Abstract
Objective / Background
Hydropuntia eucheumatoides (HE) seaweed is widely distributed in tropical seas and is a common food for coastal people. Our objectives were to prepare seaweed milk from HE, and test is possible utility as a food supplement and source of antioxidants that has an appropriate sensory profile.
Methods
We used an enzyme from Lactobacillus strains for polysaccharide hydrolysis into HE seaweed milk food supplements. We cultured five Lactobacillus strains with appropriate agarase, cellulase, and amylase enzyme activities.
Results
We optimized the enzyme concentration (mass of seaweed HE: 40 g/L), substrate concentration (2 mL enzyme/g seaweed), temperature (50 °C), pH (5), and hydrolysis time (96 h). The hydrolyzate included oligosaccharides, monosaccharides, amino acids, and minerals. The product exhibited excellent sensory details and is likely to be safe for consumption.
Conclusions
We completed our objectives. Our product is ready for initial consumer sensory testing.
Introduction
Seaweed Hydropuntia genus is a marine resource with high economic value (used in food, medicine, and agriculture) and is an indispensable raw material for agar extraction. Therefore, corresponding culturing is ideal in Vietnam. 1 Seaweed has several main components: carbohydrates, proteins, minerals, lipids, and pigments. The carbohydrates are mainly agar and some cellulose. For the human body to effectively use seaweed, it is necessary to first convert the seaweed into liquid form for easy absorption. This involves converting carbohydrates and proteins into their corresponding monomers. The present work liquefies seaweed to create a nutrient-rich suspension that is easy for the body to absorb. The Hydropuntia chemical components have nutritional value and activity. For example, agaro-oligosaccharide (AOS) has many biological activities; including prebiotic, immunomodulatory, antioxidant, and anti-tumor activities.2,3
Protein from Seaweed
Growing seaweed is not in conflict with natural resources and food. Thus, a sustainable diet can consist in part of seaweed protein. The protein content of seaweed is 47% by dry mass (eg, red seaweed). 4 Traditional protein sources, such as cow milk (3.4%), are higher.5,6 Currently, a number of protein-rich seaweeds [eg, Ulva lactuca (Chlorophyta), Undaria pinnatifida (Phaeophyceae), and Porphyra tenera (Rhodophyta)] have been approved by the European Food Safety Authority as human food. 7
Seaweed biomass consists of various proteins. These include glycoproteins (covalently linked to glycans), 8 phycobiliproteins (covalently linked open-chain tetrapyrroles known as phycobilins in red algae), 9 lectins (contain at least one noncatalytic domain that is reversibly linked to specific mono- or oligosaccharides), peptides (protein fragments containing between 3 and 40 amino acids), and cell wall-attached proteins (eg, arabinogalactan proteins; highly glycosylated proteins found in some seaweeds). 10
Therefore, seaweed milk produced from Hydropuntia eucheumatoides (HE) might be a source of nutritious and safe proteins / amino acids.
Lactobacillus
The main carbohydrate in milk is lactose. 11 Upon ingestion, it is hydrolyzed by a lactase (lactose–galactose hydrolase, a border membrane-bound enzyme). Afterward, the intestinal cells absorb glucose and galactose and transport them into the bloodstream. 12 Lactose and corresponding hydrolysis derivatives facilitate calcium absorption and protect against pathogens by aiding aid gut microbiota, and ensure feces consistency. Furthermore, galactose is the main source of white matter in the growing brain. 13
Initial colonization of the gastrointestinal tract is facilitated by Lactobacilli. Vaginal delivery of infants and breastfeeding facilitate delivery of high quantities of Lactobacilli. 14 Lactose is easily fermentable and is the primary source of energy for lactic acid bacteria (LAB) through β-galactosidase activity in many lactobacilli species.15–17 The human body is able to degrade substantial quantities of lactose in the first year of age, yet this ability declines until adulthood. 18
Lactose transport through the bacterial cell is via lactate permease of LAB. This enzyme then catalyzes hydrolysis of the β-1,4-D-glycosidic linkage of lactose, affording glucose and galactose. Next, glucose is metabolized into lactic acid. 19
Lactobacillus is capable of producing the galactosidases for cleaving the 1–4 glycoside bonds of lactose substrate. The lactose substrate has a bond between glucose and galactose; similar to the bond of agar, starch, and cellulose substrates in HE seaweed. Therefore, using Lactobacillus enzyme to hydrolyze HE is appropriate in this study.
Enzymatic Hydrolysis
Hydrolysis of red seaweed polysaccharides, facilitated by small molecules and enzymes, has presents opportunities and challenges. Chemical hydrolysis is cost-effective yet simple and fast. However, chemical hydrolysis is nonspecific, resulting in many monosaccharides and unwanted toxic compounds such as furfural. 20 Enzymatic hydrolysis has many advantages; such as producing large quantities of oligosaccharides, its environmentally friendly nature, as well as producing low quantities of monosaccharides and toxins. A disadvantage is low enzyme activity, necessitating excessive reaction times. High enzyme cost, difficulty of reuse, and eventual loss of enzyme activity are further challenges.
Although enzymatic hydrolysis requires special equipment, the end product does not contain toxins or large quantities of monosaccharides. Corresponding nzymes that produce AOS from red seaweed polysaccharides include α-agarase, 21 β-agarase, 22 β- porphyranase, 23 β-galactosidase, 24 cellulase and pectinase. 25 Microorganisms from many sources produce such enzymes; including the human intestine, marine algae, marine molluscs, seawater, and soil. Furthermore, microorganisms use various enzymes to hydrolyze red seaweed polysaccharides in marine environments. 26
Different glycosidase enzymes exhibit different mechanisms for hydrolyzing red seaweed polysaccharides. For example, α-agarase hydrolyzes α-1,3 linkages in a manner that affords AOS. Alternatively, β-agarase hydrolyzes β-1,4 linkages in a manner that affords neo-AOS. 26 β-porphyranase hydrolyzes the β-(1,4)-galactose glycosidic bonds of porphyran in a manner that produces oligosaccharides. 27 Enzymatic hydrolysis produced a reducing sugar content of 18 g/L of the seaweed Laminaria digitata at 37 °C over the course of 24 h. 24 Enzymatic hydrolysis produces a large quantity of oligosaccharides yet the reaction time is excessive. The literature indicates many reports of various enzymatic methods. Gracilaria spp. agar is hydrolyzed by recombinant β-agarase (Aga 50A) from Escherichia coli in a manner that produces higher quantities of AOS than commercial agarose. 28 Thus, we used commercial Lactobacillus strains that have been used as food. These strains will produce enzymes used to convert HE seaweed into dietary supplements. Lactobacillus enzymes are food-safe enzymes and have high specificity for agar substrate.
Biological Activity of Macroalgae Hydrolyzate
HE seaweed hydrolyzate consists mainly of oligosaccharides, monosaccharides, and amino acids. The main ingredient is AOS, such that the activity of the hydrolyzate is AOS. The literature reveals many inherent benefits of AOS on human health. These include prebiotic, 20 immunomodulatory, 29 anti-inflammatory, 30 anti-oxidant, 31 and anti-tumor, 32 effects.
Oligosaccharides can scavenge various free radicals, such as 2,2'-diphenyl-1-picrylhydrazyl (DPPH) and 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) radicals, in vitro. The C-2 and C-6 hydroxyl groups in oligosaccharides are particularly pertinent to H-atom transfer with these radicals. 33 Some AOS, made from agar and cellulose, exhibit free radical scavenging activities toward ABTS and DPPH; increasing antioxidant activities. 31
In this study, we conducted experiments on the free radical scavenging ability of HE hydrolyzate with DPPH reducing agent. The aim was to evaluate whether this hydrolyzate has antioxidant activity.
Need for the Present Research
There is no research on the hydrolysis method of HE seaweed by using Lactobacillus enzymes. Current work refers to hydrolysis of red seaweed with inorganic acids or commercial enzymes. Our research focuses on the conditions for culturing and obtaining enzymes from Lactobacillus, then using the enzyme to hydrolyze HE into a nutritious and biologically active solution.
Normally, seaweed is used as instant food such as Porphyra, Ulva, Laminaria, Uldaria; the ability to subsequently absorb substances remains limited. Therefore, we converted red seaweed into oligosaccharides, amino acids, and minerals; which are nutrients that are easily absorbed. In particular, converting seaweed into porridge by using enzyme technology does not use chemicals and toxic substances; rendering it safe for the elderly, children, and sick. Such a source of nutritional supplements can be used daily.
Our goal is to produce food supplements from HE seaweed. We evaluated the culture parameters of various Lactobacillus strains to obtain enzymes, and then quantified hydrolysis parameters for this type of seaweed. This nutritious food contains oligosaccharides, amino acids, and minerals that impart strong antioxidant activity.
Materials and Methods
Red Seaweed, Bacterial Strains
H. eucheumatoides (Harvey) 1860—Phylum, Rodophyta; class, Florideophyceae; order, Gracilariales; family, Gracilariaceae; genus, Hydropuntia, species, eucheumatoides—was collected from Hon Rua Island, Nha Trang City, Vietnam.
HE seaweed was harvested from March to June every year; over this time the seaweed has high biomass and the best quality. After harvesting, the seaweed was dried naturally with sunlight, and then impurities in the seaweed (such as sand, coral fragments, sponges, and salt) were removed. The seaweed was then milled into small pieces 0.5–2 mm in size, then packaged and preserved for processing.
The chemical composition of H. echuematoides was carbohydrate, 80.3%; protein-derived nitrogen, 6.2%; lipid, 1.5%; and ash, 12%; and (all percentages are w/w). The high mineral content is attributable to calcification of the seaweed hulls by seawater and adhesion of some of the sand particles onto the surface. The carbohydrates mainly consist of agar, as well as some cellulose and starch. Thus, the two main sugars are galactose and glucose. 34
Lactobacillus strains were Lactobacillus casei, L. bungaricus, L. plantarum, and L. fermentum from KAN Phytochemicals, India. L. rhamnosus was from Swanson Health Products (Fargo, ND, USA).
Fermentation, Obtaining Enzymes from Lactobacillus, and Enzyme Hydrolysis
Fermentation and Obtaining Enzymes from Lactobacillus
Lactobacillus strains were inoculated into standard Man–Rogosa–Sharpe (MRS) medium and 5 g dried HE seaweed was added. Anaerobic fermentation proceeded in the system in a 5-L tank volume bioreactor (Ecell, model 3000, Anyang City, Korea). The solution was treated at 120 °C for 15 min. Then it was fermented at pH 5.2, at 30 °C, 150 rpm, 10d—with an initial microbial count of 6.3 log colony-forming units (CFU)/mL.
At the end of fermentation, there was a cell suspension to centrifuge (Sorvall™ ST 8 Small Benchtop Centrifuge, Thermo Scientific) at 7200 rcf for 15 min to separate cells and crude enzyme solution. The obtained enzyme was used for hydrolysis to create oligosaccharide.
Effect of Lactobacillus Strains on Enzyme Activity
Lactobacillus casei, L. bungaricus, L. rhamnosus, L. plantarum, and L. fermentum were tested. Enzyme activity of Lactobacillus strains was determined by using agar, starch, and carboxymethyl cellulose (CMC) substrates. Afterward, a strain was selected with good enzyme activity.
Effect of Lactobacillus Cultivation Time on Enzyme Activities
The selected strain had its enzyme activity determined at 1–5 d to determine the time of optimum enzyme quality.
Enzyme Hydrolysis
Pretreatment
Dried H. eucheumatoides was mixed with water to a concentration of 10–50 g/L. Subsequent pretreatment was with 5 mM aqueous citric acid (Merck) at 120 °C for 15 min.
In the next four sections, the experimental parameters were as follows, except where noted: crude enzyme solution concentration: 2 mL/g seaweed, H. eucheumatoides concentration: 40 g dry seaweed/L; temperature: 50 °C; reaction time: 96 h.
Effect of Enzyme Concentration on Hydrolysis
Enzymatic reaction conditions were 0.5, 1, 1.5, 2, and 3 (crude enzyme solution, mL/w). The hydrolysis conditions were 40 g dry H. eucheumatoides/L, 96 h, 50 °C, 150 rpm.
Effect of H. eucheumatoides Concentration on Hydrolysis
The H. eucheumatoides concentration was as follows: 10, 20, 30, 40, 50 g/L.
Effect of Temperature on Hydrolysis
The temperature was as follows: 30 °C, 35 °C, 40 °C, 45 °C, 50 °C, and 55 °C.
Effect of Reaction Time on Hydrolysis
The hydrolysis reaction time was as follows: 24, 48, 72, 96, and 120 h.
Biological Activity
Ref. 35 reports the enzyme activities of β-galactosidase, β-glucosidase, and α-glucosidase. The reaction mixture was 1 mL enzyme, 2 mL substrate [0.05% (w/v) (agarose, CMC, and rice starch), and 0.1 M citrate buffer (pH 4.8)]. Incubation was at 50 °C for 30 min. The change in sugar concentration—agarose, CMC, and rice starch—was measured by the Somogyi method, 36 based on the absorbance at 520 nm of a colored complex between copper-oxidized sugar and arsenomolybdate. We used a UV–visible spectrophotometer (model 6405; Jenway, UK). The reaction mixture was 1 mL (carbohydrate standards and sample, 0-1 mg/mL), 1 mL copper reagent, and 1 mL arsenomolybdate reagent. One unit of the activity (U) is the quantity of enzyme that under standard assay conditions released 1 µg sugar from the substrate per minute. 36
The percent antioxidant activity (AA%) of the fluid fermentation was assessed by the DPPH radical scavenging activity, in accordance with Ref.
37
The reaction mixture was 0.5 mL sample, control solution, or blank; 3 mL ethanol; and 0.3 mL DPPH radical in 0.5 mM aqueous ethanol. The AA% was measured at 517 nm with an ultraviolet (UV)–visible spectrometer (model 6405; Jenway, UK):
Analysis
The chemical composition of the seaweed milk was determined as per AOAC. 38 The nitrogen content of the protein was assayed by the Kjeldahl method. 39 Ash was measured after heating the H. eucheumatoides at 1500 °C for 8 h. Lipid was extracted with a Soxhlet extractor (Soxhlet laboratory extractor EV6, Gerhardt Systems, Germany). Total carbohydrate followed Dubois et al. 40
The total sugar mass of the H. eucheumatoides hydrolyzate was determined as per Dubois et al 40 First, the H. eucheumatoides hydrolyzate was centrifuged. Next, the supernatant was collected for subsequent sugar analysis. The sugar concentration was measured with a UV–visible spectrophotometer (model 6405; Jenway, UK). The mass of the oligosaccharide was calculated as the total sugar mass minus the sum of the total lactose, glucose, and galactose mass.34,41
The viscosity was determined with a viscometter (model LVDV-E; Ametek Brookfield, Middleborough, MA, USA).
The lactose, glucose, galactose, and lactose masses were determined by high-performance liquid chromatography (HPLC; Shimadzu, Kyoto, Japan): LC 10AD of the pump, anion–exchange column ICE-AS1 of Dionex (250 mm × 9 mm column dimensions), detection RID 6A. The eluent was 5 mM aqueous H2SO4, flow rate 0.8 mL/min, temperature 45 °C, standards and sample (concentration 0-1 mg/mL), injection volume 10 µL, and retention time 30 min. 42
The amino acids were determined by HPLC: column YMC ODS AQ (250 mm × 4.6 mm column dimensions), detection SPD 10Apv. The eluent was 0.8 mL/min, temperature 45 °C, 0.05% (v/v) aqueous methanol solvent prepared in deionized water; detection by UV–visible spectroscopy, where λ = 205 nm, standards and sample (concentration 0-1 mg/mL), injection volume 10 µL, and retention time 30 min. 43
Metal determination in the seaweed milk refers to Ca, Mg, Na, K; which were analyzed with an atomic absorption spectrometer (Model Solaar M6 Dual Zeeman, Thermo Elemental, Waltham, MA, USA).
Sensory Analysis
Seaweed milk sensory analysis was quantitative descriptive as per ISO 8586: 2012 standard (ISO 2012). There were 10 assessors with at least 1 y of experience in descriptive sensory evaluation of milk products, selected as per their sensory sensitivity based on the correlation value. 44
The examined attributes were selected in a panel discussion; smell (red seaweed: hydrolysis, sterilization, sweet, irritant, and other), color, density, smoothness, viscosity, and taste (red seaweed: hydrolysis, sterilization, sour, sweet, salty, bitter, irritant, and other). 45
Statistical Analysis
All experiments were repeated 3×, duplicate samples. The results are presented as the mean ± standard deviation. Excel 2016 was used for all statistical analyses.
Results and Discussion
The Lactic Acid Bacteria Produced the Enzyme Activities Extracellularly
Effect of Lactobacillus Strains on Extracellular Enzyme Activity
We evaluated the enzymatic activity of the extracellular crude enzyme solution from five Lactobacillus strains with substrates found in HE seaweed: starch, agar, and cellulose (Figure 1). The enzymes that react with the substrates are as follows: β-galactosidase (agar), β-glucosidase (CMC), and α-glucosidase (rice starch). All five strains had enzymatic activity with the substrates and the enzymatic activities of the substrates differed greatly. Agar is the polysaccharide with the largest mass of 90% HE; thus, β-galactosidase had the highest activity, whereas the substrates cellulose produced β-glucosidase and starch produced α-glucosidase with low activity. Among the five strains studied, L. casei and L. rhamnosus had the strongest activity: the β-galactosidase activity was 9.5–11.2 UI/mL. Three strains (L. bungaricus, L. plantarum, and L. fermentum) had lower activity: ranging from 6.5–8.8 UI/mL. In contrast, the enzymatic activity of β-glucosidase and α-glucosidase of these three strains was better than the aforementioned two strains; the L. fermentum enzymatic activity of α-glucosidase was 2.5 UI/mL and that of β-glucosidase was 1.2 UI/mL. Thus, this strain is more suitable for hydrolyzing seaweeds with higher starch and cellulose content than the HE species. We found that L. casei had the highest β-galactosidase activity; thus, it is the most suitable strain for HE hydrolysis and we used it for further studies.
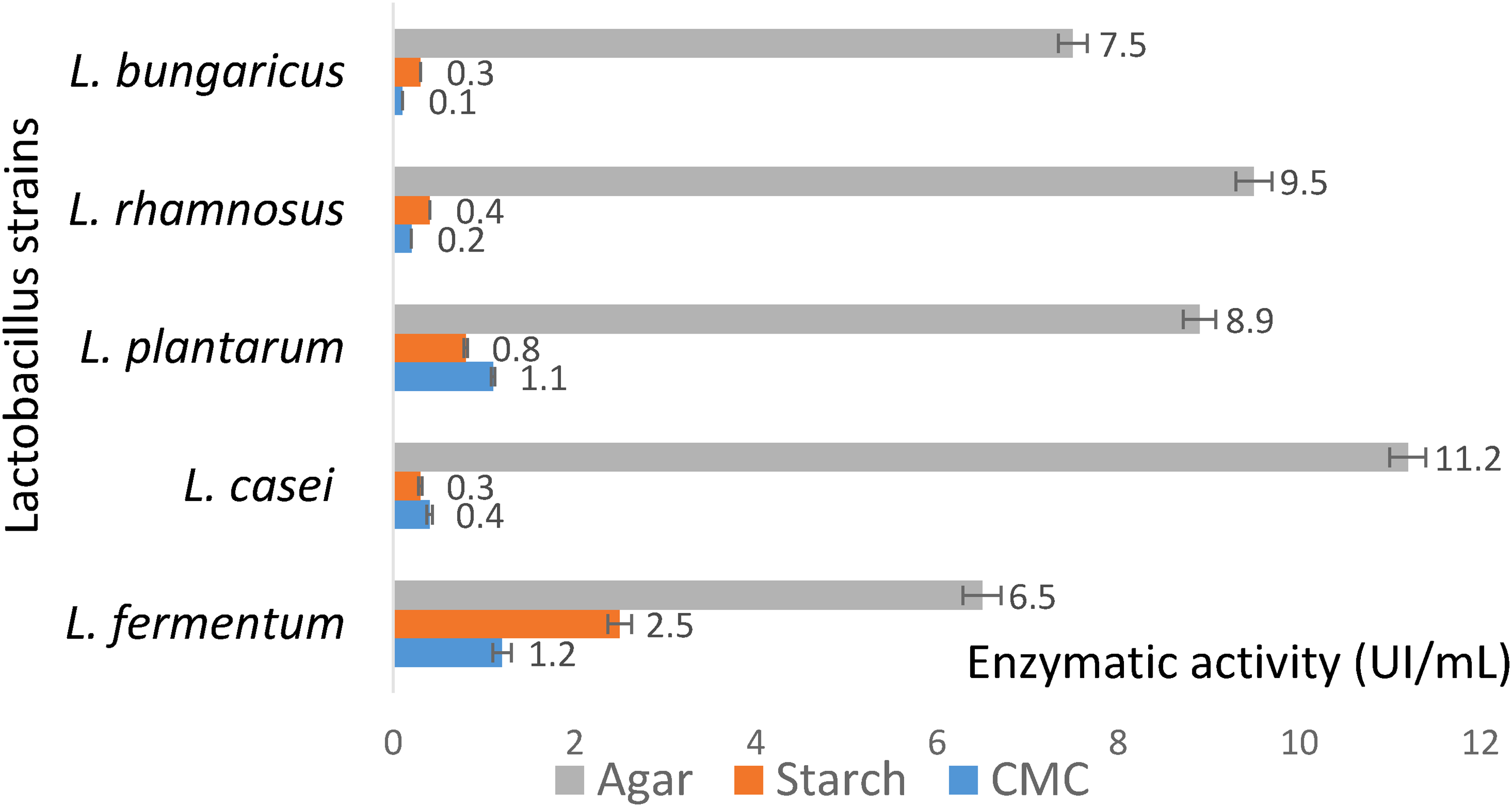
Enzymatic activity of the extracellular crude enzyme solution.
Effect of Culture Time on Extracellular Enzyme Activity
During growth of L. casei, we found that the total enzymatic activity gradually increased over time (Figure 2). From 0–48 h, L. casei only increased biomass production and did not produce enzymes. We observed enzyme synthesis after 24 h, at which time the total enzyme activity was still low: 2.8 UI/mL. From 48–72 h, the enzyme production increased sharply with a total activity of 10 UI/mL. L. casei enzyme production was highest after 96 h of fermentation and then no longer increased; the total activity was 12 UI/mL. During this process, the activity of agar-hydrolyzing enzyme (β-galactosidase) was the strongest at 11.2 UI/mL, whereas the two enzymes that hydrolyze starch (α-glucosidase) and cellulose (β-glucosidase) had low activity: 0.7–0.8 UI/ml. The enzymes α and β glucosidase break down the cellulose capsule layer in a manner that enables β galactase to function. In this study, we chose the time to collect enzymes as after 96 h of fermentation.

Extracellular crude enzyme solution over time.
This study indicates that Lactobacillus strains grew well in MRS medium containing HE seaweed substrate and produced various enzymes. Among them, Lactobacillus casei is the strain that afforded the highest enzymatic activity after 96 h of culture. The highest-quantity enzyme was β-galactosidase, an enzyme that hydrolyzes agar substrate well. The Lactobacillus strains and culture time are two important factors for producing HE hydrolytic enzymes. These two factors have been previously described in Lactobacillus fermentation studies producing β-galactosidase enzyme from milk.17,18 Therefore, enzymes from Lactobacillus casei are suitable for studying the saccharification of HE seaweed.
Factors Affecting Enzymatic Hydrolysis of HE Seaweed
We evaluated factors affecting enzymatic hydrolysis to find suitable conditions. We used standard conditions: ratio of crude enzyme solution to dry seaweed weight (renzyme/seaweed) 2 mL/g, pH 5, HE substrate concentration 40 g/L, 96 h, and temperature 50 °C.
Effect of renzyme/seaweed on Hydrolysis
We evaluated renzyme/seaweed at 0.5, 1, 1.5, 2, 3, 4, and 5 mL/g (Figure 3). The hydrolysis varied with renzyme/seaweed. At an renzyme/seaweed of 0.5 mL/g, hydrolysis afforded a low monosaccharides solution of 9.1 g/L with a high viscosity of 8000 cP. An renzyme/seaweed of 1–1.5 mL/g of monosaccharides resulted in an increase of the monosaccharides content to 20–26 g/L and a decrease in the viscosity to 2500 cP. An increasing renzyme/seaweed up to 2 mL/g increased the monosaccharides content to 35 g/L and the viscosity to 1200 cP, corresponding to oligosaccharides 23.6 g/L and monosaccharides 11.8 g/L. Continuing to increase the enzyme to 3 mL/g, the sugar content no longer increased and the viscosity decreased to 700 cP, due to the increase in the monosaccharide content and the decrease in oligosaccharides. The purpose of this research was to obtain high oligosaccharide content and an appropriate viscosity; thus, we chose an renzyme/seaweed of 2 mL/g.
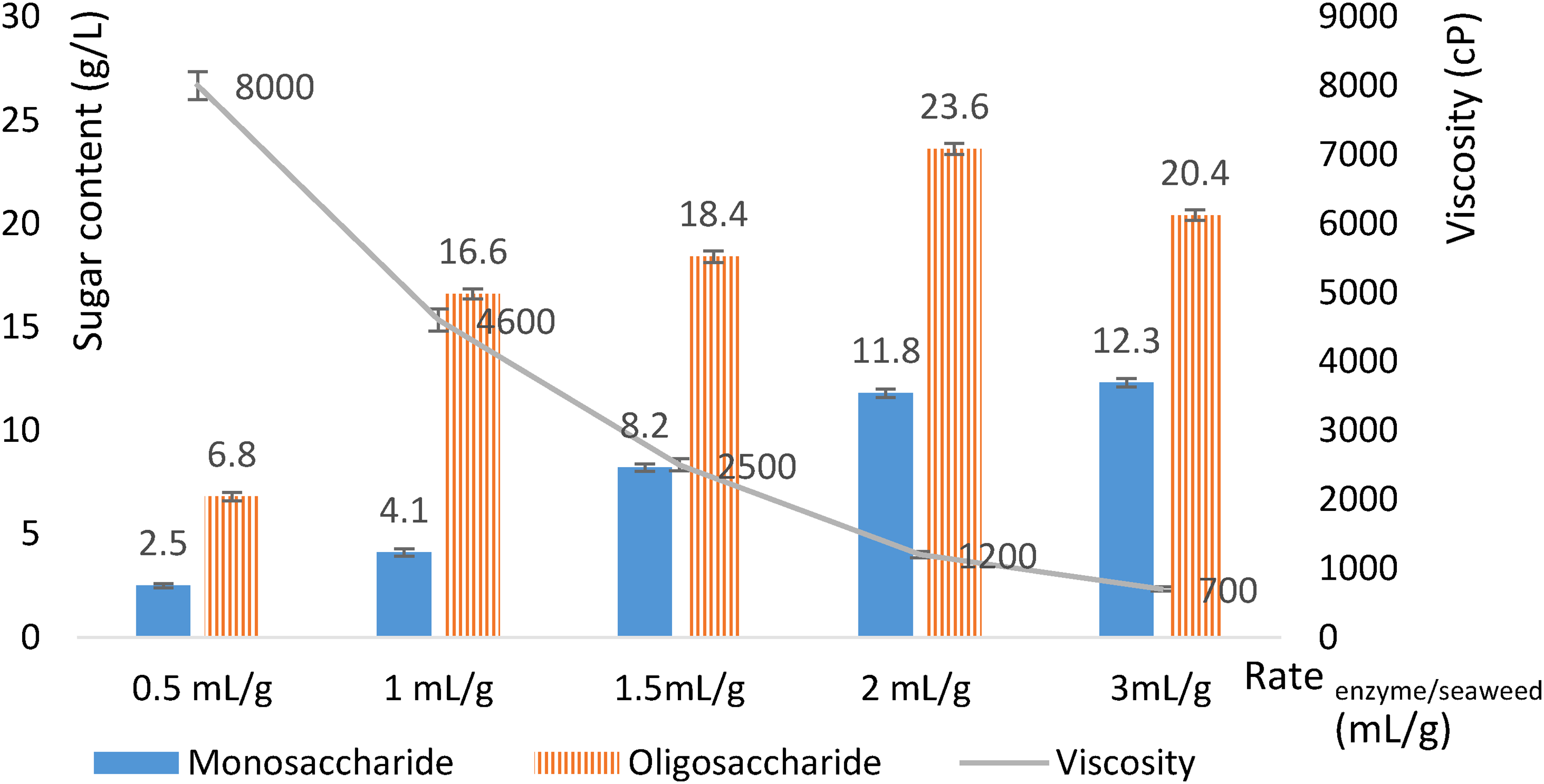
Effect of renzyme/seaweed on hydrolysis.
Effect of Substrate Concentration on Hydrolysis
We used HE substrate concentrations of 10–50 g/L (Figure 4). At an HE concentration of 10 g/L, the monosaccharides content of 6.4 g/L was much greater than the oligosaccharide content of 2.5 g/L; thus, the hydrolyzate had a low viscosity of 300 cP. At HE concentrations of 20 and 30 g/L, the oligosaccharide content was slightly higher than the monosaccharides, and the hydrolyzate had a low viscosity of 570–600 cP.

Effect of substrate concentration on hydrolysis.
At an HE concentration of 40 g/L, the oligosaccharide content of 23.6 g/L was twice as high as the monosaccharides content of 11.8 g/L and the hydrolyzate had a moderate viscosity of 1200cP.
When we increased the HE concentration to 50 g/L, the oligosaccharide and monosaccharides no longer increased and the viscosity in the solution was as high as 1900 cP; at high substrate concentrations, the reaction rate of the enzyme hydrolyzing the substrate decreased. Therefore, an HE concentration of 40 g/L is an appropriate concentration for enzymatic hydrolysis in a manner that affords oligosaccharide.
Effect of Temperature on Hydrolysis
When hydrolyzed at 30 °C to 40 °C, the low monosaccharides content was 1.8–2.9 g/L and the oligosaccharide sugar content was 18.7–19.6 g/L; many times higher than the monosaccharides (Figure 5). Upon increasing temperature from 45 °C to 50 °C, the monosaccharides content of 7.2–11.8 g/L increased rapidly and the oligosaccharide content of 23.6 g/L reached its peak. Upon increasing temperature to 55 °C, the monosaccharides and oligosaccharide content were nearly equal (16.4 and 18.2 g/L), which indicates that the oligosaccharide content no longer increased but was converted into monosaccharides. Thus, hydrolysis at a temperature of 30 °C to 40 °C resulted in minimal change, producing moderate oligosaccharide content and low sugar solubility; accordingly, the enzyme activity was low at this temperature. When we increased the temperature to 45 °C to 50 °C, the enzyme hydrolyzed well in a manner that created highly soluble oligosaccharides and sugars. Upon increasing the temperature to 55 °C, conversion of HE into monosaccharides increased and oligosaccharide decreased.

Effect of temperature on hydrolysis.
The 30 °C to 55 °C thermal process corresponds to conversion of sugar and a change of viscosity. At temperatures of 30 °C to 40 °C, the high viscosity of −4800–6800 cP was due to the high oligosaccharide content and negligible monosaccharides. At a temperature of 45 °C to 50 °C, the viscosity decreased to 1200–2700 cP, the oligosaccharide content was high, and the sugar solubility increased slightly. At a temperature of 55 °C, the viscosity was low (450 cP) and the monosaccharides as well as oligosaccharide content were nearly equal. Thus, as the temperature increased, the oligosaccharide content decreased, the monosaccharides content increased, and the viscosity decreased. The sugar content was approximately linear with viscosity.
Effect of Time on Hydrolysis’
We hydrolyzed HE seaweed for 120 h (Figure 6). Every 24 h we extracted samples to evaluate the content of monosaccharides, oligosaccharides, and viscosity. At 0–24 h, the oligosaccharide content was <10 g/L and the viscosity was high: > 10,000 cP. By 48 h, the oligosaccharide content increased to 18 g/L and the viscosity gradually decreased to 6000 cP. By 72 h, the highest oligosaccharide content was 28 g/L but the viscosity was still high: 2100 cP. By 96 h, the oligosaccharide content no longer increased but the monosaccharides content increased to 11.8 g/L and the viscosity decreased to 1200 cP. By 120 h, the dissolved sugar continued to increase to 15.8 g/L and the viscosity decreased to 450 cP. Thus, the viscosity was inversely proportional to the dissolved sugar and oligosaccharide. An increasing hydrolysis time corresponded to an increasing conversion into monosaccharides. Therefore, an appropriate time to obtain high oligosaccharide content was 96 h of hydrolysis.
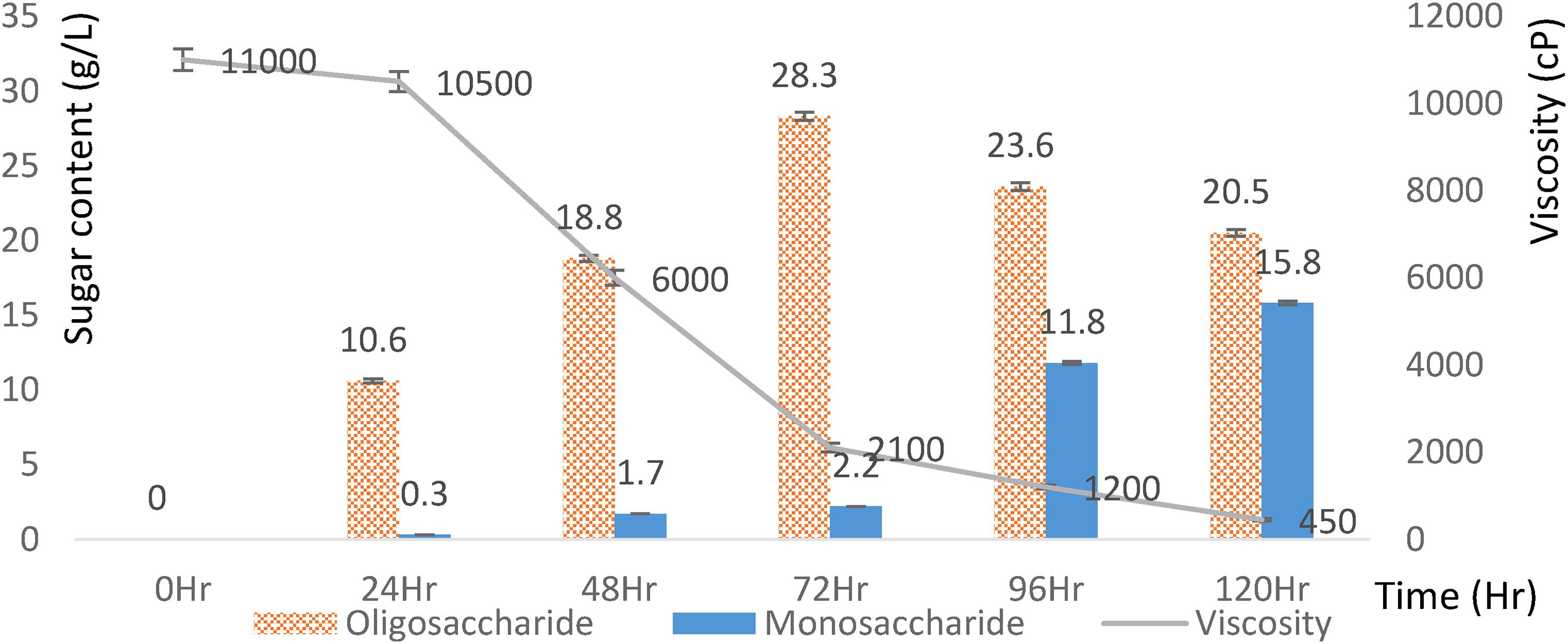
Effect of time on hydrolysis.
We determined the parameters of hydrolysis of HE seaweed with Lactobacillus enzymes for producing seaweed milk. This process was mainly influenced by the seaweed concentration, renzyme/seaweed, temperature, and time. The parameters were as follows: mass of seaweed HE 40 g/L, 2 mL of enzyme/g seaweed, temperature 50 °C, pH 5, for 96 h; at the end of hydrolysis and heating to 80 °C for 10 min. The results of this study differ from the literature. Ariga et al (2014) reported characterization and purification of α-neo-AOS hydrolase from enzyme of Cellvibrio sp. OA-2007 at an agar concentration 4 g/L, temperature 35 °C, and pH 7.0–7.2 for 30 min. 21 Lee C. H et al (2015) reported saccharification of agar by hydrothermal pretreatment and enzymes supplemented as follows: seaweed Saccharophagus degradans concentration of 50 g/L, β-galactosidase of Vibrio sp. EJY3, temperature 35 °C, pH 7.0 for 12 h. 24 We hypothesize that the reason for the differences between our study and the literature lies in the research objective. In particular, our research used enzymes from commercial Lactobacillus strains, which are safe and used as food. However, the enzyme source of Ariga et al (2014) and Lee C. H et al (2015) were obtained from Vibrio sp. This is a strain that has not been revealed to be safe for consumption. The limitation of my study is similar to other studies: the production time for a batch of seaweed milk is up to 96 h. The reason is that the saccharification process of seaweed takes a long time. However, seaweed milk is safe for users.
Chemical Properties
We hydrolyzed HE seaweed by using Lactobacillus casei enzymes to create a solution containing mainly oligosaccharides, monosaccharides, amino acids, and minerals (Table 1). These are compounds that are easily absorbed by the human body and have biological activity.
Chemical Properties of Seaweed Milk of 1 L.

Seaweed HE's main chemical composition is carbohydrates; which can be hydrolyzed to produce oligosaccharide, lactose, galactose, and glucose. The contents were as follows: oligosaccharide 23.6 g/L, lactose 10.7 g/L, glucose 0.6 g/L, and galactose 0.5 g/L. In this research, the goal was to create a high content of oligosaccharides such that this product had good biological activity. The lipid content in seaweed milk is low: 0.6 g/L (1.5% dry mass) and easily absorbed by the human body.
Nitrogenated protein was the second-largest content. We found that with a HE seaweed mass of 40 g/L, the total protein content was 3.04 g/L (7.5% dry mass) and the amino acid content in the solution was 0.89 g/L. The total protein content was low compared with some seaweed species such as Mastocarpus stellatus of 16.9% dw, 46 Durvillaea antarctica of 9.7% dw, 47 and Ulva rotundata of 10% dw. 48 The main amino acid composition of the present hydrolyzate was aspartic acid 400 mg/L, glutamic acid 200 mg/L, glycine 100 mg/L, and arginine 100 mg/L. Although the types of amino acids were not diverse and the content was not high, it might be a suitable product for dieters.
The seaweed milk contained calcium 205 mg/L, magnesium 110 mg/L, sodium 260 mg/L, and potassium 82.4 mg/L. These minerals are of natural origin; thus, they are easily absorbed and might be safe for consumers.
Currently, there are many types of milk; including cow and plant-based milks (Table 2). The nutritional composition of each type of milk depends on the plant source as well as the production process and the level of nutrient fortification. Most dairy products are fortified to provide trace quantities of minerals and vitamins, although the quantity of other micronutrients is often unknown. Besides flavor and texture, protein and added sugars are perhaps the most variable attributes among plant-based milks. The biggest difference between seaweed milk and other plant-based milks is the oligosaccharide content of agar, amino acids, and saccharose sugar; not added during production of seaweed milk. This is a clear difference in seaweed milk compared with cow and other plant-based milks.
Nutrition Composition of various Types Milk for a 240-mL Cup.

Information obtained from the USDA Food Composition Databases. 49 N/A = not available
The importance of producing seaweed milk in terms of nutritional benefit to consumers is as follows. Seaweed milk is a new type of beverage. It provides more choice for daily drinks for users. Seaweed milk contains the same sugars glucose, galactose, and lactose and minerals as cow's milk; but it has much fewer lipids than cow's milk. Therefore, it is easily absorbed by children and sick people. In Vietnamese hospitals, in the first 12 h after surgery, doctors often give sick people bird's nest beverage. This is a beverage with main ingredients agar and carragenan. Therefore, seaweed milk will likely be suitable for sick people. Galactose in seaweed milk is the main source of white matter in the growing brain and is essential for young children. Furthermore, seaweed milk can be mixed with syrup to create fun drinks.
Seaweed milk meets the requirements for industrial production because seaweed raw materials have been cultivated to obtain biomass. Seaweed milk production technology is not complicated. It includes two main processes of enzyme production and saccharification; the technological parameters have been shown in this article. The production equipment is a bioreactor system that controls temperature, pH, dissolved oxygen, and mixing. This is a common system in beverage production; thus, seaweed milk is likely to be profitable from an industrial perspective.
Antioxidant Activity
We evaluated the DPPH free-radical results of seaweed milk in terms of AA%. The AA% coefficients were 72.32 ± 0.3%; standard sample 2 mg/L ascorbic acid, 95.5 ± 0.7%. The two samples substantially differed from one another yet were still within ∼23% of each other. The antioxidant capacity of seaweed milk was also high. This mainly depended on the quantity of oligosaccharide as well as amino acid in the fluid. Seaweed milk has high antioxidant capabilities. For example, Kang et al (2014) reported the antioxidant activity of agaro-oligosaccharide (24% with 500 µL of sample), 31 and Cheong et al (2018) reported antioxidant activity of galactose oligosaccharide (65% with 200 µL of sample). 50 We hypothesize that the different antioxidant activity of this study compared with other studies is because of the amino acid and lipid composition.
Sensory of Food Supplement from Macroalgae: Qualitative Characteristics
The assessors’ sensory evaluation results for the seaweed milk indicate some main characteristics of this product (Table 3). The density, color, viscosity, and smoothness of the seaweed milk were typical. The density is dense/runny, the color is similar to that of white agar, the viscosity is ductile, and the smoothness is sandy/floury/smooth. The smell is that of light agar: similar to honey and sugar. The taste is slightly sweet.
Sensory of Food Supplement from Macroalgae: Qualitative Characteristics.

Note: Attribute in terms of intensity strengths: no"-”, little"+”, moderate"++”, high"+++”
The smell, color, and viscosity of this drink constitute its main distinctions from rice milk and nest beverage. The smell of this product is characteristic of agar saccharification. The color is similar to that of agar. The product is brighter than rice milk, and the viscosity is the same as rice milk but lower than nest beverages. Future work will focus on consumers’ sensory acceptance of red seaweed milk.
Conclusions
We produced a food supplement based on H. eucheumatoides. The seaweed was primarily agar and had a high protein as well as mineral content. We cultivated Lactobacillus strains that are suitable for obtaining appropriate enzymes. We maximized th hydrolysis yield by optimizing the enzyme formulation, H. eucheumatoides concentration, time, and temperature. We determined the contents of the active ingredients in seaweed milk products. They might be useful as supplements for the human body and have antioxidant activity.
Thus, we confirmed our hypothesis that seaweed milk from macroalgae Hydropuntia eucheumatoides might be useful as food supplements and are antioxidants. Future study will focus on the seaweed milk's oligosaccharide activity as well as consumers’ sensory acceptance.
Seaweed milk is likely to be safe because it is produced from natural ingredients that are similar to food that has already been approved for human consumption. The production process uses biological methods and biological strains that are currently found in commercialized food. Nevertheless, rigorous tests for safety require additional studies.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Vietnam Academy of Science and Technology, (grant number CSCL18.02/23-24).
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.

