Abstract
Golden camellia, a group of herbal materials belonging to the Theaceae family, was widely distributed in Asian countries, particularly Vietnam and southern China. Numerous pharmacological effects of various golden camellia species have been reported in reputable databases such as PubMed and Google Scholar. These effects included anti-tumor, lipid-lowering, anxiolytic, antidepressant, neuroprotective, antioxidant, antibacterial, and anti-inflammatory properties. Furthermore, toxicity studies on golden camellia species have demonstrated their safety for use. Our systematic review provides a comprehensive understanding of the pharmacological effects and safety of various golden camellia species, aiming to provide reliable evidence for the clinical use of this herbal material.
Introduction
The trend of using medicinal herbs and herbal medicines in combination with Western medical therapies for disease management is one of the prominent treatment strategies developed and recommended by the World Health Organization (WHO) in 2013. 1 This strategy has been applied within the health systems of many countries worldwide. The use of medicinal herbs in conjunction with pharmaceutical drugs not only enhances treatment efficacy but also allows for a reduction in the dosage of pharmaceutical drugs, thereby reducing or neutralizing their unwanted side effects.
Golden camellia species, belonging to the Camellia genus, are precious and expensive medicinal herbs, often referred to as the “queen of teas”, and naturally distributed in several Asian countries such as Indian, China, and especially Vietnam. According to the statistics by Le Nguyet Hai Ninh, there were approximately 34 species of Golden Camellia tea found in various provinces and cities in Vietnam. 2 Vietnamese folk medicine has used various types of teas, particularly golden camellia species, as a refreshing beverage to clear the heat, reduce diarrhea, and prevent cancer.
Regarding the chemical profile, nearly 60 chemical compounds have been reported to be presented in golden camellia leaves and flowers, especially in Camellia nitidissima, including tannins, flavonoids, polyphenols, saponins, polysaccharides, essential oils, sterols, and more. 3 Moreover, the phytochemical constituents of golden camellia species were believed to possess numerous biological effects, including antioxidant, lipid-lowering, antibacterial, anti-inflammatory, and anticancer properties. Gaining knowledge about the pharmacological activities, dosages, applications, pharmacological mechanisms, and toxicity of golden camellia species will contribute to the enhancement of the efficacy of utilizing these materials and facilitating the development of related products. However, there is currently a lack of reviews that has reported on the bioactivities of golden camellia species. Therefore, our study aims to fill this gap by conducting a systematic review that summarizes the available evidence on the biological activity and safety of golden camellia species and their compounds. We will include various types of study designs, such as in vitro and in vivo, to provide a comprehensive assessment of the topic.
Materials and Methods
The systematic review was conducted by following the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) checklist. 4 Our review methodology was listed in the International Prospective Register of Systematic Reviews (PROSPERO) (ID number CRD42023478835).
Inclusion Criteria
All studies on the pharmacological effects (in vitro, in vivo, ex vivo) and toxicity of the leaves and/or flowers of golden camellias and/or isolated chemical compounds from golden camellias have been published in English up to October 1, 2023.
Exclusion Criteria
Review and pharmacological researches on the species of Camellia other than golden camellias, as well as unreliable data such as articles with only abstracts, conference reports, theses, posters, and so on will be excluded.
Search Strategy
The search process was conducted on two databases: PubMed and Google Scholar, using the search terms presented in Table 1. Other relevant studies were manually searched using the reference lists of selected studies.
Search Terms on the Databases.

Study Selection and Data Collection
We utilized the WebPlotDigitizer tool available at https://automeris.io/WebPlotDigitizer/ to extract data from the graphical results presented in the research papers. To eliminate duplicate entries, we employed Endnote X9 (Clarivate Plc, London, United Kingdom) for automatic filtering of the search results. TLP and TAMP independently searched and screened relevant studies to meet the requirements. The data was then evaluated by MNT and VDT and was further verified by LTV and TMNH. In cases of disagreement between the results, the two reviewers were asked to carefully review the conflicting content and reconfirm their assessments. If reviewers could not reach an agreement, the supervisor (LTNH) was consulted.
The primary focus of the extracted data was on the preparation methods of golden camellia genus species and their pharmacological activities. The data were categorized based on the pharmacological activity and study design.
Risk of Bias
For in vitro studies, the Quality Assessment Tool For In Vitro Studies (QUIN Tool) checklist was utilized. This tool is easy to use, efficient, and effective for assessing the risk of bias in in vitro studies. It includes 12 criteria that evaluate the study's clarity and transparency, covering aspects such as the details of the comparison group, a thorough explanation of the methodology, statistical analysis, and the method of measuring the outcomes. The risk of bias was determined based on the final scores assigned to each study. A score > 70% indicated a “low risk of bias”, 50–70% indicated a “moderate risk of bias”, and <50% indicated a “high risk of bias”.
5

Results
A total of 245 research articles were found in the 2 databases. Among them, 200 articles were identified as duplicates and removed. The remaining 32 research articles and 12 manual finding researches underwent further screening, resulting in the selection of 35 articles for the final analysis. The PRISMA diagram is presented in Figure 1.

PRISMA flow diagram of study selection.
The pharmacological activities and safety of golden camellia species were listed in Table 2.
Pharmacological Activities and Safety of Golden camellia species.

(*: Data were extracted by the WebPlotDigitizer tool; 3-CT: 3-cinnamoyltribuloside, 5-FU: 5-fluorouracil; 5-HT: serotonine; 8-OHdG: 8-hydroxy-2′-deoxyguanosine; ABTS: 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid); ACC: Acetyl-CoA carboxylase, ADP: Adenosine Diphosphate; ADR: Adriamycin; AEW: Acetone/Ethanol/Water (2:2:1, v/v/v); AGEs: Advanced Glycation End-products; AKT: Protein kinase B; ALT: Alanine transaminase; AST: Aspartate transaminase; ATGL: Adipose triglyceride lipase; BDNF: Brain-derived neurotrophic factor; BUN: Blood urea nitrogen; CACNA1D: Calcium voltage-gated channel subunit alpha 1 D; CAT: Catalase; C/EBP: CCAAT enhancer-binding protein; CLS: saponin rich extract of Camellia nitidissima leaves; cort: Corticosterone; CRE: Creatinine; CREB: CAMP responsive element binding protein; DA: Dopamine; DDP: Cisplastin; DMSO: Dimethyl sulfoxide; DPPH: 2,2-diphenyl-1-picrylhydrazyl; EGFR: Epidermal growth factor receptor; FAS: Fatty acid synthase; FST: Forced swimming test; FRAP: Ferric reducing antioxidant power; GFR: Glomerular filtration rate; GLP-1: Glucagon-like peptide 1; Glycerol-3-phosphate 1-O-acyltransferase; HDL-C/LDL-C: High density lipoprotein-cholesterol/Low density lipoprotein-cholesterol; HFR: Hydroxyl free radical; HMGCR: 3-hydroxy-3-methyl-glutaryl-coenzyme A reductase; HO-1: Heme oxygenase 1; HOMA-IR: Homeostatic Model Assessment for Insulin Resistance; HSL: Hormone-sensitive lipase; IC50: 50% inhibition concentration; IGF1R: Insulin-like growth factor 1 receptor; IKBα: Nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor alpha; IL: Interleukine; INHBB: Inhibin subunit beta B; iNOS: Nitric oxide synthase; ITGB8: Integrin Subunit Beta 8; JHC-4: n-Butanol extract of Camellia nitidissima Chi; LD50: Lethal dose 50%; LPS: Lipopolysaccharide; MAPK: Mitogen-activated protein kinase; MDA: Malondialdehyde; ME: Material equivalent; MIC: Minimum inhibitory concentration; mTOR: Mammalian target of rapamycin; NE: Norepinephrine; NF-κB: Nuclear factor kappa-light-chain-enhancer of activated B; NO: Nitric oxide; Nrf2: Nuclear factor erythroid 2-related factor 2; NVB: Navelbine; OD: Optimal density; OGTT: Oral glucose tolerance test; ORAC: Oxygen radical absorbance capacity; OSC-PLS-DA: Orthogonal signal correction-Partial least squares-Discriminant analysis; PAI: Press-shear assisted interaction; PARP: Poly [ADP-ribose] polymerase; P-GSK-3β: Glycogen synthase kinase-3 beta; PI3K: Phosphoinositide 3-kinase; PKA: Protein kinase A; PKI3R3: Phosphoinositide-3-Kinase Regulatory Subunit 3; PPARγ: Peroxisome proliferator-activated receptor gamma; P-PTEN: Phosphatase and tensin homolog; PS: Press shear; QCG: Quercetin 7-O-(6″-O-E-caffeoyl)-β-D-glucopyranoside; RBC: Red blood cell; ROS: Reactive oxygen species; SD: Standard deviation; SHR/cp: Obese spontaneously hypertensive SHR/NDmcr-cp/cp rat; SOD: Superoxide dismutase; SREBP-1c: Sterol regulatory element-binding protein 1; SRC: Proto-oncogene tyrosine-protein kinase; TAX: Paclitaxel; TC: Total cholesterol; TE: Trolox equivalent; TEAC: Trolox equivalent antioxidant capacity; TG: Triglyceride; TGFB2: Transforming Growth Factor Beta 2; TNFα: Tumor necrosis factor alpha; TPT: Topotecan; TrkB: Tropomyosin receptor kinase B; TST: Tail suspension test; WBC: White blood cell; WKY: WKY/Izm rat).
Anticancer Effects
The phytochemical constituents in golden camellia species exhibited anticancer effects on various mutated cell lines. The compound quercetin 7-O-(6″-O-E-caffeoyl)-β-D-glucopyranoside, isolated from the ethyl acetate fraction of Camellia nitidissima flowers, demonstrated inhibitory activity on the growth of U937 human lymphoma cells (IC50 = 140.1 μM). This compound also increased the proportion of cells in the early apoptosis phase and activated the caspase-3 enzyme. 7 Figure 2 and 3 illustrated the chemical formula of cytotoxic phytochemical compounds in golden camellia species.

Chemical formula of quercetin 7-O-(6″-O-E-caffeoyl)-β-D-glucopyranoside.

Chemical formula of 3 cytotoxic triterpenoid compounds in leaves of Camellia nitidissima.
The leaves of Camellia nitidissima contain three chemical compounds that exhibited cytotoxic effects on four cancer cell lines: lung cancer cells (A549 and NCI-H1975), gastric cancer cells (HGC-27), and colorectal cancer cells (SW620). These compounds were 3-acetyl-13β-hydroxy-6α-methyl-olean-7-one, A1-barrigenol-22a-angelate, and 3β,6α,13β-trihydroxyolean-7-one. Among them, the compound A1-barrigenol-22a-angelate showed the best activity at a concentration of 10 μM. The IC50 value of A1-barrigenol-22a-angelate on NCI-H1975 lung cancer cells with EGFR resistance after 48 h of incubation was 13.37 μM. 8
The compounds luteolin and quercetin in Camellia nitidissima exhibited strong inhibition of cell growth in HCT117 colorectal cancer cells, and their effectiveness depended on the incubation time (IC50 values after 72 h of incubation were 18.2 μM and 160.7 μM, respectively). Additionally, luteolin and quercetin stimulated the apoptosis process in cells and prevented cell cycle progression at the G2/M phase. 9 Moreover, the compound 3β,6α,13β-trihydroxyolean-7-one demonstrated inhibitory effects on the growth of SGC7901 gastric cancer cells (IC50 = 91.7 μg/mL). 10
In terms of the anticancer effects of leaves of golden camellia species in vitro, the ethanolic extract of Camellia tunghinesis demonstrated superior cytotoxicity against MDA-MB-231 breast cancer cells (around 80% inhibition at a concentration of 800 μg/mL). 11 On the other hand, the n-butanol fraction of Camellia nitidissima showed significant growth inhibition and induced cell death in human gastric cancer cells (IC50 values of 147.62 μg/mL and 157.68 μg/mL after 72 h of incubation). 12 Furthermore, the n-butanol fraction of Camellia nitidissima exhibited synergistic effects with paclitaxel, by enhancing the sensitivity of gastric cancer cells to paclitaxel and significantly augmenting the growth-inhibitory effects of paclitaxel. 12 The ethanol extract of Camellia nitidissima has been shown to possess anti-tumor effects on human colorectal cancer cell lines HCT116, SW480, and HCT115 in vitro, through mechanisms involving cytotoxicity, stimulation of apoptosis, and cell cycle arrest at the G2/M phase in a time-dependent manner (IC50 values of 108.6 μg/mL, 97.98 μg/mL, and 84.38 μg/mL after 72 h of incubation, respectively). 13 The water extract of Camellia nitidissima also demonstrated inhibitory effects on SK-MES-1 lung cancer cells. 14
The water extract of Camellia nitidissima flowers exhibited inhibitory effects on human gastric epithelial cells (Eca109) through the activation of apoptosis in a time-dependent manner (IC50 values of 513.64 μg/mL, 326.88 μg/mL, and 217.31 μg/mL after 24, 48, and 72 h of incubation, respectively). It also disrupted the cell cycle by arresting cells at the G0/G1 phase. 15
In terms of the anticancer effects in vivo, the n-butanol and the water extract of Camellia nitidissima leaves exhibited anti-inflammatory colorectal cancer properties in mice, the n-butanol extract showed better efficacy compared to the water extract. Fractionated extracts rich in saponins and polyphenols from the leaves and flowers of Camellia nitidissima demonstrated inhibitory activity against non-small cell lung cancer both in vitro and in vivo, particularly the saponin-rich leaf extract showed positive signals on EGFR-resistant lung cancer cell lines. Furthermore, the saponin-rich leaf extract inhibited tumor growth in mice after 9 days of treatment (at 100 mg/kg), while doses of 200 mg/kg and 400 mg/kg showed effects starting from the fifth day of treatment. 16
Metabolic Process Effects
The water extract of leaves of Camellia nitidissima at a dose of 100 mg/kg exhibited effects in preventing metabolic syndrome in vivo through the inhibition of blood flow sluggishness, improvement of mean arterial pressure and systolic blood pressure, improvement of blood clotting time and platelet aggregation ability (not significantly), reduction of 3-nitrotyrosine, 3-chlorotyrosine, 8-OHdG, and blood lipid peroxidation levels, without affecting body weight, blood glucose, triglyceride, free fatty acid levels, and vascular dilation capacity. 22
The ethyl acetate fraction obtained from the ethanol extract of leaves and flowers of Camellia nitidissima demonstrated inhibitory effects on the formation of advanced glycation end products (AGEs) in vitro. Three pure compounds isolated from this golden camellia flowers, including kaempferol 3-O-[23,4-tri-O-acetyl-α-L-rhamnopyranosyl-(1→3)-2,4-di-O-acetyl-α-L-rhamnopyranosyl-(1→6)]-β-D-glucopyranoside, kaempferol 3-O-[23,4-tri-O-acetyl-α-L-rhamnopyranosyl-(1→3)-4-O-acetyl-α-L-rhamnopyranosyl-(1→6)]-β-D-glucopyranoside, and catechin also inhibited the formation of AGEs with the same mechanism (illustrated in Figure 4).17,18
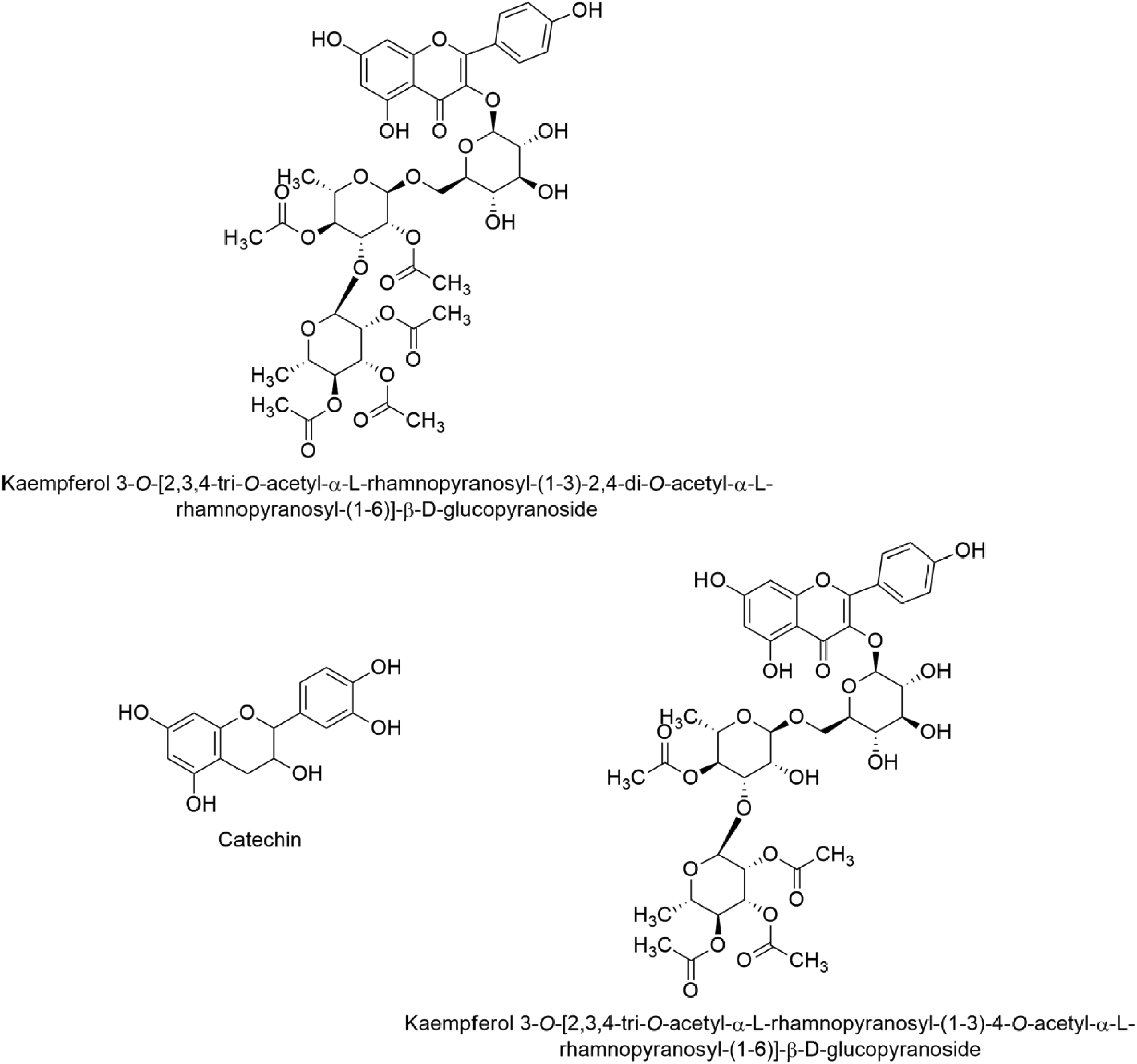
Chemical formula of 3 AGEs formulation inhibition compounds.
The 59% ethanol extract of leaves and flowers of Camellia nitidissima pigmented flower, protein, and polysaccharide fractions exhibited inhibitory effects on pancreatic lipase activity in vitro. The flower extract of Camellia nitidissima also demonstrated better inhibitory effects on cholesterol esterase enzyme compared to pancreatic lipase in vitro. These inhibitory effects were further observed in in vivo experiments, where they resulted in reducing digestion and food consumption in mice, as well as a significant increase in triglyceride and total cholesterol concentrations in the feces.19,20
The water extract of Camellia euphlebia flowers exhibited lipid-lowering effects in vivo by reducing the levels of total cholesterol, triglycerides, and LDL-C; increasing HDL-C levels; enhancing the activity of antioxidant enzymes such as SOD and GSH-Px; reducing the levels of MDA and serum ALT; reducing lipid droplet accumulation in liver cells; decreasing the activity of the enzyme glutamic-pyruvic transaminase; and reducing the mRNA expression levels of fatty acid synthase, 3-hydroxy-3-methylglutaryl CoA reductase, and glycerol-3-phosphate acyl transferase. However, there was no significant difference in food consumption between the treatment and the model groups. 21
The ethanol extract of flowers of Camellia nitidissima golden was proven to demonstrate anti-obesity effects and related complications in vivo by significantly reducing body weight, food intake, Lee index, liver index, epididymal fat index, and retroperitoneal fat index. It also decreased fasting blood glucose levels, fasting serum insulin levels, and HOMA-IR index, while increasing ADP enzyme and GLP-1 levels. Additionally, it reduced total cholesterol, triglycerides, leptin, MDA, AST, and ALT levels. It mitigated microvesicular steatosis and lipid infiltration in cells and inhibited the expression of genes related to fatty liver conditions such as C/EBPα, C/EBPβ, C/EBPδ, PPARγ, SREBP-1C, and ACC, which were involved in cholesterol and fatty acid accumulation in the liver. On the other hand, it increased the expression of genes ATGL and HSL, which were associated with lipid breakdown and neutral fat metabolism. Moreover, it significantly reversed dysbiosis by regulating the reduction of Firmicutes bacteria and increasing the abundance of Bacteroidetes and Verrucomicrobia strains. 23
Finally, the saponin triterpenoid compound, 21β,22α-di-O-angeloyl-15α,16α,28-trihydroxyolean-12-en-3β-O-α-L-rhamnopyranosyl-(1→3)-α-D-xylopyranosyl-(1→3)-β-D-glucopyranoside isolated from Camellia nitidissima (depicted in Figure 5), exhibited inhibitory effects on α-glucosidase enzyme activity in vitro, with an IC50 value of 185.9 μmol/L. 40

Chemical structure of 21β,22α-di-O-angeloyl-15α,16α,28-trihydroxyolean-12-ene-3β-O-α-L-rhamnopyranosyl-(1→3)-α-D-xylopyranosyl-(1→3)-β-D-glucopyranoside.
Effects on the Central Nervous System
Anxiolytic and Antidepressant Effects
The water extract of leaves of Camellia euphlebia exhibited anxiolytic and antidepressant effects in an in vivo model. It increased the time spent in the light area (light-dark box model), reduced immobility time (forced swim and tail suspension tests), effectively counteracted ptosis and body temperature decrease induced by reserpine, enhanced head twitch response induced by 5-hydroxytryptophan, increased the levels of brain monoamines such as serotonine, dopamine, and noradrenaline, and decreased serum corticosterone and plasma ACTH levels. 24
Neuroprotective Effects
The ethanol extract of flowers of Camellia euphlebia demonstrated neuroprotective effects on pheochromocytoma (PC12) cells by reversing the apoptosis process induced by corticosterone. This was evaluated through the assessment of parameters such as cell viability, lactate dehydrogenase enzyme activity, Hoechst 33342 and propidium iodide staining, dual AV-FITC/PI staining, and DNA fragmentation analysis. 25 Furthermore, the combination of fine powder leaves of Camellia euphlebia and 2% shellfish powder exhibited the strongest neuroprotective effects. 41
The ethyl acetate fraction of leaves of Camellia nitidissima exhibited neuroprotective effects on human SH-SY5Y neuroblastoma cells treated with hydrogen peroxide at a concentration of 150 μg/ml. It increased the viability of SH-SY5Y cells (from 53.5% to 99.4%), reduced extracellular LDH activity and intracellular ROS levels, decreased the number of apoptotic cells, and enhanced the expression of phosphorylated cAMP response element-binding protein (pCREB) and brain-derived neurotrophic factor (BDNF), which were involved in neuronal survival and function. 26
Antioxidant Effects
The antioxidant effects of leaves of golden camellia species were evaluated in vitro using the peroxyl radical scavenging capacity assay (ORAC). The antioxidant activity decreased in the following order: Camellia tunghinesis > Camellia nitidissima > Camellia chrysantha > Camellia microcarpa > Camellia euphlebia > Camellia impressinervis (with IC50 values ranging from 327.1 to 2270.9 μmol TE/g). 27 The water extract of Camellia chrysantha leaves exhibited the best free radical scavenging activity against DPPH radicals, with a percentage inhibition of 78.5% at an equivalent concentration of 1 g of the herbal material per mL. This extraction was performed using a soaking time of 30 min, an extraction time of 5 h, and a solvent-to-herb ratio of 3:2 (mL/g). 28
The ethanolic extract of leaves of Camellia nitidissima exhibited the antioxidant activity better than the essential oil and flower essential oil, as well as ascorbic acid, in the DPPH free radical scavenging assay (with IC50 values of 17.4, 164.8, and 720.3 μg/mL, respectively) and the ABTS assay (with IC50 values of 28.8 μg/mL for the leaf extract and 889.6 μg/mL for the flower essential oil). 29 Furthermore, the 30% ethanol extract of Camellia nitidissima demonstrated the free radical scavenging activity against reactive oxygen species (ROS) better than Camellia euphlebia and Camellia insularis (with IC50 values of 311.70, 332.64, and 917.60 μg of herbal material/mL, respectively). 30
The ethyl acetate fraction of flowers of Camellia nitidissima exhibited better antioxidant activity in vitro than the petroleum ether, dichloromethane, n-butanol, and water fractions on three free radical scavenging models: DPPH, ABTS, and hydroxyl radicals, at a concentration of 5 mg/mL. Furthermore, compounds such as okicamelliaside, gallic acid, catechin, and salicylic acid presented in flowers of Camellia nitidissima reduced the activity of the enzymes SOD (superoxide dismutase) and CAT (catalase) in a concentration-dependent manner in 293 T cells. 31
The ethyl acetate fraction of Camellia nitidissima leaves partitioned from a 75% ethanol extract exhibited better antioxidant activity than the n-hexane, n-butanol, and water fractions on three antioxidant models: DPPH, ABTS, and FRAP (with antioxidant capacities of 4.0, 6.7, and 3.7 mmol Trolox/g, respectively). 32
The essential oil from the leaves of Camellia euphlebia demonstrated the highest antioxidant activity compared to Camellia nitidissima and Camellia tunghinensis on the DPPH and ABTS models (with IC50 values of 42.8 ± 3.7 and 102.5 ± 2.6 µg/mL, respectively). 33
Kombucha preparation based on Camellia petelotii showed significant antioxidant effects. In particular, the FRAP and TEAC values of the water extract with tea residue demonstrated higher antioxidant activity than the water extract without tea residue. 34
Antibacterial Effects
The dichloromethane fraction partitioned from the ethanol extract of Camellia nitidissima flowers exhibited antibacterial activity against Pseudomonas aeruginosa PAO1. It demonstrated bactericidal effects with a minimum inhibitory concentration (MIC) of 2.50 mg/mL. It also reduced bacterial virulence through various mechanisms, including the inhibition of procyanin production (with an IC50 of 0.158 mg/mL), inhibition of swarming motility (with an IC50 of 0.139 mg/mL), inhibition of swimming ability (with an IC50 of 0.334 mg/mL), and downregulation of the expression of genes such as asI, lasR, rhlI, and rhlR. 35
The ethanol extract of leaves of Camellia nitidissima exhibited better antibacterial activity than the leaf essential oil and flower essential oil against two Gram-positive bacteria, Staphylococcus aureus (MIC = 0.625 mg/mL) and Bacillus subtilis (MIC = 1.25 mg/mL). 29
The essential oil from leaves of Camellia nitidissima had a better inhibitory effect on various bacterial strains compared to the leaf essential oils of Camellia tunghinensis and Camellia euphlebia. It exhibited stronger inhibitory activity against Staphylococcus aureus (MIC = 1.25 mg/mL), Bacillus subtilis (MIC = 1.25 mg/mL), and Escherichia coli (MIC = 0.625 mg/mL). 29
Anti-Inflammatory Effects
The compound 3-cinnamoyltribuloside (depicted in Figure 6), isolated from flowers of Camellia nitidissima, exhibited anti-inflammatory effects in vitro on RAW246.7 cells by inhibiting the production of nitric oxide (NO), reducing the mRNA expression of the iNOS gene, and decreasing the expression of inflammatory cytokines (TNF-α, IL-1β, IL-6) at both mRNA and protein levels. 36 Similarly, the compound camellioside I, isolated from the leaves of Camellia bugiamapensis, demonstrated inhibition of NO production from RAW264.7 cells with an average IC50 value of 49.42 μM without exhibiting cytotoxic effects on the cells. 37

Chemical structure of 3-cinnamoyltribuloside.
The 30% ethanol extract of Camellia nitidissima demonstrated better anti-inflammatory effects in vitro compared to Camellia euphlebia and Camellia insularis. It inhibited the production of nitric oxide (NO) from RAW246.7 cells, with IC50 values of 754.68, 1182.39, and 1527.83 μg of herbal material/mL for Camellia nitidissima, Camellia euphlebia, and Camellia insularis, respectively. 30
Hepatoprotective Effects
The aqueous extract of leaves of Camellia nitidissima at a dose of 160 mg/kg demonstrated hepatoprotective effects in a mouse model of acute liver injury induced by CCl4. This was evidenced by reducing liver-to-body weight ratio, decreasing levels of ALT, AST, MDA, and ROS, increasing levels of SOD and GSH enzymes, alleviating hepatocellular necrosis, inhibiting production of pro-inflammatory cytokines TNF-α, IL1β, IL6 through downregulation of mRNA and protein expression, decreasing nuclear expression of p65, cellular material, p-IκBα, and IκBα in mouse liver, and enhancing mRNA expression of Nrf2 and HO-1 genes. 38
Toxicity
The aqueous extract of leaves of Camellia euphlebia did not exhibit acute toxicity, with an LD50 value greater than 5000 mg/kg.24,39 However, doses of 4000 and 5000 mg/kg resulted in drowsiness and increased gas production in mice. No mortality or significant changes in food intake, water consumption, body weight, AST, ALT, creatinine, red blood cell count, and white blood cell count were observed after a 14-day sub-acute toxicity test. However, mice treated with a dose of 1600 mg/kg of the extract showed behavioral changes including sedation, increased gas production, and piloerection, as well as mild cellular material degeneration in the liver. Similarly, in a 28-day sub-chronic toxicity model, no mortality or significant changes in food intake, water consumption, body weight, AST, ALT, creatinine, white blood cell count, or histopathological abnormalities in organs were observed. However, mice treated with a dose of 600 mg/kg of the extract for 28 days showed a statistically significant increase in red blood cell count compared to the control group. Finally, the genotoxicity assessment of the extract showed negative results for chromosomal aberration assay in CHL cells and no statistically significant differences between the control and treatment groups in terms of micronucleus formation ability in mouse femur bone marrow cells. 39
Risk-of-Bias of Included Studies
Out of the 29 studies on the in vitro effects of golden camellia species, 21 reports were evaluated as having a low risk of bias, 2 studies had a moderate risk of bias, 6 studies had a high risk of bias, and none of the studies were excluded. On the other hand, all in vivo studies were deemed to have a moderate risk of bias since many areas did not have sufficient comprehensive information supplied to appropriately assess the risk of bias. The assessment of the risk of bias was shown in Table 3 and Table 4.
Quality Assessment of in Vitro Studies According to the Items of the QUIN Tool Checklist.
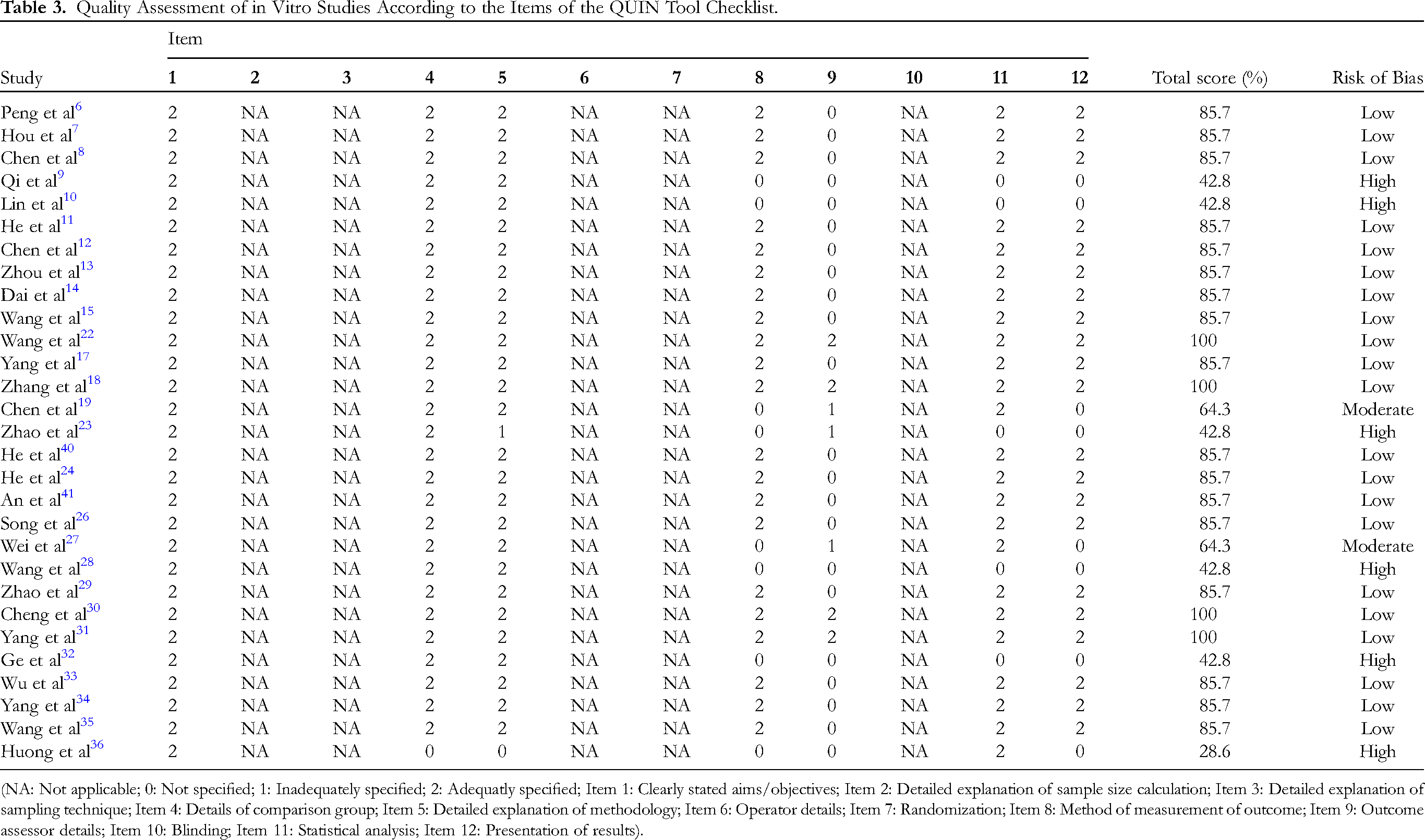
(NA: Not applicable; 0: Not specified; 1: Inadequately specified; 2: Adequatly specified; Item 1: Clearly stated aims/objectives; Item 2: Detailed explanation of sample size calculation; Item 3: Detailed explanation of sampling technique; Item 4: Details of comparison group; Item 5: Detailed explanation of methodology; Item 6: Operator details; Item 7: Randomization; Item 8: Method of measurement of outcome; Item 9: Outcome assessor details; Item 10: Blinding; Item 11: Statistical analysis; Item 12: Presentation of results).
Quality Assessment of in vivo Studies According to the Items of the SYRCLE's Tool.
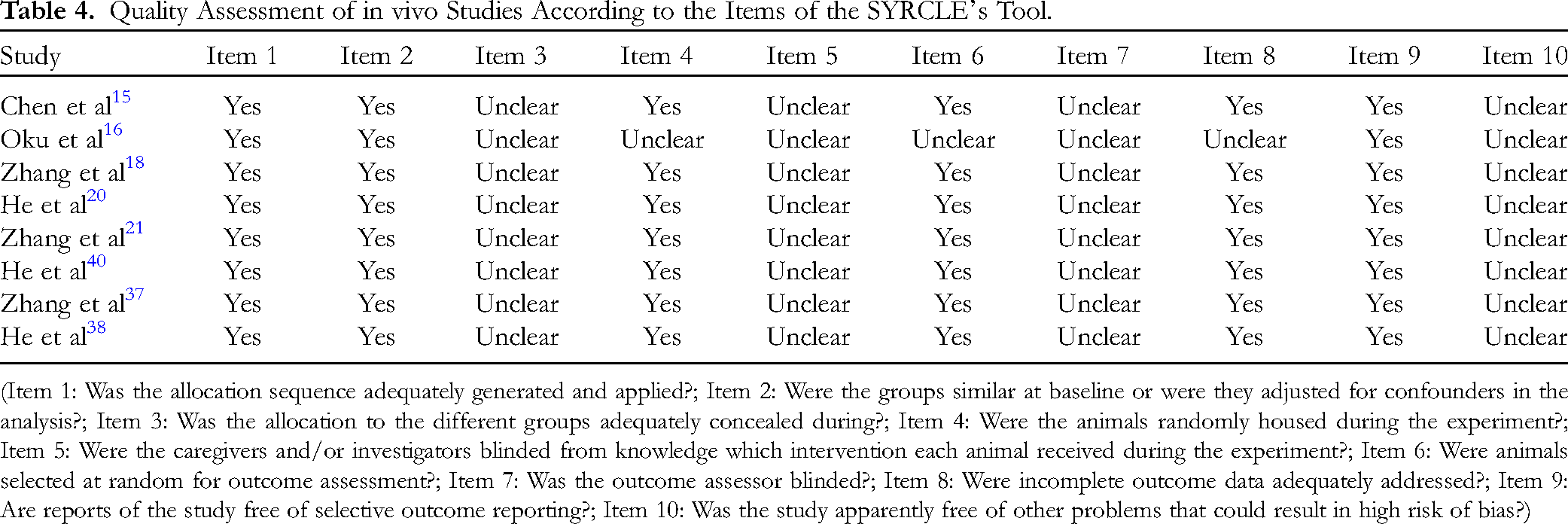
(Item 1: Was the allocation sequence adequately generated and applied?; Item 2: Were the groups similar at baseline or were they adjusted for confounders in the analysis?; Item 3: Was the allocation to the different groups adequately concealed during?; Item 4: Were the animals randomly housed during the experiment?; Item 5: Were the caregivers and/or investigators blinded from knowledge which intervention each animal received during the experiment?; Item 6: Were animals selected at random for outcome assessment?; Item 7: Was the outcome assessor blinded?; Item 8: Were incomplete outcome data adequately addressed?; Item 9: Are reports of the study free of selective outcome reporting?; Item 10: Was the study apparently free of other problems that could result in high risk of bias?)
Discussion
Golden camellia species is a precious herbal material unique to Vietnam and some Asian countries. Currently, the application of various parts of golden camellias in healthcare and the support of certain diseases is being promoted. A comprehensive overview of the in vitro and in vivo pharmacological effects of golden camellia species would contribute to providing scientific evidence for its medicinal use.
The anticancer and antitumor effects are among the many biological effects that have been studied and are of great interest in herbal medicine in general and specifically in golden camellia species, especially Camellia nitidissima. Numerous cancer cell lines have been found to be inhibited by this herb, such as human lymphoma cells U937, lung cancer cells A549 and NCI-H1975, gastric cancer cells HGC-27, colorectal cancer cells SW620 and HCT117, breast cancer cells MDA-MB-231, human colon carcinoma cells HCT116, SW480, and HCT115, and lung cancer cells SK-MES-1.7–16
Many phytochemical compounds belonging to the flavonoid and saponin groups have been successfully extracted, isolated, and structurally identified from the leaves and flowers of golden camellias (such as luteolin, quercetin, quercetin 7-O-(6″-O-E-caffeoyl)-β-D-glucopyranoside, A1-barrigenol-22a-angelate, and 3β,6α,13β-trihydroxyolean-7-one).7–10 The main mechanism of tumor growth inhibition by these phytochemical compounds is through the induction of programmed cell death (apoptosis). Additionally, other mechanisms have been discovered, including cell cycle arrest at the G0/G1, G2/M, and S phases. Luteolin and quercetin also had regulatory effects on the PI3K-Akt signaling pathway by reducing the expression of proteins such as EGFR, SRC, AKT, IGF1R, P-GSK-3β, and P-PTEN in cancer inhibition. In molecular docking experiments, luteolin exhibited strong binding to the VEGFA target of cancer cells by forming π bonds with the amino acid Phe47 and hydrogen bonds with the amino acids Cys60 and Cys26. 9 Moreover, Camellia nitidissima also enhanced the inhibitory effect of paclitaxel on tumor growth by inducing autophagy. 12
Glycation is a chemical reaction that occurs between proteins and lipids or carbohydrates, resulting in the formation of reactive carbonyl compounds. The products of advanced glycation end-products (AGEs) are known to contribute to various diseases such as diabetes, aging, Alzheimer's disease, atherosclerosis, and more.42–45 The bioactivity compounds in golden camellia species, particularly the phenolic group, have been demonstrated to inhibit the process of advanced glycation, thereby potentially assisting in the treatment of metabolic disorders. The mechanism of inhibition of advanced glycation by golden camellias involved the scavenging of methylglyoxal radicals. Kaempferol glycoside exhibited less inhibitory activity against the glycation process compared to kaempferol aglycone, and the addition of an acetyl group at position 2’” of the kaempferol glycoside compound did not significantly affect its inhibitory effect on glycation.17,18
In addition, the leaves and flowers of Camellia nitidissima also impacted the metabolic process through various pathways such as (1) reducing the risk of metabolic syndrome formation through the presence of antioxidant compounds; (2) inhibiting pancreatic lipase enzyme by binding to free enzymes or complexes of enzymes and substrates at non-catalytic sites, while also modifying the secondary protein structure of the enzyme; (3) inhibiting fat accumulation in the liver and regulating the gut microbiota, leading to anti-obesity effects and related complications; (4) preventing lipid disorder by antioxidative activity, reducing liver damage in mice by decreasing the activity of glutamic-pyruvic transaminase enzyme and inhibiting lipid accumulation by regulating the expression of fatty acid synthase, 3-hydroxy-3-methylglutaryl CoA reductase, and glycerol-3-phosphate acyltransferase enzymes; and (5) preventing blood glucose elevation by inhibiting the activity of the α-glucosidase enzyme.19,20
One of the biological effects of golden camellia species that has received research attention is its impact on the central nervous system. In addition to Camellia nitidissima, the leaves and flowers of Camellia euphlebia had the potential to exhibit anxiolytic, antidepressant, and neuroprotective effects. The antidepressant effect of leaves of Camellia euphlebia may be related to the apoptosis process through the intermediate tyrosine kinase and the PKA-CREB-BDNF signaling pathway, while the neuroprotective mechanism of leaves of Camellia nitidissima involved enhancing the coordination between intrinsic antioxidant defense capabilities and neuronal signaling pathways.24–26,41
The antioxidant effect is a pharmacological effect commonly evaluated in the process of studying herbal medicines. There were 16 phytochemical compounds that significantly contributed to the antioxidant effect of leaves of Camellia chrysantha through the mechanism of scavenging the DPPH free radical. Among them, 6 compounds have been identified with decreasing DPPH free radical scavenging activity, including quercetin-7-O-β-D-glucopyranoside, catechin, epicatechin, kaempferol, vitexin, and isovitexin. The differences in DPPH free radical scavenging ability can be explained by the higher number of hydroxyl radicals in the B-ring of the flavonoid structure of the three compounds quercetin-7-O-β-D-glucopyranoside, catechin, and epicatechin (2-hydroxyl groups) compared to kaempferol, vitexin, and isovitexin. Additionally, on the C-ring of quercetin-7-O-β-D-glucopyranoside, there is a hydroxyl group at position C3, resulting in superior antioxidant activity compared to the other two compounds. 31 The flavonoid compounds catechin, catechin-4 alpha,8-catechin, quercetin, isoquercitrin, and kaempferol in the ethyl acetate fraction of Camellia nitidissima flowers exhibited potential antioxidant activity. Furthermore, the antioxidant effect of these compounds was influenced by the o-catechol group on the B-ring and steric hindrance caused by glycoside groups. 32
Antibiotic resistance is currently a challenging issue in the treatment of bacterial infections. Therefore, in the process of studying herbal medicines, the antimicrobial effect is an important aspect of interest. The ethyl acetate fraction of Camellia nitidissima can reduce the virulence factors of Pseudomonas aeruginosa PAO1 bacteria by modulating the representative sensing system. Six chemical compounds identified in the ethyl acetate fraction were gallic acid, catechin, ellagic acid, chlorogenic acid, quercetin, and kaempferol, among which ellagic acid inhibited procyanin production, inhibited biofilm formation, and reduced the swimming ability of bacteria most effectively. Although they have potential antimicrobial effects, the overall antimicrobial activity of golden camellia species was still not as effective as pharmaceutical drugs. 35
For the anti-inflammatory effect, several potential compounds have been isolated from golden camellia species, such as camellioside I, 3-cinnamoyltribuloside, and others. The compound 3-cinnamoyltribuloside affected the metabolism of 35 endogenous compounds such as choline, glucose, phenylalanine, etc, thereby demonstrating anti-inflammatory effects through the cholinergic pathway, oxidative stress, energy metabolism, and amino acid metabolism. Additionally, both Camellia nitidissima and Camellia euphlebia exhibited similar and significantly better inhibitory activity against the production of NO and ROS compared to Camellia insularis.36,37
The hepatoprotective effect of the aqueous leaves extract of Camellia nitidissima has been demonstrated through in vivo experiments, with a mechanism related to the protection of the liver involving the Nrf2 and NF-κB signaling pathways. 38 Lastly, the toxicity of golden camellia leaves and flowers is an important aspect that requires research attention to predict any adverse effects when using herbal medicine. A study by He and Xu in 2022 showed that the leaves of Camellia euphlebia exhibited no significant acute or sub-acute toxicity and did not induce genotoxicity. However, it had the potential to cause slight erythrocyte increase and mild hepatocellular degeneration. 39
Finally, the toxicity evaluation of golden camellia indicated a high level of safety when used as a herbal medicine (no mortality, abnormal signs, or genotoxicity observed in in vivo testing). Some adverse effects noted in mice when using golden camellia included drowsiness, increased flatulence, and increased red blood cell count, which should be monitored when used in humans.
Conclusion
The phytocompounds found in the leaves and flowers of golden camellia species exhibited various pharmacological effects such as cancer inhibition, regulation of metabolic processes, antioxidant activity, antibacterial properties, anti-inflammatory effect and so on without causing toxic. This highlights the potential use of these herbal materials in supporting the treatment of clinical conditions. Our systematic review provides a comprehensive understanding of the pharmacological effects of golden camellia species. However, our study did not include Chinese-language reports from the CNKI database and grey literature. Additionally, the keywords used for searching in PubMed and Google Scholar may not have covered all the existing golden camellia species worldwide. Therefore, we recommend future researches updating the literature on the pharmacological effects of golden camellia species to gain a better understanding of the efficacy of this herbal medicine.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Trial Registration
Not applicable, because this article does not contain any clinical trials.
