Abstract
Purpose
Evaluate the clinical efficacy of “Oriza tablet” (OT) for patients with neurologic bladder or overactive bladder affected by xerostomia due to receiving antimuscarinic drugs.
Material and methods
This was a randomized clinical trial. Twenty-eight patients (median age = 64 years) with xerostomia were divided into two groups: OT (intervention group) and placebo (control) group. Both groups comprised 14 patients each.
Objective
We assessed severity using a questionnaire for the subjective evaluation of xerostomia. Objective evaluation was undertaken for the stimulated salivary flow rate (SSFR). Xerostomia evaluation took place before treatment (baseline) as well as 2 weeks and 4 weeks after treatment.
Results
The xerostomia score decreased significantly (p < 0.05) in the OT group compared to the placebo group, with scores of 2.75 versus 4.4 at 2 weeks and 2.19 versus 4.13 at 4 weeks, respectively. The SSFR increased significantly (p < 0.05) in the OT group compared to the placebo group, with SSFR of 2 versus 1.2 at 2 weeks and 1.85 versus 1.1 at 4 weeks, respectively. Throughout the trial, no patient discontinued taking antimuscarinic medications. There were no adverse effects noted.
Conclusion
Continuous daily use of OT for 4 weeks reduced xerostomia symptoms and increased saliva production in patients suffering from neurologic bladder or overactive bladder who received antimuscarinic medications.
Clinical trial registration
The study was registered with The Thai Clinical Trials Registry (TCTR). ID: TCTR20240715015.
Introduction
Neurogenic bladder (NB; also known as “neurogenic lower urinary tract dysfunction”) refers to loss of voluntary control of the bladder. NB can be secondary to injury to the central nervous system or neurological diseases. NB can present with incontinence and incomplete emptying of the bladder. However, the major concerns for patients with NB are recurrent infection of the urinary tract (affects 29.2%–36.4% of cases), urosepsis (2.6%–4.7%), urinary retention (9.0%–14.0%), and hydronephrosis, which leads to renal failure (0.8%–2.2%) because of high-intravesical pressure. 1 These conditions increase the risk of death.
Overactive bladder (OAB) is a syndrome of urinary urgency, often with urinary frequency and nocturia, in the absence of a pathologic cause. The prevalence increases with age. In adults, OAB prevalence ranges from 7% to 27% in men and 9% to 43% in women. OAB symptoms have a significant negative impact on quality of life (QoL), such as significantly higher levels of anxiety and depression, reduced general health status, and worse sleep quality.2,3
Treatment of NB and OAB is behavioral therapy, such as bladder-training strategies, pelvic-floor physiotherapy, fluid management, and medications. 4 The latter include antimuscarinic drugs, which block acetylcholine from binding to muscarinic receptors to reduce bladder spasms and urinary incontinence. Muscarinic receptors have five subtypes (M1-5), which are distributed in the detrusor muscle, salivary glands, gastrointestinal tract, brain, and heart. 5 The most common side effects of antimuscarinic drugs are dry mouth (xerostomia) and constipation. Other side effects of this class of medication include tachycardia, mental confusion, and short-term memory loss.6,7
Xerostomia is a major factor affecting patient compliance for taking antimuscarinic drugs. The complications of xerostomia can lead to speech problems, taste disorders, chewing difficulties, swallowing difficulties, and ill-fitting dentures. Consequently, these complications lead to a poor QoL and increase the risk of developing infectious oral diseases such as caries, oral mucositis, periodontitis, and oral candidiasis.8–10 Treatment for xerostomia includes systemic therapies (eg, cholinergic agonists), topical interventions (eg, saliva stimulants (sialogogue) and saliva substitutes), and non-pharmacologic interventions (eg, acupuncture and electrostimulation).11–13
To improve patient compliance and avoid the side effects of xerostomia, an oral sialogogue, “Oriza tablet” (OT), was developed by cross-department collaboration from Dentistry, Pharmacology, and Medicine within Mahidol University (Bangkok, Thailand) and approved by the Food and Drug Administration of Thailand. Oryza sativa L., also known as black rice, is the source of OT. This rice species contains tocopherols and anthocyanins. These compounds have several biological effects, including antioxidant properties, antineoplastic properties, and anti-inflammatory reactions. Anthocyanins from Oryza sativa L. inhibited several regulatory proteins and prevented 5-fluorouracil from activating NF-κB, resulting in antiproliferative and antimicrobial effects, including restoring healthy salivary gland cell proliferation.14–17
Here, we investigated the efficacy of OT in improving xerostomia in patients with NB or OAB who received antimuscarinic drugs.
Methods
Participants
Patients were recruited from the outpatient Department of Urology within Ramathibodi Hospital (Bangkok, Thailand).
Inclusion Criteria
The inclusion criteria were: (i) age >18 years; (ii) diagnosed with NB or OAB; (iii) receiving antimuscarinic medications (oxybutinin, solifenacin, trospium, tolterodine, imidafenacin, flavoxate); (iv) having xerostomia.
Exclusion Criteria
The exclusion criteria were: (i) a history of radiotherapy to the head or neck; (ii) surgery of the salivary glands; (iii) suffering from Sjögren's syndrome, scleroderma, sarcoidosis, rheumatoid arthritis, polyarteritis nodosa, systemic sclerosis, lupus erythematosus, autoimmune disease, or infection by the hepatitis C virus or human immunodeficiency virus; (iv) had received treatment for xerostomia.
Calculation of Sample Size
The sample size was calculated using different variables from a double-blind, randomized clinical trial 13 that met the same objective. Differences in the xerostomia score were analyzed using the two-sample means test. Using mean of 1 = 36.4, mean of 2 = 29.1, standard deviation = 7.1, alpha = 0.05, and power = 0.80, we suggested that our study needed 32 participants, who would be divided into two groups of 16. Participants underwent four-block randomization using Stata 14.1 (which is licensed to the Section for Clinical Epidemiology and Biostatistics in Ramathibodi Hospital).
Ethical Approval of the Study Protocol
The study protocol was approved by Ethics Review Board of Mahidol University. The study protocol was in full compliance with international guidelines for human research, such as the Declaration of Helsinki 1964 and its later amendments, Belmont Report, Council for International Organizations and Medical Sciences guidelines, and the International Conference on Harmonization in Good Clinical Practice. All participants provided written informed consent before data collection.
Study Design
This was a prospective, single-blind, randomized clinical trial. To investigate the possible properties of “Oriza tablet” (OT), a pre-post-treatment design was used. Data were collected from June 2021 to December 2022. All participants who met the inclusion criteria provided written informed consent at the outpatient department. We randomly divided the patients into an intervention (OT) group and a placebo (control) group by using a concealed envelope. Patients sucked the medication (OT or placebo tablet) three times per day before a meal and once at bedtime (ie, four times a day). The signs and symptoms of xerostomia were assessed using the xerostomia score and stimulated salivary flow rate (SSFR) after taking medicine for 2 and 4 weeks, respectively, and compared to baseline data.
Tablets
Each packet of OT contained 10 tablets. The medication was prepared by Herbtec International Company in Nonthaburi, Thailand. Each OT had a size of 2 cm × 0.8 cm. Each packet had a date of manufacture and an expiration date. Both OT and placebo were comparable in size and shape.
Measurements
The primary outcome measure was the xerostomia score. The secondary outcome measures were the SSFR, adverse effects, and compliance with taking antimuscarinic drugs. The xerostomia score was calculated using a questionnaire containing eight questions about the severity of xerostomia symptoms in a scale ranging from 0 (“not troublesome”) to 10 (“most troublesome”) (Table 1). 18 The xerostomia score at baseline was collected at the outpatient department concomitant with the measurement of the SSFR. The xerostomia score 2 weeks and 4 weeks after treatment was collected by telephone. A SSFR was “low” at values between 0.7 mL/min and 1 mL/min. We assessed the SSFR before and after treatment using the spitting method. Patients were prohibited from eating and drinking for ≥60 min before saliva collection. They chewed a paraffin cube for 30 s, then spit into a tube for 5 min, after which the saliva volume was measured. The spitting tubes were 50 mL and 15 mL in size. Participants spat into the larger tube, and then we transferred the saliva to the smaller tube for precise measurement.19,20 The collection and measurement of saliva were made by patients under the instruction of a dental assistant before treatment at the outpatient department. After treatment, saliva collection was made by the participant at home based on an instruction note. Information was collected at the outpatient department before treatment. Post-treatment information was collected by telephone 2 weeks and 4 weeks after treatment. Adverse events were reported.
Subjective Xerostomia Questionnaires (Severity Score 0–10).

Statistical Analyses
The Pearson chi-square test and Fisher's exact test were used to ascertain differences in categorical data. The Wilcoxon rank-sum test was employed to find differences between two groups of continuous data. Statistical analyses were undertaken using SPSS 20.0 (IBM, Armonk, NY, USA). Differences were considered statistically significant if p < 0.05.
Results
Characteristics of Participants at Baseline
Initially, 29 patients participated in our study. Finally, data from 28 patients were obtained. One patient withdrew before starting treatment due to contracting coronavirus disease 2019 (COVID-19). The demographic data of patients are shown in Table 2. Nineteen patients (67.86%) were women (median age = 64 years). Twenty patients (71.43%) had OAB, and eight patients (28.57%) had NB. A single antimuscarinic drug was used in 25 patients (89.28%) and the remainder of patients used a combination of two antimuscarinic drugs.
Demographic Data.

*The Pearson chi-square test, **Fisher's exact test and ***The Wilcoxon rank-sum test.
Xerostomia Score
There was no significant difference in the xerostomia score for all questions at baseline (Table 3). A significant decrease in the xerostomia score was noted 2 weeks after treatment regarding DRYTHR, DRYTNG, and LVLTHR (p < 0.05 for all) and 4 weeks after treatment with respect to DIFSPK, DRYMOU, DRYTHR, DRYLIP, and LVLTHR (p < 0.05 for all) compared with that in the placebo group (Table 3). Overall, the xerostomia score revealed a significant improvement in xerostomia symptoms (p < 0.05) in OT compared to placebo, with a score of 2.75 versus 4.4 at 2 weeks and 2.19 versus 4.13 at 4 weeks, respectively. (Figure 1).
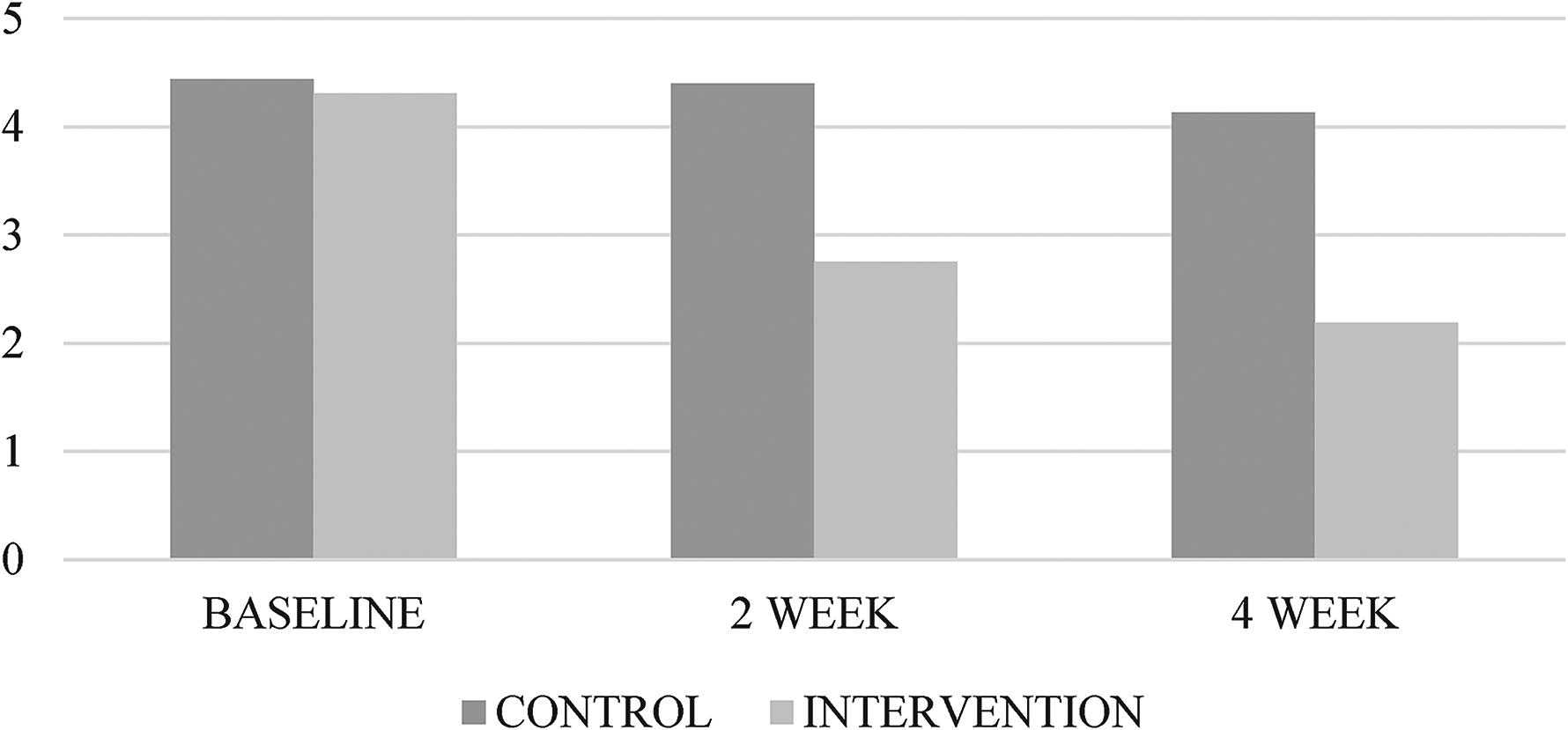
Overall xerostomia scores.
Subjective Xerostomia Scores and Stimulated Salivary Flow Rate.

SSFR
Before treatment initiation, the SSFR between the OT group and the placebo group was not significantly different. The SSFR 2 weeks and 4 weeks after using OT (intervention group) and placebo (control group) is shown in Table 3 and Figure 2. There was a significant increase (p < 0.05) in the SSFR in OT compared with the placebo group, with an SSFR of 2 versus 1.2 at 2 weeks and 1.85 versus 1.1 at 4 weeks, respectively.

Stimulated salivary flow rate (ml/min).
Adverse Events and Comments from Participants
There were no adverse effects noted. One patient complained that the tablet was too large and dissolved too slowly. One patient suggested adding flavoring to the medication. In 28 patients, the mean number of tablets remaining after completion of treatment was 8.9. No participants stopped taking antimuscarinic drugs.
Discussion
Patients with NB or OAB are typically prescribed antimuscarinic medications. Two such medications—oxybutynin and trospium—are commonly used in the urology department of Ramathobodi Hospital. Xerostomia is a primary cause of patients ceasing to take prescribed antimuscarinic drugs. A systematic review of the safety of antimuscarinic medication found the incidence of antimuscarinic-induced xerostomia to be 64.1%, which was 3.5 times greater than in the placebo groups. The same study found that the withdrawal incidence from adverse events was up to 15% and increased 1.37 times compared to the placebo. 21
Xerostomia can lead to oral infections, taste dysfunction, problems with chewing and swallowing, speech disturbances, and poor QoL. The fewer adverse effects of topical therapy make it preferable to systemic therapy when treating xerostomia. Antimuscarinic drug-induced xerostomia is easily treatable because the patient's salivary glands are still able to produce saliva. Therefore, patients are told to suck on sialagogue tablets, which is an easy and convenient solution.
Recently, OT was approved by the Food and Drug Administration of Thailand. Our study evaluated 28 patients with NB or OAB who were experiencing antimuscarinic medication-induced xerostomia. The appropriate sample size was calculated to be N = 32 with 80% power; at N = 28, the power was calculated to be 78.2%, which is acceptable. Participants were randomly divided into an intervention group and a placebo group. Xerostomia severity was evaluated before and after treatment using a severity score and SSFR.
Continuous daily use of OT for four weeks led to an improvement in overall subjective xerostomia symptoms and saliva production (p < 0.05). Longer use (two weeks and four weeks) of antimuscarinic medications was found to lead to improvements in lip dryness and speaking difficulty. An SSFR of 0.7–1.0 mL/min was considered low, and the SSFR of all patients at baseline was 1.13 mL/min, which is within the normal range. After treatment, improvements in saliva production were observed, which correlated with improvements in xerostomia symptoms. All patients took the medication four times a day. After completion of the study, the average number of remaining tablets was 8.9, suggesting a need for studies on the optimal daily dose (eg, three times per day or as needed) for improved patient satisfaction. No participant stopped taking antimuscarinic drugs in either group. Studies examining a longer duration of treatment (eg, 6 or 12 months) may reveal trends in NB or OAB patients’ treatment compliance.
Plenty of evidence confirms that xerostomia negatively affects QoL.8–10 The results of our study follow those obtained by Gomez-Moreno et al12,13 and Niklander et al 22 using 1% magic acid spray as a sialogogue in elderly and antidepressant-induced xerostomia, which demonstrated improvement of xerostomia symptoms and increased salivary flow rate. Also, the studies by Dalodom et al 19 and Femiano et al 23 using salivary substitution obtained the same positive outcomes.
Our study has three main limitations. First, Thailand experienced an uncontrolled resurgent outbreak of COVID-19 in April 2021, with 1000 + new cases per day. The government's policies of curfew and lockdown, and improvements in telemedicine, caused the number of patients with NB or OAB visiting the outpatient department of our hospital to decrease, so our study cohort was small. Second, time constraints on follow-up care prevented determination of the long-term outcomes of patient compliance with antimuscarinic drug treatment. The third limitation concerns the method of data collection; we planned to collect post-treatment data in the outpatient department, but, due to COVID-19, these data had to be collected by telephone.
Conclusions
Continuous daily use of OT for 4 weeks reduced xerostomia symptoms and increased saliva production in patients suffering from NB or OAB who received antimuscarinic medications.
Footnotes
Acknowledgements
The authors are thankful to all study participants. We thank Ms. Wirunchana Thiapthaisong for research assistance, Ms. Kornkanok Somboonpun for statistical analyses, and Ms. Malinee Rungsawang for editing the manuscript.
Funding
This work was supported by Faculty of Medicine, Ramathobodi Hospital and Faculty of Dentistry at Mahidol University, Thailand.
Ethical Approval
Ethical approval to report this case was obtained from the Human Research Ethics Committee, Faculty of Medicine Ramathibodi Hospital, Mahidol University (COA. MURA2020/1770).
Declaration of Conflicting Interests
The authors whose names are listed immediately below certify that they have NO affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Statements and Declarations
The authors formally declare that the content of this paper is the original work of themselves. It has not been published previously in any media including journals, conferences, or websites.
Surawach Piyawannarat: Methodology, data collection, data analysis, data interpretation and writing manuscript.
Pokket Sirisreetreelux: Data collection and analysis.
Wattanachai Rattapornsompong: Data collection and analysis.
Salunya Tancharoen: Project administration and manuscript revision.
Wit Viseshsindh: Conceptualization, project administration and manuscript revision.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Human Research Ethics Committee, Faculty of Medicine Ramathibodi Hospital, Mahidol University (COA. MURA2020/1770) approved protocols
Statement of Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
