Abstract
Objective
Essential oils extracted from Syzygium species are known for various pharmacological purposes. Syzygium fluviatile (Hemsl.) Merr. & L.M.Perry is a flowering plant found in China and Vietnam. The current study aims to offer a comparison of chemical compositions in essential oils of S. fluviatile fruits and leaves, collected from five regions of Vietnam. The obtained oils were also taken into antimicrobial consideration, which was further aided by in silico approaches.
Methods
Phytochemical analysis of essential oils was carried out using GC-FID/MS (gas chromatography-flame ionization detection/mass spectrometry) analysis. An antimicrobial assay was performed using broth micro-dilution for in vitro screening. In silico considerations are mainly based on docking studies and toxicity assessments using the AutoDock Vina v1.2.3 program and the ProTox 3.0 web server, respectively.
Results
Hydro-distillation of S. fluviatile fresh fruits and leaves can lead to the production of yellow essential oil with yields of 0.21–0.32% v/w. In general, the obtained oils were dominated by monoterpene and sesquiterpene derivatives, as well as (E)-caryophyllene (8.40-47.12%) being the principal compound. The oil samples showed strong antimicrobial activity against the Gram (+) bacteria Enterococcus faecalis ATCC51299, Staphylococcus aureus ATCC29213, and Bacillus cereus ATCC11778, and the yeast Candida albicans ATCC 60193 with the MIC and IC50 values 16–64 µg/mL and 5.12–24.68 µg/mL. Docking results indicated that (E)-caryophyllene exhibited binding affinities from −6.728 kcal/mol to −5.729 kcal/mol with important amino acid residues in the DNA gyrase, PBP3, and SAP2 targets. The toxicity profile of (E)-caryophyllene is also discussed.
Conclusion
The isolation of (E)-caryophyllene from Vietnamese Syzygium essential oils as a purified compound is necessary. In vivo antimicrobial studies and molecular mechanisms of action are needed.
Introduction
The genus Syzygium contains about 1800 species of flowering plants in the family Myrtaceae. Its native regions range from Asia to Africa, Madagascar, and the Pacific islands. 1 The fruits of some species can be eaten fresh or used in jelly and jam. 1 Specifically, S. aromaticum, also known as the clove, plays an important role in food chemistry, and pharmacological aspects. 2 Phytochemical studies on Syzygium plants resulted in the isolation and determination of various phytochemical classes, but terpenoids and phenolics are predominant. 1 Syzygium constituents are also known for their pharmacological values in the treatment of diseases, such as anticancer, antioxidant, anti-inflammatory, antimicrobial, antidiarrheal, and hepatoprotective activities. 1
In another aspect, Syzygium plants have been recognized as a good reservoir of essential oils, and monoterpenes, sesquiterpenes, and their derivatives are the main constituents. 3 The obtained values of Syzygium essential oils in pharmacology are related to their actions to control bacteria, cancer, inflammation, insects, etc3–5 For instance, eugenol is recorded to account for 50% at least in the clove essential oil. 4 Antioxidant and antibacterial activities of the essential oil of S. cumini leaves might be due to the abundance of α-pinene (32.32%), β-pinene (12.44%), and trans-caryophyllene (11.19%). 6 The leaf essential oil of S. myrtifolium containing three main compounds δ-cadinol (29.53%), caryophyllene oxide (26.25%), and cyclocolorenone (7.7%) showed anti-inflammatory activity in lipopolysaccharide-activated RAW 264.7 macrophage cells via the inhibition of nitric oxide production. 7
Syzygium fluviatile has been found only in China and Vietnam.8–10 Chromatographic separation indicated the presence of phlorogucinols and terpenoids in its twigs and leaves.8, 9 To date, there has been no report on identifying chemical compounds in the essential oils of this species. In this study, we first describe a chemical analysis of essential oils from its fruits and leaves, collected from five different locations in Vietnam. The obtained essential oils were also taken into consideration for their antimicrobial ability. Experimental results were further aided by in silico approaches.
Materials and Methods
Plant Materials
The fresh fruits and leaves have been collected from different regions of Vietnam. The yields of extraction, colors, voucher specimens, and coordinates are outlined in Table 1 and Fig. 1. All samples were collected in November 2021. The botanical identification was carried out by co-author Do Ngoc Dai, and the voucher specimens were deposited in the plant herbarium department of Nghe An University of Economics. The fresh material (2.0 kg, each sample) was subjected to hydro-distillation using a Clevenger-type apparatus for 3.0 h. The obtained essential oils were dried over Na2SO4 and maintained in small sealed vials at 5 oC before further analysis. The yield (fresh weight/volume-w/v) was calculated by an arithmetic mean value in triplicate (Table 1).

The collection regions of Syzygium fluviatile in Vietnam.
Plant Collection and Hydro-Distillation Details of Five Vietnamese S. fluviatile Samples.

The GC-FID/MS (gas Chromatography-Flame Ionization Detection/Mass Spectrometry) Analysis
Chemical constituents in essential oils were analyzed using the GC-FID/MS analysis.11–13 The GC-FID was performed on an Agilent Technologies HP 7890A Plus Gas chromatograph (USA) coupled with the FID detector, and HP5-MS column (column dimension of 30 m×0.25 mm and a film thickness of 0.25 μm). The GC was run under a setting condition of the carrier gas He (flow rate of 1.0 mL/min), injector temperature (250 °C), and detector temperature (260 oC). The column rises from 55 °C (with 2.5 min hold isothermally) to 220 °C (held for 9 min) at 4 °C/min. Essential oil (1.0 μL) was injected singly at a split ratio of 9:1. The inlet pressure was 6.0 kPa. Quantification was performed using an external standard approach utilizing calibration curves established by doing the GC analysis of sample chemicals.
Regarding the GC/MS analytical procedure, a mass spectrometer HP 5973 was interfaced with the GC using the HP5-MS column (30 m×0.25 mm, film thickness 0.25 μm). Furthermore, the GC analytical parameters were the same as previously mentioned.11–13 An ionization voltage of 70 eV and an emission current of 40 mA were the operating conditions of the mass spectrometer. At a sampling rate of 1.0 scan/s, the mass spectra were obtained within a scan mass range of 40–450 amu. The GC-MS spectrum was used to identify chemical compounds in essential oils. This was also carried out by comparing their retention indices (RI) with homologous series of n-alkanes (C7-C30). Chemical structural identification has been matched with the W09N08 library, Adams book, 14 and NIST Chemistry WebBook. 15
Antimicrobial Assay
Microbial strains used in this study consist of three Gram (+) bacteria Enterococcus faecalis ATCC51299, Staphylococcus aureus ATCC29213, and Bacillus cereus ATCC11778, three Gram (
Molecular Docking
The crystal structures of DNA gyrase B (PDB ID: 3G7B) and penicillin-binding protein 3 (PBP3, PDB ID: 3VSL) from S. aureus, and secreted aspartic proteinase (SAP2, PDB ID: 1EAG) from C. albicans were retrieved from the RCSB Protein Data Bank (https://www.rcsb.org/).17–19 The protein files were prepared using AutoDockTools software by adding missing polar hydrogen atoms, removing water molecules, and computing Kollman partial charges.20, 21 The chemical structure of (E)-caryophyllene was drawn using Marvin JS software, and energy optimized with MMFF94 s force field using Avogadro software. Subsequently, this compound and the selected proteins were prepared as the PDBQT files for docking program input using AutoDockTools. The grid box parameters were set based on the active site of the specific proteins under study: 3G7B (X = 50.613, Y = –3.651, Z = 19.927 and X x Y x Z = 24 × 24 × 24), 3G7B (X = 50.905, Y = –31.733, Z = 25.613 and X x Y x Z = 24 × 24 × 24), 1EAG (X = 41.899, Y = 25.601, Z = 11.368 and X x Y x Z = 24 × 24 × 24), and the exhaustiveness parameter was set to 400. All molecular docking processes were performed using AutoDock Vina v1.2.3, and molecular interaction analysis was performed using Discovery Studio Visualizer software. 22
Toxicological Profile
To evaluate the toxicity, the ProTox 3.0 web server (prediction of toxicity of chemicals) was utilized. 23 The chemical structure of (E)-caryophyllene was converted into SMILES format for input into this web server using OpenBabel software. 24 Then, the toxicity of this compound and the positive controls were elucidated, including the LD50 value, toxicity class, and organ toxicity (hepatotoxicity, neurotoxicity, nephrotoxicity, respiratory toxicity, and cardiotoxicity).
Statistical Analysis
Data are processed using Microsoft Excel and represented as Mean ± SD (Standard Deviation). The difference was statistically meaningful with p < 0.05.
Results and Discussion
Phytochemical Analysis
Hydro-distillation of the fresh fruits (the sample SF-1), collected from Dakrong Natural Reserve, gave a yellow essential oil with a yield of 0.21 (v/w, based on the fresh material). The GC-FID/MS analysis of this sample resulted in the identification of 49 compounds, which represented 90.05% (Table 2). Sesquiterpene hydrocarbons and their oxygenated derivatives were predominant at 23.97 and 61.00%, respectively. On the other hand, monoterpene hydrocarbons and their oxygenated derivatives were found to reach less than 3.00%. Besides, non-terpenic compounds occurred in a minor amount of 0.16%. As shown in Table 2, the major compounds included epi-cedrol (26.53%), caryophyllene oxide (9.75%), (E)-caryophyllene (8.40%), and spathulenol (6.95%). Other compounds were identified with more than 1.00%, such as α-cadinol (3.69%), α-humulene (2.89%), epi-α-cadinol (2.63%), and curzerenone (2.50%).
The Identified Compounds (%) in Essential Oils of S. fluviatile Samples.
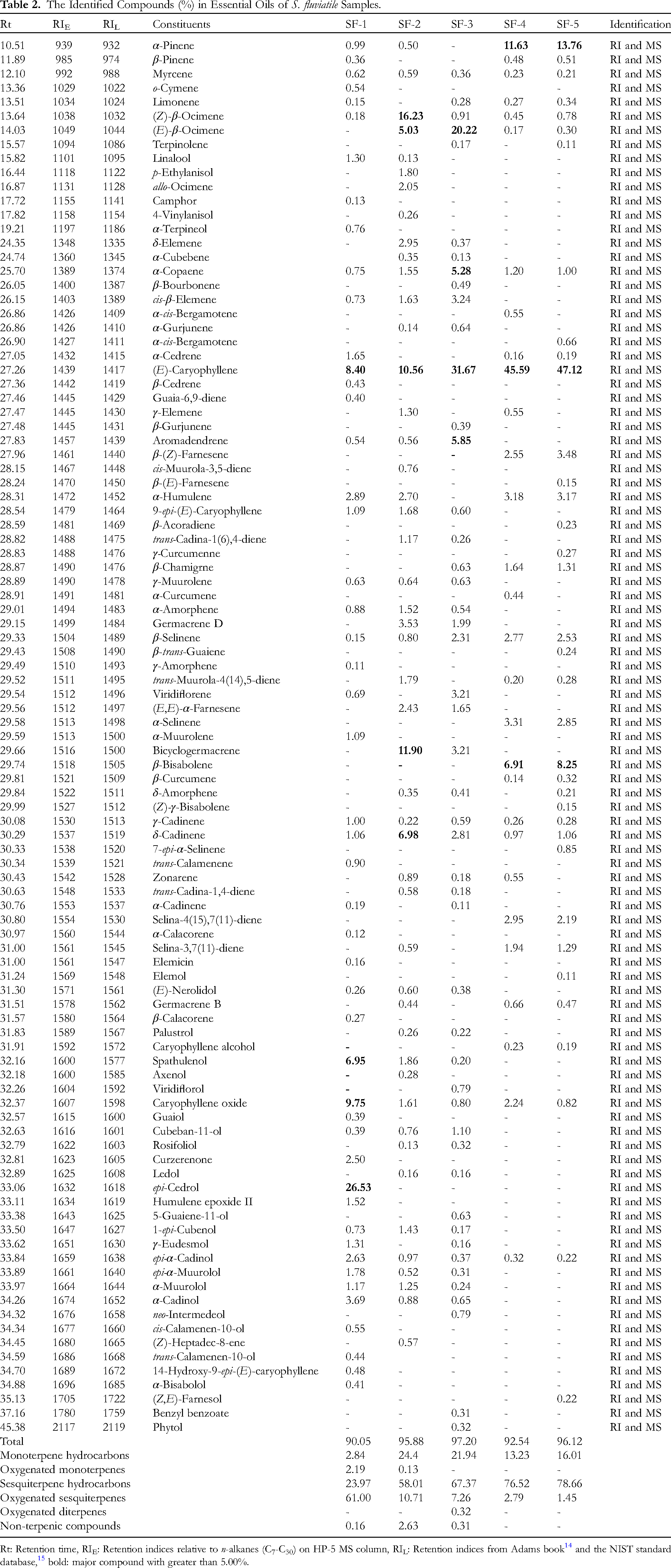
The sample SF-2 was collected from Pu Hoat Natural Reserve, and its yellow essential oil (0.26%, v/w) was extracted from the fresh leaves. A total of 48 compounds were identified, which accounted for 95.88%. The studied essential oil contained sesquiterpene hydrocarbons (58.01%), monoterpene hydrocarbons (24.4%), and oxygenated sesquiterpenes (10.71%). Oxygenated monoterpenes and non-terpenic compounds were present in trace percentages of 0.13 and 2.63%, respectively. The major compounds in this sample have encompassed (Z)-β-ocimene (16.23%), bicyclogermacrene (11.90%), (E)-caryophyllene (10.56%), δ-cadinene (6.98%), and (E)-β-ocimene (5.03%). Some compounds possessed the percentages exceeding 1.00%, such as germacrene D (3.53%), δ-elemene (2.95%), α-humulene (2.70%), (E,E)-α-farnesene (2.43%), and allo-ocimene (2.05%).
Hydro-distillation of the fresh leaves (the sample SF-3) collected from Vu Quang National Park also resulted in a yellow essential oil with a yield of 0.30, v/w. By the GC-FID/MS analysis, 48 compounds were identified, which was calculated to be 97.20%. This essential oil was characterized by sesquiterpene hydrocarbons (67.37%) and monoterpene hydrocarbons (21.94%). The remaining classes encompassed oxygenated sesquiterpenes (7.26%), oxygenated diterpenes (0.32%), and non-terpenic compounds (0.31%). The principal compounds were identified as (E)-caryophyllene (31.67%), (E)-β-ocimene (20.22%), aromadendrene (5.85%), and α-copaene (5.28%). Other compounds of note were cis-β-elemene (3.24%), viridiflorene (3.21%), bicyclogermacrene (3.21%), δ-cadinene (2.81%), β-selinene (2.31%), germacrene D (1.99%), and cubeban-11-ol (1.10%).
Considering the sample SF-4 collected from Pu Luong Natural Reserve, its yellow essential oil was obtained with a yield of 0.27% v/w. 29 Identified compounds were tabulated in Table 2, which represented 92.54%. Phytochemical classes identified in this oil were sesquiterpene hydrocarbons (76.52%), monoterpene hydrocarbons (13.23%), and oxygenated sesquiterpenes (2.79%). (E)-Caryophyllene (45.49%), α-pinene (11.63%), and β-bisabolene (6.91%) could be the primary compounds, as well as various compounds possessed exceeding 1.00%, comprising α-selinene (3.31%), α-humulene (3.18%), selina-4(15),7(11)-diene (2.95%), β-selinene (2.77%), β-(Z)-farnesene (2.55%), caryophyllene oxide (2.44%), selina-3,7(11)-diene (1.94%), β-chamigrne (1.64%), and α-copaene (1.20%).
The extraction of the fresh leaves (the sample SF-5 collected from Ke Go Natural Reserve) also induced a yellow essential oil with the highest yield of 0.32%, v/w. There have been 36 identified compounds in the sample, which accounted for 96.12%. Similar to the SF-4, this sample was characterized by three phytochemical classes sesquiterpene hydrocarbons (78.66%), monoterpene hydrocarbons (16.01%), and oxygenated sesquiterpenes (1.45%), whereas oxygenated derivatives of monoterpenes and diterpenes and non-terpenic compounds were absent. The main compounds still included (E)-caryophyllene (47.12%), α-pinene (13.76%), and β-bisabolene (8.25%). Other significant compounds were also recorded, such as β-(Z)-farnesene (3.48%), α-humulene (3.17%), α-selinene (2.85%), β-selinene (2.53%), selina-4(15),7(11)-diene (2.19%), β-chamigrne (1.31%), selina-3,7(11)-diene (1.29%), δ-Cadinene (1.06%), and α-copaene (1.00%).
In general, essential oils derived from Vietnamese S. fluviatile were associated with the presence of monoterpene hydrocarbons, sesquiterpene hydrocarbons, and oxygenated sesquiterpenes. The contents of sesquiterpene hydrocarbons are found to be increased from sample SF-1 to sample SF-5, but their oxygenated derivatives are in contrast. Monoterpene hydrocarbons are abundant in the leaf essential oil but are present much less in the fruit essential oil. Various compounds were only found in one sample (Table 2). (E)-Caryophyllene is likely to be a characteristic compound in Vietnamese S. fluviatile essential oils, in which it reaches the highest percentage in the SF-5 and the lowest proportion in the SF-1. epi-Cedrol, caryophyllene oxide, and spathulenol naturally occur as the main compounds of the fruit essential oil, but they are insignificant or absent in the leaf essential oil. Among essential oils from the leaves, the major compound (E)-β-ocimene (5.03-20.22%) is present in the high contents of the SF-2 and SF-3, but it is not remarkable in the remaining samples. α-Pinene (11.63-13.76%) and β-bisabolene (6.91-8.25%) are found to be characteristic compounds of the SF-4 and SF-5, but they are less important in other samples. Although (Z)-β-ocimene, bicyclogermacrene, and δ-cadinene might be classified as the main agents in the SF-2, they are not remarkable in the remaining samples. Similarly, α-copaene and aromadendrene are characteristic compounds in the SF-3, but they are present in trace amounts or were absent in the remaining studied samples.
The current research is broadly consistent with the results obtained previously. Monoterpenes, sesquiterpenes, and their oxygenated derivatives are now available in the essential oils of various Vietnamese Syzygium species. (E)-Caryophyllene reached up to 18.21–64.53% in the leaf essential oils of S. boisanum, S. corticosum, and S. lineatum, which were also collected from Vu Quang National Park, Pu Hoat Nature Reserve, and Ke Go Nature Reserve, respectively.25–27 Besides eugenol, (E)-caryophyllene was found to account for 23.87% of Java-Indonesian clove leaf oil. 28 Eugenol (51.51%) and (E)-caryophyllene (36.20%) were the main compounds of essential oil from different brands of Oman S. caryophyllatum. 29 The hydro-distilled extraction of S. guineese leaves, collected from Benin, induced an essential oil containing (E)-caryophyllene (20.1%). 30 Essential oil from S. kanarense aerial parts, which were gathered from India, was dominated by sesquiterpene hydrocarbons (49.5%). 31 Hence, it can be concluded that Syzygium plants could be a good source of monoterpenes and sesquiterpenes, especially eugenol and (E)-caryophyllene.
Antimicrobial Activity
Five essential oils were further subjected to antimicrobial examination. As shown in Table 3, all tested samples showed activity against the Gram (+) bacteria B. cereus, S. aureus, and E. faecalis with the MIC and IC50 values of 16–64 µg/mL and 5.12–24.04 µg/mL, when streptomycin was used as a positive control with the MIC and IC50 values of 32 µg/mL and 20.45–50.34 µg/mL. Especially, all five samples were able to compare the positive control in the inhibitory treatment of the bacterium S. aureus (Table 3). However, S. fluviatile essential oils were inactive against the Gram (
Antimicrobial Activity of the Studied Essential Oils.

“-“: Inactive.
Essential oils from Syzygium species seem to have potential effects on antimicrobial treatments. The leaf essential oils of four Vietnamese Syzygium species S. formosum, S. syzygioides, S. megacarpum, and S. chantaranothaianum were associated with the MIC values of 16–128 µg/mL against the Gram (+) bacteria S. aureus and B. cereus, and Gram (
Molecular Docking Study
In this section, a molecular docking approach was applied to consider interactions between the major compound of S. fluviatile essential oil, (E)-caryophyllene, with the main targets DNA gyrase and PBP3 from S. aureus and SAP2 from C. albicans. Docking protocols were validated before conducting the results shown in Fig. S1 with the calculated RMSD value using the DockRMSD program as 1.564 Å, which is less than 2 Å indicating a high reliability of prediction. 35 Subsequently, the compounds were docked with the selected proteins using this protocol, and the corresponding binding affinities were determined as shown in Table 4. The interaction energies were compared in terms of binding modes, and molecular interactions with the positive controls streptomycin and cycloheximide.
The Binding Affinity of major Compound (E)-Caryophyllene and their Potential Molecular Interactions with Amino Acid Residues of Target Proteins.

In the docking study regarding the binding position of DNA gyrase, (E)-caryophyllene exhibited a binding affinity of −6.728 kcal/mol, which is close to the reference compound cycloheximide at −6.748 kcal/mol, and showed a slight difference in binding affinity compared to streptomycin at −0.635 kcal/mol. The molecular interaction pattern of (E)-caryophyllene indicated alkyl and pi-alkyl interactions with three residues Ile175, Ile102, and Ile86 as depicted in Fig. 2. It is noteworthy that Ile175 and Ile86 were two important amino acids in the active site of DNA gyrase. 17
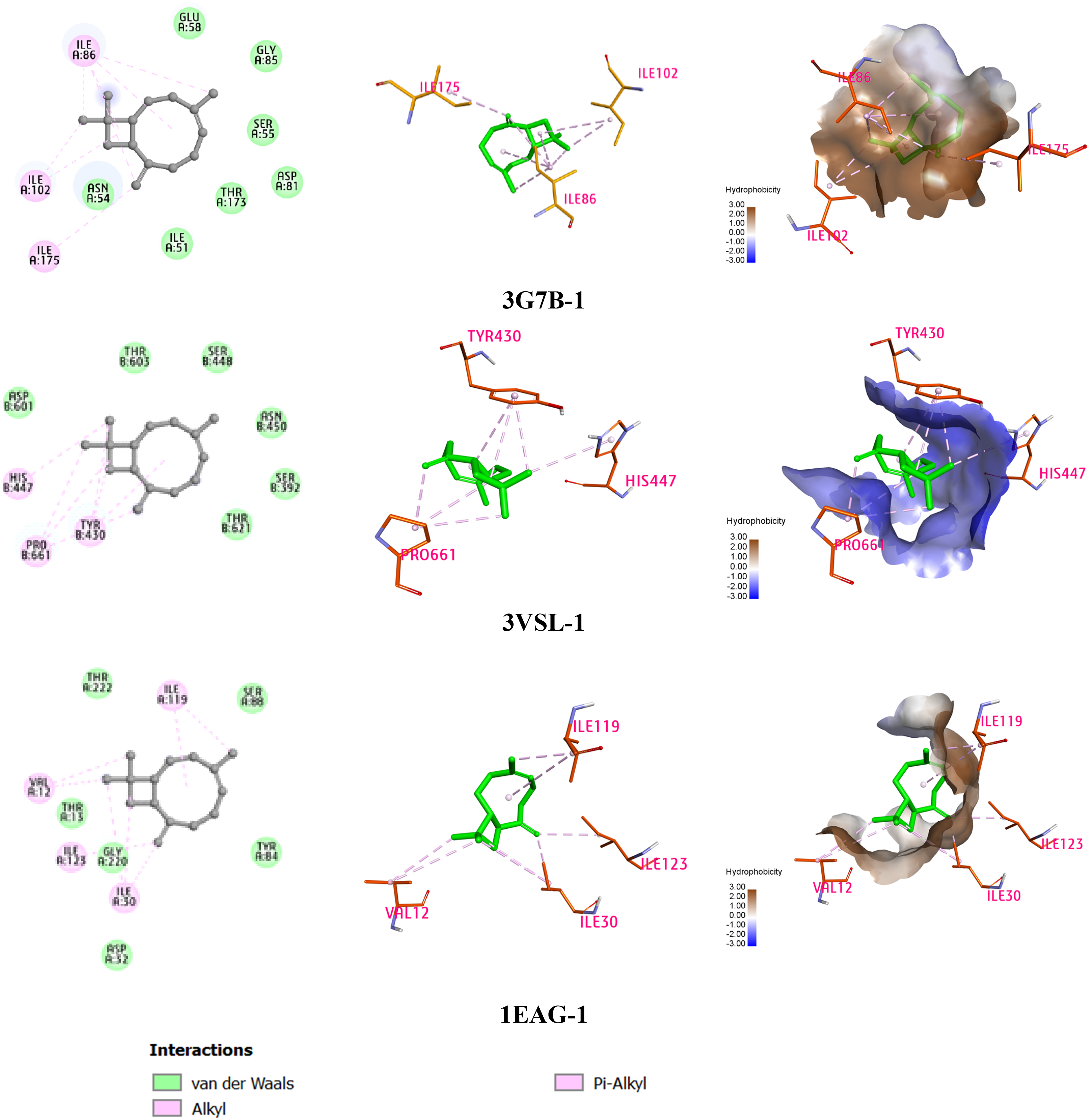
2D and 3D interactions of (E)-caryophyllene with the amino acid residues in the active-site gorge of the studied proteins.
For the binding ability to PBP3, (E)-caryophyllene exhibited a good binding affinity of −5.729 kcal/mol, compared to those of cycloheximide (–6.935 kcal/mol) and streptomycin (–7.725 kcal/mol). The interaction pattern is similar to the DNA gyrase, in which (E)-caryophyllene formed interactions with three amino acid residues Tyr430, His447, and Pro661 (Fig. 2). Moreover, the residue His447 was considered an important amino acid in the active site of PBP3. 18
Considering the antifungal potential on the SAP2 target of (E)-caryophyllene, it showed a binding affinity of −6.127 kcal/mol, compared to those of cycloheximide (–7.176 kcal/mol) and streptomycin (–7.464 kcal/mol). (E)-Caryophyllene also interacted with residues Val12, Ile119, Ile123, and Ile30 (Fig. 2). Among these residues, Ile119 was considered an important amino acid in the active site of SAP2. 19
Toxicological Prediction
Predicting the toxicity of compounds is considered one of the crucial steps in drug discovery. 36 In this study, (E)-caryophyllene was predicted through the ProTox 3.0 web server and compared with streptomycin and cycloheximide. The predicted results are presented in detail in Table 5. It can be observed that the LD50 value of (E)-caryophyllene was predicted to be 10.6 and 2650 times higher than those of streptomycin and cycloheximide, respectively. Based on the Globally Harmonized System classification, (E)-caryophyllene with a toxicity class of less than 5 is considered to have low toxicity and less impact when ingested, while the two control compounds are highly toxic and pose a danger. Additionally, the prediction accuracy and average similarity of (E)-caryophyllene are good with values of 70.97 and 98.96%, respectively (Table 5).
The Oral Toxicity Prediction of (E)-Caryophyllene and the Positive Controls.

*p: probability.
Organ toxicity assessment was performed considering the inactive and active targets, including hepatotoxicity, neurotoxicity, nephrotoxicity, respiratory toxicity, and cardiotoxicity. It noted that (E)-caryophyllene showed inactivity for all surveyed targets. Among these, the probability (p) value of (E)-caryophyllene with nephrotoxicity was the highest (p = 0.92), indicating a high accuracy prediction, followed by cardiotoxicity (p = 0.81), hepatotoxicity (p = 0.80), respiratory toxicity (p = 0.63), and neurotoxicity (p = 0.51). Overall, (E)-caryophyllene exhibited low toxicity and did not show organ toxicity. Therefore, further biological testing studies are needed to clarify the prediction results.
Conclusions
The current research first provides a phytochemical analysis of essential oils from S. fluviatile, collected from five different regions of Vietnam. It was noted that the collection location is responsible for the difference in the chemical results. In general, monoterpene hydrocarbons, oxygenated sesquiterpenes, and especially sesquiterpene hydrocarbons were the main phytochemical constituents. (E)-caryophyllene appeared as the main compound in the fruits and leaves. All studied samples showed remarkable results in antimicrobial activity against the Gram (+) bacteria B. cereus, S. aureus, and E. faecalis, and the yeast C. albicans. From an in silico approach, (E)-caryophyllene indicates good binding capability with the protein targets DNA gyrase, PBP3, and SAP2. Additionally, alkyl and pi-alkyl interactions significantly contribute to its binding affinity. Toxicological calculation suggests that (E)-caryophyllene is not toxic to organs.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241280876 - Supplemental material for Essential Oil Compositions and Antimicrobial Activities of Syzygium fluviatile (Hemsl.) Merr. & L.M.Perry: A Comparative Study on Collection Regions
Supplemental material, sj-docx-1-npx-10.1177_1934578X241280876 for Essential Oil Compositions and Antimicrobial Activities of Syzygium fluviatile (Hemsl.) Merr. & L.M.Perry: A Comparative Study on Collection Regions by Do Ngoc Dai, Do Thi Xuyen, Le Thi Huong, Nguyen Thi Hoang Anh, Nguyen Xuan Ha and Ninh The Son in Natural Product Communications
Footnotes
Acknowledgments
The authors are grateful to the boards of directors of Dakrong Nature Reserve, Pu Hoat Nature Reserve, Pu Luong Natural Reserve, Ke Go Natural Reserve, and Vu Quang National Park, for allowing sample collection.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Figs. S1-S7
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
