-α levels under a concentration of 10 μM. The phenomenon likely indicates that GBD has a targeted inhibitory effect on IL-1β. This could mean that GBD may block the synthesis of IL-1β protein, inhibit the gene expression of IL-1β, or interfere with IL-1β related signal transduction pathways. 19 The differential impact on IL-1β compared to TNF-α suggests that GBD acts more selectively on certain inflammatory mediators.20,21 While our findings on GBD's selective anti-inflammatory effects are novel, they can be contextualized within the broader literature on GBD's immunomodulatory properties. 22 Although previous studies have not specifically highlighted this selectivity, they have reported various anti-inflammatory effects of GBD.22-24 For instance, El-Ashmawy et al, 23 demonstrated GBD's capacity to downregulate iNOS gene expression in the rat colon, while Weng et al, 24 reported inhibitory effects on iNOS expression in BV2 cells. Our study extends these findings by revealing GBD's differential effects on specific inflammatory mediators, particularly its selective impact on IL-1β and iNOS without affecting TNF-α. The consistency of GBD's inhibitory effects across iNOS across different cell types and experimental models, as observed in our study and previous research, strengthens the credibility of our findings. However, the selective modulation of IL-1β, iNOS, and NO by GBD without affecting TNF-α, as we’ve uniquely identified, underscores the distinct regulatory mechanisms governing these inflammatory mediators. This discrepancy highlights the complexity of the inflammatory response and suggests that GBD may have differential effects on different cytokines. Further comprehensive studies on GBD's mechanism of action are needed to fully understand its differential effects on various inflammatory mediators and to explore its potential therapeutic applications.
The ability of GBD to modulate multiple signaling pathways simultaneously points to its potential as a multi-target anti-inflammatory agent. This broad-spectrum activity could be particularly beneficial in addressing complex inflammatory conditions where multiple pathways are dysregulated. Our findings reveal a complex interplay between GBD and key inflammatory signaling pathways, namely JAK2/STAT3 and AMPK. The observe inhibition of LPS-induced phosphorylation of these proteins by GBD suggests a multi-faceted mechanism of action in its anti-inflammatory effects. The suppression of JAK2 and STAT3 phosphorylation is particularly significant given the central role of the JAK2/STAT3 pathway in inflammatory cytokine signaling. Recent studies have shed light on the complexity of JAK/STAT signaling in inflammatory conditions. For instance, Liang et al 25 discovered a novel compound that effectively inhibits both JAK/STAT and NF-κB pathways, showing potent anti-inflammatory effects in inflammatory bowel disease (IBD) models. This finding aligns with our observations on GBD's ability to modulated multiple pathways. However, the landscape of JAK/STAT signaling in inflammation is more complex that initially thought. Stolzer et al 26 highlighted the differential contribution of JAK signaling in ulcerative colitis (UC) and Crohn's disease (CD), indicating that the lack of effect on JAK2/STAT3 signailing in CD may be due to the non-redundant functions of STAT1 and STAT2 in regulating inflammation in different parts of the intestine. Furthermore, Cordes et al 27 demonstrated an imbalance between STAT1 and STAT3 activation in UC monocytes, leading to stronger inflammatory responses. These findings collectively suggest that the differential activation and function of STAT proteins in various inflammatory conditions may underlie the varying effects observed in different models. This inhibition likely contributes to the reduced levels of IL-1β and iNOS expression observed in our earlier experiments, highlighting a potenital mechanism through which GBD attenuates the inflammatory response.
AMPK activation plays a crucial role in mediating the anti-inflammatory effects of natural compounds through various mechanisms. AMPK activation has been shown to inhibit the STAT1, thereby suppressing vascular inflammation. 28 Additionally, AMPK exerts anti-inflammatory effects by activating SIRT1, PGC-1α, p53, FoxO3a, and p300, while down-regulating NF-κB and AP-1 activity, ultimately reducing inflammation. 29 These findings highlight the diverse and interconnected mechanisms through which AMPK activation contributes to the anti-inflammatory properties of natural compounds. In general, inflammatory stimuli induce the production of reactive oxygen species and increase intracellular calcium concentration, 30 leading to the activation of AMPK-activating kinases. 31 While AMPK activation is often associated with anti-inflammatory effects, our results suggest that GBD's anti-inflammatory action may operate through AMPK-independent mechanisms in this context. As shown in Figure 3C, LPS treatment significantly increased AMPK phosphorylation; however, pretreatment with GBD inhibited LPS-induced AMPK phosphorylation. Interestingly, several reports have shown that AMPK promotes cell death in a variety of cell types, including neurons, and that inhibiting AMPK protects cells from ATP depletion-induced cell death following exposure to various stressors.32,33 In the present study, we identified AMPK as a potent counter-regulator of macrophage inflammatory function. The inhibition of LPS-induced AMPK phosphorylation by GBD, coupled with its anti-inflammatory effects, suggests a novel mechanism of action for GBD in the context of inflammation regulation. This finding challenges the conventional understanding of AMPK's role in inflammation and highlights the complex nature of cellular responses to inflammatory stimuli. Further research is needed to elucidate the precise molecular pathways through which GBD mediates its anti-inflammatory effects, potentially revealing new targets for anti-inflammatory therapies.
In the present study, we also emphasized the action of GBD on the inflammation-controlling pathway, particularly highlighting the interconnectedness between JAK2 and AMPK in the cellular processes. We demonstrated that AG490, a JAK2 inhibitor, and CC, an AMPK inhibitor, attenuated IL-1β and iNOS protein expression in LPS-treated RAW264.7 cells. This result is consistent with several previous studies showing that AG490 and CC inhibited IL-1β and iNOS release in various cells.34-37 Additionally, according to the effect of the inhibitors, our data suggests that GBD may influence LPS-induced inflammation by initially inhibiting AMPK, followed by an impact on JAK2/STAT3 signaling. This distinguishes the study by revealing the intricate network of signaling events modulated by GBD. However, the exact mechanism by which AMPK inhibition leads to the inhibition of JAK2 and STAT3 requires further elucidation. The findings compared to previous studies suggested several potential mechanisms. First, AMPK may directly phosphorylate STAT3, altering its transcriptional activity.
12
Second, AMPK inhibition could affect the activity of negative regulators such as SOCS proteins.38,39 Moreover, it's crucial to recognize the constraints that come with the use of a particular cell line
Conclusion
In conclusion, the present study has unveiled novel insights into the anti-inflammatory mechanisms of GBD in LPS-stimulated RAW264.7 macrophages. Our findings highlight GBD's ability to modulate AMPK, JAK2, and STAT3 signaling cascades. This multi-target action of GBD represents a significant advancement in our understanding of natural anti-inflammatory compounds. The intricate interplay between AMPK and JAK2/STAT3 pathways modulated by GBD suggests a more complex mechanism of action than previously recognized. This complexity enhances our understanding of inflammatory signaling networks and may lead to more effective strategies for treating inflammatory disorders. These findings not only contribute to our fundamental understanding of inflammatory processes but also pave the way for a new paradigm in natural drug development against inflammation-related ailments. This study provides valuable insights into the potential of GBD as an anti-inflammatory agent and opens up new avenues for research in the field of inflammation and immunomodulation.
Materials and Methods
Chemicals and Reagents
GBD (≥ 98%) and L-NAME were procured from Cayman Chemical (Ann Arbor, MI, USA). Dulbecco's modified Eagle's medium (DMEM), fetal bovine serum (FBS), and L-glutamine penicillin/streptomycin were obtained from Invitrogen (Thermo Fisher Scientific, Inc., Waltham, MA, USA). LPS, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), and dimethyl sulfoxide (DMSO) were sourced from Sigma-Aldrich (St. Louis, MO, USA). Santa Cruz Biotechnology, Inc. (Dallas, TX, USA), supplied the anti-iNOS polyclonal antibody (pAb) and anti-α-tubulin monoclonal antibody (mAb). Affinity Biosciences (Cincinnati, OH, USA) provided antiphospho-JAK2, antiphospho-STAT3, and antiphospho-AMPK pAbs. Anti-TNF-α and anti-IL-1β pAb were obtained from Proteintech Group (Chicago, IL, USA). Horseradish peroxidase-conjugated donkey antirabbit immunoglobulin (Ig) G and sheep antimouse IgG antibodies were supplied by Amersham, GE Healthcare Life Sciences (Chalfont, UK). Hybond™-P polyvinylidene difluoride (PVDF) blotting membranes and enhanced chemiluminescence for Western blotting were provided by GE Healthcare Life Sciences.
Cell Culture
The RAW264.7 cell line was acquired from the Bioresource Collection and Research Center (BCRC) in Hsinchu, Taiwan. Cells were cultured in DMEM supplemented with 10% FBS, 100 U/mL penicillin G, and 100 mg/mL streptomycin at 37 °C in a humidified incubator containing 5% CO2. GBD was resuspended in DMSO to achieve a stock concentration of 100 mM and stored at −20 °C. DMSO served as a vehicle control in all GBD experiments, and the amount of DMSO (0.1%, v/v) used was standardized across all samples.
Cell Viability Assay
RAW264.7 cells (1 × 105 cells per well) were seeded into 24-well culture plates with DMEM containing 10% FBS for 24 h. The cells were treated with various concentrations of glabridin (1-10 μM) or solvent control (0.1% DMSO) for 20 min and then stimulated with LPS (1 μg/mL) or left unstimulated for 24 h. Cell viability was measured using the MTT assay. Formazan crystals were dissolved in DMSO, and sample absorbance was determined at 570 nm using an MRX absorbance reader (Dynex Technologies, Chantilly, VA, USA).
Determination of Nitric Oxide Production
To evaluate NO production, the levels of nitrite/nitrate, stable oxidative end products of nitric oxide, were measured with slight modifications to a previously described method. 40 RAW264.7 cells (1 × 105 cells/well) were plated in 24-well culture plates with DMEM containing 10% FBS for 24 h. Following a 30 min treatment with GBD (1-10 μM), L-NAME (20 μM) or the solvent control (0.1% DMSO), the cells were either stimulated with LPS (1 μg/mL) or left unstimulated for an additional 24 h. After the 24-h incubation period, the cell culture supernatants were collected and combined with equal volumes of Griess reagent (Cayman Chemical, Ann Arbor, MI, USA). The absorbance of the samples was measured at 540 nm using an MRX absorbance reader. Nitrite/nitrate concentrations were determined using a standard curve generated by linear regression based on absorbance measurements of standard solutions (sodium nitrite dissolved in the same culture medium).
Immunoblotting
A Western blotting analysis was performed to determine protein expression in cells as previously described. 12 RAW264.7 cells (8 × 105 cells/dish) were seeded on 6-cm dishes with DMEM containing 10% FBS for 24 h. The cells were pretreated with GBD or 0.1% DMSO for 30 min and either stimulated with LPS (1 μg/mL) or left unstimulated according to the experimental design. Proteins were extracted using lysis buffer, and protein concentrations were quantified using a Bradford protein assay (Bio-Rad, Hercules, CA, United States). Fifty micrograms of protein samples were applied to sodium dodecyl sulphate (SDS)-polyacrylamide gel electrophoresis, and separated proteins were electrophoretically transferred onto PVDF membranes (0.45 μm). Membranes were blocked with 5% skimmed milk in TBST buffer (10 mM Tris-base, 100 mM NaCl, and 0.01% Tween 20) for 30 min. Membranes were incubated with the targeting primary antibodies against phospho-JAK2, phospho-STAT3, phospho-AMPK, iNOS, IL-1β, TNF-α, and α-tubulin for 2 h and then subjected to HRP-conjugated donkey anti-rabbit IgG or sheep anti-mouse IgG for 1 h at room temperature. The ECL system was used to detect immune-reactive bands, and densitometry of protein bands was performed using the Biolight Windows Application, V2000.01 (Bio-Profil, Vilber Lourmat, France).
Statistical Analysis
Results are presented as means ± standard error (SEM) with the number of observations (
Footnotes
Abbreviations
Acknowledgements
The National Science and Technology Council of Taiwan, Cathay General Hospital, Chi Mei Medical Center and Taipei Medical University, and Shin Kong Wu Ho-Su Memorial Hospital provided funding for this work.
Author Contribution
SMH, JRS and CHH were involved in conceptualization; SMH, SWH, KCL, CWH, and WCH were involved in formal analysis and investigation; TLY was involved in methodology; CHH written the original draft preparation. JRS and CHH reviewed and edited the manuscript. All author have read and agreed to the publication of the final version of the manuscript.
Data Availability
The data that support the findings of this study are available within the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article.
Funding
This research was supported by the National Science and Technology Council of Taiwan (MOST 110-2320-B-341-001-MY3 and NSTC 112-2320-B-038-037-MY3), Cathay General Hospital (CGH-MR-A11226), Chi Mei Medical Center-Taipei Medical University (110CM-TMU-06) and Shin Kong Wu Ho-Su Memorial Hospital (2023SKHAND006 and 2024SKHAND008).
Shin Kong Wu Ho-Su Memorial Hospital, Cathay General Hospital, Chi Mei Medical Center-Taipei Medical University, National Science and Technology Council, (grant number 2023SKHAND006, 2024SKHAND008, CGH-MR-A11226, 110CM-TMU-06, MOST 110-2320-B-341-001-MY3, NSTC 112-2320-B-038-037-MY3).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
