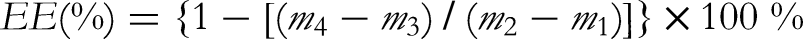Abstract
Introduction
Ziziphi Spinosae Semen (ZSS), derived from the dried seeds of Ziziphus jujuba Mill. var. spinosa, Rhamnaceae, is a renowned traditional Chinese medicine with high pharmaceutical value. 1 It is rich in phytochemicals and exhibits a variety of biological activities. Fatty oil accounts for approximately 30% of its main components. 2 Ziziphi Spinosae Semen oil (ZSSO) is a fatty oil mixture extracted from ZSS and it mainly comprises unsaturated fatty acids (89.16%), saturated fatty acids (10.55%), hydrocarbons, and esters. Sun et al identified 20 main fatty acids by gas chromatography and they found that methyl oleate (44.23%) and methyl linoleate (45.33%) collectively accounted for 89.56% of the fatty acids in ZSSO. 3 Since unsaturated fatty acids are not only a very important nutrient but also an essential component of cell membranes, their content and type determine the nutritional and healthy value of ZSSO. 4 Oleic acid (24.46%-56.375%) is present in the largest amount among ZSSO's unsaturated fatty acids extracted by various methods such as Soxhlet extraction and supercritical fluid extraction. 5 Olive oil exerts modulatory effects on a wide range of physiological functions. Oleic acid has anticancer 6 and anti-inflammatory 7 activities and can facilitate wound healing. 8 Xie et al studied the in vivo activity of the lotion prepared from fatty oil in ZSS and the results revealed that the lotion had a cardiotonic effect on isolated toad heart. 9 ZSSO not only has the biological activity of unsaturated fatty acids but also is closely related to various unique effects of ZSS. As a medicinal and edible homologous traditional Chinese medicine, ZSS has various pharmacological effects such as sedation, hypnosis, antianxiety, antidepression, and memory improvement.10-12 ZSSO (10.8 g/kg bodyweight) can substantially inhibit the autonomous activity of normal mice (P < 0.05), prolong the sleep time induced by pentobarbital sodium (P < 0.01), and shorten the subthreshold dose sleep latency of pentobarbital sodium (P < 0.05). 13 These results suggest that ZSSO has anti-insomnia activity. Sun et al revealed that the terpenoids present in ZSSO targeted different sleep-aid pathways to varying degrees and exhibited potential for preventing central nervous system diseases. The sleep-promoting assay discovered that the terpenoids present in ZSSO enhanced gamma-aminobutyric acid (GABA) and downregulated glutamate (Glu), IL-1β, and norepinephrine expression. 3 ZSSO is a kind of multifunctional and healthy edible oil. It is nontoxic and harmless to the human body and shows huge potential for development. However, the use of ZSSO as a food ingredient is limited because of its susceptibility to oxidation and lack of corresponding pharmacological research. Microcapsulation is originally a technology for packaging solids, and it involves the incorporation of food ingredients and the release of liquids embedded in sealed capsules. The encapsulation of microcapsules can reduce the evaporation rate of their core materials in the external environment. 14
Complex coacervation is a liquid-liquid separation phenomenon that typically occurs between two different aqueous mediums. For these two mediums, there are multiple available options and various types of intermolecular interactions (electrostatic interaction, hydrogen-bonding interaction, cation-π interaction and multivalency) between each pair of the participants. 15 Complex coacervation also is a microencapsulation technique for producing polymers, and it involves the interaction of water-soluble polymers with opposite charges. 16 Natural polymers (including gelatin, sodium carboxymethyl cellulose, sodium alginate, etc) have been widely and intensively investigated in the field of micro-/nano-encapsulation technologies via complex coacervation in the food and cosmetic industries. Compared to positively charged components, more negatively charged components can be available. 15 Microcapsules prepared by complex coacervation can maintain a stable structure even at high temperature, so they are suitable for embedding volatile and perishable fatty oil. 17
The selection of wall material combinations is crucial for the preparation of microcapsules by complex coacervation. Common wall material combinations include gelatin-sodium alginate, 18 gelatin-arabic gum, 19 gelatin-chitosan, whey protein-arabic gum, gelatin-carboxymethyl cellulose, 17 lactoferrin-sodium alginate, 20 and maltodextrin-whey protein. Gelatin is a common amphoteric polymer excipient that can be used in various pharmaceutical preparations and has been widely used in microcapsule production. 21 Sodium alginate is an anionic polyelectrolyte that can interact with cationic polymers, form composite materials with good film-forming properties, and exhibit slow-release and protection effects. 22 Notably, the gel formed by sodium alginate and cation (such as Ca2+ and Zn2+) under mild conditions can enable some drugs to achieve sustained release. Sodium alginate has been applied in the pharmaceutical and food industries because of its unique structure. In addition, as a surfactant with hydrophilic and oleophilic groups, emulsifiers can increase system dispersion and maintain emulsion stability. Therefore, various nontoxic and biodegradable emulsifiers have been widely used in the modern food industry.
This study mainly aimed to prepare microcapsules embedded with ZSSO by complex coacervation. ZSSO was encapsulated within gelatin-sodium alginate, which protected it from deteriorating environmental and processing conditions. The general physicochemical properties, microstructure, and storage stability of microcapsules were measured by scanning electron microscopy (SEM), Fourier transform infrared spectrometer (FTIR), and thermogravimetric analysis (TGA). Additionally, the sleep improvement activity of ZSSO and ZSSO microcapsules (ZSSO-MPs) was preliminarily explored through in vivo pharmacological experiments.
Materials and Methods
Chemicals and Reagents
Analytically pure Gelatin (CAS:9000-70-8; LOT: J11HS188235) was procured from Shanghai Yuanye Bio-Technology Co., Ltd China. Analytically pure Alginate (CAS:9005-38-3; LOT: P2098391) was purchased from Shanghai Titan Scientific Co., Ltd China. Food Grade Glutamine transaminase (LOT: 2023020201) was purchased from Jiangsu Yiming Biological Technology Co., Ltd China. Analytically pure SP80 (CAS: 1338-43-8; LOT: 20200508), analytically pure acetic acid (CAS: 64-19-7; LOT: 20230102), analytically pure sodium hydroxide (CAS: 1310-73-2; LOT: 20200908) and analytically pure petroleum ether (CAS: 8032-32-4; LOT: 20220402) were provided by Tianjin Tianli Chemical Reagents Co., Ltd China. PCPA (CAS: 7424-00-2, LOT: C15212228) was purchased from Shanghai Macklin Biochemical Technology Co., Ltd China. Mice 5-HT (LOT: 202310), GABA (LOT: 202310) and Glu (LOT: 202312) ELISA kits were purchased from Jiangsu Meimian Industrial Co., Ltd China. Furthermore, deionized water was used in all experiments.
Experimental Animals
SPF male Kunming mice weighing 22 ± 2 g were provided by the CHENGDU DOSSY Experiment Animals CO., LTD (production license number: SCXK2020-0030, Chengdu, Sichuan). The mice were kept under the following conditions: 23 °C ± 2 °C, 50%-65% relative humidity, and a 12 h light and 12 h dark cycle (light on 9:00-21:00). They were given a 1-week acclimatization period before the experiment began. The mice were provided unrestricted access to food and water, and their weight is monitored daily. All the experimental procedures for the animals complied with the guidelines of the laboratory animal ethics committee at Shaanxi University of Traditional Chinese Medicine (NO. SUCMDL20231024004).
Extraction of ZSSO
The ZSS used in this study was harvested from Zhongshan Mountain, Chunhua County, Xianyang, Shaanxi Province, and its identity was confirmed by Director Pharmacist Zhongxing Song of Co-construction Collaborative Innovation Center for Chinese Medicine Resources Industrialization by Shaanxi. The ZSS was dried in the baking oven at 40 °C for 4 h, then crushed and passed through a 50-mesh sieve. Approximately 20.0 g of powdered ZSS was collected, and 100 mL of petroleum ether (60 °C-90 °C) was added and extracted by ultrasonication (300 W, 40 kHz) for 1 h. Then, the extract was filtered, and the solvent was evaporated under reduced pressure for ZSSO production.
Preparation of ZSSO-MPs
ZSSO-MPs were prepared by complex coacervation. The effects of wall material concentration, wall-to-core material ratio, and emulsifier concentration (based on response surface method) on the preparation of microcapsules were examined. 23 Gelatin and sodium alginate (3:1, w/w) were dissolved separately by heating in a water bath at 45 °C. ZSSO and Span80 were added to the sodium alginate solution and dispersed at 10,000 rpm for 10 min with LC-SFJ-10-type handheld homogenizer at 160 W. Subsequently, gelatin solution was added to the emulsion and dispersed at 12,000 rpm for 10 min to form an oil-in-water emulsion. The pH value of the solution was adjusted to 4.4 with 10% glacial acetic acid and stirred at 45 °C and 600 rpm for 1 h. The pH value was measured by LPH-A-type desktop pH meter. The mixture was cooled to low temperature (<10 °C) with an ice bath and stirred 5 times, each time lasting for 5 min. The pH was then adjusted to 6.0 with 10% sodium hydroxide. After adding glutamine aminotransferase, the mixture was stirred at low temperature (<10 °C) at 600 rpm with HCJ-2E-type magnetic stirring water bath pot for 3 h to prepare a microcapsule solution. Pour the microcapsule solution into a glass culture dish and pre cool at −80 °C for 6 h. Finally, the surface cured lotion was freeze-dried in vacuum to prepare ZSSO-MPs with LGJ-10-type freeze-drying machine at 970 W. 24
Encapsulation Efficiency (EE %)
Amount of entrapped oil in the microcapsules was determined using the method showed by Xu et al 25 The total amount of ZSSO was determined as follows: 0.5 g of powder was placed in a conical flask, 25 mL of petroleum ether (30 °C −60 °C) was added, ultrasonic extraction performed for 40 min, the mixture was filtered to a known weight (m1) of the conical flask, and then the solvent was evaporated to a constant weight (m2) with water bath. (m2-m1) is the total oil content in the powder.
The surface oil content was determined as follows: 0.5 g powder was added to a conical flask, 20 mL of petroleum ether was then added (30 °C −60 °C). After gently shaking the mixture for 30 s, centrifuge for 1 min (4000 rpm). The supernatant was collected and added to a conical flask of known weight (m3), and the solvent was evaporated to constant weight (m4) in a water bath. (m4-m3) was the surface oil content in the powder. EE % was calculated using the following equation:
Particle Size and zeta Potential
The particle size and zeta potential for each microcapsule solution was measured with a Nano-ZS90 (Malvern Instruments Ltd, UK) at 25 °C, and distilled water was used as the dispersant. Each measurement was repeated three times. 26
SEM
The surface microstructure of the ZSSO can be observed intuitively by SEM (ZEISS Sigma 300, USA). Spread a small amount of sample evenly on the conductive tape pasted on the sample stage, and perform gold spraying treatment in a vacuum environment. The acceleration voltage during photos shooting were 3 kV, with a magnification of 2000 times. 27
FTIR
The functional group characteristics of the sample were analyzed by FIIR spectroscopy. An appropriate amount of freeze-dried sample and KBr (spectral pure) powder were grinded thoroughly in a dry agate mortar. Samples were mixed well before pressing into transparent thin sheets. The thin film was placed in an infrared spectrometer for spectral scanning with a wavelength scanning range of 4000–400 cm−1 and a resolution of 4 cm−1. 28
TGA
TGA was performed on a HITACHI STA200 simultaneous thermal analyzer. The carrier gas was nitrogen with a flow rate of 20 mL/min. The initial temperature was 35 °C and then raised to 500 °C at a rate of 20 °C/min. Mass changes were continuously recorded as a function of temperature. 29
Oxidative Stability Analysis
The oxidative stability of ZSSO and ZSSO-MPs was assessed using an accelerated oxidation test at 60 °C for 18 d. The oxidative level was determined by peroxide value (PV) tests. The samples were stored in amber bottles sealed to protect them from exposure to light and then placed in a pharmaceutical integrated stability incubator at 60 °C and 50% RH for 18 d. Oxidative testing was conducted every 3 d throughout the 18 d testing period.30,31 The determination method was based on GB/T 5009.227–2016 “Method for analysis of hygienic standard of edible oils.” Each sample (2.0 g) was mixed with 30 mL of trichloromethane–ice acetic acid (V: V = 2:3) in a 250 mL iodine flask. Furthermore, the mixture was shaken until the sample completely dissolved. Then, 1 mL of saturated potassium iodide solution was added. The flask was covered with the stopper, shaken slightly for 30 s, and left in the dark for 3 min. After pure water (100 mL) was added, the mixture was shaken well and then titrated with 0.01 mol/L sodium thiosulfate standard solution immediately. Oscillation and titration were performed simultaneously. A starch indicator (1%; 1 mL) was added after the solution turned light yellow. Titration with sodium thiosulfate standard solution and oscillation were performed until the blue color of the solution disappeared as the end point of the titration. A blank test was performed. Each experiment was repeated three times. The PV was calculated according to the following formula:
Establishment of PCPA-Induced Insomnia Mice Model
After 5 d of acclimatization, 90 mice were arbitrarily divided into nine groups: control, model, diazepam positive control (Dp, 2.0 mg/kg), low-dose ZSSO (ZSSO-L, 1.8 g/kg), medium-dose ZSSO (ZSSO-M, 3.6 g/kg), high-dose ZSSO (ZSSO-H, 7.2 g/kg), low-dose microcapsule (ZSSO-MPs-L, 9.0 g/kg), medium-dose microcapsule (ZSSO-MPs-M, 18.0 g/kg), and high-dose microcapsule (ZSSO-MPs-H, 36.0 g/kg). The control group received CMC-Na solution at a dosage of 0.1 mL/10 g through gavage. The remaining groups received PCPA at a dosage of 350.0 mg/kg for 3 d through gavage. PCPA was dissolved in 0.5% CMC-Na solution to replicate the insomnia model. After 3 d, the model group exhibited disheveled hair, considerably decreased body weight, and irritability, indicating that the modeling of insomnia was successful. 32 On the fourth day of modeling, the treated groups received the corresponding samples for successive 7 d.
Sample Collection and Preparation
After 1 h of the last administration, the blood was collected from the orbits of mice and centrifuged at a speed of 6000 r/min for 10 min to obtain the serum samples, which were stored at −80 °C for reserve. The mice euthanized through cervical dislocation. The scalp was cut along the anterior midline of the mouse skull, and the skull was carefully pried open. The entire brain tissues were taken out and washed with normal saline. Three brain tissues of each group were taken into 50 mL centrifuge tubes containing 4% tissue fixative for HE staining. The other brain tissues were taken into 5 mL EP tubes and immediately placed on dry ice, followed by transferring to −80 °C refrigerator for content determination.
Sleep Induction Test
The pentobarbital sodium-induced sleep test is commonly used in evaluating whether a drug has sedative–hypnotic activity. On the sixth day of administration, 30 min after gavage, sleep induction experiments were performed by intraperitoneal injection of sodium pentobarbital (50.0 mg/kg) in each group of mice. After inducing sleep, sleep latency and sleep time were measured. The sleep latency was measured until movement ceased, and sleep time was measured as the time it took for the mouse in a supine position to perform a righting reflex, ie, the time it takes to return to a prone position on all fours after waking. The control group was given the same volume of normal saline (0.9%) by gavage as the administration group. 33
Assay for Brain 5-HT, GABA and Glu
Mice brain tissues were removed from −80 °C refrigerator and thawed at room temperature. After weighing, brain tissues were placed in a 2 mL EP tube filled with steel beads, 9 times normal saline was added and grinded tissues into homogenate by grinder. The homogenate was centrifuged at 3000 r/min for 10 min and the supernatant was taken. Finally, the levels of 5-HT, GABA and Glu in the tissues were determined according to ELISA.
Histological Observation
The brain tissues of each mice group were fixed in a 10% formalin solution. The processed and washed brain tissues were sequentially dehydrated with gradient ethanol (50%-100%), washed with xylene, and embedded in paraffin. Then they were sliced into sections (3-5 μm) to perform routine hematoxylin and eosin (H&E) staining, sealed with neutral resin, and evaluated using a light microscope at a magnification of 300. The hypothalamic tissue lesions in mice were assessed using H&E staining.
Statistical Analysis
Statistical analysis was performed using the Statistical Package for the Social Sciences (SPSS, version 19.0). The normally distributed data with equal variances were expressed as the mean ± standard deviation. Multiple comparisons were performed using a one-way analysis of variance (ANOVA) followed by the least significant difference (LSD) test. A P < 0.05 was considered statistically significant for the analyses while a P < 0.01 was considered extremely significant. Data statistical analyses were performed by SPSS 19.0 software and graphics were drew by Origin 2021. All software ran in the operating system of Microsoft Windows 10 Professional.
Results and Discussions
Encapsulation Efficiency, Particle Size and zeta Potential
In this study, microcapsules were prepared by the complex coalescence method, and the effects of wall material concentration, wall-core ratio, and emulsifier concentration on the EE of microcapsules were investigated. According to the results of response surface optimization, the encapsulation process of microcapsules was determined as follows: wall material concentration of 2.0%, wall-core ratio of 3:1, emulsifier concentration of 0.5%, and the EE of the prepared microcapsules was 56.08% ± 0.19%. The particle size and zeta potential are the important indicators for evaluating the quality and application of microcapsules. As shown in Figure 1, the ZSSO-MPs prepared had a uniform particle size distribution with an average particle size of 166.17 nm. The zeta potential of ZSSO-MPs was 38.2. when the zeta potential of the particles was ≥|30|, indicating that they were stable.

Particle size and zeta potential of ZSSO-MPs (n = 3).
Results of SEM
Under the electron microscope, the ZSSO-MPs showed smooth and dense spherical surfaces with adhesion between particles (Figure 2). The appearance of adhesion can be attributed to the incomplete cross-linking and polymerization of high concentrations of gelatin and sodium alginate in solution, and their colloidal properties causing adhesion between ZSSO-MPs particles during freeze-drying, resulting in adhesion phenomenon. The capsule walls of ZSSO-MPs were relatively complete without cracks, indicating that the wall material had a favorable protective effect on the core material.

Scanning electron micrographs of ZSSO-MPs.
Results of FTIR
FTIR spectroscopy analyses were performed to characterize the functional group characteristics of gelatin, sodium alginate, ZSSO and ZSSO-MPs (Figure 3).

FTIR spectroscopy analysis.
The spectrum of gelatin presented characteristic peak at 3448 and 1636 cm−1 attributed to the water molecule O-H vibration mode, indicating that the gelatin contains a large amount of bound water. In addition, the width of absorption peaks at 3448 and 1636 cm−1 increased, containing stretching vibrations of N–H and C = O in the amide group, respectively.
The spectrum of sodium alginate showed absorption bands of hydroxyl and N-H stretching vibration (3433 cm−1); Symmetry and asymmetry of methylene stretching (2921 cm−1); C = O stretching (1626 cm−1); The absorption peak intensity near 1405 cm−1 is relatively high, and its composition is complex, mainly including methyl/methylene deformation vibration, carboxylic acid group C = O symmetric stretching vibration.
ZSSO exhibited C = H stretching peak at 3008 cm−1, which was attributed to unsaturated fatty acid. The peaks at 1745 and 1711cm−1 were due to carboxylate group C = O stretching vibrations. The absorption peak at 1462 cm−1 contained the C-H bending vibrations of methylene and methyl groups.
The spectrum of the prepared ZSSO-MPs exhibited peaks for gelatin, sodium alginate, and ZSSO-related absorption bands. The absorption peaks of microcapsules at 2927, 2855, and 1720cm−1 are due to the C-H and C = O stretching vibrations in ZSSO, indicating that the MP products contained ZSSO. The shift in the C = O absorption peak of the amide group at 1648 cm−1 demonstrated a polyelectrolyte reaction between gelatin and sodium alginate in the wall material. In addition, the infrared absorption spectra of the ZSSO-MPs did not show any characteristic absorption peaks different from those of gelatin, sodium alginate, and ZSSO, indicating that no new chemical bond formed during the coacervation reaction. This behavior indicated that ZSSO was successfully encapsulated.
Results of TGA
Weight loss in different samples in function of temperature and to evaluate the thermal stability of the ZSSO and ZSSO-MPs through TGAs (Figure 4). The ZSSO started to degrade slowly after 140 °C and nearly completely decomposed at approximately 500 °C; approximately 1.2% of the mass remained. ZSSO-MPs decomposed slowly before 240 °C, which indicate that the water content of the prepared microcapsules is relatively low. When the temperature is between 239.1 °C and 361.4 °C, the ZSSO-MPs decomposed rapidly. At this stage, the wall material started to decompose, including the decomposition of bound water and volatile substances. When the temperature increased to 500 °C, and 23.1% of the ZSSO-MPs mass remained. Comparing the fusion temperature of ZSSO to that of loaded microcapsules, it was found that pure ZSSO has a greater mass loss, which may prove that the wall material formed by gelatin and sodium alginate has a significant protective effect on ZSSO. Therefore, the TGA results showed that the heat loss of ZSSO-MPs was lower than that of ZSSO, which indicated ZSSO-MPs thermal stability during the heating process.

Thermogravimetric analysis.
Oxidative Stability
ZSSO has a high content of unsaturated fatty acids and is highly susceptible to oxidative deterioration. 34 PV reflects the degree of fatty oil oxidation, which forms primary oxidation products. The PV changes of ZSSO and ZSSO-MPs are shown in Figure 5. Initially, the PV of ZSSO was 3.32 mmol/kg and that of microcapsules was 3.15 mmol/kg. After 18 d of storage, The PV of ZSSO rapidly increased, ultimately reaching 15.63 mmol/kg. Conversely, the ZSSO-MPs had a slower rate of oxidation and showed maximum PV of 6.20 mmol/kg. During storage, the slow oxidation rate of the ZSSO-MPs was due to the encapsulation of the wall material, which prevented its contact with air. The results indicated that microencapsulation could effectively reduce the oxidative deterioration of ZSSO, improve its storage stability and extend its shelf life.

Changes in peroxide value for free and encapsulated ZSSO during storage at 60 °C (n = 3).
Analysis of Sleep Induction Test Results
Whether the drug had the effect of improving sleep was evaluated with a pentobarbital sodium-induced sleep experiment. After 1 week of drug treatment in each group of mice, pentobarbital sodium was injected intraperitoneally into the mice, and the sleep latency and sleep time of the mice in each group were observed and recorded (Figure 6). Compared with the control group, the model group showed significant increase in sleep latency and significant reduction in sleep time (P < 0.01). The sleep latency of mice in each administration group was significantly shorter than that of the model group (P < 0.05, P < 0.01), and sleep time was significantly prolonged (P < 0.01, P < 0.001). The results indicated that ZSSO and microcapsules shortened the sleep latency and prolonged the sleep time of the insomnia model mice, thus improving sleep condition.
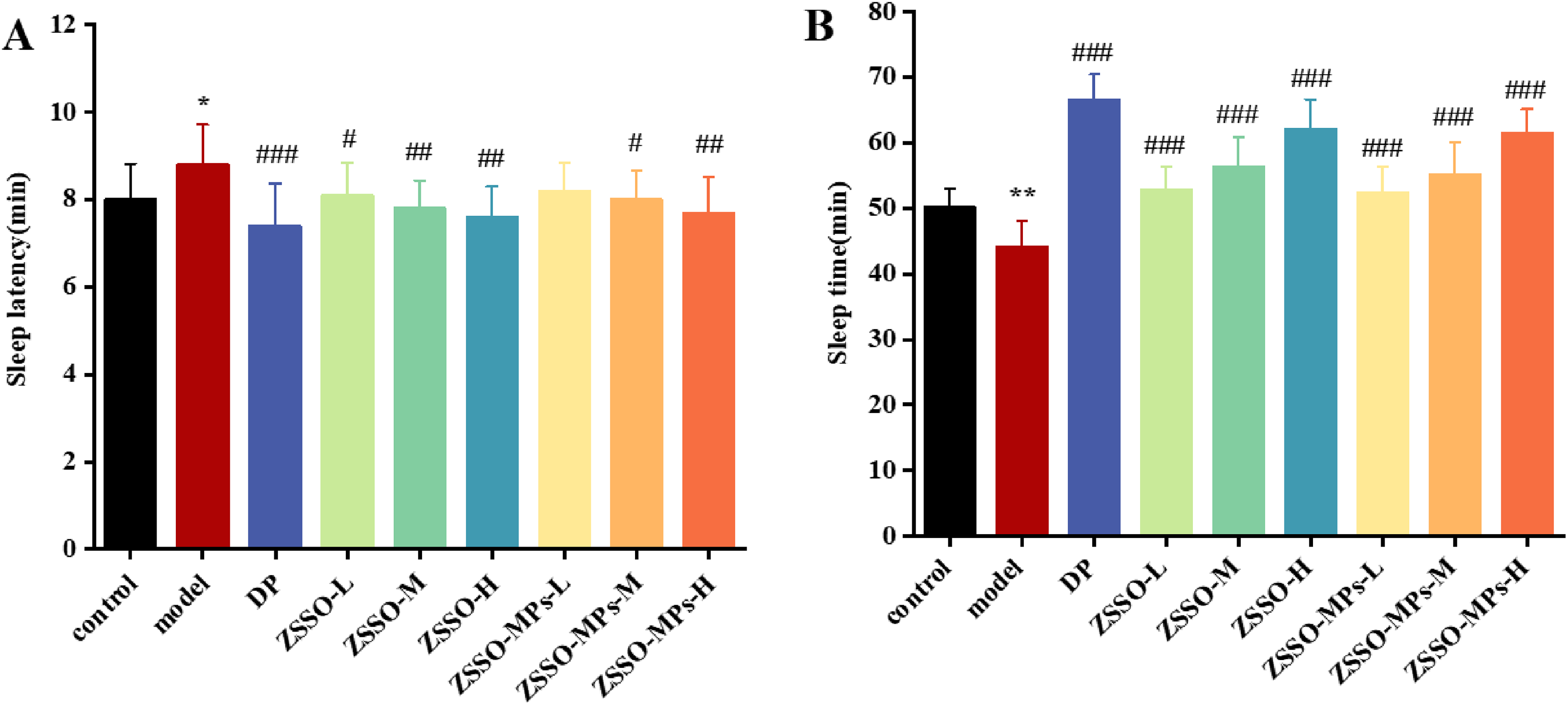
Analysis of sleep latency and sleep time in mice. Date are presented as the mean ± SD (n = 6). *P < 0.01, **P < 0.01 **P < 0.01 versus control group; #P < 0.05, ##P < 0.01, ###P < 0.001vs model group.
The Effects on 5-HT, GABA and Glu in the Brain of PCPA-Induced Insomnia Mice
As shown in Figure 7, Model group showed a significant decrease in 5-HT and GABA content (P < 0.001, P < 0.01), and a significant increase in Glu content and Glu/GABA (P < 0.001, P < 0.01), indicating that the replication of the PCPA insomnia model mice was successful. After 1 week of drug treatment in each group of mice, the levels of 5-HT and GABA were significantly increased and the levels of Glu were significantly decreased in each treatment group (P < 0.05, P < 0.01). Correspondingly, the ratio of Glu/GABA was significantly decreased. These results indicate that ZSSO and ZSSO MPs can improve sleep by regulating the levels of 5-HT, GABA, and Glu in mice brains.

Comparison of 5-HT, GABA, Glu content and Glu/GABA in brain tissue of mice in each group. Date are presented as the mean ± SD (n = 6). *P < 0.01, **P < 0.01 **P < 0.01 versus control group; #P < 0.05, ##P < 0.01, ###P < 0.001vs model group.
H&E Staining
As shown in Figure 8, the hypothalamic neurons in the control group were abundant in number, uniformly distributed, and regular in shape, with clear nuclei and without obvious necrosis or inflammatory cell infiltration. The model group had mild hemorrhage in the meninges, with many neuronal nuclei wrinkled and deeply stained, cell bodies shrunk and deformed, and unclear boundaries between nuclei and cytoplasm. The hypothalamic neurons in the administered groups were abundant in number and evenly distributed, with occasional individual neuronal nuclei wrinkled and deeply stained. This indicates that ZSSO can repair the neuronal cells of insomnia mice to improve their sleep condition.
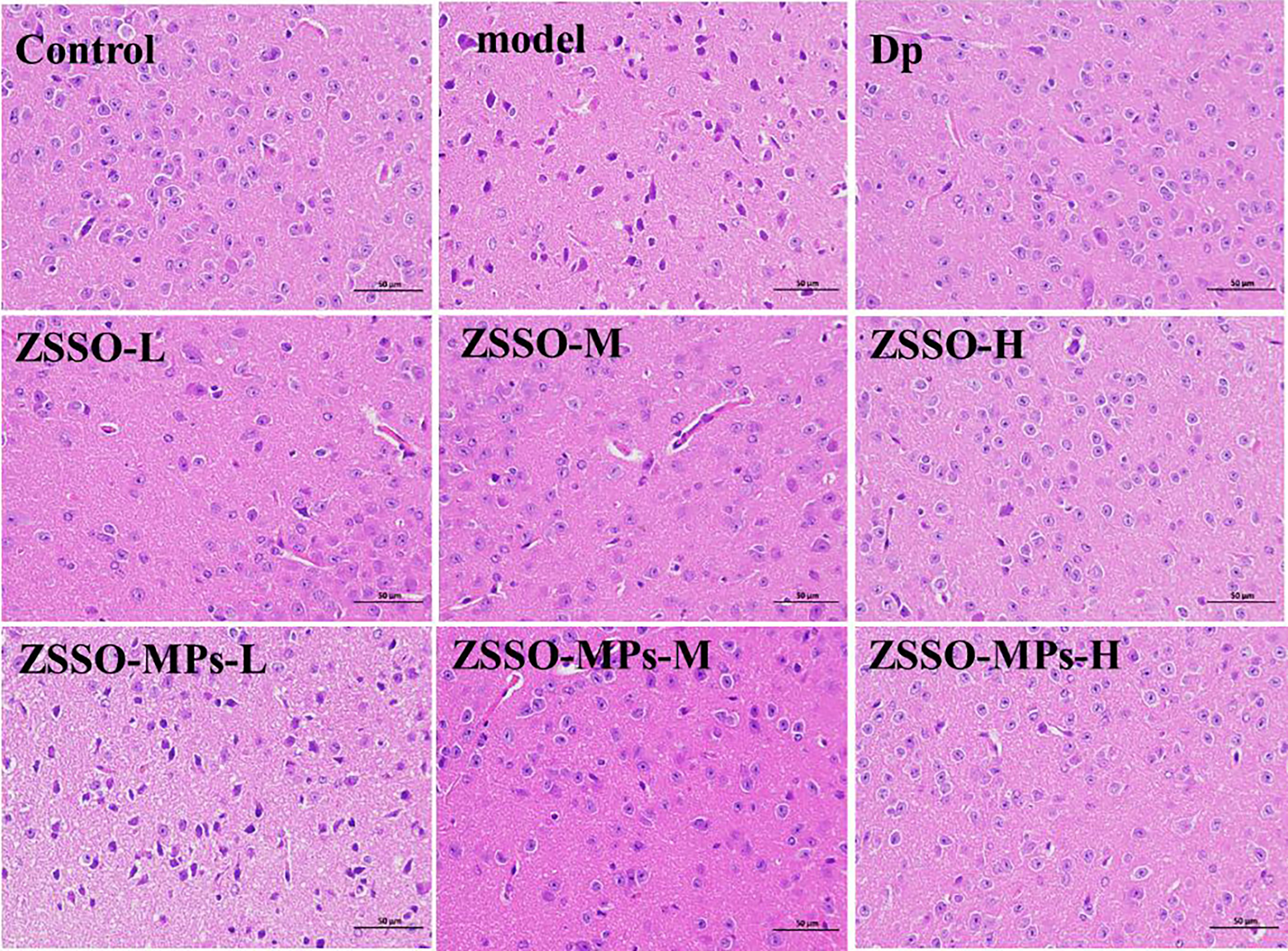
Histopathological observation of hypothalamic slices in mice of PCPA-induced insomnia. (H&E stain 400).
Conclusion
ZSSO is a kind of natural plant oil with high nutritional and healthy value. It has various pharmacological activities such as sedation, hypnosis, and lipid-lowering. ZSSO has broad development prospects due to its unique biological activity and low toxicity, but the lack of research on ZSSO product development limits its widespread application.
Microencapsulation provides a physical barrier to protect the core materials from external environmental influences such as light, oxygen, and temperature, helping to extend the shelf life and control the release of the core materials. 35 At the same time, it can also mask the unpleasant odor of some drugs, facilitating storage, transportation, and product processing. Microencapsulation has been proven to be a good method for stabilizing biologically active compounds. With the natural oil ZSSO as a core material, this study successfully prepared a small particle (166.17 nm) microcapsule, which showed excellent thermal and oxidative stability and had a certain effect on sleep improvement by increasing 5-HT and GABA content and decreasing Glu content in the brain of insomnia mice. The changing trend of neurotransmitters (GABA and Glu) in the mice brain was similar to the research results of Sun et al 3 Studies also have shown that ZSS can enhance pentobarbital induced sleep in mice and rats, which is characterized by a decrease in sleep latency and an increase in sleep duration. This effect is related to the content of neurotransmitters 5-HT 36 and GABA. 37 In this experiment, both ZSSO and ZSSO-MPs exhibited similar pharmacological effects, indicating that ZSSO may be closely related to its sleep improvement effect. 37 However, There is still significant space for optimization in the laboratory preparation process of ZSSO microcapsules to meet the requirements of industrial production. The mechanism of ZSSO microcapsules in improving sleep is complex and requires further in-depth research combined with more modern technological methods.
Sleep aids derived from natural plants have emerged as a popular choice for enhancing sleep quality due to their exceptional safety. In the future, basic research on increasing the extraction rate of ZSSO should be strengthened to enhance resource utilization. On the one hand, research on the active ingredients of ZSSO's sedative and hypnotic effects is a focus of future research. On the other hand, the mechanism of ZSSO's sedative and hypnotic effects is of great significance for the development of ZSSO drugs to improve suboptimal health conditions. In addition, the deep development of ZSSO as a health product to improve the sleep quality of insomnia sufferers can be one of the potential values of ZSSO.
Footnotes
Acknowledgments
The technical assistance and equipment support of Shaanxi University of Chinese Medicine, Co-Construction Collaborative Innovation Center for Chinese Medicine Resources Industrialization by Shaanxi and Education Ministry, Shaanxi, China, are appreciated for this study.
Author Contributions
Jiaxin Chen wrote the first draft of the paper; Zhishu Tang provided funding support for research; Xinbo Shi edited the paper; Zhongxing Song designed the research; Guolong Li performed the research; Hongbo Liu analyzed the data and wrote the final version of the paper.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by key project at central government level: Major Science and Technology Special Plan of Xianyang City (2018k01-41), China agriculture research system of MOF and MARA (CARS-21) and Key Research and Development Program of Shaanxi Province(2019SF-313).
Ethical Approval
This study was approved by the Administration Committee of Experimental Animals, Shaanxi Province, China. All the experimental procedures for the animals complied with the guidelines of the laboratory animal ethics committee at Shaanxi University of Traditional Chinese Medicine (NO. SUCMDL20231024004).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the “Ethics Committee of Shaanxi university of Chinese medicine approved the animal protocol (Approval number: 20231024004)” approved protocols. Statement of Informed Consent There are no human subjects in this article and informed consent is not applicable.