Abstract
Essential oils of two Asteraceae plants Blumea riparia DC. and Pluchea pteropoda Hemsl. ex Hemsl., collected from Vietnam, have been studied by hydro-distillation and GC/MS-FID (gas chromatography/mass spectrometry-flame ionization detector) analysis. Oil yields of B. riparia fresh leaves and twigs were in the range of 0.1–0.2%, whereas essential oil in the fresh aerial parts of P. pteropoda reached about 0.5%. Sesquiterpene hydrocarbons and oxygenated sesquiterpenes were the main chemical classes in the essential oils of B. riparia. The leaves and twigs of this species gave essential oils containing germacrene D (33.6-42.6%), (E)-β-caryophyllene (11.2-11.6%), and bicyclogermacrene (9.3-12.1%) as the major components. Oxygenated monoterpenes and sesquiterpene hydrocarbons achieved the highest amounts in the essential oil of P. pteropoda aerial parts; 2,5-dimethoxy-p-cymene (43.5%), β-maaliene (14.0%), and α-isocomene (9.0%) were characteristic compounds. Particularly, as compared with related Blumea and Pluchea species, B. riparia and P. pteropoda, collected from Vietnam, might be good sources of germacrene D and 2,5-dimethoxy-p-cymene, respectively. The essential oil of B. riparia leaf successfully controlled the growth of the fungus Fusarium oxysporum, with an MIC value of 50.0 µg/mL, and the essential oil of P. pteropoda aerial parts showed significant antimicrobial activity against the Gram positive bacterium Bacillus subtilis and the yeast Candida albicans, with the same MIC value of 50.0 µg/mL.
Introduction
Blumea riparia, family Asteraceae, locally named Day bau rung, is an evergreen shrub or subshrub with woody stems at the base. It is mainly distributed in Vietnam, Laos, Indonesia, Thailand, India, Malaysia, and China. 1 In Chinese traditional medicine, the plant is used to treat headache, hypertension, colic, and gynecological diseases. 2 In Malaysia, the root decoction is used to treat cough, stomachache, and edema. 3 Essential oils from Blumea plants have shown a wide range of pharmacological activities, such as antimicrobial, antioxidative, and insecticidal.4-6 Hence, phytochemical studies focusing on identifying the chemical composition of the essential oils have drawn much interest. Xanthoxylin (44.9%) was a major component of the essential oils of Chinese B. balsamifera leaves. 7 Essential oil of Nigerian B. perrottetiana aerial parts was dominated by 2,5-dimethoxy-p-cymene (30.0%) and 1,8-cineole (11.0%). 6 β-Caryophyllene (23.5%), 2,5-dimethoxy-p-cymene (14.7%), and germacrene D (13.2%) were the main constituents of the essential oils of Indian B. oxyodonta aerial parts. 8
P. pterpoda, locally named Sai ho nam, is also a flowering plant belonging to the Asteraceae family. 9 This plant is now available in South China and Indochina. 10 In Vietnamese traditional medicine, its roots were used to reduce fever, lower blood pressure, and treat malaria, headache, thirst, chest tightness, breath shortness, cough, and typhoid. 9 Leaves were used to treat back pain and dysentery. 10 Essential oils of Pluchea plants are associated with the presence of terpenoids. The principal components of the essential oil of Caribbean P. carolinesis aerial parts were selin-11-en-4α-ol (17.7-33.4%), and β-caryophyllene (5.5-21.1%), 11 whereas α-maaliene (18.84%), berkheyaradulen (14.0%), and dehydro-cyclolongifolene oxide (10.3%) were predominant in the essential oil of Egyptian P. diocoridis aerial parts. 12 Pluchea essential oils have great value in drug development due to their anticancer, antiaging, antimicrobial, and insecticidal activities.11,12
In this current study, we report the chemical compositions of the essential oils from B. riparia and P. pterpoda collected from Quangtri, Vietnam. The obtained essential oils have been further subjected to antimicrobial activity.
Results and Discussion
Regarding the essential oil of B. riparia leaves, thirty-six compounds were identified, which accounted for 91.8% of the total (Table 1). Among them, sesquiterpene hydrocarbons (69.8%, nineteen compounds) and oxygenated sesquiterpenes (18.4%, eleven compounds) were the main chemical classes. Germacrene D (33.6%), (E)-β-caryophyllene (11.2%), and bicyclogermacrene (9.3%), were the principal components. Some oxygenated sesquiterpenes were obtained in significant amounts, such as caryophyllene oxide (5.9%), α-cadinol (2.7%), epi-α-cadinol (2.4%), and spathulenol (2.3%). Besides these two main chemical classes, the leaf oil also contained one oxygenated monoterpene, linalool (0.5%), one sulfated sesquiterpene, mintsulphide (0.2%), one oxygenated diterpene, phytol (1.8%), two aldehydes, pentadecanal (0.4%) and nonanal (0.1%), and one fatty acid, hexadecanoic acid (0.6%).
Chemical Compositions of Essential Oils from Blumea Riparia Leaves and Twigs.

Retention indices relative to n-alkanes (C7-C30) on an HP5-MS column.
Thirty compounds were identified in the essential oil of B. riparia twigs, which represented 94.2% of the total. Sesquiterpene hydrocarbons (82.5%, twenty compounds) and oxygenated sesquiterpenes (10.3%, seven compounds) were the major components. The amounts of the principal sesquiterpene hydrocarbons germacrene D (42.6%), bicyclogermacrene (12.1%), and (E)-β-caryophyllene (11.6%), in the twig oil were higher than those in the leaf oil. However, as compared with the leaf oil, the percentages of the sesquiterpene hydrocarbon α-humulene, the oxygenated sesquiterpene caryophyllene oxide, and the oxygenated diterpene phytol in the twig oil tended to decrease (Table 1).
γ-Elemene (0.7%), α-ylangene (0.1%), spathulenol (2.3%), rosifoliol (0.4%), 14-hydroxy-α-muurolene (0.3%), 14-hydroxy-9-epi-(E)-caryophyllene (0.2%), mintsulfide (0.2%), hexadecanoic acid (0.6%), and nonanal (0.1%) were only found in the leaf oil. In the same manner, β-cubebene (0.7%), α-amorphene (0.7%), and allo-aromadendrene (0.1%) were characteristics of the twig oil. There is a remarkable difference in the essential oils from Vietnam Blumea species. Dung et al reported that the essential oil of B. lanceolaria fresh leaves and stems, collected from the Hanoi area, was characterized by methyl thymol (95.0%). 15 With percentages from 10.0 to 20.0%, β-caryophyllene, thymolhydroquinon-dimethylether, and caryophyllene oxide were the major components of the essential oil of Blumea lacera fresh aerial parts, gathered from Nghean-Vietnam. 16 Germacrene D is a well-known sesquiterpene hydrocarbon that has been detected in many plants, but it was not a major component of the other Blumea species essential oils, to date. As a consequence, B. riparia from Vietnam could be a good resource for this metabolite.
Thirty-one compounds (90.4%) were separated from the essential oil of P. pteropoda aerial parts using a GC-MS analytical approach (Table 2). Oxygenated monoterpenes (five compounds) and sesquiterpene hydrocarbons (nine compounds) were present in the highest amounts of 44.6 and 35.1%, respectively. The other chemical classes were also detected, comprising seven oxygenated sesquiterpenes (6.8%), six monoterpene hydrocarbons (2.9%), one alcohol (0.3%), and one tricyclic hydrocarbon (0.4%). The oxygenated monoterpene 2,5-dimethoxy-p-cymene (43.5%) was found to be the most abundant compound, followed by the sesquiterpene hydrocarbons β-maaliene (14.0%) and α-isocomene (9.0%). The essential oil of P. pteropado aerial parts is a mixture of various compounds, many of which were obtained in trace mounts (< 1.0%). However, some compounds, including limonene, silphin-1-ene, α-copaene, (E)-β-caryophyllene, α-humulene, δ-cadinene, neryl isovalerate, and geranyl valerate, appeared with percentages of 1.0–3.9%. From a chemotaxonomic point of view, there is a close relationship between this plant and P. rosea since 2,5-dimethoxy-p-cymene (33.7%) was also determined as the predominant compound in the oil of Cuban P. rosea stems. 17 This is the first time that the chemical constituents of the essential oil from P. pteropado have been reported. Therefore, it is expected that P. pteropado might be a good resource of 2,5-dimethoxy-p-cymene.
Chemical Compositions of Essential Oil from Pluchea pteropoda Aerial Parts.
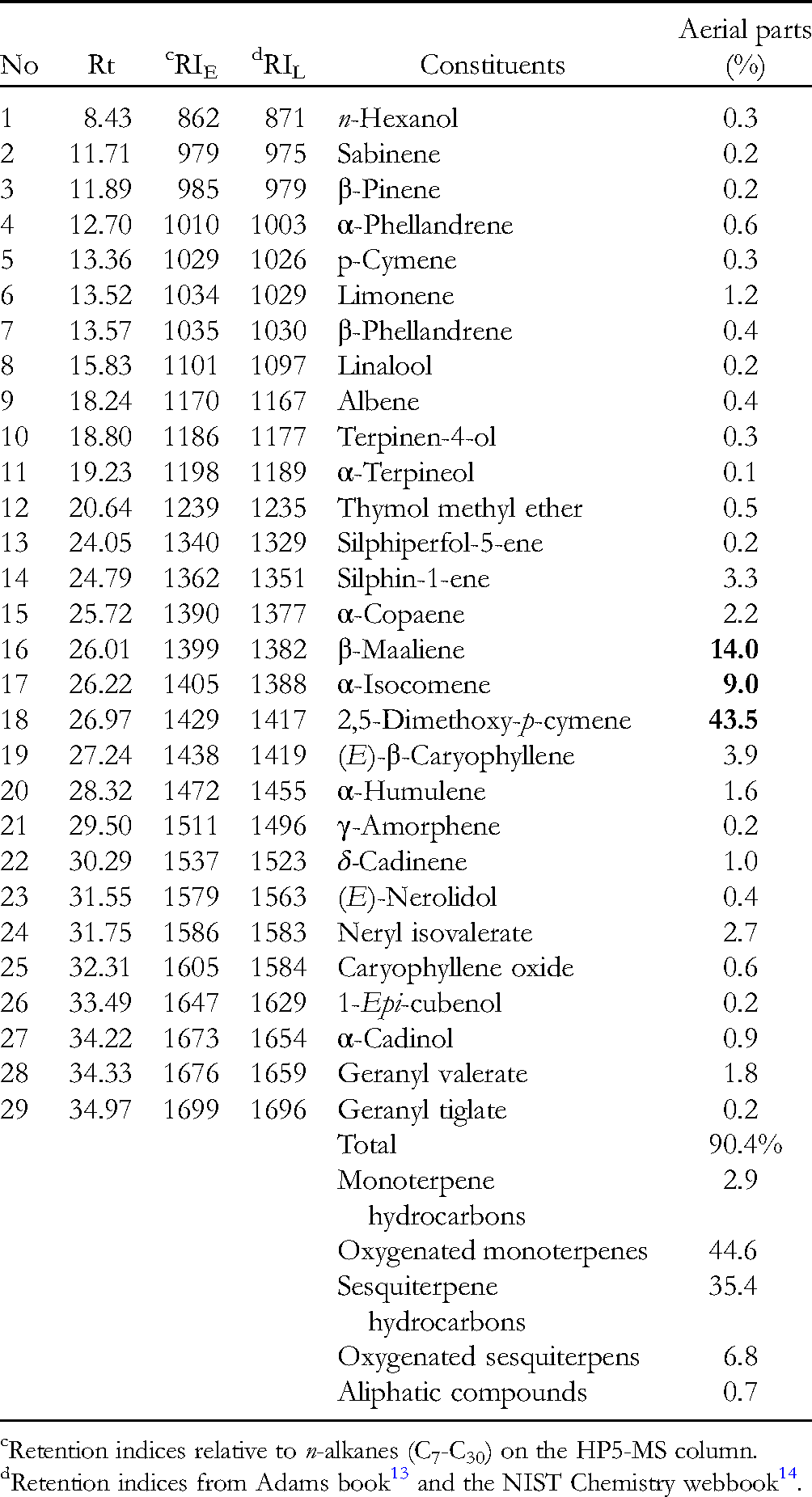
Retention indices relative to n-alkanes (C7-C30) on the HP5-MS column.
The essential oils of these two Asteraceae plants were further subjected to antimicrobial activity testing. As seen in Table 3, B. riparia twig oil possessed an MIC value of 100 μg/mL against the Gram-positive bacterium Bacillus subtilis, but B. riparia leaf oil failed to do so. This result is in line with that of Blumea mollis leaf oil (MIC value of 125 μg/mL). 18 By contrast, the leaf oil exerted an MIC value of 200 μg/mL against the Gram-negative bacterium Escherichia coli, but the twig oil was inactive. The leaf oil had MIC values of 50 and 100 μg/mL against the fungus Fusarium oxysporum and the yeast Saccharomyces cerevisiae, respectively. Meanwhile, the twig oil produced the same MIC value of 200 μg/mL against these pathogenic strains.
Antimicrobial Activity of Essential Oils from Blumea Riparia and Pluchea Pteropoda Plants.

There have been several reports on the antimicrobial activity of Pluchea essential oils. For example, the twig essential oil of P. arabica suppressed the growth of B. subtilis, Staphylococcus aureus, and Candida albicans, with inhibitory zones of about 8 mm. 19 The oil of P. pteropado aerial parts induced an MIC value of 50 μg/mL towards the Gram positive B. subtilis and the yeast C. albicans, when streptomycin (MIC = 7.25 μg/mL) and nystatin (MIC = 11.70 μg/mL) were used as the positive controls, respectively. This oil was also found to inhibit the fungi Aspergillus niger and Fusarium oxysporum, with respective MIC values of 200 and 100 μg/mL, but it was inactive against pathogenic strains of S. aureus, E. coli, Pseudomonas aeruginosa, and S. cerevisiae (Table 3).
Conclusion
Our current study first provides new information on the identification of essential oils from two Asteraceae plants, B. riparia and P. pteropoda, collected from the north central area of Vietnam. Sesquiterpene hydrocarbons and oxygenated sesquiterpenes were the main chemical classes in the essential oils of B. riparia fresh leaves and twigs, in which germacrene D was the principal compound, accounting for 33.6–42.6%. The main chemical classes in the essential oil of P. pteropoda fresh aerial parts were oxygenated monoterpenes and sesquiterpene hydrocarbons, as well as the oxygenated monoterpene 2,5-dimethoxy-p-cymene, which remarkably reached the highest percentage of 43.5%. The essential oils from the two Vietnamese Asteraceae plants were active against several pathogenic bacterial strains. The oil of B. riparia leaf showed moderate activity against F. oxysporum with an MIC value of 50.0
Materials and Methods
Materials
The leaves and twigs of B. riparia were gathered from Huonghoa, Quangtri, Vietnam (16°41′19.2''N and 106°41′21.0''E) in 03/2022, whereas the aerial parts of P. pteropoda were collected from Trieuphong, Quangtri, Vietnam (16°53′46.1''N and 107°12′17.0''E) in 03/2022. The leaves and twigs were collected from one B. riparia plant, whereas for P. pteropoda, the aerial parts were derived from several plants. The taxonomic identification was performed by taxonomist Vu Tien Chinh, Vietnam National Museum of Nature, VAST. The herbal specimens [BRL-2022 (B. riparia leaves), BRS-2022 (B. riparia twigs), and PPAP-2022 (P. pteropado aerial parts)] have been deposited at the Department of Analytical Chemistry, Institute of Natural Products Chemistry, VAST.
Extraction of Volatile Compounds
The fresh leaves of B. riparia (1.0 kg) were placed in a pressure cooker containing 3.0 L of water. Hydro-distillation was then carried out using a Clevenger-type apparatus for 2.0–2.5 h. The obtained yellow oil (0.2% yield, v/w) was centrifuged to eliminate the remaining water before being placed in a small vial, and refrigerated until analyses. The same procedure was applied to B. riparia fresh twigs and P. pteropado fresh aerial parts, in which oil yields of twigs and aerial parts reached 0.1 and 0.5%, respectively.
GC/MS-FID Procedure
The essential oils were analyzed using an Agilent Technology HP5-MS column (30 m × 0.25 mm, film thickness 0.25 m), and an Agilent Technologies HP7890A GC with a flame ionization detector.20,21 Temperatures for the injector and detector were set to 250 °C and 280 °C, respectively. The temperature of the column was started at 60 °C, and gradually increased to 240 °C at a rate of 4 °C/min. Helium was used as the carrier gas, with a flow rate of 1.0 mL/min. Splitting was used to inject the sample (1.0 mL essential oil with a split ratio 1:100). An ionization voltage of 70 eV, an emission current of 40 mA, and an acquisitions scan mass range of 40–450 amu under full scan were the settings for the mass selective detector (MSD). The relative percentage of each component was estimated without correction using the GC peak area (FID response). To match mass spectra and RI, MassFinder 4.0 software was utilized in conjunction with the HPCH1607 and W09N08 libraries, as well as the Adams book 13 and the NIST Chemistry WebBook. 14
Antimicrobial Activity
Antimicrobial activity of essential oils was determined using two Gram-positive bacterial strains Bacillus subtilis ATCC 6633 and Staphylococcus aureus ATCC 25923, two Gram-negative bacterial strains Escherichia coli ATCC 25922 and Pseudomonas aeruginosa ATCC 15442, two fungal strains Aspergillus niger ATCC 1015 and Fusarium oxysporum ATCC 46591, and two yeast strains Candida albicans ATCC 10231 and Saccharomyces cerevisiae ATCC 4098.22-24 These ATCC strains were obtained from American Type Culture Collection, Manassas, USA. The stock solutions of the three oil samples were prepared in DMSO (dimethylsulfoxide), and the dilution series ranged from 400 to 12.5 µg/mL. The bacteria, grown in Mueller-Hinton broth (Sigma Aldrich), and the filamentous fungi in double-strength Saboraud-2% dextrose broth (SDB) (Merck, Germany) were standardized to about 5 × 106 colony-forming units (CFU)/mL and 1 × 103 CFU/mL, respectively. The oil samples (400-12.5 µg/mL) were introduced to 96-well microtiter plates containing fresh cultures. They were then incubated for 24 h at 37 oC. The minimum inhibitory concentration (MIC) of a sample is the lowest concentration at which bacterial growth is completely inhibited. The positive controls for the Gram positive and Gram negative bacteria were streptomycin and tetracyclin, respectively, while for fungi and yeasts it was nystatin. DMSO 5% was used as a negative control. Each experiment was repeated three times.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221110662 - Supplemental material for Essential Oils of the Asteraceae Plants Blumea riparia DC. and Pluchea pteropoda Hemsl. ex Hemsl. Growing in Vietnam
Supplemental material, sj-docx-1-npx-10.1177_1934578X221110662 for Essential Oils of the Asteraceae Plants Blumea riparia DC. and Pluchea pteropoda Hemsl. ex Hemsl. Growing in Vietnam by Son Ninh The, Anh Le Tuan, Thuy Dinh Thi Thu, Nguyen Dinh Luyen and Tuyen Tran Thi in Natural Product Communications
Footnotes
Author Contributions
SN The: Formal analysis and manuscript writing; AL Tuan: Plant collection; TDT Thu and TT Thi: GC-MS analysis; and N DLuyen: Biological experiment.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
