Abstract
Background
Carica papaya L. is a dioecious plant with leaves known for their medicinal and nutritional value due to their rich phytochemical content, including flavonoids. Phytochemical variability arising from plant maturity and sex differences may influence the bioactivity of the plant.
Objectives
This research aims to investigate the variations of flavonoids in C. papaya leaves across different maturation stages and the impact of sex differences on the accumulation of these compounds.
Methods
An ultra-high performance liquid chromatography–tandem mass spectrometry method was developed to quantify 6 flavonoids (manghaslin, clitorin, rutin, nicotiflorin, quercetin, and isorhamnetin) in the leaves simultaneously. The acquired data were further analyzed using chemometric tools.
Results
Significantly higher concentrations of flavonoids were observed in shoots and young leaves in comparison to older leaves. Furthermore, distinct variations in flavonoid composition between male and female plants were also observed, especially in young leaves, as evidenced by hierarchical clustering analysis. Further analysis of flavonoid content indicates that clitorin, nicotiflorin, and rutin could serve as chemical markers to differentiate maturation stages and sexes.
Conclusion
The study revealed significant variations in flavonoid concentrations during leaf development and between male and female plants, highlighting the importance of understanding these differences for unlocking the full potential of C. papaya leaves in medical and nutritional applications.
Keywords
Introduction
Papaya, scientifically known as Carica papaya Linn., is a fruit-bearing plant found in various tropical regions worldwide. In natural populations, papaya trees are exclusively dioecious, with plants being either male or female, as is evident by the distinct flowers borne by each sex. 1 Conversely, cultivated papaya is predominantly gynodioecious, with the coexistence of female and hermaphrodite flowers within the population, which affects fruit production, shape, and yield.2,3 Papaya fruit serves as a major tropical crop consumed globally, while the leaves are valued for their medicinal properties. They contain substantial essential nutrients and diverse phytochemicals.4,5 Among these phytochemicals, flavonoids in papaya leaves have garnered scientific interest due to their pharmacological properties, including antioxidant, anti-inflammatory, and anticancer activities.6,7
However, to harness the potential of plant leaves as herbal remedies, understanding how their maturity affects the variability of bioactive flavonoids is crucial. As regulators of plant growth and development, flavonoid biosynthesis activities are influenced by both intrinsic and extrinsic factors, resulting in a diverse array of flavonoids in plant tissue.8-10 It has been observed that younger leaves generally have higher flavonoid concentrations than older ones. For example, in Camellia sinensis, leaf maturation not only reduces moisture content but also alters phenolic profiles and diminishes flavonoid concentrations. 10 Similarly, a study on Blumea balsamifera found that immature leaves contain higher total phenolics and total flavonoid content, particularly quercetin, displaying remarkable antibacterial properties. 9 These findings underscore the significance of leaf age in the accumulation of valuable bioactive compounds, influencing their potential applications in various fields.
Furthermore, a recent study into Broussonetia papyrifera leaves suggested that male and female leaf plants may exhibit differences in flavonoid composition, possibly due to the distinct physiological roles of each sexual form. 8 The study found that flavonoids accumulated gradually with developmental time, with female plants having a higher content than male plants. These findings highlight the noteworthy effect of sex-specific variations in plant biochemistry. Hence, investigating these factors in relation to flavonoid variability is vital for the quality control of papaya leaf-based herbal medicines and dietary supplements. Likewise, the maturation of papaya leaves involves a complex interplay of biochemical changes, impacting their flavonoid compositions. These changes are subject to seasonality, the circadian cycle, environmental conditions, and other intrinsic plant attributes.11,12
Currently, there are no studies demonstrating flavonoid variations in leaf maturation of C. papaya leaves and their association with sexual differentiation. Therefore, our study aims to investigate the variations of flavonoids in C. papaya leaves throughout the 3 maturation stages (shoot, young, and old leaves) and the impact of sexual variety (plants with female and male flowers) on flavonoid accumulation. Ultra-high-performance liquid chromatography (UHPLC) coupled with tandem mass spectrometry (MS/MS)-based targeted metabolomics was employed to evaluate the flavonoid profiles, that is, clitorin, manghaslin, rutin, nicotiflorin, quercetin, and isorhamnetin, of 90 C. papaya leaf extracts.
Results and Discussion
Method Performance
The total ion chromatogram and multiple reaction monitoring (MRM) chromatogram of the mixed standards show good chromatographic separation among all flavonoids (Supplemental Figure S1). A rapid 10 min gradient was developed to separate the 6 flavonol-type flavonoids, which are structurally similar to each other and an internal standard (IS), cynaroside. The chemical structures of these flavonols are depicted in Supplemental Figure S2. The MRM method demonstrated excellent linearity, with correlation coefficients (R2) ranging from 0.9958 to 0.9987 for all compounds. Limit of detection (LOD) and limit of quantification (LOQ) were achieved, ranging from 0.001 to 0.015 µg/mL and from 0.004 to 0.019 µg/mL, respectively, showing that the method is sufficiently sensitive (Supplemental Table S1). The intraday and interday precision, ranging from 1.19% to 4.79%, indicated high reproducibility. In a single-day analysis, good accuracy of the method is demonstrated by the average recovery of the spiked reference standards, which ranged from 95.33% to 98.88%, demonstrated the good accuracy of the method in a single-day analysis. However, during 3 consecutive days of analysis, recoveries were lower, ranging from 94.13% to 96.34% for certain samples, indicating potential degradation within the samples at 4 °C storage. Hence, all samples were prepared freshly, and the analyses were completed within the same day. For high throughput analysis that requires longer run time, samples need to be stored at lower temperatures. The precision and accuracy analysis for both intraday and interday measurements are presented in Supplemental Table S2. This validated analytical method was employed for the simultaneous quantification of all flavonoids in C. papaya leaf extracts.
Flavonoid Content in C. papaya Leaves
A total of 90 extracts, encompassing shoot, young, and old leaves, as well as female and male varieties were analyzed by an external standard calibration method. It revealed significant variations in the concentrations of 6 flavonoids, as detailed in Supplemental Table S3. The selections of these flavonoids were based on their potency as antioxidants and their suitability as chemical markers for quality control purposes.13-16 Overall, Supplemental Table S3 presents concentrations of clitorin (3710.8 to 17 086.1 µg/g), manghaslin (2052.4 to 8725.7 µg/g), rutin (917.3 to 4889.0 µg/g), nicotiflorin (305.2 to 4837.5 µg/g), quercetin (5.2 to 56.3 µg/g), and isorhamnetin (2.1 to 8.7 µg/g). A comparative analysis with previous reports highlights considerable differences in compound concentrations.17-19 Nevertheless, these differences are still within the range of concentrations observed in the current study. A study using leaves sourced from Kalimantan, Indonesia reported the concentrations of clitorin at 7230.0 µg/g, manghaslin at 3110.0 µg/g, rutin at 970.0 µg/g, and nicotiflorin at 810.0 µg/g. 19 Another study found rutin concentrations ranging from 1128.0 to 1242.0 µg/g in leaves at different maturity stages. 18 Although quercetin was below the quantification limit in the aforementioned studies, Canini et al 17 and Nugroho et al 19 detected the presence of quercetin in leaf samples from West Cameroon (Africa) at a concentration of 40.00 µg/g by sample derivatization and gas chromatography–mass spectrometry analysis. A previous high-resolution mass spectrometry study suggested the presence of isorhamnetin in the freeze-dried leaf juice extract of papaya, 4 but there are no published reports that have quantified the levels of this compound in papaya leaves. In our case, the highly sensitive targeted mass spectrometry method of MRM allowed us to measure isorhamnetin, even if it was minuscule in comparison to other flavonoids.
The biosynthesis of these flavonoids in plants involves the phenylpropanoid pathway, where phenylalanine is converted to 4-coumaroyl-CoA, which then enters the flavonoid pathway. 20 The flavonoid pathway leads to the formation of flavonoid aglycones such as quercetin, kaempferol, and isorhamnetin. Quercetin and kaempferol are synthesized from dihydroflavonols by the action of flavonol synthase. Isorhamnetin is a methylated derivative of quercetin, synthesized by the action of O-methyltransferase. 20 The aglycones may undergo further modifications into various glycoside forms. For instance, manghaslin and rutin are 3-O-glycosylated derivatives of quercetin, while clitorin and nicotiflorin are 3-O-glycosylated derivatives of kaempferol. 20 The biosynthesis of these flavonoids involves various enzymes, including phenylalanine ammonia-lyase, cinnamate 4-hydroxylase, 4-coumarate-CoA ligase, chalcone synthase, chalcone isomerase, flavanone 3-hydroxylase, flavonoid 3′-hydroxylase, dihydroflavonol 4-reductase, flavonol synthase, and O-methyltransferase. 15 These flavonol glycosides and aglycones could serve various roles in the biology of C. papaya, including reproduction, ultraviolet (UV) protection, regulating internal plant cell functions, and supporting the plant's immune system by acting as free radical scavengers. 16
In an effort to produce papaya fruit that is suitable for local tastes and environmental conditions, breeding initiatives for this economically viable crop have produced a multitude of varieties and cultivars. The var. Sekaki and var. Eksotika, for instance, are the 2 most important varieties in Malaysia due to their high fruit yield and esthetic appeal. 21 An untargeted metabolomics analysis of these varieties using fruits sampled at different ripening stages showed that var. Eksotika and var. Sekaki may be differentiated by their organic acid and amino acid profile. The metabolite profiles of the leaves, an underutilized plant component, have not, however, been compared. Our unpublished data from earlier investigations indicate that the phenolic and flavonol glycoside fingerprints are similar across different sexes (Supplemental Figure S3). Still, this may not comprehensively reflect the similarity of individual compounds in terms of quantitative measurements. At the very least, evidence from Nisa et al 22 showed homogeneous total flavonoids across 5 Indonesian cultivars, ranging from 30 to 40 µg/g. In other quantitative studies, most reports do not authenticate down to the specific varietal level.17-19 Apart from genetic differences, we acknowledged that variations in the production and accumulation of flavonoids may be attributed to various factors such as geographical location, instrumental limitation, extraction method, maturity, or sex differences in the tree, emphasizing the need for standardized criteria to facilitate the development of targeted applications. In this study, we opted to focus on Sekaki, a variety which we could source reliably from our research facility which allowed us to control variations related to plant source.
Variation in Flavonoid Concentrations Across Maturation Stages
In relation to leaf maturity, Figure 1 reveals 2 distinct trends of temporal variation in the individual flavonoid concentrations. Notably, the concentrations of clitorin, rutin, and nicotiflorin exhibited an inverted V-shaped pattern, reaching their peak in young leaves. After reaching the peak, there is a significant decrease (p < .05) in clitorin and rutin concentrations. This phenomenon is potentially associated with elevated biosynthetic activities characteristic of the early growth phase. 23 With the aging of leaves, there is a gradual decline in metabolic rates, resulting in the lowest flavonoid concentrations in old leaves. 24 On the contrary, manghaslin, quercetin, and isorhamnetin demonstrated significant reduction (p < .05) in concentrations when leaves grew, marking a divergent trend from the former group of flavonoids. These trends imply a complex nature in flavonoid biosynthesis and accumulations during different maturation phases.
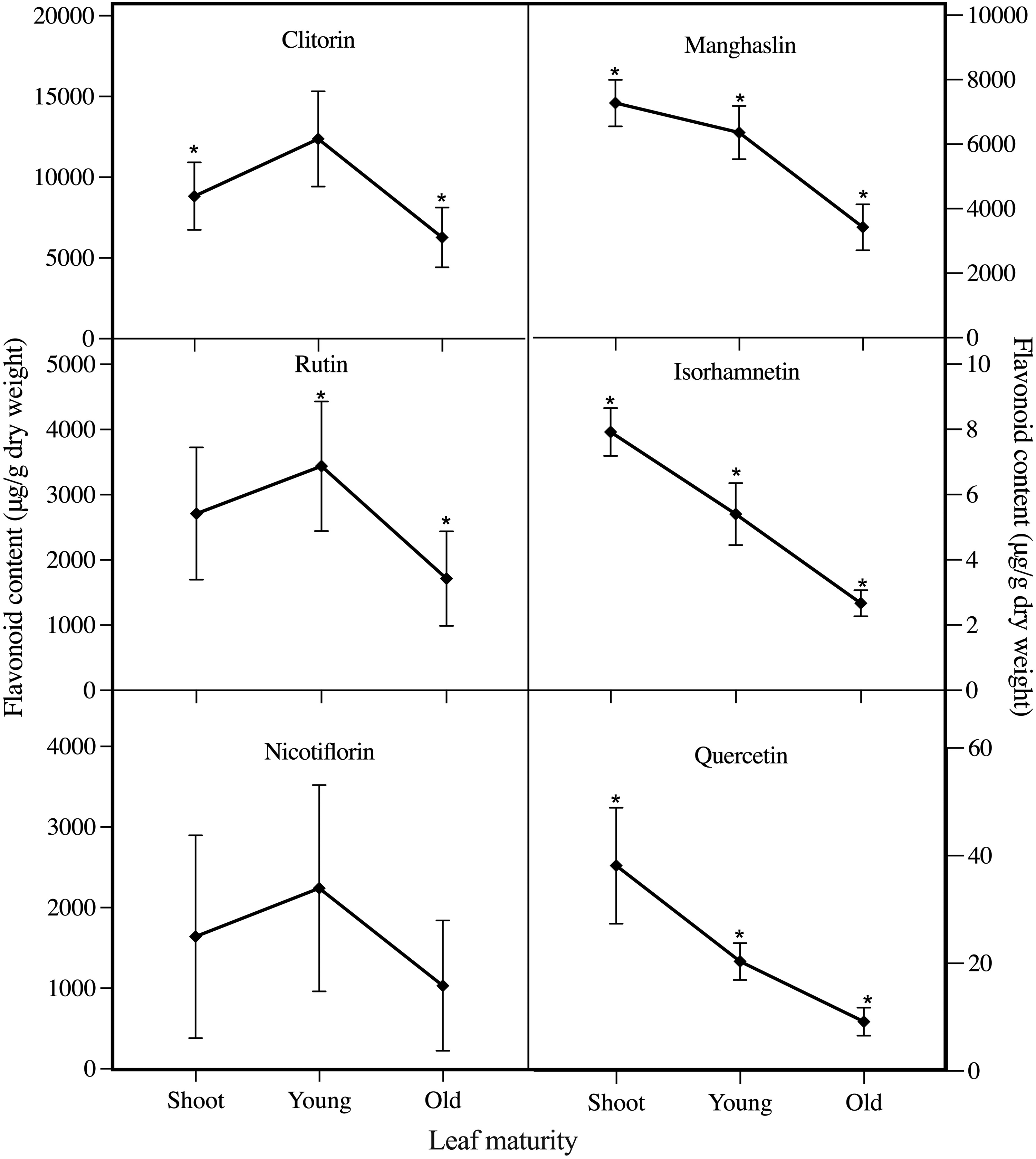
Concentration changes of various flavonoids in C. papaya leaves as a function of leaf maturity levels. The asterisk (*) indicates a significant difference within the developmental stage, according to the ANOVA and Tukey HSD test (p < .05).
Principal component analysis (PCA) was utilized to simplify the complex associations among different age groups of leaves and their respective flavonoid compositions. Figure 2 further elucidates these findings by projecting the data onto the first 2 principal components (PCs), which collectively explain ∼87% of the total variance in the dataset. The score plot reveals distinct clusters of leaf samples based on their flavonoid profiles, thereby highlighting the inherent variability in flavonoid content across different developmental stages. PC1 effectively spans the old leaf samples in the negative region. PC2, on the other hand, distinguishes young leaves from shoot leaves, and this distinction is contributed by the elevated concentrations of clitorin, rutin, and nicotiflorin in young leaves, as well as manghaslin, quercetin, and isorhamnetin in shoot leaves.

Principal component analysis (PCA) biplot. The clustering of different maturation stages was observed based on 6 flavonoids.
The accumulation of certain flavonoids in different growth stages suggests that these compounds have specific roles in the development and stress response of the plant. 25 Nicotiflorin, rutin, and clitorin, which are abundant in young leaves, are glycosylated flavonoids. This may indicate that these compounds are involved in the growth and development of the plant, as glycosylated compounds are often associated with cellular growth and division. 13 On the other hand, the elevated presence of manghaslin, quercetin, and isorhamnetin in shoot leaves is likely attributed to their polyphenolic nature and specific arrangements of hydroxyl and methoxy groups on the flavonoid backbone. Quercetin and isorhamnetin are glycoside-free molecules, suggesting that they may have different functions than the glycosylated flavonoids found in young leaves. The absence of glycoside molecules may make these compounds more hydrophobic, allowing them to interact with cell membranes and potentially contribute to the structural integrity of the plant. 26 The distinct chemical structures and biosynthetic pathways of these flavonoids likely reflect their specialized functions in different stages of leaf development. The dynamic nature of plant secondary metabolism allows for the production of a diverse array of compounds that can respond to the changing needs of the plant throughout its development.27,28
The observations of the flavonoid content in C. papaya leaves agree with previous research by Gogna et al, 18 which reported higher concentrations of secondary metabolites, including kaempferol and rutin, in young papaya leaves compared to older leaves. The distinct flavonoid profiles associated with different maturation stages underscore their crucial roles in leaf development and maturation. Flavonoids, synthesized in various plant parts, play key roles in regulating cell growth, attracting pollinators, and providing defense against environmental stresses. 14 For instance, flavonols, a subclass of flavonoids, significantly impact leaf physiology and development, including processes such as gas exchange through stomata. 25 Additionally, flavonoids participate in signaling pathways that govern cell growth and differentiation, vital for leaf development. 29 The accumulation of flavonoids in leaves has also been linked to defense mechanisms against UV filtration and symbiotic nitrogen fixation, crucial for maintaining leaf health and resilience. 30 Relevant studies conducted on different plants, such as Cabernet sauvignon, Manihot esculenta, and Brassica napus, highlight the dynamic nature of flavonoid profiles during leaf growth stages, further validating the findings of this study.31-33
Association between leaf maturity and sexual differentiation
Without accounting for the plant's maturity stages, the difference in flavonoid content of male and female plants is summarized in Table 1. The results show that male samples exhibited significantly higher concentrations (p < .05) for all tested flavonoids except for manghaslin and quercetin. The higher concentrations may contribute to specific physiological roles related to male reproductive structures. The most significant difference is shown in the level of nicotiflorin, with a fold change of nearly 2.18 and a significantly higher concentration in male plants. Clitorin concentrations are ∼1.47 times higher in male plants compared to females and rutin concentrations show a similar trend, being ∼1.44 times higher in males. Meanwhile, there are only marginal differences in isorhamnetin, manghaslin, and quercetin between male and female plants, with fold changes slightly above or close to 1. The elevated levels of flavonoids such as clitorin, nicotiflorin, and rutin in male plants, suggest they could be chemical markers distinguishing between genders, irrespective of leaf maturity.
Comparative Analysis of Flavonoid Concentrations in Male and Female Plant Samples Disregarding Maturation Stages. Concentrations are Expressed in μg/g Dry Weight.

*t-test indicates no significant difference between male and female groups across all plant samples (p > .05).
When comparing flavonoid levels across different sex varieties at various stages of maturity, the bar charts further reveal a significantly higher manghaslin level in male plants during the shoot phase (Figure 3). In contrast, quercetin content remains consistent across all maturity groups, with no significant difference between sexes (p > .05). This highlights the robustness and uniformity of quercetin levels irrespective of tree sexual dimorphism. For clitorin and rutin, the significant difference (p < .05) was particularly evident during the shoot and young leaf phase in the male compared to the female variety. Meanwhile, the levels of nicotiflorin and isorhamnetin were significantly higher in males only during the shoot phase. Conversely, in old leaves, no significant differences (p > .05) in all flavonoid levels across both male and female varieties. This observation indicates potential regulatory shifts in flavonoid metabolism as leaves mature. 34
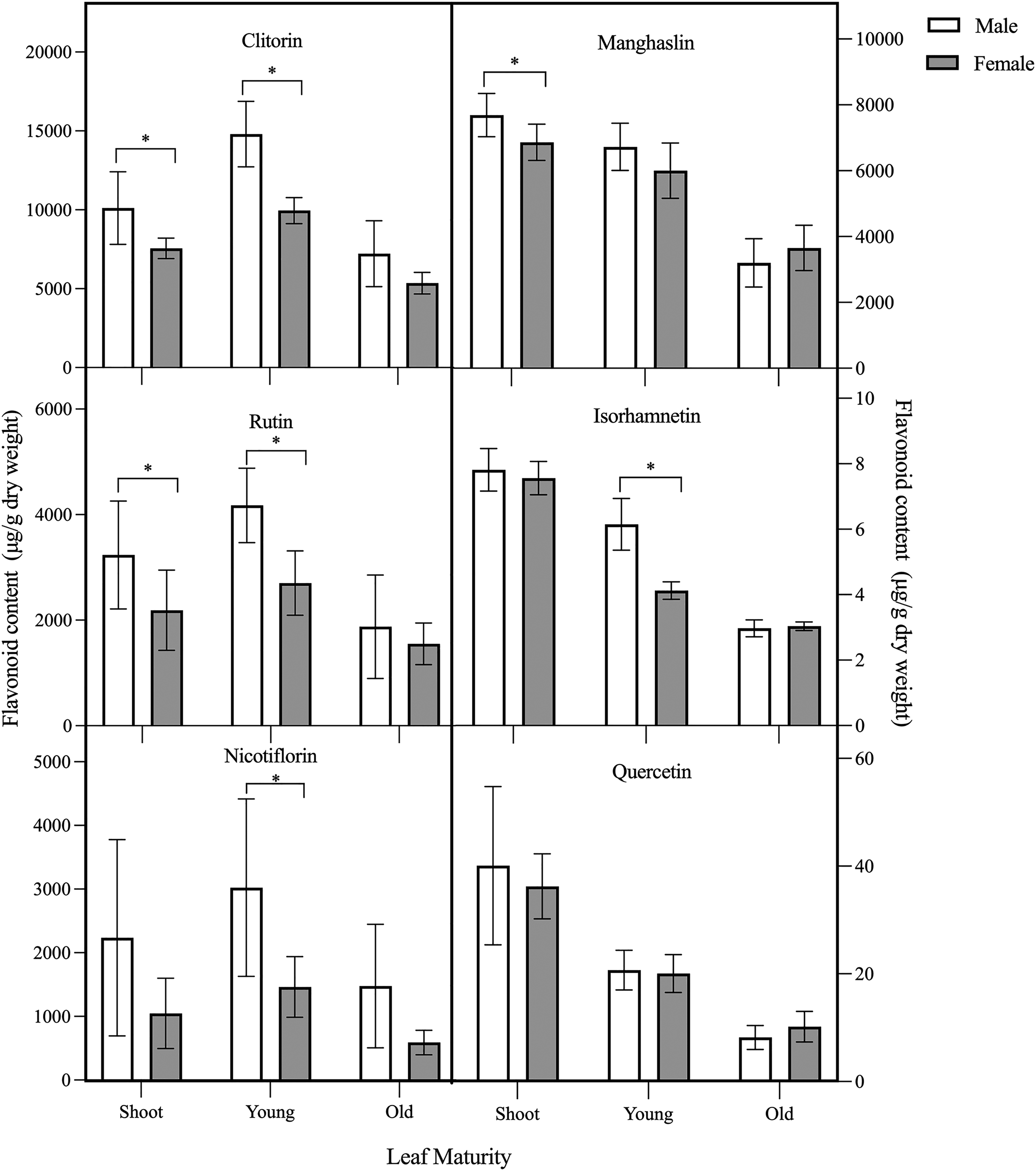
Flavonoid content in different sexes of C. papaya across shoots, young leaves, and old leaves. Each data point represents the mean of 5 replicates (mean ± SD). Asterisk (*) indicates a significant difference between sexes within the developmental stage, according to the ANOVA and Tukey HSD test (p < .05).
Hierarchical clustering analysis (HCA) was performed to visualize the similarity or dissimilarity between male and female groups based on their flavonoid profiles and the associations between the 6 flavonols (Figure 4). Based on the dendrogram generated by HCA of the 3 maturation phases, differentiation between male and female leaves is only evident during the phase of young leaves, with male plants showing overall higher flavonols (indicated by red and grey). For the shoot and old leaf groups, 2 male populations (Figure 4, see dotted square) appeared to cluster separately from the other male populations, with higher levels of nicotiflorin, manghaslin, quercetin, and isorhamnetin in both shoot and old leaves but different trends of clitorin and rutin levels between these 2 maturity phases. The same applies to the shoot and old leaves of 1 female population. The shoot and old leaf groups exhibited similar correlations among flavonols, resulting in 2 major flavonoid clusters. This clustering suggests that certain flavonoids tend to co-occur in similar proportions across these groups. Specifically, clitorin and rutin were found in the same cluster, indicating a strong correlation or similar expression patterns between these 2 flavonoids. In contrast, the remaining flavonoids formed a separate cluster. As seen in other investigations, the tendency of flavonoids clustering does not seem to be explained by the position of the hydroxyl group in the aglycone or the degree of glycosylation.31,33 From the HCA, identifying specific flavonoids that contributed to sex differentiation across shoot and old leaves proved to be challenging, as the pattern was unclear in leaves at all maturation stages. Inadequate meta-data gathered throughout plant collections prevent us from explaining these anomalies. Factors such as sunlight exposure, when samples were planted, could influence the data outcomes. 35 Nonetheless, the cause is not related to instrument stability, as appropriate quality control procedures were followed.

Heat-map and hierarchical clustering analysis (HCA) analyses of flavonoid levels in different sexes of C. papaya throughout various stages of leaf maturation.
The observation of sex-based differences in flavonoid content among dioecious plants is well documented.8,36 However, flavonoid concentrations are not consistently higher in male plants across species, potentially due to variations in gene expression within the flavonoid biosynthesis pathway. Previous studies suggest that overexpression of specific genes (CHS2, CHS7, CHI, and DFR) is associated with higher flavonoid accumulation in female plants, including garbanzol, naringenin 7-O-glucoside, hesperetin, and kaempferol. 8 Besides genetic factors, hormonal differences could potentially contribute to the variation of flavonoid biosynthesis. Hormonal factors such as auxin and cytokinin are known to regulate secondary metabolites, including flavonoids in plants. Auxin and cytokinin act synergistically or antagonistically to regulate development and alter growth in response to environmental cues. 37
Furthermore, differences in flavonoid accumulation between sexes may be impacted by numerous additional factors. This study recognizes limitations, such as potential environmental influences, not including all of the flavonoid compounds, and the exclusion of hermaphrodite plants, which are commonly found in commercial plantations. This research provides practical recommendations for refining harvesting practices to ensure the quality of C. papaya leaf extract for therapeutic use. Despite mature leaves being larger with potentially higher extraction yields than young leaves and shoot leaves, it is suggested to harvest medium-sized leaves from the middle level of the tree to attain optimal total flavonoid concentrations. Moreover, we propose a preference for male plants to maximize flavonoid concentrations compared to female plants. Differences in sex forms and maturation stages may also impact other metabolites, including the alkaloids in C. papaya. There is an opportunity to enhance this research by employing untargeted metabolomics to comprehensively analyze all metabolites.
Conclusion
This study employed a validated UHPLC-MS/MS method combined with multivariate data analysis to investigate the association between 6 flavonoid contents across different maturation stages and sexes of C. papaya leaves. Results indicate that younger leaves generally possess the highest flavonoid levels compared to shoots and mature leaves. Additionally, significant differences in flavonoid composition based on the sex of the trees, with male trees showing higher flavonoid levels, particularly in shoots and young leaves. Further analysis reveals that clitorin, nicotiflorin, and rutin exhibit distinct differences between male and female samples, as well as across various maturation stages, despite the initial challenges posed by the unclear patterns observed. These flavonoids could serve as chemical markers for distinguishing both maturation stages and sex differentiation in the leaves of C. papaya trees. The findings may contribute to understanding the variability of bioactive compounds in C. papaya leaves and suggest the potential for optimizing flavonoid content through selective harvesting methods. Furthermore, the study suggests expanding research to explore alkaloid composition, aiming to develop standardized extracts with potential therapeutic and dietary benefits.
Materials and Methods
Reagent and Chemicals
All analytical solvents were obtained from Merck (Darmstadt, Germany). Reference materials, namely rutin, nicotiflorin, and isorhamnetin, were purchased from Extrasynthese (Genay, France), while quercetin and cynaroside were obtained from Sigma-Aldrich (Missouri, United States). Manghaslin and clitorin, isolated from C. papaya leaf extracts, were characterized using mass spectrometry and nuclear magnetic resonance before undergoing quantitative analysis, as described in our previous work. 38
Sample Materials
Fresh green leaves of C. papaya L. var. Sekaki were sampled from the herbal garden at the Institute for Medical Research Malaysia (3°10′7.3″N, 101°42′1.4″E). A voucher specimen was deposited at the Forest Research Institute Malaysia with the reference Voucher No. 007/10. The plants were cultivated for research purposes in our facility, with the duration from planting to harvesting at 8 months old across all samples. All plants were cultivated in a controlled environment to minimize variability from extrinsic factors. The growth stages were classified as follows: shoot leaves (small light green leaves budding at the tip of the trunk, with leaf size ranging from 150 to 220 mm), young leaves (medium-sized dark green leaves located at the middle level—third levels from the top with sizes ranging from 250 to 350 mm), and old leaves (faded-green leaves with a slight yellowish tint, growing lower down from the 7th level from the top, measuring between 350 and 450 mm). Supplemental Figure S4 provides the representative leaf characteristics for each growth stage. Male and female trees were differentiated by observing flower shapes and fruit production, following the description from The World Flora Online. 39 The differences between the male and female papaya trees are shown in Supplemental Figure S5. For this study, 10 trees were carefully selected for leaf collection, comprising 5 male and 5 female trees at 3 different growth stages. To ensure data reliability, only healthy leaves free from ring spots were considered. Three leaves were collected from each tree, resulting in 90 samples for the extraction and UHPLC-MS/MS analysis.
Sample Preparation
The leaf samples were initially washed with tap water to remove surface contaminants, such as wax, soil, and fertilizers, followed by a further rinse with reverse osmosis water. They were then sectioned into 2 mm × 2 mm pieces and lyophilized using an Alpha 1-2 LDPlus freeze dryer (Martin Christ, Osterode am Harz, Germany). The resulting lyophilized samples were then powdered, homogenized, and stored at −40 °C before further analysis. For the extraction of flavonoids, the lyophilized leaves were macerated in 85% methanol (5 mg/mL) and continuously sonicated at room temperature for 30 min. Subsequently, the extract was centrifuged at 10,000 rpm for 10 min, and the supernatants were collected. Prior to solid-phase extraction, 1.0 mg/mL cynaroside (IS) was added to the supernatants at a 1:100 ratio (v/v). This process entailed passing the sample solution through a 40–60 μm, 70 Å, HyperSep C18 column (Thermo Fisher Scientific, Massachusetts, United States) under a vacuum manifold at a constant pressure of ∼22 mm Hg. The eluate was collected and subsequently dried using a centrifugal vacuum concentrator (MiVac Quattro concentrator, Genevac Ltd., Ipswich, United Kingdom) at 35 °C for 120 min. The dried eluates were reconstituted with 1 mL of methanol for UHPLC-MS/MS analysis.
UHPLC-MS/MS Analysis
Samples were analyzed using an Agilent 1290 Infinity LC, coupled to an Agilent 6490 triple quadrupole system via an electrospray ionization (ESI) source (Agilent Technologies Inc., California, United States). Data acquisition and processing were executed using MassHunter Workstation software (version B.06.00). With an injection volume of 5 μL, chromatographic separation was achieved on an Agilent ZORBAX Eclipse Plus C18 column (2.1 mm × 50 mm, 1.8 µm) at 40 °C. The gradient elution was conducted at a constant flow rate of 0.3 mL/min using a mobile phase comprised of a mixture of 0.1% formic acid in ultra-pure water (A) and 0.1% formic acid in acetonitrile (B). The elution program was structured as follows: 0 min, 5% B; 0.5 min, 10% B; 8 min, 45% B; 9 min, 100% B; and 10 min, 100% B. Prior to the subsequent injection, the mobile phase was reset to 5% B (95% A) over 2 min, followed by a 3 min column re-equilibration period. Detection and quantification were performed in MRM mode using negative ionization. The ESI conditions were set as follows: N2 drying gas temperature at 200 °C with a flow rate of 11 L/min, nebulizer pressure at 45 psi, sheath gas temperature at 200 °C, and a capillary voltage of 4 kV. Analyte-specific MRM parameters, such as collision energy and cone voltage, were optimized by infusing an appropriate standard solution to ensure the most specific, abundant, and stable ion transitions. A summary of retention times and MRM transitions for each analyte is provided in Table 2. The analytical performances, including linearity, LOD, LOQ, precision, and accuracy, were established in accordance with the guidelines outlined by the International Conference on Harmonization. 40
MRM Parameters for Targeted Metabolites and Internal Standard.

Abbreviations: MRM, multiple reaction monitoring; IS, internal standard.
Data Analysis
Statistical analysis was performed using JMP Pro 16 (SAS Institute) data analysis software. An analysis of variance (ANOVA) and Tukey's honestly significant difference (HSD) test were employed to test the difference in flavonoid concentrations across the designed groups, with a significance level set at 0.05. To further explore the underlying pattern in the flavonoid dataset, PCA and HCA were utilized for dimension reduction. The PCs were derived from the correlation matrix, and the HCA dendrogram was generated by Ward's linkage method based on squared Euclidean distance. 41
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241260175 - Supplemental material for Flavonoid Variability in Carica papaya L. var. Sekaki Leaf Maturation and Its Association With Sexual Differentiation Using Targeted Metabolomics
Supplemental material, sj-docx-1-npx-10.1177_1934578X241260175 for Flavonoid Variability in Carica papaya L. var. Sekaki Leaf Maturation and Its Association With Sexual Differentiation Using Targeted Metabolomics by Norazlan Mohmad Misnan, Adlin Afzan, Maizatul Hasyima Omar and Kah Hin Low in Natural Product Communications
Footnotes
Acknowledgments
The authors would like to thank the Director-General of Health, Ministry of Health, Malaysia, and the Director of the Institute for Medical Research for permitting the publication of this paper. NMM also acknowledges the Ministry of Health Malaysia for funding postgraduate studies in the Department of Chemistry, Universiti Malaya, through the Educational Scholarship for Master of Science.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable to this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Ministry of Health Malaysia (NMRR-14-569-21235).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
