Abstract
Most initial liver cancers are hepatocellular carcinomas (HCC), which make up the vast majority of cases. Hepatitis B or C virus infection as well as alcohol consumption is among the key risk factors. The significance of the most intriguing soluble factors as indicators for early diagnosis and as suggested targets for therapy in light of the increasing challenges in precision medicine. The development of HCC is influenced by a complex combination between pro-inflammatory and anti-inflammatory cytokines and their signalling cascades. Recently,researchers are aims to assess the potential of a number of distinct molecular cascade/cascade including cytokines to function as key players with particular underlying etiologies. Increasing our knowledge of the signaling network that links retro differentiation and inflammationmay help us find novel therapeutic targets and develop combined therapies or treatments that work against tumors with a significant degree of heterogeneity. With nursing processes at its center, comprehensive nursing care is a new nursing paradigm that combines the benefits of primary and group nursin g as well as a perfect synthesis of many nursing metrics like nursing philosophy, nursing plan, and nursing quality evaluation. In order to treat patients with serious liver diseases like cancer, it can conduct nursing interventions item by item in accordance with the unique disease conditions of each patient and combine efficient therapeutic approaches with high-quality nursing modes. Dietary natural products, including fruits, vegetables, and spices, may prevent and treat liver cancer by inhibiting tumor growth, protecting the liver, and enhancing chemotherapy.
Keywords
Introduction
Hepatocellularcarcinoma (HCC), a distinct term for liver cancer, is a malignancy that begins in the liver cells. Most cases of primary liver cancer can be attributed to this subtype. Hepatocytes, the most prevalent cell type in the liver, are the cells of origin for HCC. 1 It's important to note that there are other types of primary liver cancers, although they are less common than HCC. These include intrahepatic cholangiocarcinoma and hepato-blastoma. HCC is the fourth most catastrophic kind of cancer and the sixth most frequentcancersoverall. 2 East Asian countries including China, Taiwan, and Korea have the world's highest HCC rates. 3 HCC is more common in certain areas because chronic hepatitis B virus (HBV) infection is so common there. Sub-Saharan Africa also has a high burden of HCC, primarily due to a high prevalence of chronic HBV, hepatitis C virus (HCV) and hepatitis delta virus (HDV) infections. North America, Western Europe, and many others high-income countries have relatively lower incidence rates of HCC compared to East Asia and Sub-Saharan Africa.
Chronic HCV infection, alcoholic liver disease, NAFLD, and other metabolic diseases are major risk factors for HCC in certain parts of the world. 4 The involvement of inflammatory cytokines in liver cancer is crucial throughout its life cycle, from diagnosis to treatment. Immunological cells along with various cytokines, which control immunological responses and inflammation. In liver cancer, the production and release of inflammatory cytokines are often increased, contributing tumor growth, angiogenesis, invasions, and metastasis. Some of the key inflammatory cytokines involved in liver cancer include interleukin-6(IL-6), tumor necrosis factor-alpha (TNF-α), interleukin-1beta (IL-1β), and transforming growth factor-beta (TGF-β). 5
The variation in HCC incidence rates is closely linked to the prevalence of specific risk factors in different regions. Countries like Japan and Egypt endure a higher prevalence of chronic HCV infection than East Asia and Sub-Saharan Africa have incedents of chronic HBV infection. In certain areas of Africa and Asia, eating food tainted with aflatoxin is a major cause of HCC. Disparities in healthcare access and resources between high-income and low-to middle-income countries contribute to variations in HCC outcomes. Lower-income countries often face challenges in early detection, access to effective treatment, and supportive care, resulting in higher HCC mortality rates. Efforts to address the burden of HCC globally involve acomprehensive approach that includes prevention, early detection, and treatment. Vaccination against HBV, effective antiviral therapy for HBV and HCV, public health campaigns to raise awareness about the risks of alcohol abuse and aflatoxin exposure, and promoting healthy life styles are some of the strategies employed to shrink the prevalence and mortality of HCC worldwide. 6
Comprehensive nursing care for patients with serious liver diseases like cancer involves a patient-centered approach, individualized care planning, multidisciplinary collaboration, evidence-based interventions, and a focus on quality evaluation and improvement. By combining efficient therapeutic approaches with high-quality nursing modes, nurses can provide optimal care and support to the patients throughout their liver cancer journey. 7
Consequently, this review underscores the significance of molecular cascades in the progression and management of HCC through nursing care. It also endeavors to clarify the downstream cascades associated with HCC management through the use of a variety of natural products and phytoconstituents.
Understanding HCC in Relation with Liver Cirrhosis and Dysplastic Nodules (DNs)
Most instances of HCC occur in people who already have liver cirrhosis. Liver inflammation and fibrosis are the results of chronic liver injury, which can be caused by things like chronic viral hepatitis, alcohol isuse, or NAFLD (Figure 1). Adis organized liver architecture is a hallmark of cirrhosis, which develops from fibrosis overtime. Dysplastic foci or nodules areclusters of defective, immature hepatocytes that can form within the cirrhotic liver. 8 Dysplastic nodules area form of precancerous lesion that raises the likelihood of hepatocellular carcinoma. The presence of atypia and other morphological traits isused to categorize DNsa slow-grade or high-grade. It can be difficult to tell the difference between high-grade dysplastic nodules and HCC in its early stages.

Relationship between liver cirrhosis, DNs, and HCC.
Strategies to establish this dichotomy can be found in a consensus guideline developed at the international level. It is widely agreed that the occurrence of stromal invasion, in which tumor cells penetrate the surrounding stroma, is the defining characteristic of early HCC as opposed to dysplastic nodules. While dysplastic nodules of any grade have the potential to progress into HCC, the risk is greatest for high-grade dysplastic nodules. Close observation for HCC progression is warranted in patients with cirrhosis and dysplastic nodules. 9
Association of HCV and HBV Infection in HCC
Cirrhosis fosters in a sizable percentage of HCV-infected people as a consequence of them chronic liver illness. Cirrhosis comprises one of the most significant preludes to HCC. Overexpression of TGF-α, which stimulates cell proliferation and survival, is one example of a mitogenic cascade that can be engaged by HCV infection. 10 HCV infection contributes to oxidative assault in the liver. Oxidative assault leads to DNA damage, genetic instability, and cellular transformation, all of which can promote the development of HCC. Chronic liver inflammations have been triggered by the compromised immune response mediated via HCV infections. Prolonged inflammation and immune dysregulation can contribute to the development of HCC. 11 HCV infection can activate NF-кβ which promotes cell proliferation and survival, contributing to HCC development. HBV DNA can integrate in to the host genome, leading to genomic instability and genetic aberrations. Integration events can occur near tumor suppressor genes, such as p16, affecting their function. HBV produces a viral protein called HBx, which acts as an oncoprotein and contributes to cellular transformation. HBx can disrupt cellular signaling cascades, promote cell proliferation, and inhibit DNA repair mechanisms. Aflatoxin B1 is a potent mutagen found in certain food products, particularly in areas with poor storage conditions. Mutations in the p53gene (especially at codon249) are strongly correlated with exposure to aflatoxin B1 in places with endemic exposure, such as Asia and portions of Africa. Aflatoxin exposure and chronic HBV infection each raise the risk of HCC, but together they exacerbate it substantially.12,13
Association of HDV Infection in HCC
HDV, a defective virus dependent on HBV for assembly and persistence, harbors a 1.7 kb circular, single-stranded RNA genome that encodes two delta antigens. 14 HDV infection is associated with the highest mortality rate among hepatitis viruses, leading to severe liver diseases such as acute liver failure, cirrhosis, and HCC. HDV evades immune responses, induces oxidative assault, and promotes epithelial-mesenchymal transition (EMT) and fibrosis through interactions with TGF-β, AP-1, and STAT3 cascades. 15
Chronic inflammation from persistent viral infections, shared by HBV and HCV, exacerbates genetic alterations and liver damage, driving HCC progression. 16 HDV mediates hepatocarcinogenesis through modifications of innate and adaptive immune responses, epigenetic changes, lncRNA alterations, and ROS production. The L-HDAg facilitates these mechanisms via interactions with signalingcascades involved in cell growth, survival, apoptosis, and wound healing. L-HDAgactivates the TGF-β and AP-1 pathways by binding to Smad3, STAT3, and c-jun, promoting EMT, fibrosis, and cell transformation. 17 It also promotes oxidative assault in the ER through interaction with NOX-4, leading to ROS release and activation of STAT3 and NF-κβ signaling. 18 Additionally, L-HDAg stimulates pro-inflammatory NF-κβ activity via TNF-α induction. The S-HDAg downregulates glutathione S-transferase P1 mRNA, increasing ROS levels and apoptosis. 15 Epigenetic modifications, such as histone H3 acetylation by both delta antigens, enhance clusterin gene expression, aiding in HDV-infected cell survival and upregulation in cancerous cells. 19
Immune Intervention and Chronic Inflammation in HCC
The formation and progression of HCC are greatly influenced by chronic hepatitis, which causes inflammation that does not resolve. This inflammation is a characteristic of malignancy. 20 Cirrhosis, caused by hepatocyte damage and chronic inflammation, accounts for about 80% of HCC cases.21 However, additional carcinogenic pathways may be at work since 10%–20% of HBV-related HCC can develop in the absence of cirrhosis.From an HBeAg-positive chronic infection to a functional cure (HBsAg-negative), there are five clinical phases of chronic HBV infection. 22 In the chronic hepatitis phase, an overactive immune response causes a drop in HBV DNA levels, inflammation of the liver, and fibrosis. This response is marked by pro-inflammatory cytokines and HBV-specific CD8+ T cells. 23 Necroinflammation, hepatocyte regeneration, and modification of the hepatic microenvironment are outcomes of immune-mediated liver injury that persist over time. This damage is aided by natural killer cells and T cells, which generate pro-inflammatory cytokines and ROS. 24 Epigenetic changes, cancer-causing mutations, shortened telomeres, and genomic instability cause fibrosis, cirrhosis, and cell transformation in this never-ending loop of necroinflammation and inadequate wound healing reactions.23,24
Since HDV replicates only with HBV, co-infection dramatically worsens liver damage. 25 HDV infection suppresses HBV replication and causes severe liver destruction and illness. HDV avoids interferon-alpha-mediated innate immune responses by inhibiting the antiviral defense-critical JAK/STAT pathway. Superinfection with HDV in chronic HBV patients generates the worst liver disease outcomes due to the patient's preceding liver damage and HDV's propensity to reduce HBV viral load. 26 Liver necrosis and disease progression are induced by CD8+ T cells recognizing and eliminating HDV-infected cells and CD4+ T cells producing cytokines. 27
The majority of instances (75%–80%) of chronic hepatitis are caused by HCV infections, which are usually contracted in adulthood. With age, gender, obesity, diabetes, alcohol consumption, and HIV/HBV co-infection as risk factors, chronic hepatitis C virus infection causes moderate inflammation of the liver but can advance to fibrosis, cirrhosis, HCC. 28 Intrinsic type I and III interferon responses are activated by HCV, leading to the production of antiviral genes. Nevertheless, hepatocyte metamorphosis occurs as a result of immune-mediated damage caused by adaptive CD8+/CD4+ T cells and natural killer cells, which leads to ROS generation, DNA damage, lipid peroxidation, and epigenetic alterations. 29 Fibrosis, cirrhosis, and HCC form when HCV evades the immune system and causes persistent inflammation in the liver. The leading cause of HCC is cirrhosis, an incurable disease that typically does not cause symptoms until it has progressed to a severe degree. In chronic inflammation and fibrosis, stellate cells and liver sinusoidal endothelial cells are activated by pro-inflammatory cytokines and ROS generated by hepatocytes and Kupffer cells, leading to an increase in collagen synthesis and remodeling of the extracellular matrix. 30 Cirrhosis, the end result of increasing fibrosis, produces a hypoxic environment in the liver, which favors angiogenesis and selects for cancer cells. 31
Somatic Mutations Observed in HCC
In comparison to other solid tumors, the mutation rate in HCC is typically between 20 and 100 per genome. But the mutation rates per genome are highest in melanoma and lung cancer. 32 Liver cancer's underlying cause, like HBV or HCV infection, might affect which mutations manifest. HBVRT, the enzyme responsible for replicating HBV, does not have a proofreading role in its RNA-mediated reverse transcription replication process. This causes HBV- related mutations to occur at a relatively high rate. Unlike HBV, HCV is an on retro viral RNA virus that is not able to replicate by inserting its genome in to the host's. Double stranded DNA breaks are rare, but HCV can produce them, leading to a higher mutation rate in HCV-infected cells. Some genes are more likely to undergo mutations in HCV-infected cells than others. This includes immunoglobulin genes, BCL-6, TP53, and β-catenin(CTNNB1). 33
Moreover, ahyper-mutated genotype is being found in several HCC cases. This genotype shares a mutational spectrum with those exposed to aristolochic acid, the poisonous element inatraditional Chinese medicine called Chinese wildginger. The study investigated the mutational spectrum in cases of HCC, both initial and early-recurrent, and discovered that hypermutated tumor samples had a dominant T > A transversion pattern. 34 Whilethe mutation all and scape of HCC is diverse, there are several commonly observed somatic mutations. Some of these mutations affect genes such as TP53, CTNNB1, AXIN1, ARID1A, and TERT promoter,and others. The advancement of HCC is often attributed to mutations that impair essential signaling cascades responsible for regulating cell cycle progression, apoptosis, and DNA repair. 35
Telomerase Promoter Mutations in HCC
The chromosome ends are protected by structures called telomeres. They prevent chromosome degradation and fusion. The protein complex telomerease is responsible for the steady extension of telomeres at the ends of chromosomes. 36 In most cases, telomerase activity is down regulated in fully differentiated adult cells, leading to a steady shortening of telomeres with each cell cycle. Telomere shortening is accelerated by chronic liver damage, which is associated with rapid cell turnover. When telomeres get too short, the cell enters a state of senescence or apoptosis because of DNA dmage. 37 The regenerative capacity reduced in hepatocytes, which promotes the progression of fibrosis and cirrhosis. Inherited genetic variations in the TERT and TERC genes that reduce telomerase activity can amplify telomere shortening in chronic liver damage. Liver fibrosis and cirrhosis can progress more quickly as a result of this. Cirrhosis contributes to the onset of HCC by shortening telomeres and increasing genomic instability. The majority of telomerase activation occurs due to mutations in the TERT promoter region, making HCC a common mutation carrier. Mutations in this region upstream of the TERT gene produce unique binding sites for transcription factors, resulting in elevated levels of TERT mRNA and protein. Mutations in the TERT promoters are found in 30%–60% of HCC patients. 38 The presence of these mutations in preneoplastic cirrhotic lesions supports their status as driving mutations. The frequency of mutations in the TERT promoter is lower in HCCs caused by HBV. However, a further mechanism for telomerase activation is the frequent integration of HBV sequences into the TERT gene locus that is observed in these tumors.38,39
The Association Between TP53 Gene Mutations and HCC
The tumor suppressor gene p53 (or TP53) is well-known. Apoptosis, DNA repair, and cell cycle advancement are among processes that it controls. As a protective measure against tumorigenesis, wild-type p53 induces apoptosis and cell cycle arrest in response to DNA damage. Hepatocytes can become carcinogenic and activate oncogenic cascades if the p53 gene or other components of the TP53 cascade are mutated in a way that renders them inactive. Mutations in genes involved in the p53 or TP53 cascade have been tentatively linked to a more aggressive form of HCC. Mutations in the p53 gene are found in head and neck cancer at varying rates, depending on the cause of the disease.40,41 From 18% to 50%, it can be found. Also indicating variability in the aetiology of HCC across locations is a geographically variable p53 mutation rate. Mutations in p53 have been linked to ingesting the fungus toxin aflatoxin (AFB1), specifically at codon 249. As it is present in AFB1-exposed individuals’ normal livers in addition to HCC tumors, this mutation is being called a driving mutation. Exposure to aflatoxin and chronic HBV infection have a synergistic effect on the progression of HCC. The production of HBx protein is linked to an uptick in the prevalence of certain mutations, especially G/C-to-T/A transversions, in hepatitis B-infected patients who have been exposed to aflatoxin. This suggests that HBV infection and HBx expression may enhance the mutagenic effects of aflatoxin exposure. Apart from TP53 itself, other genes in the p53 cascade, such as ATM and RPS6KA3, are recurrently mutated in HCC. Alterations in these genes may contribute to the disruption of p53-mediated tumor suppression and the development of HCC. 42
Alteration in Various Downstream Cascades Contributes to HCC
The development of HCC is multifactorial, involving various risk factors and etiological factors. The primary risk factor for HCC is chronic liver disease, particularly cirrhosis, which is responsible for the majority of cases. Approximately 80% of HCC instances occur in people who already have liver cirrhosis. Numerous phases, comprising genetic and epigenetic changes, occur during the onset and progression of HCC, contributing to the disease's high molecular complexity. The pathogenesis of HCC involves a series of genetic and epigenetic alterations that occur in hepatocytes or hepatic stem cells, triggered by external stimuli. 43 These alterations can lead to apoptosis, cell proliferation, dysplasia, and neoplasia. Mitotic signaling cascades are activated in the preneoplastic phase, providing growth advantages to the afflicted cells and ultimately triggering the selection of dysplastic cell clones. 44 Dysplastic nodes, which are clusters of cells enclosed by connective tissue, are formed as these clones of dysplastic cells multiply. These dysplastic lymph nodes, however, are not yet cancerous. Point mutations, chromosomal arm gains or losses, and aberrant promoter methylation of important genes are all examples of genomic insults that contribute to the acquisition of the malignant phenotype. These genetic and epigenetic alterations contribute to the transformation of dysplastic nodes into fully malignant hepatocellular carcinoma. The dysregulated signaling cascades and disrupted gene expression patterns in HCC cells further promote tumor growth, invasion, and metastasis. Different mechanisms might be associated with the initiation and spread of liver cancer depending on the etiologies involved. 45 The molecular alterations involved in the development of HCC are recapitulated in Table 1.
Molecular Alterations Involved in the Development of Solid Tumors.

Molecular Cascade of the Wnt/β-Catenin Cascade in HCC
In order HCC in development, the Wnt/β-catenin cascade is indispensable. The Frizzled receptors on the surface of cells are regulated by Wnt ligands, which are secreted signaling molecules. 48 Genetic modifications or mismanagement of Wnt cascade components can cause inappropriate activation of the system, which in turn can lead to HCC. Protein complex consisting of glycogen synthase kinase-3β (GSK-3β), adenomatous polyposis coli (APC), and Axin phosphorylates β-catenin in the absence of Wnt ligands. Whenever β-catenin is phosphorylated, the proteasome will eat it up. The activity of GSK-3β is suppressed when Wnt ligands bind to Frizzled receptors.48,49 That way, β-catenin doesn’t get phosphorylated and degraded. β-catenin translocates from the cytoplasm to the nucleus when stimulated by Wnt ligands. APC and Axin's impairment of the destruction complex makes this possible. T-cell factor/lymphoid enhancer factor (TCF/LEF) binds to stabilized β-catenin once it has entered the nucleus. 50 Cell proliferation, survival, and differentiation are all regulated by the expression of target genes, which are regulated by this complex (Figure 2). In HCC, target genes implicated in cell proliferation, anti-apoptosis, and angiogenesis are uncontrollably activated due to dysregulation of the Wnt/β-catenin cascade.
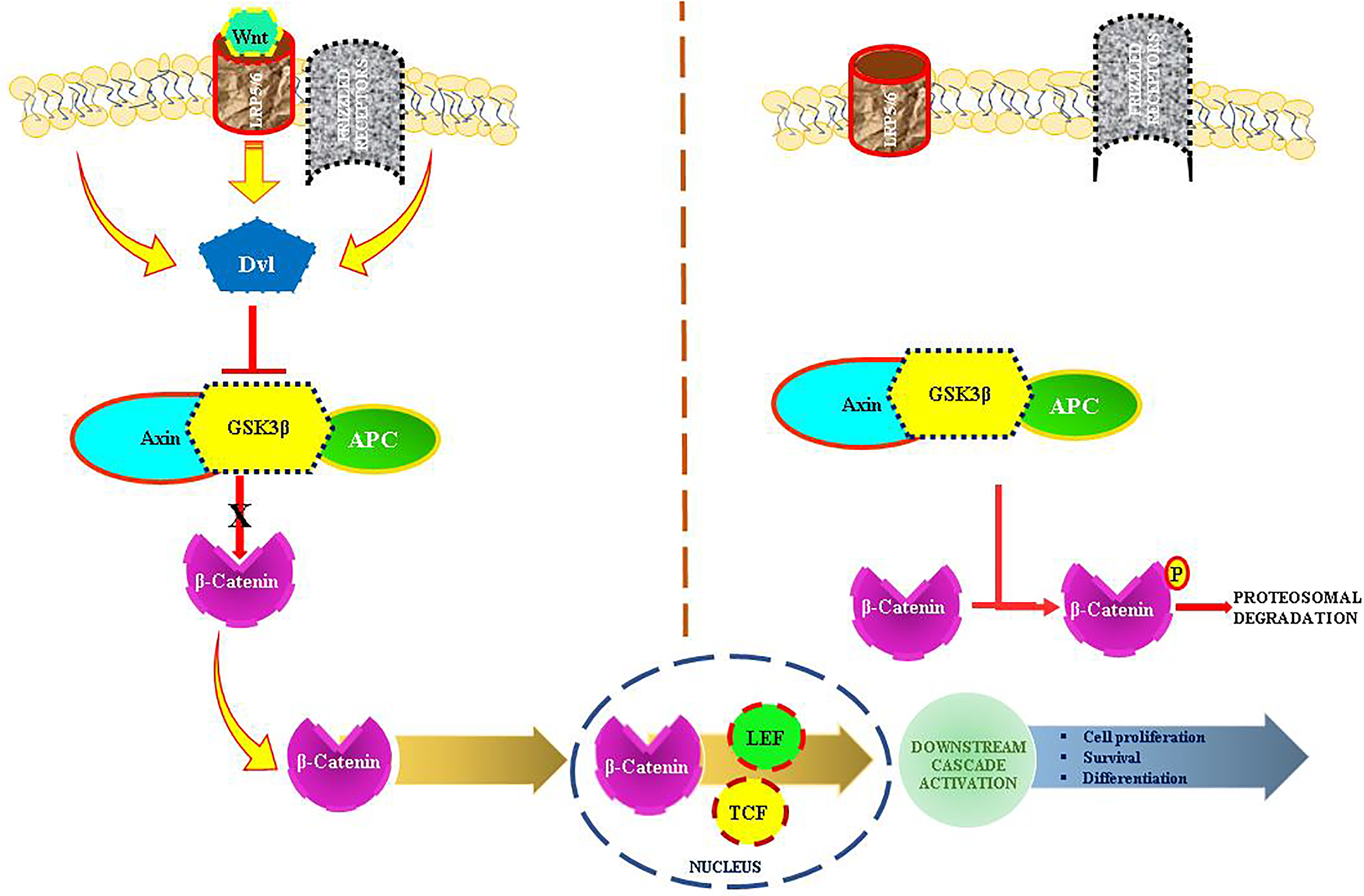
Schematic diagram of molecular cascade of the Wnt/β-catenin pathway in HCC.
Molecular Cascade of PI3K/Akt/mTOR Cascade in HCC
Once it comes to the emergence of HCC, the phosphoinositide 3-kinases (PI3K)/Akt/mammalian target of rapamycin (mTOR) signaling cascade is pivotal. 51 Epidermal growth factor (EGF) and insulin-like growth factor (IGF) are two examples of growth factors that bind to specific cell surface receptors. PI3K becomes triggered as the consequence. Phosphatidylinositol 34,5-trisphosphate (PIP3) is produced when phosphatidylinositol 4,5-bisphosphate (PIP2) is phosphorylated by an active PI3K. 52 The second messenger PIP3 is responsible for Akt recruitment and activation. When PIP3 binds to Akt, phosphoinositide-dependent kinase 1 (PDK1) and mammalian target of rapamycin complex 2 (mTORC2) phosphorylate and activate Akt (Figure 3). Tuberous sclerosis complex 2 (TSC2) is phosphorylated and inhibited by active Akt, which in turn activates mTORC1. The mTOR complex 1 plays a critical role in controlling cell size, biotransformation, and protein synthesis. Multiple downstream effectors, such as p70S6 kinase (p70S6K) and eukaryotic translation initiation factor 4E-binding protein 1 (4E-BP1), are phosphorylated and activated after mTORC1 activation.53,54 These effectors promote protein synthesis and cell proliferation. Dysregulation of the PI3K/Akt/mTOR cascade in HCC leads to constitutive activation of mTORC1, promoting uncontrolled cell growth, survival, and angiogenesis. It also contributes to metabolic reprogramming and resistance to apoptosis. 55

Schematic diagram of molecular cascade of HGF/MET pathway in HCC.
Molecular Cascade of IGF Cascade in HCC
In the pathogenesis of HCC, the IGF signaling cascade undergoes dysregulation, with overexpression and activation of IGF-1 receptor (IGF-1R) and increased expression of IGF-II. 56 Additionally, several modulators of the cascade, such as insulin-like growth factor-binding proteins (IGFBPs), are significantly downregulated. In HCC, there is often overexpression of IGF-1R, leading to an increased number of receptors on the cell surface. This overexpression can result from genetic alterations or epigenetic modifications. Activation of IGF-1R initiates downstream signaling events. IGF-II, one of the ligands for IGF-1R, is frequently upregulated in HCC. The increased expression of IGF-II can occur through various mechanisms, including gene amplification and loss of imprinting. IGFBPs, including IGFBP-1, IGFBP-3, and IGFBP-4, serve as modulators of the IGF signaling cascade by binding to IGFs and regulating their availability and activity.56,57 In HCC, these IGFBPs are often significantly downregulated, potentially leading to increased free IGF levels and enhanced activation of the IGF-1R. Once activated by ligand binding, IGF-1R triggers downstream signaling cascades that promote cell growth, survival, and proliferation.58,59
Molecular Cascade of HGF/MET in the Pathogenesis of HCC
Hepatocyte growth factor (HGF) is a key molecule involved in liver regeneration, and it transmits signals by binding to the MET receptor. 60 Deregulation of both HGF and MET is commonly observed in HCC. HGF is primarily produced by stromal cells in the liver, such as hepatic stellate cells and endothelial cells. Upon liver injury or in response to certain stimuli, HGF is released into the liver microenvironment. HGF binds to the MET receptor, leading to receptor dimerization and activation.60,61 Autophosphorylation of certain tyrosine residues within the intracellular domain of MET is triggered by this interaction. Initiation of the PI3K/Akt/mTOR cascade, the Ras/Raf/MEK/ERK cascade, and the STAT cascade all ensue upon MET expression. 62 These signaling cascades promote cell survival, proliferation, migration, and invasion. HGF/MET signaling stimulates cell proliferation and survival in HCC. It enhances the cell cycle progression, inhibits apoptosis, and supports the growth of cancer cells. It promotes tumor growth by encouraging the development of new blood vessels by inducing the expression of pro-angiogenic factors including VEGF. Deregulation of HGF and MET in HCC can occur through various mechanisms, including genetic alterations, such as gene amplification or mutations, as well as epigenetic modifications (Figure 3). 63 Increased expression of HGF and/or overactivation of MET receptor signaling have been associated with aggressive tumor behavior, enhanced metastasis, and poor prognosis in HCC.62,63
Molecular Cascade of JAK/STAT Cascade in HCC
Signal transduction through the Janus kinase/signal transducer and activator of transcription (JAK/STAT) cascade is triggered by the obligatory binding of cytokines, hormones, or growth factors to their receptors. 64 The receptor's associated Janus kinases (JAKs) become engaged as part of this association. The signal transducer and activator of transcription (STAT) proteins, most notably STAT3 and STAT5, include tyrosine residues that are phosphorylated when a JAK is activated. STATs can become homo- or heterodimers after being phosphorylated. When a STAT protein is phosphorylated, it might form a dimer with another phosphorylated STAT protein or another transcription factor. The dimers undergo a nuclear translocation process. When STAT dimers get to their nuclear destination, the promoter regions of target genes, they bind to particular DNA sequences called STAT response elements (SREs).65,66 This binding determines whether these genes will be actively activated or repressed at the transcriptional level. Cell proliferation, migration, differentiation, and death are all activities that are controlled by STATs that are activated and, in turn, govern gene transcription. The JAK/STAT cascade can be dysregulated in HCC, which can lead to unchecked cell proliferation and the advancement of the tumor. Activated STATs generate suppressors of cytokine signaling (SOCS) proteins such as SOCS1 and SSI-1 to prevent the cascade from becoming overactivated. When these proteins attach to phosphorylated JAKs and their receptors, they block the signaling cascades. 67
Molecular Cascade of Ubiquitin-Proteasome in HCC
To degrade proteins and control their levels, HCC relies heavily on the ubiquitin-proteasome system. In the first step, the enzyme E1 acts as an activator of ubiquitin. 68 Enzyme 1 (E1) is responsible for passing on the active form of ubiquitin to enzyme 2 (E2). The ubiquitin- conjugating enzyme (E2) interacts with a specific ubiquitin ligase enzyme (E3), which serves as a substrate recognition component. Through recognizing and adhering to specific target proteins, E3 ligases increase the process's specificity. It is the E3 ligase's job to take the ubiquitin molecule from the E2 enzyme and attach it to the appropriate protein. 69 This exchange can happen in a one-step process or during the assembly of multi-ubiquitin chains. The 26S proteasome, a big protein complex composed of a 20S core particle and 19S regulatory particles, is able to recognize the ubiquitinated target protein. Ubiquitinated proteins are destroyed by proteases once they are unfolded and transported to the 20S core particles. 70 The ubiquitin-proteasome system regulates the abundance and activity of various cancer-related proteins involved in cell cycle control, tumor suppression, and oncogenic signaling cascades. For example, tumor suppressor proteins like p53, p27, pRb, and PTEN can be targeted for ubiquitin-mediated degradation by specific E3 ligases, thereby modulating their levels and function. 71
Circumventing Immune Surveillance and HCC
In HCC, immune escape mechanisms play a crucial role in evading immune surveillance and promoting tumor growth. Tregs are a type of CD4+ T cell that serve to moderate the immunological response by suppressing the expression of effector T cells. 72 It is frequent to detect an increase in Tregs in both tumor tissues and peripheral blood of patients with HCC. Both their presence and their activity limit the immune system's ability to fight tumors. Increases in immunosuppressive cytokines (such as IL-4, IL-5, IL-8, and IL-10) and deficits in immune-activating cytokines (such as IL-1, TNF, and interferon gamma [IFγ]) are observed in the tumor microenvironment of HCC. The immunosuppressive milieu caused by this cytokine imbalance is detrimental to mounting an efficient anti-tumor immune response. In this instance, high IL-10 levels are linked to a worse prognosis in HCC. 73 Two molecules called immunological checkpoints control immune responses; these are the programmed cell death-1 (PD-1) receptor and its ligand, PD-L1. Tumor cells in HCC may upregulate expression of PD-L1, which then interacts with PD-1 on T cells to cause T cell fatigue and apoptosis. This cooperation dampens the immune system's attack on the tumor. Immune checkpoint inhibitors that target the PD-L1/PD-1 cascade, such as nivolumab, have shown clinical benefit in the treatment of HCC. 74
Cytokines and HCC
A varied class of tiny proteins known as cytokines serves as a messenger between immune system cells. They can be pro- or anti-inflammatory and are generated by a variety of cell types, including stromal cells, immune cells, and even cancer cells. Numerous biological processes, including immunological responses, cell proliferation, tissue healing, and inflammation, are regulated by these molecules. Cytokines are important mediators in the course of cancer because they have the ability to either stimulate or inhibit the growth, invasion, as well as metastasis of tumors. Because these signaling molecules are essential to the growth, development, and immunomodulation of this aggressive malignancy, interest in the role of cytokines in HCC has grown. We shall examine the role of cytokines in HCC, their influence on tumor growth, and their potential as therapeutic targets in this extensive essay. Most often, cirrhosis or infections with hepatitis B and C are associated with chronic liver disorders, which can lead to the development of HCC. 75 HCC has a complex and multifaceted pathophysiology that includes environmental, genetic, and epigenetic variables.75,76 Tumor cells and the surrounding tissue can be impacted by cytokines, which can also have an impact on tumor cells’ proliferation, differentiation, migration, and survival. We shall explore the role of cytokines in HCC in this article, beginning with those linked to the cytokine landscape of HCC. One of the main features of the development of cancer is inflammation, which is also a major factor in the pathogenesis of HCC. Liver cancer can start and spread more easily in a microenvironment that is created by chronic inflammation, which is frequently brought on by underlying liver illnesses. Immune cells, fibroblasts, and cytokines that encourage cell division and tissue remodeling are present in this milieu.77,78 HCC frequently develops in the context of cirrhosis and chronic liver disease. The development of HCC is aided by the liver's ongoing fibrosis and inflammation. The next section discusses cytokines, which are important mediators of the inflammatory response and have a role in initiating hepatocarcinogenesis 79 and promoting liver damage:
IL-6
As a pleiotropic cytokine, IL-6 is essential to the development of HCC. It is made by a variety of liver cell types, such as Kupffer cells and hepatocytes. The activation of STAT3, a transcription factor that stimulates angiogenesis, cell proliferation, and resistance to apoptosis, is facilitated by IL-6. Raised levels of IL-6 are linked to larger tumors and a worse prognosis in HCC.80,81
TNF-α
This pro-inflammatory cytokine has the ability to stimulate inflammation and apoptosis in the liver. Exposure to TNF-α over an extended period of time may be linked to the development of liver cirrhosis, fibrosis, and eventually HCC. The NF-κB cascade, which is linked to the advancement of HCC, is triggered by TNF-α signaling.81,82
TGF-β
TGF-β is a multipurpose cytokine that contributes to the development of HCC in two ways. It functions as a tumor suppressor in the early phases of hepatocarcinogenesis by preventing cell division and triggering apoptosis. Nevertheless, in more advanced phases, HCC cells frequently become resistant to TGF-β's growth-inhibitory activities, which might encourage tumor invasiveness and metastasis. 83
IL-1
A potent pro-inflammatory cytokine called IL-1 contributes to the early stage of HCC known as liver fibrosis. It induces the synthesis of more pro-inflammatory cytokines and chemokines, which makes the hepatic milieu more pro-tumorigenic. Other signaling cascades, such as the IL-6/STAT3 cascade, which is known to be implicated in HCC, can interact with IL-1 through crosstalk. This intercommunication may help HCC grow and advance even further. IL-1 may encourage HCC cell growth directly or indirectly. It has the ability to stimulate signaling cascades linked to cell survival and proliferation, such as the NF-κB cascade. This might facilitate the development of tumors with HCC. The process of EMT gives cancer cells a more migratory or invasive character. The promotion of EMT in a variety of cancer types has been linked to IL-1, and this transformation is necessary for the spread of HCC to other bodily regions. Since the liver is an organ with a distinct immune system, HCC frequently arises in the setting of chronic liver disorders. IL-1 influences immune cell activity and fosters an immunosuppressive tumor microenvironment, which can aid in immune evasion strategies in HCC. 84
IL-10
In HCC, the anti-inflammatory cytokine IL-10 can both stimulate and inhibit tumor growths. It has the ability to both reduce anti-tumor immune responses and prevent HCC cell multiplications. 84 Its function in HCC is complicated and context-dependent. It's crucial to remember that each patient's response to IL-10 for HCC may be different and context- dependent. While some research has shown that IL-10 might be a target for treatment, other research has suggested that it may have a protective function through the regulation of inflammation. Our knowledge of the function of IL-10 in HCC is constantly expanding along with our comprehension of the molecular cascades behind this illness. These cytokines and their corresponding signaling cascades have complex and frequently interconnected interactions. For instance, IL-6 can increase TGF-β and TNF-α production, aggravating the inflammatory and pro-tumorigenic milieus in the liver. Depending on the patient's unique genetic and immunological makeup as well as the disease's stage, the overall impact of these cytokines on the development of HCC can change. Angiogenesis, or the growth of new blood vessels, is influenced by IL-10. Angiogenesis is necessary for the development and spread of tumors in HCC. Pro-angiogenic factor production may be impacted by IL-10, which might encourage angiogenesis inside the tumor. HCC cells can be directly encouraged to grow and survive by IL-10. It could raise the burden of tumors by promoting the growth of cancer cells and preventing apoptosis, or programmed cell death. It is well known that IL-10 suppresses the immune system. It may help the tumor in HCC avoid detection by the immune system. Immune cells that are essential for identifying and eliminating cancer cells, such as T cells and natural killer (NK) cells, can have their activity inhibited by IL-10. This may encourage the development and spread of tumors.85,86
VEGF
New blood vessel growth, or angiogenesis, is a sign of progressing tumors. One important cytokine in angiogenesis is VEGF. Overexpression of VEGF in HCC facilitates the growth and metastasis of the tumor by encouraging the development of new blood vessels. VEGF expression is frequently elevated in HCC. 87 Numerous biological processes, including genetic mutations, epigenetic modifications, and the activation of certain signaling cascades, might cause this. The overproduction of VEGF causes the tumor's angiogenesis to grow. Hypoxia, or low oxygen levels, is a characteristic shared by quickly growing solid tumors such as HCC. In response to hypoxia, the HIF cascade is triggered in cells. In response to low oxygen levels, HIF transcription factors induce the synthesis of VEGF and other angiogenic factors, which aid in the development of new blood vessels. On the surface of endothelial cells in blood arteries, VEGF attaches itself to its receptors. Intracellular signaling cascades, including the VEGF receptor-2 (VEGFR-2) cascade, are activated by this interaction. By encouraging endothelial cell survival, migration, and proliferation, activation of these cascades eventually results in the development of new blood vessels that provide the tumor with oxygen and nutrients. VEGF can work in two ways: autocrine, which stimulates the tumor cell that generates it, and paracrine, which stimulates neighboring cells, in addition to encouraging angiogenesis. The HCC cells themselves may proliferate and survive longer as a result.87,88
PDGF
Hepatic stellate cells (HSC) proliferation may be induced by PDGF ligand overexpression and PDGFR stimulation. It's interesting to note that stimulation of PDGFRα and overexpression of PDGF-C in hepatocytes cause liver fibrosis as well as the eventual formation of HCC, indicating the important function of PDGFRα in the carcinogenesis of HCC and hepatic fibrosis. It is still unclear, nevertheless, what exactly the cellular target of PDGF-C is, where PDGFRα should be prevalent. PDGFRα expression is observed on non- parenchymal liver cells, mostly HSCs, in non-cancerous conditions. Thus, it is conceivable that overexpression of PDGF-C might activate HSCs’ PDGFRα, which would then cause the cells to become activated and proliferate. In HCC tumor locations, PDGFRα over-expression may be a result of these activated HSCs. It is still unclear, nevertheless, how HSC activation, proliferation, and HCC development are related. 89
HGF
Hepatocyte growth factor (HGF) has been demonstrated to be overexpressed in HCC in contrast to normal and cirrhotic livers devoid of neoplastic indications. When compared to the distinct paracrine mechanism found in normal liver tissue (in the absence of cancer), expression of HGF and its receptor supports the existence of both autocrine and paracrine mechanisms of HGF action in HCC, suggesting that it also plays a role in tumor development and progression. Tumor cell products enhance the secretion of HGF by myofibroblasts and stellate cells, which in turn promotes the invasiveness of tumor cells. Higher blood HGF levels have been found in recent studies to favorably correlate with tumor growth and adversely correlate with patient survival time.90,91
Chemokines
It has been demonstrated that CCL5, a chemoattractant of memory T cells and other immune cell types, is essential for managing persistent viral infections. It has also been demonstrated that CCL5 is linked to hepatic inflammation when chronic HCV and HBV are present. Only one study has examined blood CCL5 levels in relation to HCC identification to date, and that study took place in a European environment. This study demonstrated that blood CCL5 levels were higher in HCC patients, 61 HCC cases compared to 78 controls. CCL5 levels more than 0.86 ng/mL were linked in a multivariate forward stepwise regression analysis to the development of HCC (odds ratio = 3.63). Furthermore, CCL5 demonstrated an AUC of 0.72, corresponding to a sensitivity of 71% and a specificity of 68% in the identifications of HCC. 92
IL-17
One pro-inflammatory cytokine that has been linked to the development of liver fibrosis and inflammation is IL-17. In individuals with HCC, higher levels of IL-17 have been linked to poorer outcomes. Immune cells, such as neutrophils and other immune cells, can be drawn to the liver by IL-17. This influx of immune cells may intensify the inflammatory milieus in the liver. Angiogenesis, the development of new blood vessels inside the tumor, is crucial for the tumor's growth and spread, and IL-17 can encourage this process. Tumor microenvironment modulation by IL-17 can affect the behavior of different cells both inside and outside of the tumor. It may promote the synthesis of more inflammatory cytokines and chemokines, which may aid in the development of tumors and the suppression of the immune system. Suppression of the anti-tumor immune response may be aided by IL- 17. It could prevent T cells from doing their vital job of identifying and eliminating cancer cells. The tumor may be able to avoid the immune system as a result of this immunosuppression. EMT, a process that makes cancer cells more invasive and capable of migrating to other areas of the body, has been linked to IL-17. 93
Cytokines and the Tumor Microenvironment in HCC
The tumor microenvironment (TME) is a complex and dynamic ecosystem that includes tumor cells, immune cells, stromal cells, and extracellular matrix components. Cytokines are essential regulators of the TME in HCC, shaping the immune response and influencing tumor behavior.
Tumor-Infiltrating Lymphocytes (TILs)
Cytokines are essential for controlling immune cell penetration into the HCC TME. When properly activated, TILs, such as natural killer cells and cytotoxic T lymphocytes, can have anti-cancer effects. TNF-α and IFN-γ, which are cytokines generated by TILs, can be deadly to tumor cells. TILs are a subset of immune cells that have entered the tumor microenvironment and are there. TILs are essential for the anti- tumor immune response when it comes to HCC, the most prevalent form of liver cancer. TIL- related molecular cascades in HCC can be complex and multidimensional. 94 Here are some salient features of their participation: TILs are essential for recognizing and eradicating tumor cells, with CD8+ T cells and natural killer (NK) cells playing crucial roles. These cells initiate an immune response against cancer by identifying antigens particular to tumors. Nevertheless, HCC tumors often escape detection by the immune system through many tactics, such as upregulating immune checkpoint molecules like CTLA-4 and PD-1/PD-L1, which hinder the action of TILs. 95 Immune checkpoint inhibitors, which are immunotherapies that specifically target these checkpoints, can reactivate TILs. The liver's distinct immunological milieu, which is affected by chronic liver disorders such as hepatitis and cirrhosis, frequently results in immunosuppression and decreased activity of TILs. Regulatory T cells and tumor-associated macrophages have a role in causing this immunosuppression. Adoptive cell therapy, such as TIL treatment, entails the ex vivo expansion and activation of TILs derived from a patient's tumor tissue. This process enhances the immune response against the tumor when the TILs are reintroduced into the patient. The presence of a tumor considerably affects the functioning of TILs. HCC tumors frequently exhibit fibrosis and inflammation. The tumor stroma, which consists of immune cells, extracellular matrix components, and cancer-associated fibroblasts, has an impact on the infiltration and activation of TILs. The tumor microenvironment releases chemokines and cytokines, such as CXCL-9 and CXCL-10, which attract T lymphocytes to the tumor site.96 Additionally, activated TILs create cytokines like IFN-γ, which enhance the anti-tumor response. Efficient presentation of antigens by dendritic cells and other cells that present antigens within the tumor microenvironment is essential for the activation of TILs. The effectiveness of this process directly impacts the potency of the anti-tumor immune response. Due to the diverse nature of HCC, various sections of the tumor may display distinct features. Therefore, it is crucial to comprehend the distribution and behavior of TILs within the tumor to devise efficient immunotherapeutic strategies.
Immune Checkpoints
Immune checkpoint molecules include cytotoxic T-lymphocyte- associated proteins 4 and programmed cell death protein 1 (PD-1). HCC cells use cytokines as a major means of evading the immune system. To inhibit the immune system, cancer cells can release immunosuppressive cytokines including TGF-β and IL-10. These cytokines hinder the ability of natural killer (NK) and cytotoxic T lymphocytes (CTLs) to efficiently target and eliminate HCC cells. In addition, HCC cells have the ability to express PD-L1 in response to immune cells’ production of IFN-γ. Immune checkpoint suppression results from PD-L1 binding to the PD-1 receptor on immune cells. Through this strategy, cancer cells are able to evade immune response and monitoring. Comprehending the immune evasion processes mediated by cytokines is crucial in order to devise immunotherapeutic approaches, such immune checkpoint inhibitors, which have the capacity to counteract these effects and augment the anti-tumor immune response in hepatocellular carcinoma. A family of immunotherapies known as immune checkpoint inhibitors (ICIs) has completely changed the ways that cancer is treated. They specifically target molecules involved in immune checkpoint suppression, such as PD-1 and PD-L1. Inhibitors of PD-1 and PD-L1 have demonstrated potential in HCC in stimulating the immune system to attack cancer cells.95,96 The goal of these treatments is to strengthen the immune response against tumors by combating cytokine-mediated immune evasion.
Gender Disparities Associated with HCC
Hepatitis virus infections (HBV and HCV in particular), drinking, aflatoxin exposure, autoimmune hepatitis, fatty liver disease, and being male are all recognized as key risk factors for HCC. A greater number of males than females are diagnosed with HCC, have a worse prognosis, and ultimately die from the disease. Females with HCC have a higher chance of survival, both without treatment and after surgery. 97 However, these benefits reduce after menopause because of the higher incidence of cirrhosis and HCC among older women. In Europe, Liou WL et al (2023) investigated gender disparities in the management and prognosis of HCC. The study analyzed data from 1051 patients with HCC from 12 European countries and discovered that women were less likely to obtain curative treatment and had poorer survival outcomes than men. 98 Male HCC instances are more common than female HCC cases overall; however, the ratio changes dramatically between younger and older individuals. The clinical manifestations of HBV and HCV differ between the sexes. The sex difference in HBV patients is present as early as the chronic hepatitis stage, suggesting an early role for causal variables in the carcinogenic process. Reactivation of latent HBV infection in male carriers has been linked to an increased risk of developing cirrhosis and HCC. Asymptomatic HBeAg-negative HBV carriers have similar tendencies. Researchers have found that females are more likely to attain HBeAg and HBsAg seroconversion than males are, suggesting that females may have more robust immune responses against HBV. 99 Further investigation into estrogen's role in controlling the immune response is warranted. Patients infected with HCV are more likely to experience rapid fibrosis progression, a worse response to IFN therapy, and premature decompensation if they are male. This indicates that gender has a role in hepatocarcinogenesis prior to the development of HCC. Hepatocarcinogenesis has been found to be more common in men than women, but the reasons for this are still being researched. Epidemiological and experimental research implies that sex steroid hormones (androgen and estrogen) may function as key regulating elements, in addition to social context and lifestyle factors including smoking and alcohol consumption. Guo et al (2022) sought to investigate the role of the estrogen axis in sex disparities observed in HCC. Targeting the estrogen axis may be a promising strategy for the prevention and treatment of HCC, particularly in women, according to the findings of this study. 100 The role of viral factors is also suggested since male dominance is more evident in HBV-related HCC compared to HCV-related HCC. Overall, the passage emphasizes the male dominance in HCC incidence, prognosis, and mortality, highlights the impact of sex-specific factors in hepatitis virus-related liver diseases, and suggests the association of sex steroid hormones and viral factors in the regulation of hepatocarcinogenesis.99,100
Management of HCC
Comprehensive Nursing Care
It is a nursing paradigm that places an emphasis on the whole person and their needs (Figure 4). Care for patients with complicated disorders, such as life-threatening liver diseases like cancers, necessitates the integration of multiple nursing measures, such as nursing philosophy, nursing plans, and nursing quality evaluation. Patients with liver cirrhosis and liver cancer who had interventional therapy were the focus of a study by Yuli GOU et al (2019). 7 The study gazed at data from 120 patients and showed that patients’ physical and mental health, as well as their happiness with the care they received, were much better after receiving comprehensive nursing care. In order to improve the outcomes of patients with liver cirrhosis and liver cancer who undergo interventional therapy, the study found that comprehensive nursing care is an effective intervention. Yang et al (2019) provides a comprehensive overview of the global burden of HCC, including its trends, risk factors, prevention, and management. 101 The study emphasizes that all these risk factors are potentially preventable, highlighting the considerable potential of risk prevention for decreasing the global burden of HCC. The study also discusses the various treatments for HCC, including surgical resection, liver transplantation, and ablation, which are associated with long-term survival. The study concludes that a multidisciplinary approach is needed for the prevention and management of HCC, including primary prevention, early detection, and appropriate treatment and management of the disease. Nursing care stipulates holistic assessment which involves analyzing their religious or spiritual affiliations, meaning and purpose, disease and healing beliefs, and spiritual anguish or support. Physical assessment evaluates the patient's vital signs, body systems, pain, mobility, nutrition, and physical complaints or restrictions. It detects health abnormalities, tracks changes, and recommends treatments. Psychological Assessment involves understanding the patient's emotional state, coping methods, and mental health issues. It requires psychological assessments. It assesses mood, anxiety, cognitive function, sickness perception, psychological distress, and support requirements which helps nurses provide emotional support, find coping methods, and work with other healthcare experts for specialized care. Social Assessment care requires understanding the patient's social situation. It evaluates them support system, family dynamics, culture, socioeconomic level, living environment, and resources. This examination identifies social determinants of health, support networks, barriers to care, and social needs and supportive environment solutions. 102

Nursing paradigm for patient care and well-being.
Individualized Care Planning
After the comprehensive assessment, nurse care plans are created to meet patient requirements and goals. HCC patients’ individualized care programs often include these factors: Liver cancer stage affects treatment and intervention. Surgery, liver transplantation, radiation therapy, chemotherapy, targeted therapy, and immunotherapy should be included in the care plan. The care plan should incorporate nursing considerations and side effects for each treatment method. Liver dysfunction, ascites, hepatic encephalopathy, bleeding issues, and infection can result from liver cancer and its treatment. Monitor and treat these issues in the treatment plan. Pain, weariness, anorexia, nausea, and jaundice are common symptoms of liver cancer. To improve patient comfort and quality of life, the care plan should incorporate pharmacological and non-pharmacological symptom management. Cancer can have serious psychological and social effects. The patient's emotional well-being, counseling or support services, and anxiety, depression, and distress management should be addressed in the treatment plan. Care planning requires patient education. Educational interventions should help patients comprehend their disease, treatment options, self-care, and lifestyle changes. Active patient participation improves outcomes and self-management. The care plan should include regular monitoring of disease progression, treatment response, and recurrence. It should include appointment, laboratory, imaging, and other evaluations schedules. 103
Multidisciplinary Collaboration
Comprehensive liver cancer nursing care requires it. A diversified healthcare team ensures that all areas of the patient's treatment are addressed, enhancing patient results and holistic well-being. Multidisciplinary collaboration has numerous advantages including treatment planning. 104 Collaboration with physicians, oncologists, surgeons, and other experts allows for a complete assessment of the patient's condition, appropriate disease staging, and a well- rounded treatment plan that takes into account all available alternatives and patient preferences. While coordinated Care diagnostic tests, therapy, medication management, symptom control, and follow-up care. Coordination reduces gaps in care, improves efficiencies, and reduces errors and omissions. Expertise from different disciplines in which each diverse team member adds their own expertise and knowledge which helps the care team grasp the patient's situation and incorporate diverse treatment techniques and supportive treatments. Patients and their families need psychosocial help from social workers, psychologists, and other mental health specialists. These specialists help patients address their emotional and psychological issues, give counseling and support, and access community resources. Dietitians are vital members of the multidisciplinary team that advise on dietary changes, nutritional support, and therapeutic side effects. These experts help patients meet their nutritional demands, improving their health and treatment outcomes. Rehab and Hospice: Rehabilitation and palliative care teams must work together when liver cancer is advanced or untreatable. These experts manage symptoms, pain, and end-of-life care. The multidisciplinary team educates patients and their families about disease, treatment, self-care, and lifestyle changes. Healthcare professionals can collaborate to deliver consistent and accurate information, encouraging patients to actively participate in their treatment and make educated decisions. 105
Palliative care encompasses interventions that do not attempt to cure HCC but rather to improve patients’ symptoms, lengthen their lives, and enhance their quality of life. In order to track the disease's course and guarantee the best possible outcomes, these treatments frequently require significant follow-up care. 106 The following are examples of commonly used palliative care approaches for HCC:
The most effective treatment for HCC is still surgical resection. 107 The perioperative mortality rate is now around 5% because of improvements in surgical technologies. Despite advances in postoperative recurrence rates and survival, the 5-year survival rate for HCC patients are still only approximately 50%. When choosing patients for partial hepatectomy, baseline hepatic function is a key factor. Patients are evaluated and peri- and post-operative mortalities are predicted using the Child-Pugh-Turcotte grading system. Patients are classified as Child A, B, or C based on the severity of abnormalities in measures such as albumin, bilirubin, prothrombin time, ascites, and encephalopathy. Extrahepatic lesions, remaining residual liver size, and surgical team experience are also important in assessing resectability. Cirrhosis raises the risk of bleeding during surgery because it causes thrombocytopenia, coagulopathy, and varices, all of which contribute to increased perioperative mortality and postoperative morbidity.107,108
In transarterial chemoembolization (TACE), chemotherapeutic drugs are combined with an embolic substance and delivered directly into the hepatic artery, where they specifically target tumors in the liver while sparing healthy tissue. 109 Tumor necrosis can be produced by cutting off blood flow to the tumor from the hepatic artery, either partially or totally. The basic objective of TACE is to provide a highly concentrated dosage of chemotherapy to tumor cells while sparing healthy liver tissue. There have been large increases in long-term survival, however, these results have not been confirmed by randomized trials. Multiple trials contrasting TACE with untreated controls have failed to find a statistically significant difference in mortality rates between the two groups. However, despite substantial research, systemic chemotherapy for HCC has been proven to have a lower than expected response rate and no survival benefit compared to supportive treatment alone. 110 An emerging modality for the management of HCC is selective internal radiation therapy (SIRT). 111 Delivering large doses of radiation to the tumor while protecting normal liver tissue, this method entails injecting radiolabeled microspheres directly into the arteries supplying the tumor. Studies using these microspheres for SIRT have shown one-year survival rates of roughly 63%, which is on line with what has been shown using TACE. Mild stomach pain, nausea, and fever are the most commonly reported adverse effects of SIRT. These symptoms are also commonly seen after other localized treatments, such as TACE. 112 With its ability to effectively inhibit tumor growth while sparing healthy liver tissue, SIRT is a viable therapeutic option for HCC. The appropriateness of SIRT as a therapy option must be determined on a case-by-case basis, taking into account circumstances unique to each patient.
Natural Products
Many dietary natural products, including grapes, black currant, plum, pomegranate, cruciferous veggies, French beans, tomato products, asparagus, cloves of garlic, the spice turmeric, ginger, soy, rice bran, as well as some edible macro fungi, may be useful in the prevention and treatment of liver cancer, according to mounting data. These dietary natural products and their active ingredients may have a variety of impacts on the onset and course of liver cancer, including preventing the growth and spread of tumor cells, shielding the liver from carcinogens, immunomodulating the body, and boosting the effects of chemotherapy. These dietary supplements, including their active ingredients, may have a variety of impacts on the onset and course of liver cancer, including preventing the growth and spread of tumor cells, shielding the liver from carcinogens, immunomodulation of the body, and boosting the effects of chemotherapy. Specific focus areas should include the separation of active compounds, the demonstration of action mechanisms, bioavailability, possible toxicity, and undesirable effects. 113
Oxidative assault has been shown to be a significant factor in the development of hepatocarcinogenesis. Among the proven effective treatments for chronic hepatitis, iron depletion therapy, glycyrrhizin, ursodeoxycholic acid, vitamin E, Sho-Saiko-To, and glycyrrhizin may all be classified as antioxidant therapies. It is predicted that these treatments have some anti-inflammatory effects. However, only hepatitis has shown that these therapies can prevent cancer to this point. However, these treatments have been shown to have anti- inflammatory and anti-fibrotic properties in other liver illnesses, suggesting that they could be useful in preventing cancer. In Table 2, we discuss several herbal plants and their phytoconstituents, highlighting their mechanisms of action against HCC.
Natural Compounds Against HCC.

Conclusion
In conclusion, HCC serves as a significant subtype of liver cancer that emerges from hepatocytes. Tumor growth, angiogenesis, invasion, and metastasis are all aided by cytokines, which play a pivotal role in the onset and course of HCC. Cytokines and growth factors such as IL-6, TNF-α, IL-10, IL-17, VEGF, and TGF-β are essential inflammatory cytokines in liver cancer. Besides, growth factors play a role in the aggression of HCC. Recognizing consequently why there is geographical diversity in HCC incidence rates requires knowledge of the distribution of certain risk variables. Preventative measures, early diagnoses, and effective treatment are crucial for reducing the global impact of HCC. Vaccination against HBV, antiviral therapy for HBV and HCV, public awareness campaigns regarding the dangers of alcoholism and aflatoxin exposure, and encouragement of healthy lifestyles all fall under this category. Patients with liver disorders such as HCC require intensive nursing care. Integral parts of this method are putting the patient first, tailoring care to each person's needs, working together with specialists, using scientifically proven methods, and constantly assessing and bettering the care provided. The best care for patients with liver cancer can be provided by nurses when they combine effective therapeutic techniques with high-quality nursing modalities. Insight into the molecular cascades and genetic changes that lead to HCC, as well as the connection between liver cirrhosis, dysplastic nodules, and HCC, can lead to better ways for preventing and treating the illness. Better patient outcomes can be achieved through the examination, diagnosis, and treatment of HCC if healthcare providers from different fields work together. Besides, many dietary natural items and antioxidants may be useful in the protection or management of liver cancer, according to mounting data. Among them, the ones that follow are noteworthy for their possible anti-hepatoma capabilities, and hence in the future, further research is needed to determine the therapeutic effectiveness of dietary natural products and their bioactive components.
Footnotes
Abbreviations
Acknowledgements
The authors are highly thankful to people who provided support to complete this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Interventional Radiology, Harbin Medical University Cancer Hospital, Harbin Heilongjiang, 150080, China (grant number NA).
