Abstract
In western Algeria, Thymelaea hirsuta aerial parts are traditionally used topically for the treatment of human skin infections. For a better knowledge of its biological activities, this study aimed at evaluating the in vitro antifungal activity and antiaging properties of several extracts of the aerial parts by in vitro assays. Antifungal activity was investigated by using the reference microbroth dilution method (National Committee for Clinical Laboratory Standards) against the dermatophytes (Microsporum audouinii, Nannizzia gypsea, Trichophyton interdigitale, and Trichophyton rubrum) and the yeast (Candida albicans). Antiaging (inhibition of 1,1-diphenyl-2-picrylhyorazyl radical scavenging and tyrosinase, lipoxygenase, elastase, and hyaluronidase activities) assays were performed in 96-well plates. All the extracts of T. hirsuta exhibited antifungal activity against M. audouinii, T. rubrum, and C. albicans, while no activity was found against N. gypsea and T. interdigitale. The highest activity was observed on M. audouinii for the aqueous leaf (minimum inhibitory concentration [MIC] of 8 µg/mL) and stem extracts (MIC = 31 µg/mL) and the methanolic leaf extract (MIC = 8 µg/mL). Additionally, some marked morphological alterations were observed on M. audouinii hyphae with all aqueous extracts. Concerning antiage bioassays, the major activity was found against hyaluronidase for the aqueous and methanolic extracts from leaves and the methanolic extract from flowers. Analysis of this last extract by high-performance liquid chromatography, electrospray ionization-mass spectrometry, and nuclear magnetic resonance, allowed us to identify daphnoretin and daphnetin as major components of this extract, and, therefore, responsible for the antiaging activity. In conclusion, this study provides additional experimental data supporting the traditional use of T. hirsuta extracts on some cutaneous dermatophytosis, as well as new perspectives on the potential use of T. hirsuta to ensure better healing of the skin.
Fungal infections of the skin and nails affect millions of people worldwide and require a costly and lengthy treatment. 1 These infections are mainly caused by some highly specialized filamentous fungi, called dermatophytes, exhibiting high affinity for keratinized tissues such as superficial layers of the skin, hair, and nails. 2 Antifungals used against dermatophytes exhibit several side effects (digestive or skin disorders) and have limited efficacy. Hence, there is a need for the development of safer and more effective antifungal drugs. 3 About 80% of the populations in developing countries are unable to afford pharmaceutical drugs and therefore resort to traditional medicines, mainly plant based, to sustain their primary health care needs. 4 Herbal medicines are in great demand in all countries for primary healthcare because of their wide biological and medicinal activities, higher safety margins, and lower cost. 5 Recent studies demonstrated that plants used in traditional medicine contain a wide range of bioactive compounds that can be used to treat infectious diseases. 6
Among them, Thymelaea hirsuta, commonly known as “Methnane” or “Al metnen” in Algeria and Tunisia, 7,8 is an evergreen shrub belonging to the Thymelaeaceae family, which is native to the Mediterranean region, north of central Europe and east of central Asia. Thymelaea hirsuta has been used in folk medicine for its antimelanogenesis, 9 antioxidant, 10 and hypoglycemic and antidiabetic 11 properties and against sterility. 8 Besides, anticancer properties were reported by Kawano et al 9 on B16 murine melanoma cells.
In western Algeria, T. hirsuta aerial parts are also claimed to treat human skin infections. Thymelea hirsuta is described as “a local plant used by our grandparents,” 12 without relying on experimental data. The dried flowering tops of the plant are traditionally ground and then boiled in water to obtain a viscous solution which is spread daily over the skin for treatment of cutaneous infections and noninfectious skin disorders such as eczema. 13 It is also reported as having leishmanicidal and vermifuge properties. 14
The aerial parts of T. hirsuta are also known as a potential antioxidant resource, mainly because of their huge phenolic and flavonoid content. 15 -17 These secondary metabolites, which are considered to be important phytochemicals, also exhibit antifungal properties. 17 Trigui et al 15 described the antifungal effect of ethanolic extracts of T. hirsuta leaves on food-borne pathogens, while Deramchia and Belhakem 12 reported an in vitro antifungal activity against some dermatophytes for T. hirsuta extracts and its essential oils. Several authors have isolated and identified daphnane diterpenoids (2 hirseins and five 12-hydroxy daphnanes) in some extracts of this plant. 9,18 Here, to provide data supporting the traditional use of this plant, we investigated the antifungal and antiskin enzyme activity of aqueous and methanolic extracts of T. hirsuta leaves, flowers, and stems. Antifungal activity was evaluated against 5 common human fungal pathogens, that is, the dermatophytes Microsporum audouinii, Nannizzia gypsea, Trichophyton interdigitale, and Trichophyton rubrum, and the yeast Candida albicans, which may also affect the epidermis. 2,19 Likewise, some in vitro cosmetic bioassays were performed on these extracts. In addition, one of the most active extracts was fractionated by semipreparative high-performance liquid chromatography-ultraviolet-visible spectroscopy (HPLC-UV-VIS), which allowed us to identify 2 major coumarins, daphnoretin and daphnetin belonging to the daphnanes family.
Results and Discussion
Antifungal Activity
First, the fungistatic and fungicidal activities of aqueous and methanolic extracts from the aerial parts (leaves, flowers, and stems) of T. hirsuta were investigated by the reference microbroth dilution method against 5 fungal strains. The minimum inhibitory concentrations (MICs) and minimum fungicidal concentrations (MFCs) of the extracts against the fungi tested are shown in Table 1. For all the extracts, no activity was found on T. interdigitale and N. gypsea. Conversely, these extracts were active against the dermatophytes M. audouinii and T. rubrum and the yeast C. albicans. The antifungal activity was variable between aqueous and methanolic extracts, as well as from 1 part of the plant to another. Aqueous extracts of flowers, leaves, and stems exhibited the highest antifungal effect with MICs values between 8 and 250 µg/mL and MFCs values between 250 and 1000 µg/mL. Surprisingly, the leaf extract appeared to be highly active on M. audouinii only, with a MIC value of 8 µg/mL. Likewise, all methanolic extracts were mainly active on M. audouinii, showing, however, higher MIC values compared with aqueous extracts (62.5-500 µg/mL depending on the part of the plant vs 8-250 µg/mL for the aqueous extracts).
Fungistatic and Fungicidal Activities of Aqueous and Methanolic Extracts From Flowers, Leaves, and Stems of Thymelea hirsuta, Expressed as MIC and MFC Values in µg/mL.

Abbreviations: MFC, minimum fungicidal concentration; MIC, minimum inhibitory concentration.
No activity was found against Trichophyton interdigitale and Nannizzia gypsea.
Comparatively, in the work carried out by Deramchia and Belhakem, 12 0% inhibitory concentration (IC90) values between 850 and 1000 µg/mL were obtained for T. rubrum and M. audouiini for the leaf and flower extracts of this species. Our extracts are overall more active on these strains, and especially the aqueous extracts, perhaps in relation to differences in the extraction process.
There is no normalization of the level of acceptance for the antifungal activity of plant extracts compared with licensed drugs. Some authors consider only activities directly comparable to those drugs, while others also consider higher values. 20 Antifungal drugs are generally active at lower concentrations, about 100‐1000 fold lower compared with the lowest MICs reported for plant extracts. 21 According to Ríos and Recio, 22 a MIC100 <100 µg/mL should be considered as a promising value for a crude extract (vs 10 g/mL for pure compounds). Likewise, based on MFC results for plants materials, Mathieu et al 23 proposed the following classification for plant materials: very high activities (MFC between 1.526 and 780 µg/mL), high activities (MFC between 780 and 6250 µg/mL), average activity (MFC between 6250 and 50000 µg/mL), and very low or low activity (MFC ≥50 000 µg/mL). Therefore, following this classification, the active extracts of T. hirsuta showed strong fungistatic activity against the fungi tested. The varying degree of sensitivity of the tested fungi may be due to the intrinsic tolerance of microorganisms and the nature of the different compounds present in the crude extracts. 24 Indeed, a phytochemical analysis of the aqueous extracts revealed higher amounts of each class of compounds in the flower extract, 16 which could explain the higher antifungal activity exhibited by this extract. Carotenoids were not detected in the stem extract and proteins in none of the extracts. Moreover, among the tested fungi, M. audouinii was more sensitive to T. hirsuta extracts.
Furthermore, phase-contrast microscopy examination of M. audouinii hyphae grown in the presence of a subinhibitory concentration of the aqueous extracts of leaves, flowers, and stems of T. hirsuta revealed an abnormal and irregular shape compared with nontreated hyphae as control (Figure 1), which suggests an impact of the aqueous extracts on the fungal cell wall. Interestingly, Trigui et al 15 reported that inhibition of the plasma membrane H+-ATPase could be responsible for the antimicrobial properties of T. hirsuta ethylacetate extract. This antiporter extrudes protons from the cell, generating an electrochemical proton gradient through the plasma membrane, which plays a key role in many essential physiological processes in the fungal cell, such as maintenance of intracellular pH homeostasis, uptake of cations and nutrients, and cell growth. 25

Morphology of Microsporum audouinii hyphae grown in the presence of a subinhibitory concentration of aqueous extract of Thymelaea hirsuta (B) showing coiled and distorted hyphae compared with the control grown in the absence of any extract (A) (×200).
Usually, it is difficult to assign the effects observed with a plant extract to a particular class of compounds, since these extracts are composed of a mixture of compounds that can act either synergistically or antagonistically. This morphological effect was not observed for the methanolic extracts, which suggests that this effect depends on compounds of higher polarity present either only or in greater amounts in the aqueous extracts. In addition, most plants produce various secondary metabolites which may be useful for their biology and also their protection against attacks from pathogenic insects or microorganisms. 23,26 The differences observed in the antifungal activity of T. hirsuta extracts from flowers, leaves, or stems could be explained by variations in the composition of the extracts and the accumulation of some components in some parts of the plant.
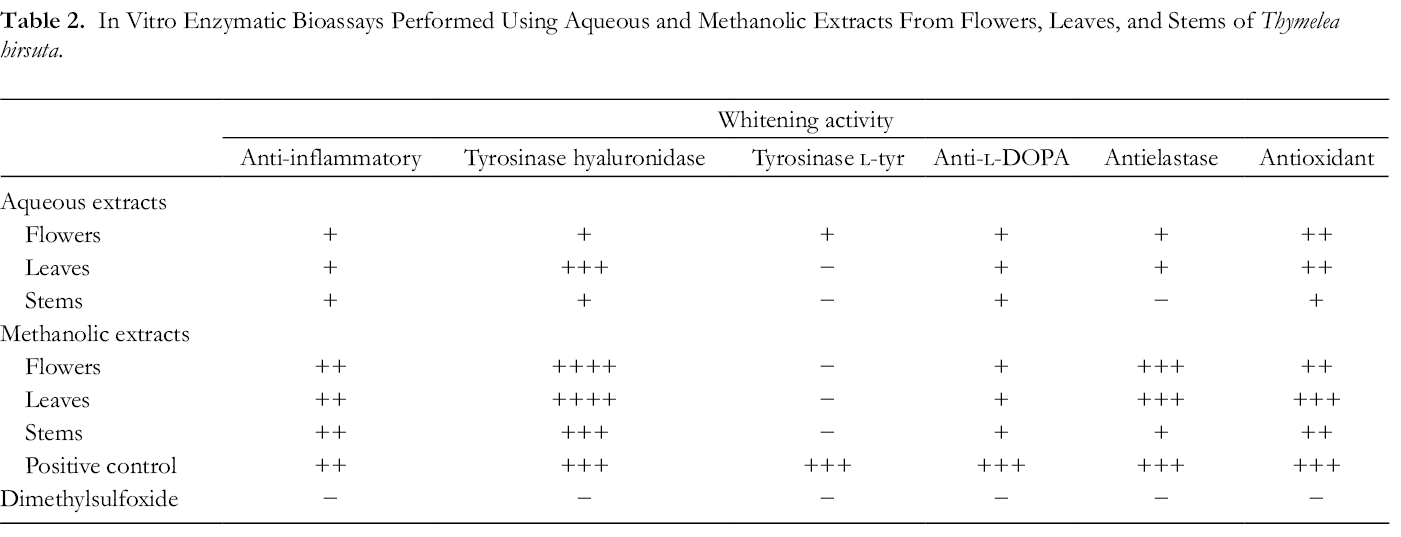
Antiskin Enzyme Activity
In parallel, the antiskin enzyme activity of the different extracts of T. hirsuta was evaluated in vitro (Table 2). As the plant was claimed to treat skin issues, we also investigated its capacity to restore and strengthen the skin barrier; these enzymatic bioassays assessed the ability of the plant extracts to improve skin elasticity and its density and to protect the skin against reactive oxygen species and therefore against inflammation. Thus, these assays may provide some insights into the traditional use of this plant for treatment of various skin disorders such as eczema, which is characterized by inflammation accompanied by redness, scales, and itching. For instance, methanolic extracts showed the highest activities in the antihyaluronidase, antielastase, and antioxidant assays and an interesting level of anti-inflammatory activity. The aqueous extract from leaves also showed some activity in the antihyaluronidase assay. Our results, therefore, not only confirm previous reports from Djeridane et al,
10
Trigui et al,
15
and Djermane et al
27
on the antioxidant properties of T. hirsuta and a possible link with phenolic compounds through hydrogen transfer reactions but also extend the spectrum of activity of this plant since in addition to antiacetylcholinesterase, antibutylcholinesterase, and anti-α-glucosidase activities,
27
our experiments also showed an inhibitory activity against some skin enzymes, especially
In Vitro Enzymatic Bioassays Performed Using Aqueous and Methanolic Extracts From Flowers, Leaves, and Stems of Thymelea hirsuta.
Microfractionation and Identification of Active Pure Compounds
Optimal profiling of each extract of T. hirsuta was developed based on analytical HPLC, and detection was ensured by UV and evaporative light scattering detectors (ELSD) to monitor the presence of UV-active metabolites and to obtain preliminary semiquantitative data on the main metabolites. All the extracts revealed the presence of a large group of polyphenols eluting between 10 and 45 minutes (data not shown). Thus, the antioxidant activity observed could be related to these polyphenols, as already reported in the literature. 27,28 In the flower methanolic extract only, 3 main peaks (Rf respectively of 19.5, 23.5, and 27.5 minutes) could be detected in the HPLC-UV-ELSD analysis and at 330 nm (Supplemental Figure S1). Thus, because of its high activity and the availability of a sufficient amount of this extract compared with the leaf extract, the bioguided fractionation was conducted using the flower extract. To obtain fractions containing sufficient amounts of metabolites from this extract, the analytical conditions were upscaled by geometric chromatographic transfer to semipreparative HPLC. 29,30 This allowed the injection of 25 mg of flower extract (FLM) and the recovery of 19 fractions designated as 1-19 (Figure 2).

Chromatogram of the methanolic extract of Thymelea hirsuta flowers obtained by semipreparative HPLC-UV-VIS and chemical structures of daphnoretin and daphnetin. Semipreparative HPLC-UV-VIS was performed on a Luna C18 column (5 µm, 150 mm × 4.6 mm) at 330 nm. HPLC-UV-VIS, high-performance liquid chromatography-ultraviolet-visible spectroscopy.
The antielastase and antihyaluronidase inhibitory activities were measured for 3 dilutions of the extract (FLM 1/2, 1/4, 1/8) and for each microfraction (Figure 3). The flower extract showed a dose-response effect in both assays. Fraction 19 (Tr = 44.58 minutes) appeared to be the most active fraction in both assays, and its activity was probably underestimated since it was tested at a 2-fold dilution (1.717 mg/mL) because of an insufficient quantity. Thus, this fraction was analyzed by negative HPLC-electrospray ionization mass spectrometry (ESI-MS), which showed a major peak at m/z 351.1 (Rt = 17.58) (Supplemental Figures S2; S3) with [2M]− = 702.5 (−), suggesting a molecular mass of 352.2 g/mol.

Antihyaluronidase and antielastase activities (expressed in percentage of inhibition of the biological activity) of the FLM of Thymelea hirsuta and some of its mcrofractions (designated F9-F19). Fraction 19 was tested after a 2-fold dilution. DMSO, dimethylsulfoxide; FLM, methanolic flower extract.
According to the literature and AntiBase database (https://application.wiley-vch.de/stmdata/antibase.php), the molecular formula of this compound was suggested to be C19H12O7, which corresponded to daphnoretin (7-hydroxy-6-methoxy-3-(2-oxochromen7yl-oxychromen-2-one). 1,31 1H-nuclear magnetic resonance (NMR) and 13C-NMR spectroscopic data (Supplemental Table S1) confirmed the identification. Even if the quantities were not sufficient to obtain a complete carbon NMR spectrum, the 2-dimensional correlations validated the chemical structure. Daphnoretin (Figure 2), a dicoumaryl ether, was isolated from the whole plant of Daphne mezereum and Wikstroemia indica C.A. Mey (Thymelaeaceae). 32,33 It has been used in Chinese folk medicine as an herbal remedy for the treatment of syphilis, arthritis, whooping cough, 34 and cancer. 35,36 A noticeable antihyaluronidase effect was also observed for fractions 12 (Tr = 12.66 minutes), 13 (Tr = 13.42 minutes), and 18 (Tr = 43.33 minutes). Fraction 13 was also analyzed because it was obtained in sufficient quantity. HPLC-ESI-MS (Supplemental Figure S4) associated with NMR spectroscopic data (Supplemental Table S2) permitted its identification as daphnetin (Figure 2) 37 : HPLC-ESI-MS data showed a peak at m/z = 178 at 8.41 minutes with a pseudomolecular ion at m/z 201.3 [M + Na]+, and 1H-NMR and 13C-NMR data analysis verified this hypothesis when compared with literature data. Daphnetin (7,8-dihydroxycoumarin) is found in the genus Daphne as well as from several other plant genera. 38 In China, daphnetin has been used clinically to treat Buerger’s disease for many years. 39 Its multiple pharmacological activities, including anti-inflammatory, antidiarrheal, and antiparasitic, have been reported by Fukuda et al 40 and Shen et al. 41 This compound is also known to exhibit antibacterial and antioxidant properties. 42,43 So far, neither toxic effects nor genetic toxicity has been found for daphnetin. 44,45 Coumarin and its derivatives are widely employed as a fragrance in cosmetics and skincare products. Skin toxicity has never been reported for coumarins 46,47 ; moreover, Pan et al 48 showed the safety of the topical application of coumarins. Since our experiments suggested a link between the presence of daphnane coumarins and the in vitro cutaneous activities observed, our results are consistent with literature data showing antioxidant and anti-inflammatory properties of daphnane diterpenes. 41,43
Experimental
Plant Material
The aerial parts (leaves, flowers, and stems) of T. hirsuta (L.) Endl. were collected from the north of Relizane state (Algeria), at about 82 m of altitude, 35°55′33″N latitude, and 0°49′23″E longitude during April 2015. The specimens were identified by Dr Sekkal Fatima Zohra, a botanist from the Plant Protection Laboratory at the University of Mostaganem (Algeria). A voucher specimen AB04-22 was deposited at the National Institute of Agronomy in Algiers (NIA, Algeria). Leaves, flowers, and stems of the plant were dried separately at room temperature (25-26 °C) away from sunlight, then ground to a fine powder using an electric blender, and stored in clean labeled airtight bottles.
Preparation of Plant Extracts
A 10% aqueous decoction was prepared separately from the different parts of the plant. 49 A total of 100 g of plant powder was mixed with 2 L of sterile distilled water, and, after shaking, the preparation was heated in a water bath at 90 °C for 1 hour. The cooled decoctions were vacuum filtered using glass microfiber filters Whatman No.4; filtrates were concentrated under reduced pressure at 40 °C using a rotary evaporator, and then lyophilized. Leaves and stem extracts were brown, and the flower extract was yellow (yields of 18%, 11%, and 8%, respectively).
Methanol extracts were also prepared by mixing separately 1 g of each explant (leaves, flowers, and stems) with 15 mL methanol, 50 under agitation for 15 minutes using an Ultraturax. The extracts were homogenized and rapidly vacuum-filtered through a sintered glass funnel. Methanol was evaporated under vacuum at 40 °C and the extracts were then lyophilized. Extracts from leaves, stems, and flowers were green, brown, and yellow, respectively (yields of 10%, 6%, and 3%, respectively).
Antifungal Activity
Antifungal activity of the different extracts was investigated on 4 dermatophyte species, that is, clinical isolates M. audouinii UA135345238-51, T. interdigitale UA130148517-02, and T. rubrum UA130142992-03 from the University Hospital of Angers (France) and the reference strain N. gypsea IHEM 7625 (from the Institute of Hygiene and Epidemiology-Mycology section culture collection in Sciensano, Brussels, Belgium), as well as on the main yeast species C. albicans ATCC 66396 (from the American Type Culture Collection in Manassas, VA USA). Fungi were cultivated for 2 days at 37 °C on yeast extract-peptone-dextrose agar (YPDA, containing in g/L: yeast extract, 5; peptone, 5; glucose, 20; agar, 20; and chloramphenicol, 0.5) plates for C. albicans and at 25 °C for 7 days on YPDA plates supplemented with 0.5 g/L cycloheximide (Sigma) for the dermatophytes. Tests were performed according to the approved reference methods for yeasts 51 and filamentous fungi. 52 Briefly, the yeast suspension was prepared in Roswell Park Memorial Institute (RPMI)-1640 culture medium and adjusted spectrophotometrically at 630 nm to reach a final concentration of ca. 0.5 × 103 to 2.5 × 103 cells per mL. For the dermatophytes, the mycelium was recovered by scraping the agar plates with 10 mL of sterile distilled water. Fungal suspensions were homogenized with a ground-glass grinder, filtered through sterile 20-µm-pore size membranes, and centrifuged; the obtained pellets were resuspended in RPMI-1640, and fungal suspensions were finally adjusted spectrophotometrically to a 0.4 absorbance at 450 nm.
Tests were performed using sterile 96-flat bottom wells microtiter plates. Serial 2-fold drug dilutions were realized in dimethylsulfoxide (DMSO): 5 µL of the obtained dilutions was distributed in triplicate into the wells to obtain final concentrations ranging from 1000 µg/mL to 8 µg/mL. After incubation for 48 hours at 37 °C for the yeast and 8 days at 25 °C for the dermatophytes, MICs were determined from the turbidimetric data as the lowest drug concentration giving rise to a fungal growth inhibition equal to or greater than 80% compared with the drug-free control (MIC80). Amphotericin B and voriconazole, 2 common antifungal agents, were used as the positive controls and DMSO as the negative control. The MFC was determined as the lowest concentration of the extracts without visible fungal growth after subculture on YPDA plates. Each strain was tested in triplicate experimental repeats.
Enzymatic Bioassays
The bioassays were performed as previously described. 28 Untreated 96-well plates were purchased from Thermo Nunc and the UV-transparent ones from Costar. During incubation, the 96-well plates were sealed with adhesive films (Greiner Bio-One). Samples for biological activity testing were prepared in 1.5 mL Eppendorf tubes, adapted to the use of the automated pipetting system epMotion 5075 (Eppendorf). Optical densities (OD) were measured using a microplate reader (Spectramax Plus 384, Molecular devices). Data were acquired with SoftMaxPro software, and inhibition percentages were calculated with Prism software. Results are presented as inhibition percentages (%), calculated as follows:

All OD were corrected with the blank measurement corresponding to the absorbance of the sample before the addition of the substrate. For all bioassays, DMSO alone was used as the negative control (ODcontrol).
DPPH Radical Scavenging Assay
The antioxidant activity of the extracts was determined based on the scavenging activity of the stable 1,1-diphenyl-2-picrylhyorazyl (DPPH) free radical according to Burger et al. 53 For this assay, 150 µL of a 0.1 M ethanol/acetate (50:50) buffer, pH 5.4, was distributed in each well, together with 7.5 µL of plant extracts. Trolox (3607.8 µM in DMSO) was used as a positive control. After measurement of the optical density at 517 nm (ODblank), 100 µL of a DPPH solution (386.25 µM in ethanol) was distributed in each well. After incubation of the sealed plate for 30 minutes in the dark at room temperature, the final OD was measured at 517 nm to determine the percentage of inhibition.
Tyrosinase Assay
Tyrosinase, a copper-containing oxidase controlling the production of the natural pigment melanin, is mainly involved in the initial rate-limiting reactions in melanogenesis: the hydroxylation of
Lipoxygenase Assay
Lipoxygenase, an iron-containing enzyme catalyzing the deoxygenation of polyunsaturated fatty acids into the corresponding hydroperoxides, is known to play a key role in inflammation. 55 For this assay, 150 µL of a 686.66 U/mL soybean lipoxygenase solution in phosphate buffer 50 mM pH 8 was distributed in each well (final concentration of the enzyme: 400 U/mL), together with 7.5 µL of plant extracts. Quercetin hydrate (1 mM in DMSO) was used as positive control. The plate was sealed and after 10 minutes of incubation, OD was measured at 235 nm. Then, 100 µL of a linoleic acid solution in phosphate buffer pH 8 was distributed in each well. After 50 minutes of incubation, OD was measured at 235 nm to assess the percentage of inhibition.
Elastase Assay
Elastase is a serine protease that preferentially digests elastin, a highly elastic protein forming a network with collagen to give its shape and firmness to the skin. 56 For this assay, 150 µL of a 0.171 U/mL porcine pancreatic elastase solution in Tris buffer 50 mM pH 8 was distributed in each well (final concentration of the enzyme per well: 0.1 U/mL), together with 7.5 µL of plant extracts. Quercetin hydrate (8583.33 µM in DMSO) was used as the positive control (final concentration of quercetin per well: 250 µM). The plate was filmed and read at 410 nm after incubation for 20 minutes at room temperature. Then, 100 µL of a 2.06 mM solution of N-succinyl-Ala-Ala-Ala-p-nitroanilide in Tris buffer (final concentration of the chromogenic peptide per well: 0.8 mM) was distributed in each well. After 40 minutes of incubation, OD was read at 410 nm to assess the percentage of inhibition.
Hyaluronidase Assay
Hyaluronidases are a family of enzymes that degrade hyaluronic acid, a high-molecular-weight glycosaminoglycan of the extracellular matrix. This macromolecule, which presents the unique capacity to bind and retain water molecules, is widely distributed in the body and notably at the periphery of collagen and elastin fibers: it, therefore, plays a major role in skin aging. 57 For this assay, 150 µL of a 13.3 U/mL hyaluronidase solution (pH 7) was distributed in each well, together with 7.5 µL of plant extracts. Tannic acid (435 µg/mL in DMSO) was used as the positive control. The plate was filmed and read at 405 nm after incubation for 20 minutes at 37 °C. Then, 100 µL of a 150 µg/mL hyaluronic acid solution (pH 5.35) was distributed in each well. After 30 minutes of incubation at 37 °C, 50 µL of a 40 mM cetyltrimethylammonium bromide solution in 2% sodium hydroxide was added to each well, and OD was measured at 405 nm to assess the percentage of inhibition (ODsample).
Results are presented as percentages of inhibition (%) calculated as follows:
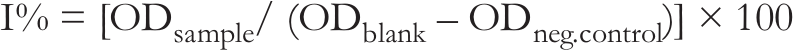
HPLC-UV-VIS-ELSD Analysis
Thymelaea hirsuta extracts were analyzed using an HPLC Agilent 1200 system (Courtaboeuf, France) equipped with a diode array detector set at 366 nm, and operating under the following conditions: injection volume 10 µL and flow rate 1.0 mL/min. Separation was performed on a C18 column (Phenomenex, Torrance, Ca; Luna 5 µm, 150 mm × 4.6 mm). The mobile phase consisted of a multistep gradient of water (HPLC grade, solvent A) and acetonitrile (ACN, HPLC grade, solvent B), both acidified with 0.1% formic acid: 5%-40% B (0-10 minutes); 40%-30% B (10-13 minutes); 30% B (13-30 minutes); 30%-95% B (30-40 minutes), and ELSD conditions were set as follows: nebulizer gas pressure 3.7 bars, evaporative tube temperature 40 °C and gain 4.
Fractionation by Semipreparative HPLC-UV-VIS
Aliquots of the flower extract (50 mg/mL) were microfractionated at room temperature (25 °C) by HPLC under the following conditions: injection volume of 25‐50 μL and flow rate of 4.0 mL/min. The preparative scale procedure and flow rate were obtained by direct geometric upscale from the analytical procedure. 29,30 Separations were performed on a C18 column (Phenomenex; Luna 5 µm, 250 mm × 10 mm). Microfractionation was achieved using a multistep gradient composed of 5%-40% B (0-10 minutes), 30% B (10-19 minutes), 30% B (19-24 minutes), and 30%-95% B (24-69 minutes). Microfractions were collected manually from 2 to 47 minutes with a fraction for every apparent peak. Dilutions of crude extract and fractions with sufficient quantities were separately evaluated for antihyaluronidase and antielastase activity.
Structural Analysis
HPLC-ESI-MS analysis
HPLC-ESI-MS was carried out in switch (positive/negative) ion mode using an Agilent 1100 Series liquid chromatography HPLC system (Agilent, Palo Alto, CA, USA) coupled with a Thermo Finnigan LCQ Advantage ion trap mass spectrometer (Thermo Fisher Scientific, San Jose, CA, USA) and equipped with an ESI source. The HPLC system included a binary pump, a thermostated autosampler, a column oven, and a diode array detector. A Gemini C18 column (150 mm × 3 mm, 3 µm; Phenomenex, Torrance, CA, USA) was used with an injection volume of 5 L and a flow rate of 0.3 mL/min. All samples were filtered through 0.2 µm sterile membranes before HPLC-MS analysis. The HPLC gradient used water (A) and ACN (B) (both acidified with 0.1% formic acid) as follows: 10%-100% B in 10 minutes and 100%-100% B in 4 minutes. The MS instrument parameters were: nebulization gas, N2, 50 a.u. sheath and 10 a.u. auxiliary gas flow; spray voltage, ±4.5 kV; capillary temperature, 275 °C; capillary voltage, −10 V; tube lens offset, −50 V.
NMR Analysis
NMR spectra of daphnetin and daphnoretin were acquired on a Bruker 500 MHz Avance III spectrometer (Bruker Biospin, Rheinstetten, Germany). Methanol-d4 (Euriso-Top, Saint-Aubin, France) was used as a deuterated solvent and its protonated residual signals were used as an internal standard at 3.31 ppm relative to trimethylsilane.
Conclusions
This study reveals that the aqueous extracts from T. hirsuta have a real impact on dermatophytes, especially M. audouinii, which is one of the main causative agents of Tinea capitis. Also, our results clearly showed the in vitro anti-inflammatory, antielastase, and antihyaluronidase activities of the methanolic extract from flowers, which could be related to a mix of daphnane coumarins. To our knowledge, these enzyme-inhibiting properties have never been described for these compounds. Thus, the present work provides experimental data supporting the traditional use of aerial parts of T. hirsuta by local populations in Africa for the treatment of some skin diseases and affords new perspectives for better healing of the skin. Further investigations should be conducted to confirm this plant as a solution to skin issues in general.
Supplemental Material
Supplementary Material 1 - Supplemental material for Antifungal and Antiaging Evaluation of Aerial Part Extracts of Thymelaea hirsuta (L.) Endl
Supplemental material, Supplementary Material 1, for Antifungal and Antiaging Evaluation of Aerial Part Extracts of Thymelaea hirsuta (L.) Endl by Nesrine Ouda Amari, Bienvenue Razafimandimby, Florence Auberon, Stephane Azoulay, Xavier Fernandez, Abdellah Berkani, Jean-Philippe Bouchara and Anne Landreau in Natural Product Communications
Supplemental Material
Figure S1 - Supplemental material for Antifungal and Antiaging Evaluation of Aerial Part Extracts of Thymelaea hirsuta (L.) Endl
Supplemental material, Figure S1, for Antifungal and Antiaging Evaluation of Aerial Part Extracts of Thymelaea hirsuta (L.) Endl by Nesrine Ouda Amari, Bienvenue Razafimandimby, Florence Auberon, Stephane Azoulay, Xavier Fernandez, Abdellah Berkani, Jean-Philippe Bouchara and Anne Landreau in Natural Product Communications
Supplemental Material
Figure S2 - Supplemental material for Antifungal and Antiaging Evaluation of Aerial Part Extracts of Thymelaea hirsuta (L.) Endl
Supplemental material, Figure S2, for Antifungal and Antiaging Evaluation of Aerial Part Extracts of Thymelaea hirsuta (L.) Endl by Nesrine Ouda Amari, Bienvenue Razafimandimby, Florence Auberon, Stephane Azoulay, Xavier Fernandez, Abdellah Berkani, Jean-Philippe Bouchara and Anne Landreau in Natural Product Communications
Supplemental Material
Figure S3 - Supplemental material for Antifungal and Antiaging Evaluation of Aerial Part Extracts of Thymelaea hirsuta (L.) Endl
Supplemental material, Figure S3, for Antifungal and Antiaging Evaluation of Aerial Part Extracts of Thymelaea hirsuta (L.) Endl by Nesrine Ouda Amari, Bienvenue Razafimandimby, Florence Auberon, Stephane Azoulay, Xavier Fernandez, Abdellah Berkani, Jean-Philippe Bouchara and Anne Landreau in Natural Product Communications
Supplemental Material
Figure S4 - Supplemental material for Antifungal and Antiaging Evaluation of Aerial Part Extracts of Thymelaea hirsuta (L.) Endl
Supplemental material, Figure S4, for Antifungal and Antiaging Evaluation of Aerial Part Extracts of Thymelaea hirsuta (L.) Endl by Nesrine Ouda Amari, Bienvenue Razafimandimby, Florence Auberon, Stephane Azoulay, Xavier Fernandez, Abdellah Berkani, Jean-Philippe Bouchara and Anne Landreau in Natural Product Communications
Footnotes
Acknowledgments
The authors gratefully acknowledge Dr Marc Pihet from ‘Groupe d’Etude des Interactions Hôte-Pathogène’ (GEIHP, EA 3142, University of Angers, University of Brest, Angers, France) for pictures of M. audouinii hyphae and Anaïs Chauvin for her experimental handling assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
