Abstract
Objective
This study presents an analytical and biological evaluation of selected vegetable oils and their unsaponifiable compounds, which are of crucial importance for the cosmetic and pharmaceutical industries, with the aim of ensuring their targeted use in dermal products.
Methods
Vegetable oils were selected based on their dominant fatty acid composition: pomegranate seed oil (punicic acid), linseed oil (α-linolenic acid), coconut oil (lauric acid), olive oil (oleic acid), poppy seed oil (linoleic acid), linden seed oil (linoleic acid), and marigold seed oil (α-calendic acid). Pharmacopoeial chemical values, fatty acid profiles, and unsaponifiable compounds were analysed. Antioxidant activity was measured using DPPH assay.
Results
Chemical values indicated good oxidative status and minimal hydrolysis. Fatty acid analysis revealed expected compositions, with rare linden and marigold seed oils exhibiting distinctive fatty acid composition, featuring sterculic and α-calendic acids. Marigold seed oil showed the richest profile of unsaponifiable compounds. Antioxidant activity was highest in olive oil and lowest in coconut oil. No significant correlation was found between the fatty acid composition, unsaponifiable matter, and antioxidant activity suggesting the synergistic or antagonistic interactions of oil components.
Conclusion
The study highlights the complex interactions among oil components that influence antioxidant activity. These findings deepen the understanding of the complex mechanisms determining the antioxidant properties of vegetable oils, crucial for informed decisions in dermal product formulation as demand for natural, effective cosmetic and pharmaceutical ingredients rises.
Keywords
Introduction
Vegetable oils play a very important role in the cosmetic and pharmaceutical industries. Chemically, they are mainly triglycerides with saturated (eg lauric, myristic, palmitic, and stearic acids), monounsaturated (eg oleic acid) and polyunsaturated (eg linoleic and linolenic acids) fatty acids. A small part in their composition (generally up to 1%) consists of unsaponifiable compounds, eg phenols, squalene, carotenoids, tocopherols, and phytosterols. 1
In terms of biological activity, both the triglyceride fraction and unsaponifiable compounds have positive effects on the skin. Vegetable oils have shown promising results in in vitro and in vivo studies in wound healing, atopic dermatitis, psoriasis, xerosis, UV-B induced erythema, molluscum contagiosum, tungiasis, scars, and striae, through their antimicrobial, anti-inflammatory, and antioxidant properties, by promoting cell proliferation, increasing collagen synthesis, stimulating dermal reconstruction, and repairing the lipid barrier function of the skin. In particular, oleic, linoleic, and linolenic fatty acids have been shown to play an important role in the synthesis of inflammatory or anti-inflammatory mediators, and are also important structural components of the lipid barrier. Medium-chain saturated fatty acids, such as lauric and myristic acids, have an antimicrobial activity.1, 2
Unsaponifiable compounds have been proven to act in vitro as antioxidant, anti-inflammatory, antitumour, immunomodulatory, and antimicrobial agents, to have antinociceptive effects and to promote wound-healing activity. Reviewed in vivo studies indicate that unsaponifiable compounds contribute significantly to the overall dermal effects of vegetable oils, act as anti-inflammatory and antitumour agents, improve the biomechanical properties of the skin (moisturize and improve elasticity), reduce transepidermal water loss (TEWL) and blood flow in the SLS-treated skin, and influence the wound healing process.1, 3
In addition to the composition and content of fatty acids and unsaponifiable compounds, the stability or quality of an oil is also very important to ensure the effectiveness and safety of the oil's use. The quality of vegetable oils as raw materials is mainly affected by oxidation, which leads to changes in organoleptic properties, such as changes in taste, odour, and color, changes in viscosity and solubility, and changes in the contents of essential fatty acids and vitamins. All this leads to reduced sensory and processing quality. 4 Factors that negatively affect the oxidative status of vegetable oils include temperature, oxygen (air), UV light, and metal ions, while antioxidants (natural or artificial) typically reduce oxidative susceptibility. 4
The primary structural elements of oxidation are unsaturated fatty acid chains. Hydroperoxides, which are the first products of lipid oxidation, do not significantly affect the sensory quality of the oil, yet they are unstable and soon transform into secondary oxidation products (ketones, aldehydes, alcohols, lactones, hydrocarbons, esters, etc) with a very low odour and taste threshold, and the degradation of the oil becomes very evident. Antioxidants, which are naturally present as the compounds of the unsaponifiable matter, eg tocopherols, carotenoids, and phenolic compounds, are known to contribute significantly to oxidative stability and slow down the rancidity process. 4
While there is a considerable number of studies dealing with oil oxidation and antioxidant activity, significantly less knowledge exists about the direct correlation between the exact composition of fatty acids and unsaponifiable compounds, and antioxidant activity. Therefore, the main objective of this research was the detailed analytical evaluation of selected vegetable oils and their unsaponifiable compounds, including the determination of pharmacopoeial chemical values (acid, iodine, peroxide, saponification, hydroxyl value, and ester values), the fatty acid composition of triglycerides and composition of unsaponifiable compounds, and the evaluation of antioxidant activity as a parameter of potential biological activity. The oils were selected based on their fatty acid composition, with one characteristic fatty acid present as the dominant fatty acid. These were (the characteristic fatty acid is written in brackets): pomegranate seed oil (punicic acid), linseed oil (α-linolenic acid), coconut oil (lauric acid), olive oil (oleic acid), and poppy seed oil (linoleic acid). In addition, two lesser-known oils were extracted, ie linden seed oil (characterized by linoleic acid) and marigold seed oil (characterized by α-calendic acid).
Results and Discussion
Extraction of Marigold and Linden Seed Oils
The extraction yield for linden seeds was 14.8%. This corresponds well to the result of a previous study in which the oil yield averaged 14.1%, 5 but is significantly higher than the yield determined by Siger et al (2.0%). 6 Marigold seeds gave a yield of 13.0%. This fits into the lower range of yields observed in a study of different genotypes of marigold originating from the Czech Republic, Germany, Italy, Latvia, Poland, and Ukraine, where Dulf et al 7 found yields between 13.6% and 21.7%. They stressed the importance of breeding when considering marigold seeds for industrial, nutraceutical or pharmacological purposes. An even higher yield of 26% was determined by Saleem et al. 8
Based on the oil content obtained, both plant species can be described as low-yielding oilseed crops compared to other commercially important oilseeds, such as poppy seeds (50%), linseed (30-45%), coconut seeds (30%), hazelnut seeds (60%), peanut seeds (50%), and macadamia seeds (75%). 9
Pharmacopoeial Chemical Values
The chemical values either corresponded to defined pharmacopoeial values (coconut, linseed, and olive oils) or the values were comparable with the results of previous studies (Table 1).
Chemical Values, and Content of Unsaponifiable Compounds.
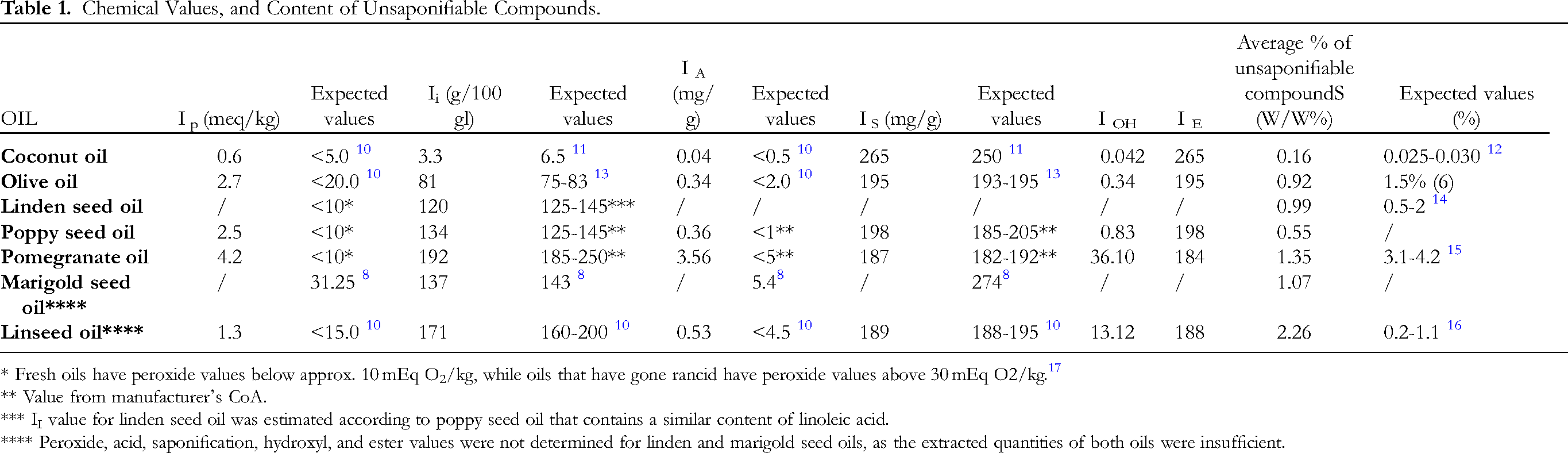
* Fresh oils have peroxide values below approx. 10 mEq O2/kg, while oils that have gone rancid have peroxide values above 30 mEq O2/kg. 17
** Value from manufacturer's CoA.
*** II value for linden seed oil was estimated according to poppy seed oil that contains a similar content of linoleic acid.
**** Peroxide, acid, saponification, hydroxyl, and ester values were not determined for linden and marigold seed oils, as the extracted quantities of both oils were insufficient.
In terms of overall oil quality, the most important chemical values are the peroxide value, which is a measure of oxidative spoilage, and the acid value, which is a measure of oil degradation/hydrolysis. The oils had a low peroxide value in the range of 0.6-4.2 meq/kg, which indicates a good oxidative status. Acid values were also low (0.04-3.56 mg/100 g), which indicates that the hydrolysis of ester bonds did not occur to a significant extent, resulting in a minimal free fatty acid content.
Two other chemical values primarily reflect the composition of the oil; the iodine value is a measure of the degree of unsaturation, while the hydroxyl value is a measure of the content of hydroxyl groups. The iodine value was highest in pomegranate seed oil (192 g/100 g), which was to be expected and is due to the high content of punicic acid, a three-times unsaturated fatty acid unique to this oil. In contrast, the lowest iodine value (3.3 g/100 g) was found in coconut oil, which contains mainly saturated fatty acids. The iodine value of marigold seed oil was 137 g/100 g, which correlates well with data found in literature, 8 while the iodine value of linden seed oil was 120 g/100 g which agrees with the value we estimated based on the linoleic acid content.
Fatty Acid Composition
The composition of the fatty acids in individual vegetable oils is summarized in Table 2. The fatty acid composition of coconut, olive, poppy, pomegranate, and linseed oils, which are widely used and studied vegetable oils, largely corresponded to data found in literature.1, 2 A slightly greater variability was found in the fatty acid composition of pomegranate seed oil, which contained a considerable amount of gondoic acid (10.3%) which is typically found in much lower concentrations (0.43-0.56%) in pomegranate seed oil.18, 19
Fatty Acid Composition.

To the best of our knowledge, the current study is one of the few available studies focusing on two rare oils, ie linden and marigold seed oils. Linden seed oil contains mainly polyunsaturated fatty acids: approximately 77% and 55% total polyunsaturated fatty acids were reported by Dowd & Farve 20 and Siger et al, 6 respectively, and 83-89% in our previous study. 5 The tested sample contained predominantly linoleic acid (about 60%), while the other two most represented fatty acids were oleic acid (23.5%) and palmitic acid (7.7%). The obtained results are comparable with previously published data found in literature.5, 6, 20 Characteristic of linden seed oil is the presence of cyclopropene fatty acids; sterculic acid typically reaches the highest content among them. In this study, sterculic acid was determined to be 3.1%, which is slightly lower than previously determined,5, 20 and close to the results of Siger et al. 6 These are the only available published scientific works that give us an insight into the fatty acid composition of linden seed oil. In terms of its general fatty acid composition, linden seed oil is close to poppy seed oil.
Similarly, the composition of marigold seed oil was found in only two scientific articles.7, 8 The fatty acid characteristic of marigold seed oil, a rare octadecatrienoic acid also known as α-calendic acid, was reported to reach a content of 51.47-57.63%. 7 Α-calendic acid was also confirmed in an early study by Saleem et al 8 in 1984, but at a very low content of 4.5%. We assume that this value could be inaccurate due to methodological issues, as the reference standard of α-calendic acid was not used. In our study, the tested sample contained a significant amount of α-calendic acid (61.3%), followed by linoleic acid (27.2%) and oleic acid (6.8%), while other fatty acids were present in a content of less than 2%. Considering this fatty acid composition, marigold seed oil is a unique vegetable oil for which there are no alternatives.
In 2022, a comprehensive review of clinical studies investigated the therapeutic and cosmetic potential of vegetable butters and oils as active ingredients for dermal applications. 2 Findings related to the effects of individual fatty acids on the skin are summarized below.
The composition of vegetable butters and oils is intricately linked to the composition of skin lipids, with fatty acids mainly found in the stratum corneum and sebum. Free saturated fatty acids dominate the stratum corneum, primarily tetracosanoic (C24) and hexacosanoic acids (C26), constituting 39 molar% and 23 molar%, respectively. The only free unsaturated fatty acids are oleic (C18:1) and linoleic (C18:2) acids, accounting for approx. 6% and 2%, respectively, while the total monounsaturated free fatty acids account for approx. 20%. The human sebum is comprised of triglycerides (41%), waxes (25%), free fatty acids (16%), squalene (12%), cholesterol and cholesterol esters (4%), and vitamin E, all of which are also components (or structurally similar components) of vegetable butters and oils. The emolliency of triglycerides in vegetable butters and oils enhances skin hydration by reducing TEWL. These substances also exhibit antimicrobial, anti-inflammatory, and antioxidant effects, through the action of glycerides, free fatty acids, and unsaponifiable matter. Moreover, the dermal application of free fatty acids facilitates their penetration into the stratum corneum, enhancing the permeation of other substances. Oleic acid, prevalent in many vegetable butters and oils, was shown to act as a skin penetration enhancer, disrupting the skin barrier, increasing TEWL and causing irritation. Linoleic acid, an essential fatty acid, plays a crucial role in the cell membrane structure and lipid barrier homeostasis. Α-linolenic and γ-linolenic acids, though not structural components of the skin, contribute to the metabolism of polyunsaturated fatty acids. A deficiency in linoleic acid, but not α-linolenic acid, has been associated with skin dysfunctions such as dryness and inflammation. 2
Unsaponifiable Compounds
The content of isolated unsaponifiable compounds of individual oils is summarized in Table 1. Unsaponifiable compounds generally account for 0.3-2% of the total oil,1–3, 21 and this is consistent with our results (0.2-2.2%).
The tested samples showed a diverse composition of unsaponifiable matter (Table 3). The richest composition of unsaponifiable matter was found in marigold seed oil, which contained 12 different compounds. These were squalene, γ-tocopherol, cholesterol, campesterol, stigmasterol, β-sitosterol, lanosterol, cholest-7-en-3-ol, 9,19-cyclolanost-24-en-3-ol, and three other components that could not be identified with the available GC-MS libraries and reference standards. This was followed by linden seed oil, with 11 different compounds: squalene, γ-tocopherol, campesterol, stigmasterol, β-sitosterol, (3-β,5-α)-cholest-7-en-3-ol, 24-methylene-9,19-cyclo-(3-β)-lanostan-3-ol, an unidentified sterol, and two other unidentified components. In addition, β-sitosterol and squalene were present in all samples. Campesterol and stigmasterol were the most abundant of all phytosterols, but were both absent in olive oil.
Unsaponifiable Compounds in the Unsaponifiable Matter of a Vegetable oil (Content 1; in %) and Unsaponifiable Compounds in a Vegetable oil (Content 2; in %), and the Numbers of Identified and Unidentified Compounds.

A review article focusing on studies about the dermal effects of unsaponifiable compounds 3 shed light on the specific dermal effects of unsaponifiable compounds found in vegetable butters and oils. These compounds were categorized by their chemical structures, covering terpenic compounds, triterpene alcohols, phytosterols, tetraterpenoids (carotenoids), tocopherols and tocotrienols (vitamin E), phenolic compounds, waxes, and phospholipids. It was summarized that compounds such as phytol exhibit various effects, eg cytotoxic, anti-inflammatory, and anti-melanogenesis effects, and squalene antioxidant and wound-healing activities. Triterpene alcohols show anti-inflammatory and antimycobacterial activities. Phytosterols, especially β-sitosterol, demonstrate angiogenic and antioxidant properties, influencing keratinocyte proliferation and differentiation. Carotenoids, tocopherols, and tocotrienols contribute to antioxidant, photoprotective and anti-inflammatory effects. Phenolic compounds such as flavonoids and ferulic acid exhibit antioxidant, antibacterial and wound-healing activities. γ-oryzanol displays antioxidant properties and inhibits mast cell degranulation. Phospholipids, including lysophosphatidic acid, show promise in wound healing. 3
These results provide an important insight into the overall benefits of unsaponifiable compounds, as well as vegetable butters and oils themselves, when applied dermally, either independently or as components in therapeutic and cosmetic formulations. While the summarized results contribute to the understanding of potential skin benefits, caution should be exercised when extrapolating findings from in vitro and animal studies to the human skin. Further research, particularly in the area of dermal applications, is required to confirm this promising potential.
Antioxidant Activity of Oils and their Unsaponifiable Compounds
Compounds with an antioxidant activity are an integral part of the unsaponifiable matter and contribute significantly to the overall stability of vegetable oils. They therefore have a wide range of applications in cooking, cosmetics, and medical treatment. 9 Each oil is characterized by a distinct composition of antioxidants, resulting in different antioxidant activity or antioxidant potential.
The results of the antioxidant activity of the studied vegetable oils and their unsaponifiable compounds are presented in Figure 1. The results revealed a high variability in antioxidant potential. This observation is consistent with published literature, in which the antioxidant properties of vegetable oils have been extensively studied. 22 Specifically, olive oil (80.0%) showed the most pronounced antioxidant activity followed by linden seed oil (63.7%), while coconut oil (2.8%) had the lowest activity.
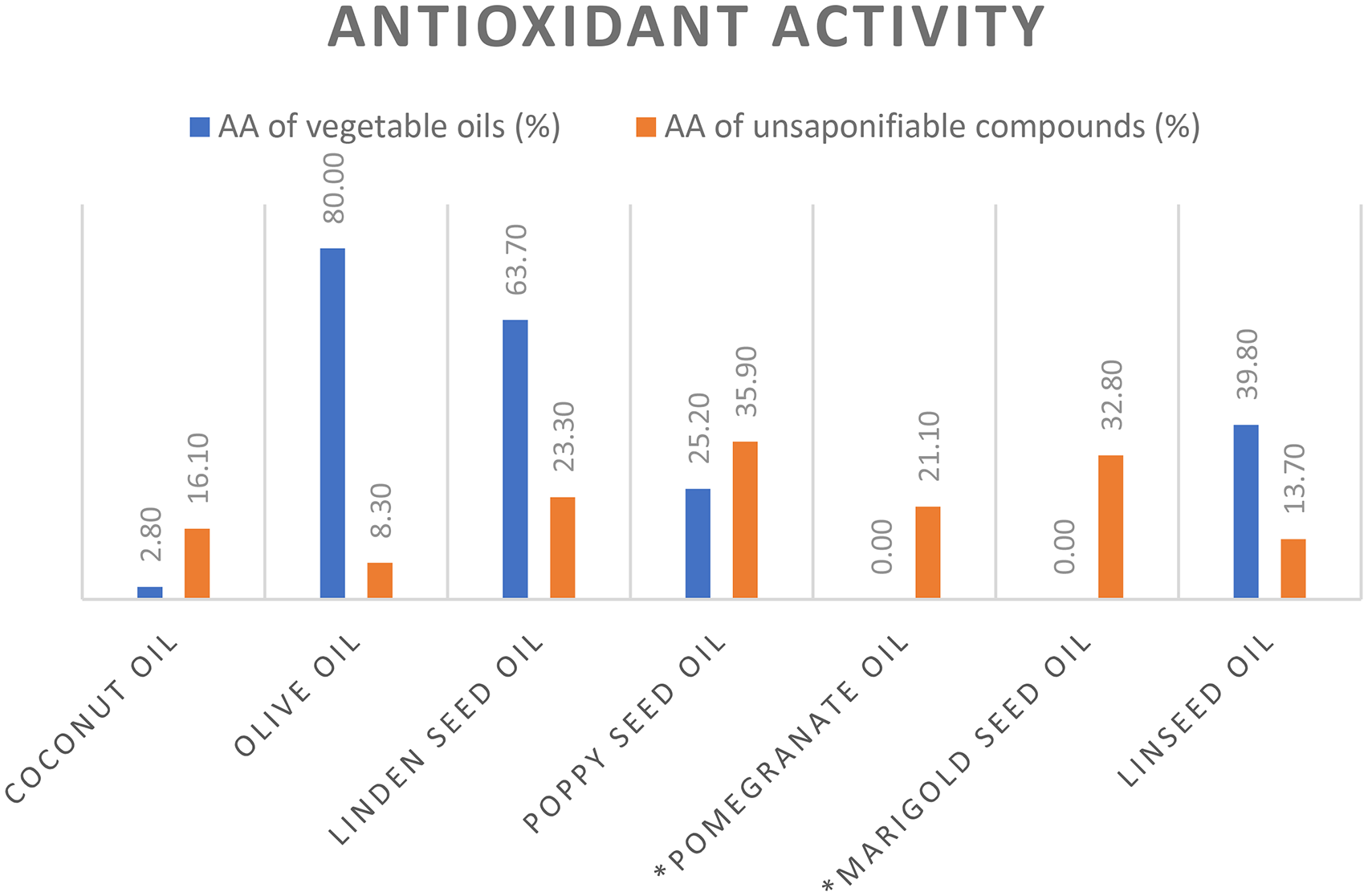
Antioxidant activity (AA) of vegetable oils and their unsaponifiable compounds. *The antioxidant activity of marigold seed oil could not be determined due to an insufficient amount of the oil, while the results for pomegranate seed oil could also not be obtained, as the measurement was hindered by the turbidity of the sample.
On the other hand, the highest antioxidant activity of the isolated unsaponifiable matter was found in poppy seed oil (35.9%) followed by marigold seed oil (32.8%), while the unsaponifiable matter from olive oil showed the lowest activity (8.3%). However, the antioxidant activity of α-tocopherol, which was used as a positive control, remained superior, reaching 80.5%.
The correlation between the fatty acid composition (% of an individual fatty acid in an oil), unsaponifiable compounds (% of the unsaponifiable matter in an oil), and antioxidant activity of an oil and the antioxidant activity of the unsaponifiable matter was then analysed statistically. No statistically significant correlations were found between the data. We therefore summarize that a high antioxidant activity of the isolated unsaponifiable matter does not necessarily translate to a high antioxidant activity of the vegetable oil. Our results suggest a more complex synergistic or antagonistic interplay between the components of a vegetable oil that ultimately characterizes its antioxidant potential. It would be interesting to further explore the complexity of these interactions.
Indeed, several studies have already dealt with this topic. In a comprehensive literature search, we found data on significant antioxidant activity (determined by the DPPH method) identified for prickly pear (Opuntia ficus-indica), sesame (Sesamum indicum), tomato (Solanum lycopersicum), date palm (Phoenix dactylifera), sour cherry (Prunus cerasus), black cumin (Nigella sativa), linseed (Linum usitatissimum), and niger (Guizotia abyssinica) seed oils. This was mainly attributed to the presence of phenolic compounds, tocopherols, and sterols.23–29
Another study showed that the addition of 0.02%, 0.05%, and 0.1% unsaponifiable compounds of sesame oil from unroasted and roasted seeds, respectively, to sunflower oil increased the oxidative stability of the oil in a concentration-dependent manner. Higher concentrations exhibited a higher antioxidant effect, which was determined as a decrease in peroxide values. The unsaponifiable compounds from roasted seeds had the strongest effect. 30 A study by Farhoosh et al 31 showed consistent results by demonstrating the stabilizing effect of the whole unsaponifiable matter of bene (Pistacia atlantica var. mutica) hull oil, eight fractions of the unsaponifiable matter, and α-tocopherol as a control on sunflower oil exposed to elevated temperatures. The highest stabilizing effect was observed for the fractions of the unsaponifiable matter, with no significant differences between them, followed by the whole unsaponifiable matter and α-tocopherol.
In addition, the results of the FRAP and DPPH methods also showed significant antioxidant activity of the unsaponifiable matter compared to α-tocopherol. However, the individual fractions behaved slightly differently in both tests. 31 Dhavamani et al demonstrated the antioxidant activity of palm (Elaeis guineensis) oil, olive (Olea europaea) oil, sunflower seed (Helianthus annuus) oil, rice bran (Oryza sativa) oil, sesame (Sesamum indicum) seed oil, and linseed (Linum usitatissimum) oil. The antioxidant activity decreased after removal of the unsaponifiable matter, 32 emphasizing their significant contribution to overall antioxidant activity. These results are also consistent with the observations of Wang et al that virgin camellia (Camellia oleifera), virgin hemp (Cannabis sativa), and virgin linseed (Linum usitatissimum) oils have higher antioxidant activity than refined oils.33, 34
Numerous studies have reported the important role of individual unsaponifiable compounds in enhancing antioxidant potential.30–32, 35–37 The antioxidant activity of the unsaponifiable compounds of Atlas pistachio (Pistacia atlantica var. mutica) seed oil correlated positively with avenasterol, 36 while the higher antioxidant activity of linden seed oils correlated with the higher content of γ-tocopherol. 5
In addition, in a study using refined sunflower oil, α-tocopherol, β-carotene, and epigallocatechin gallate, alone and in different combinations and ratios, were investigated. Among samples with different combinations and concentration ratios of these components, some exhibited synergistic effects, while others showed antagonistic effects. The highest antioxidant activity was found for epigallocatechin gallate followed by β-carotene and then α-tocopherol, while the sample with a 1:13.3 ratio of α-tocopherol and epigallocatechin gallate produced superior results. 38
Limitations
Several limitations were faced during the study. The volumes of marigold and linden oils were insufficient to determine all pharmacopoeial chemical values. Since the oils were freshly extracted and assumed to be in good oxidative condition, only the most essential tests were performed. The antioxidant activity test of calendula oil was not performed due to insufficient quantity. Additionally, during the DPPH test, we encountered difficulties measuring the absorbance of pomegranate oil due to sample turbidity, resulting in incomplete data for antioxidant activity results. It is also important to point out that in vitro tests were performed, and the results should be considered as a preliminary guide for further research. Generalization of these results is problematic due to the inherent biological variability.
Conclusion
A detailed analytical evaluation of selected vegetable oils and their antioxidant activity was conducted, with the aim of providing evidence-based insights for their use in dermal products. The study uncovered a probable complex synergism or antagonism among various oil components affecting antioxidant activity. The antioxidant strength of the isolated unsaponifiable fraction did not consistently align with that of the whole vegetable oil, suggesting complex interactions among all constituents. As demand rises for natural and effective cosmetic and pharmaceutical ingredients, these findings contribute to a deeper understanding of the interplay between oil composition and antioxidant effects, offering valuable insights for formulating dermal products with optimized stability and effectiveness. This study not only sheds light on the complex chemical profiles of various vegetable oils but also provides a foundation for the further extensive research of vegetable oil antioxidant activity, and with that, an informed selection and application of these oils in dermal formulations backed by scientific evidence.
Materials and Methods
Plant Material
The seeds of marigold (Calendula officinalis) were collected in Ljubljana, Slovenia. The seeds of linden (Tilia cordata) were collected in Kamenče, Slovenia. The seed samples were collected in 2019, in the phase of full ripeness, air-dried in a laboratory at room temperature, and stored in a dry and dark place until oil extraction.
Vegetable Oils
Coconut (Cocos nucifera) oil, cold pressed, organic (All Organic Treasures, Germany), linseed (Linum usitatissimum) oil, cold pressed, organic (Pečarič Oil Mill, Slovenia), olive (Olea europaea) fruit oil, cold pressed (the Poljšak family, Slovenia), pomegranate (Punica granatum) seed oil, cold pressed, filtered (All Organic Treasures, Germany), poppy (Papaver somniferum) seed oil, cold pressed, unrefined, and unfiltered (Pečarič Oil Mill, Slovenia)
Reagents
The following reagents were used: acetic anhydride 95% (p.a., Kemika, Croatia), anhydrous acetic acid 95% (JT Baker, Avantor, USA), anhydrous ethanol 96% (p.a., Carlo Erba, Italy), C15:0, α-calendic acid (8E,10E,12Z-octadeca-8,10,12-trienoic acid; Larodan AB, Sweden), campesterol (Carl-Roth, Germany), cholesterol, 99% (Fluka, Germany), cyclohexane 99.5% (p.a., Sigma-Aldrich, USA), diethyl ether 99% (Sigma-Aldrich, USA), DMSO (dimethyl sulfoxide; Sigma-Aldrich, USA), DPPH 95% (Fluka, Switzerland), ethanol 96% (Kefo, Slovenia), HMDS (hexamethyldisilazane) + TMCS (trimethylchlorosilane) + pyridine, 3:1:9, (Supelco, USA), hydrochloric acid 37% (Emsure (ACS, Reag. Ph. Eur.), Merck, Germany), iodine monochloride 98% (p.a., Sigma-Aldrich, USA), isooctane 95% (Emsure (ACS, Reag. Ph. Eur.), Merck, Germany), KOH 99,99% (Sigma-Aldrich, USA), methanol 95% and above (Carlo Erba, Italy), methanol 95% (Carlo Erba, Italy), n-hexane 95% (Panreac Quimica, USA), petroleum ether 90% and above (Sigma-Aldrich, USA), phenolphthalein 95% (Riedel-de Haën, Germany), potassium hydroxide 99.99% (Sigma-Aldrich, USA), potassium iodide 99.5–100.5% (Emsure (ISO, Reag. Ph Eur), Merck, Germany), purified water (Faculty of Pharmacy, University of Ljubljana, Slovenia), pyridine 99% (p.a., Merck, Germany), salicylic acid 99.5–100.5% (Farmalabor, Italy), β-sitosterol, 80% (Carl-Roth, Germany), sodium hydroxide 95% (Riedel-de Haën, Germany), sodium thiosulphate anhydrous 99.5% (Riedel-de Haën, Germany), squalene, 98% (Sigma-Aldrich, USA), standard mixture of methyl esters of fatty acids F.AME. Mix C4-C24 (Supelco, USA), starch 95% (p.a., Kemika, Croatia), stigmasterol, 95% (Carl-Roth, Germany), HMDS + TMCS + pyridine (3:1:9) (Sylon HTP, Supelco, Merck, Germany), α-tocopherol, 95.5% (Sigma-Aldrich, USA).
Oil Extraction
The oil extraction from marigold and linden seeds was carried out, as previously described. 5 Briefly, linden fruit husks were removed and the seeds separated. Dehulled linden seeds and whole marigold seeds were then ground separately into powder. Ultrasound-assisted extraction with hexane was performed for 10 min; herbal drug to solvent ratio = 1:3. The supernatant was filtered, the hexane was evaporated and the extraction process was repeated three more times. The oils obtained from all four extractions were combined and cooled in an inert atmosphere to prevent oxidative degradation until the analyses were performed.
Pharmacopoeial Chemical Values
The procedures for the determination of the peroxide value (method B), iodine value (method B), acid value, saponification value, hydroxyl value (method A), and ester value were carried out according to the instructions of the European Pharmacopoeia 10.0, and the final results were calculated according to the corresponding equations. 10
In Situ Derivatization of Fatty Acids and Determination of the Fatty Acid Composition Using GC-MS
Derivatization was performed as previously described. 5 Briefly, 10 μL of dichloromethane and 200 μL of 0.5 M NaOH/MeOH were added to 10 mg of an oil sample and mixed. After heating in a water bath (90 °C, 10 for minutes) and cooling, 200 μL of 14% BF3/MeOH were added, and then heated again (10 min). 200 μL of distilled water and 1 mL of hexane were added, and the mixture was shaken vigorously by hand (1 min), and then left for phase separation. The upper, organic phase was used for GC-MS analyses.
Chromatographic Conditions
System: GCMS-QP2010 Ultra (Shimadzu Corporation, Kyoto, Japan); column: non-polar capillary, Rxi-5Sil MS, 30 m × 0.25 mm i.d.; df = 0.25 µm; SF: 1,4-bis (dimethylsiloxy) phenylenemethylpolysiloxane (Restek, Bellefonte, Pennsylvania, USA); computer program: GC-MS Solution 4.2 (Shimadzu Corporation, Kyoto, Japan); carrier gas: helium (99.99%); gas flow: 1 mL/minute (linear velocity); injection method: “split” 1:100; injector temperature: 250 °C; ion source temperature: 200 °C; interface temperature: 300 °C; injection volume: 1 µL; voltage on the detector: 1 kV; ionization mode: EI; ionization energy: 70 eV; data acquisition frequency: 5 Hz; relative molecular weight measurement range (m/z): 40-400; temperature program: 40 → 300 °C (3 °C/minutes), 300 °C (5 min); total analysis time: 91.7 min.
Individual fatty acids were identified based on a comparison of their mass spectra and retention indices with those of the synthetic compounds in the spectral library of the National Institute of Standards and Technology (NIST14), as well as the Flavors and Fragrances of Natural and Synthetic Compounds spectral library (FFNSC3). The content (in %) was determined by comparing the area under the curve for the chromatographic peak of an individual fatty acid with the area under the curve for the peak of the reference compound of known concentration.
Isolation, Derivatization and Identification of Unsaponifiable Compounds Using GC-MS
Isolation was performed according to the instructions of Ph. Eur. 10.0. 10 In the final step, the Ph. Eur. 10.0 test for the detection of acids in isolated unsaponifiable matter was performed to verify the efficiency of the isolation of unsaponifiable compounds from the oil. 10 Only one isolation was carried out for marigold and linden seed oils, as the quantity of the two oils was limited. For the other oils, two replicates were combined and the third was used to verify the efficiency of the isolation with the acid detection test.
The derivatization of the unsaponifiable compounds was performed by silylation, as previously described. 5 Briefly, 50 μL of the Sylon reagent were added to 5 mg of a sample, stirred well, and centrifuged (13,000 rpm, 5 min). The mixture was then heated in a water bath (60 °C, 30 min) and cooled. 950 μL of heptane were added, mixed, and centrifuged as before. The supernatant was used for GC-MS analyses. The reference compounds were prepared using the same procedure. Linear calibration curves were generated for each reference compound and used to calculate the content of each unsaponifiable compound in the samples.
Chromatographic Conditions
System and general conditions as above, temperature program: 50 → 270 °C (12 min), 270 °C (55 min), total analysis time: 67.0 min.
Individual unsaponifiable compounds were identified based on a comparison of their mass spectra and retention indices with those of the synthetic compounds in the spectral library of the National Institute of Standards and Technology (NIST14), as well as the Flavors and Fragrances of Natural and Synthetic Compounds spectral library (FFNSC3). The content of each unsaponifiable compound was calculated using a calibration curve. The equations are shown below.

Antioxidant Activity of Oils and their Unsaponifiable Compounds
Antioxidant activity (AA) was evaluated using a DPPH (2,2-diphenyl-1-picrylhydrazyl) method. DPPH was dissolved in methanol at a concentration of 0.04 mg DPPH/mL. The solvent (MeOH) was used as a blank control. Samples were prepared by dissolving 100 mg of an oil or 1 mg of an unsaponifiable fraction in 1 mL of the solvent. 500 µL of each oil sample, unsaponifiable fraction sample or controls were mixed with 2 mL of the DPPH solution. After 30 minutes of incubation in the dark, absorbance was determined spectrophotometrically at 517 nm (Spectrophotometer UV/VIS, Nanocolor UV/VIS, Macherey-Nagel, Germany). Three measurements were performed for each sample and the average absorbance was calculated. The following equation was used.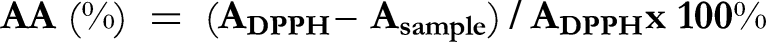
Statistical Analysis
The correlation between the fatty acid composition (% of an individual fatty acid in an oil), unsaponifiable compounds (% of the unsaponifiable matter in an oil), and the antioxidant activity of an oil and the antioxidant activity of the unsaponifiable matter was analysed statistically using the JASP 0.17.1.0 program (University of Amsterdam, The Netherlands) and Pearson correlation coefficients were determined.
Footnotes
Acknowledgments
We gratefully acknowledge the skillful laboratory assistance of Mateja Novak and Kaja Podvratnik, and the assistance of Assist. Dr Petra Ratajc with statistical analysis.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article.
Funding
This work was funded by the Slovenian Research Agency, grant number P1-0208.
Javna Agencija za Raziskovalno Dejavnost RS, (grant number P1-0208).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
