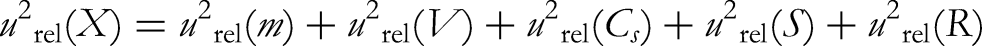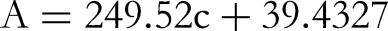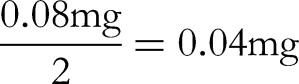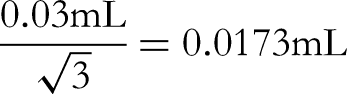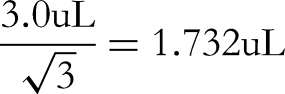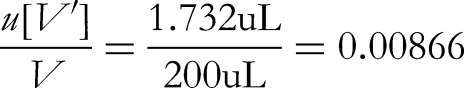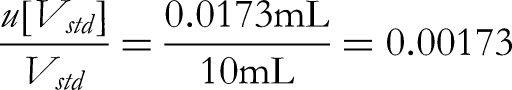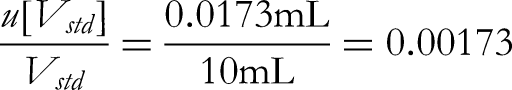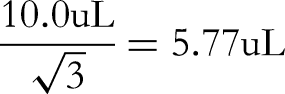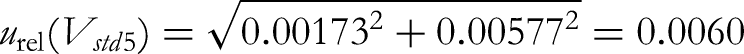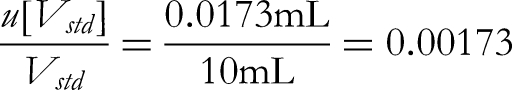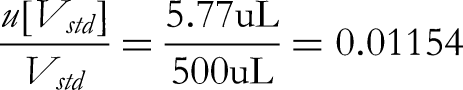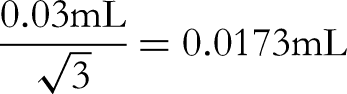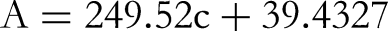Abstract
High-performance liquid chromatography-tandem mass spectrometry (HPLC-MS/MS) was used to determine the content of sibutramine in health foods according to the BJS 201701 standard Determination of Compounds such as Sibutramine in Food. By evaluating each uncertainty component, the influencing factors of the uncertainty were identified and the expanded uncertainty of sibutramine detection in health foods was provided. Using the example of illegal addition of sibutramine to health products, the HPLC-MS/MS method was used for qualitative screening and quantitative analysis of samples. The sources of uncertainty in the quantitative results were analysed, including sample weighing, sample volume determination, standard solution preparation, standard curve fitting and determination of sample repeatability. The synthetic relative standard uncertainty of the measurement results was calculated and the expanded uncertainty was reported. By optimizing the liquid phase conditions, the sample can be detected within 5 min with a linear correlation coefficient of 0.998. When the content of sibutramine in health products was 42.2 mg/kg, the extended uncertainty of the results was 4.56 mg/kg (k = 2) with a 95% confidence interval. Among the various components affecting the results of detection, the uncertainty introduced during the process of preparing the standard solution has the greatest impact on the results. A proper evaluation of the uncertainty, detailed analysis and quantification of the various sources of uncertainty can help to improve the reliability of test results.
Keywords
Introduction
With the improvement of living standards, people are paying more and more attention to their health. As a type of food that claims to have specific health functions or to supplement vitamins, minerals and other purposes, health foods are suitable for consumption by certain groups of people and have the function of regulating bodily functions, so they are increasingly favored by consumers.1,2 However, some unscrupulous companies often add illegal drug ingredients to health foods for profit, including sibutramine.3-5 Sibutramine is a serotonin and norepinephrine reuptake inhibitor developed by Boots Company in 1988. It was originally used to control appetite and was once used as an ingredient in weight-loss drugs. Its main mechanism of action is to inhibit the reuptake of noradrenaline, serotonin and dopamine in the body, increase gastrointestinal fullness and accelerate energy expenditure. It is suitable for obesity that cannot be controlled by diet and exercise to achieve weight loss.6-8 However, a large body of clinical data has shown that sibutramine can cause many adverse effects, such as an increased risk of heart disease, hypertension and arrhythmias, as well as dry mouth, nausea and even an increase in appetite. It can also cause anxiety, irritability, convulsions and epileptic seizures.9-11 In 2010, the State Food and Drug Administration announced a ban on the production, sale and use of sibutramine preparations and raw materials. Although the country has banned sibutramine, there are still many unscrupulous companies that take risks for profit and package sibutramine as various weight loss health foods for sale.12-14 Therefore, the detection of sibutramine in health foods is of great importance.
Uncertainty is an assessment of the range of values of the “measured value” and is a very important part of the laboratory measurement process.15-18 In this study, the components of measurement uncertainty introduced by different aspects were analysed, which play an important role in improving the measurement method. This study takes sibutramine as an example, according to the standard BJS 201701 Determination of Chemicals in Foods such as Sibutramine, HPLC-MS/MS was used for qualitative and quantitative analysis of the samples. By evaluating the different uncertainty components, the factors influencing the uncertainty were identified. This provides a reference for standardizing the daily control of illegal addition of sibutramine in health foods to evaluate the accuracy and effectiveness of test results.
Materials and Methods
Materials and Reagents
Sibutramine hydrochloride monohydrate standard solution (concentration 1000 µL/mL, Bepure, China). Methanol and acetonitrile (chromatographic grade, Merck Chemical Technology (Shanghai) Co., Ltd, Germany). Formic acid (LC-MS, TCI, Japan). Laboratory water is grade I water. Sibutramine quality control product was purchased from Beijing North Weiye Measurement Technology Research Institute Co., Ld
Instruments and Conditions
Waters XevoTQD high-performance liquid chromatography-tandem mass spectrometry (Waters Technology (Shanghai) Co., Ltd); XSE205 analytical balance (d = 0.1 mg, Mettler-Toledo Instruments (Shanghai) Co., Ltd). A CORTECS T3 column (Waters, 2.1 × 100 mm, 1.8 µm particle size) was used for the separation, using an aqueous phase (0.1% formic acid, solvent A) and an organic phase (0.1% formic acid/acetonitrile solution) as the mobile phase under gradient elution as follows 0–0.5 min, 95% A; 0.5–2.5 min, 95% −2% A; 2.5–4 min, 2% A; 4–4.5 min, 2% A-95% A; 4.5–5 min, 95% A. The flow rate was 0.3 mL/min with an injection volume of 1 µL. The column oven was set at 30 °C.
For the MS/MS detection, electrospray positive electrospray ionization mode was used for ionization. The desolvation gas flow was set at 1200 L/h with a source temperature of 350 °C. The capillary and cone voltages were 1.0 kV and 22 V, respectively. For each analyte of interest, two pairs of selected-reaction monitoring (SRM) were selected, one for qualifier and the other for quantifier: m/z 280.1 > 124.9 (qualifier, collision energy: 22 eV), 280.1 > 130.0 (quantifier, collision energy: 22 eV)
Sample Pretreatment
1 g (or 1 mL for liquid) of the sample was weighed accurately (to 0.001 g) and placed in a 10 mL volumetric flask. Methanol was added to make up the volume. After sealing and weighing, the flask was sonicated for 10 min. It was then cooled and weighed again. Methanol was added to make up the lost weight and the solution was then passed through a 0.22 µm filter membrane. 100 µL of the filtrate was taken and diluted in a 100 mL volumetric flask for measurement by liquid chromatography-tandem mass spectrometry.
Quantification Algorithm
The quantification module of MassLynx V4.1 was used to generate the quantification method, including peak detection, peak integration, and analyte quantification. Within the MassLynx Classic Integration Algorithm, the smoothing factor and the bunching factor were set to 3 and 1, respectively. Calibration function generation: plot the peak area ratio of an analyte against the corresponding analyte standard concentration. A weighted leastsquares method was used with a weighting factor of 1/x2.
Analysing Sources of Uncertainty
By analysing the factors affecting the uncertainty during the experimental process, the fishbone diagram of the factors affecting the uncertainty of sibutramine was drawn, which helps to avoid the repeated calculation of uncertainty factors, as shown in Figure 1. By analysing various conditions, the main factors causing uncertainty were identified, including standard solution preparation, sample weighing, sample volume determination, and repeatability of sample measurement (instrument performance, personnel operation, random error), etc.

Influencing factors of uncertainty.
Establishment of the Mathematical Model
(1) The quantitative detection formula can be expressed as follows: X: the content of the tested component in the sample, µg/kg; C: obtain the content of the tested component in the sample solution obtained from the standard curve, µg/L; V: the volumetric capacity of the sample solution, mL; m: Weight of the sample, g; f: Conversion coefficient.
(2) By analysing the sources of uncertainty, the measurement uncertainty of sibutramine is mainly composed of standard substance solution preparation, sample weighing, sample volume determination, standard curve fitting, and the uncertainty introduced by the repeatability of sample measurement. Therefore, the synthetic relative standard uncertainty formula can be expressed as follows: urel (m): the relative standard uncertainty of the sample weight; urel (V): relative standard uncertainty of the sample volume determination; urel (Cs): relative standard uncertainty of the standard solution preparation; urel (S): the relative standard uncertainty of standard curve fitting; urel (R): the relative standard uncertainty of the sample repeatability experiments.
Results and Discussion
Linearity
As for a quantitative measuring procedure, linearity experiment is an essential component of testing and confirming the detection range of the measurement procedure. In this experiment, five points were analyzed with three replicates each following the standard operating procedures of BJS 201701 Determination of Compounds such as Sibutramine in Food, and the linear range was evaluated by the polynomial regression method. Linear correlation coefficient should be larger than 0.990 for accurate quantification. Take the distribution of 5 standard working solutions of different concentrations (1, 2, 5, 8, 10 μg/L) and measure it three times. The calibration curves is shown in Figure 2 and the linear correlation coefficient is 0.998. The regression equation of the standard working curve is:

The calibration curves for sibutramine.
Matrix Correction
Matrix effect, which is independent of the presence of the analyte, influences the accurate quantification for analyte. Matrix effects can cause suppression or enhancement of the analyte signal. The magnitude of matrix effects can be evaluated through the calculation of matrix correction factor (MF), which is defined as the ratio of the intensity of the extracted blank matrix added the analytes to the intensity of the analytes in pure solution (methanol in this experiment). The matrix effect factor for the present method was 85.3%, which is between 80% and 120% is acceptable.
Carry-Over Effect
The carry-over effect was evaluated in this experiment and operatedas follows: The blank sample was injected immediately following a high concentration (10 µg/mL) of the calibration solution. The results were shown in Figure 3. At the elution time of analytes, the peak area of the blank sample was < 20% of the peak area of the LOQ sample for analytes, which is acceptable.

Chromatograms of (a) 10 µg/mL calibration solution; (2) blank sample.
Intra-day and inter-day Precision
To evaluate the intra-day and inter-day precision, five replicates of the in-house prepared QC samples at two levels of concentration (5 and 10 µg/L) were analyzed on the same day and three consecutive days. For the QC samples, the precision of each concentration should not exceed 15% CV is acceptable. In present experiment, the precision of Intra-day and inter-day were 3.96% and 8.63%, respectively, for the 5 µg/L. And 5.28% and 7.35%, respectively, for the 10 µg/L. The validation results for precision were all well within the maximum tolerated CV.
Uncertainty Introduced During the Sample Measurement Process, urel(m)
The sample is weighed on an electronic balance of 1 g and the calibration certificate shows a subdivision of 0.1 mg. The standard uncertainty of the sample quality m comes from two aspects:
First, the variability of the weighing is within 50 g, with a standard deviation of 0.08 mg for the variability;
Second, the uncertainty generated by the balance calibration is calculated as a standard deviation based on the expanded uncertainty given in the calibration certificate: 0.08 mg (k = 2), converted into standard deviation:
Uncertainty Introduced by Sample Volumetric and Dilution Procedures, urel(V)
The glass volumetric flasks used for sample volumetric and dilution procedures are both Class A. For sample volumetric and dilution processes, a 10 mL volumetric flask is used once, a 100 mL volumetric flask is used once and a 200 µL pipette is used once.
The acceptable difference in capacity of a 10 mL Class A volumetric flask is ±0.03 mL, converted to standard deviation based on uniform distribution:
Uncertainty in the Preparation of Standard Solution Concentration, urel(Cs)
Relative Uncertainty Introduced by the Purity of the Reference Materials, urel(P)
According to the certificate provided by the producer, the relative uncertainty introduced by the purity of the standard substance is:
urel(P) = 0.03
Uncertainty in the Preparation of the Control Stock Solution
(1) Relative standard uncertainty of 1000 µg/L standard stock solution preparation
The uncertainty of preparing the reserve solution using a 100 mL volumetric flask consists of two parts:
First, the uncertainty of the volume of the volumetric flask. According to the verification regulations for glass measuring instruments, the allowable deviation of the capacity of a 100 mL Class A volumetric flask used for the standard reserve liquid is ±0.03 mL, which is converted into the standard deviation based on uniform distribution:
The uncertainty of preparing the stock solution using a 10 mL volumetric flask includes two parts:
First, the uncertainty of the volume of the volumetric flask. According to the verification regulations for glass measuring instruments, the allowable deviation of the capacity of a 10 mL Class A volumetric flask used for the standard stock liquid is ± 0.03 mL, which is converted into the standard deviation based on uniform distribution:
The uncertainty of preparing the stock solution using a 10 mL volumetric flask consists of two parts:
First, the uncertainty of the volume of the volumetric flask. According to the verification regulations for glass measuring instruments, the allowable deviation of the capacity of a 10 mL Class A volumetric flask used for the standard stock liquid is ± 0.03 mL, which is converted into a standard deviation based on uniform distribution:
The uncertainty of preparing the stock solution using a 10 mL volumetric flask includes two parts:
First, the uncertainty of the volume of the volumetric flask. According to the verification regulations for glass measuring instruments, the allowable deviation of the capacity of a 10 mL Class A volumetric flask used for the standard stock liquid is ±0.03 mL, which is converted into a standard deviation based on uniform distribution:
The uncertainty of preparing the stock solution using a 10 mL volumetric flask includes two parts:
First, the uncertainty of the volume of the volumetric flask. According to the verification regulations for glass measuring instruments, the allowable deviation of the capacity of a 10 mL Class A volumetric flask used for the standard stock liquid is ±0.03 mL, which is converted into a standard deviation based on uniform distribution:
The conversion from uniform distribution to standard deviation is:
The uncertainty of preparing the stock solution using a 10 mL volumetric flask consists of two parts:
First, the uncertainty of the volume of the volumetric flask. According to the verification regulations for glass measuring instruments, the permissible deviation of the capacity of a 10 mL Class A volumetric flask used for the standard stock liquid is ±0.03 mL, which is converted into the standard deviation based on uniform distribution.
The uncertainty of preparing the stock solution using a 10 mL volumetric flask includes two parts:
First, the uncertainty of the volume of the volumetric flask. According to the verification regulations for glass measuring instruments, the allowable deviation of the capacity of a 10 mL Class A volumetric flask used for the standard reserve liquid is ±0.03 mL, which is converted into the standard deviation based on uniform distribution.
Uncertainty Introduced by Fitting Standard Working Curves, urel(Cs)
Take the distribution of 5 standard working solutions of different concentrations (1, 2, 5, 8, 10 μg/L) and measure it three times. The measured peak area is given in Table 1. The regression equation of the standard working curve is:
Uncertainty of Standard Curve Fit for Sibutramine Hydrochloride.

The sample was repeated 6 times and the peak area was entered into the linear regression equation to calculate the concentration of sibutramine in the sample solution, which were 4.15381, 4.25404, 3.96771, 4.03032, 4.03664, 3.98891 μg/L, respectively, and the mean concentration C sample = 4.07191 µg/L. Representative ion chromatograms of sample containing sibturamine is shown in Figure 4.

Representative ion chromatograms of sample containing sibturamine.
The standard uncertainty introduced by the least-squares fit of the standard working curve is: SA - standard deviation of the residual peak area measured with standard working solution; Sc - the sum of the squared residuals of the standard working solution concentration; n - the number of standard working solution measurements, take 15 here; P - number of sample solution measurements, take 6 here.



Therefore, the relative standard uncertainty introduced by the standard working curve fitting process is:
Uncertainty Introduced by Repeatability Measurement, urel(R)
The actual content of sibutramine in the health products obtained from 6 repeat measurements of the samples are 43.0091, 44.0468, 41.0821, 41.7304, 41.7958 and 41.3016 mg/kg, with an average value of 42.1610 mg/kg. The uncertainty introduced by the repeatability measurement is of Class A and should be evaluated using the Bessel method. This uncertainty result includes the uncertainties introduced by operator handling, instrument performance, sample uniformity, random error, etc The calculation procedure is as follows
Standard deviation of repeatability:


Synthetic Standard Uncertainty, Expanded Uncertainty and Uncertainty Results
A summary of the uncertainty components for the determination of sibutramine in foods by high-performance liquid chromatography-tandem mass spectrometry is given in Table 2.
Measurement Results of Uncertainty Components.

The composite relative standard uncertainty is calculated as follows:



Discussion
The influencing factors of uncertainty in the quantitative results were analysed, including sample weighing, sample volume determination, standard solution preparation, standard curve fitting and determination of sample repeatability. From the formula, we could see that, among the factors, the uncertainty generated by standard solution preparation made the largest contribution in the relative standard uncertainty, followed by standard curve fitting and sample repeatability. The uncertainty generated by sample weighing and sample volume determination was the lowest.
Conclusion
Uncertainty assessment is a very important part of the laboratory measurement process. This article provides a relatively objective method for assessing the uncertainty of sibutramine content based on actual detection data. By analysing and quantifying the various sources of uncertainty in detail, it helps to better understand the possible errors that may occur during the detection process. The appropriate evaluation of uncertainty can correctly assess whether the configuration of analytical instruments is appropriate, which helps to improve the reliability of detection results. In this article, the detection of sibutramine in health foods by high-performance liquid chromatography tandem mass spectrometry was investigated, the factors affecting the uncertainty were systematically analysed, and a mathematical model was developed. By analysing the measurement uncertainty components introduced in various aspects, including sample weighing, sample volume determination, standard solution preparation, standard curve fitting and sample repeatability determination, the key factors affecting the detection of sibutramine are identified, which plays an important role in improving measurement methods. In this experiment, the uncertainty generated by standard solution preparation was the highest, followed by standard curve fitting and sample repeatability. The uncertainty generated by sample weighing and sample volume determination was the lowest. In day-to-day experimental testing, it is necessary to standardize the use of equipment and personnel, and to regularly maintain and calibrate equipment to ensure the reliability and accuracy of test results. In addition to the factors discussed in this article, which believed have significant influence on the the calculation of uncertainty, other factors such as temperature or inter-day precision of the measurement, may also have an impact on the uncertainty. That is a limitation of this article.
Footnotes
Acknowledgments
This work was financially supported by Tianjin Municipal Association of Higher Vocational & Technical Education National Natural Science Foundation of China (2024-H-038).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable for this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Tianjin Municipal Association of Higher Vocational & Technical Education National Natural Science Foundation of China, (grant number 2024-H-038).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.