Abstract
Background
Hyperuricemia is a metabolic disorder caused by abnormalities in purine metabolism that increase serum uric acid. Some traditional medicine remedies have been proven to cause hypouricemia by inhibiting xanthine oxidase. In Vietnam, some remedies that eliminate wind, cold, and dampness have been used to treat gouty arthritis, but there is still a lack of scientific evidence regarding the underlying biological mechanisms involved. This study aimed to assess the xanthine oxidase inhibitory activity in vitro and the hypouricemic effects of several Vietnamese remedies used to treat Bi syndrome in traditional medicine.
Methods
The chosen remedies that met our criteria were successively extracted with 70% ethanol. A preliminary investigation of the chemical composition of the extracts was performed by common chemical reactions. The in vitro xanthine oxidase inhibitory activity of the extracts was investigated using the procedure of Noro et al (1983). Acute toxicology and hypouricemic effect of the remedy that showed the highest xanthine oxidase inhibitory activity were investigated in vivo using a potassium oxonate-induced hyperuricemic mouse model.
Results
Five remedies that met the criteria were chosen. Phytochemical screening of all the extracts revealed the presence of flavonoids, saponins, alkaloids, tannins, and carbohydrates. The Bai nghiem phuong 2 extract did not show xanthine oxidase inhibitory activity whereas the Thang Tri Te Thap extract, which exhibited the highest inhibitory xanthine oxidase activity with an IC50 value of 48.33 ± 0.41 µg/mL, was selected for the subsequent in vivo experiments. The Thang Tri Te Thap extract did not cause acute toxicity, with a Dmax of 31.67 g/kg. At doses of 1.5 g/kg and 3 g/kg, Thang Tri Te Thap significantly reduced the serum uric acid concentration by 59.84 ± 5.79% and 65.01 ± 12.73%, respectively (p < 0.05).
Conclusions
Thang Tri Te Thap showed xanthine oxidase inhibitory activity in vitro and hypouricemic effects in vivo.
Introduction
Hyperuricemia is a common metabolism disorder that affects 2.6–47.2% of people in different populations. The accumulation of uric acid leads to the formation and accumulation of monosodium urate crystals in the body. These crystals not only cause gout flares but also stimulate inflammatory reactions, which, in turn, contribute to the development of some common metabolic disorders, such as obesity, diabetes mellitus, and hypertension.1–3 Although hyperuricemia is asymptomatic in most cases, a serum urate concentration ≥ 9 mg/dL significantly increases the risk of developing gout flares and/or results in complications, such as chronic kidney disease and metabolic disorders. Therefore, current guidelines recommend conditionally starting urate-lowering therapy (ULT) when the serum urate concentration is ≥ 9 mg/dL.3–5
The first-line drug of ULT is allopurinol, which inhibits the enzyme xanthine oxidase (XO) – a key enzyme in the metabolism of uric acid, that reduces uric acid production. However, allopurinol has many adverse effects such as Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome, and Stevens-Johnson syndrome, and the risk for adverse effects increases in populations where the incidence rate of the HLA-B*5801 allele is high, such as in the Asia–Pacific region.3,5,6
In traditional medicine, many herbal drugs and remedies that are commonly used to treat Bi syndrome (drugs that eliminate wind, cold, and dampness) have been found to possess XO inhibitory effects in vitro and/or hypouricemic effects in vivo. 7 In Vietnam, there are many highly efficacious remedies that have been used to treat Bi syndrome. However, the use of these remedies still lacks scientific evidence. In our effort to clarify the correlation between traditional uses and modern pharmacological activities of the Vietnamese formulas, this study was conducted to assess the in vitro xanthine oxidase inhibitory effect and in vivo hypouricemic effects of several potential traditional Vietnamese remedies.
Results
The Chosen Books and Remedies
We only assessed documents written in Vietnamese. From our criteria, eight traditional Vietnamese books were chosen. As shown in Table 1, all the books were published in the late 20th and 21st centuries after the Vietnam War. Older documents cannot be assessed or were not published by a reputable publisher. Among the chosen books, “Hải Thượng Y tông tâm lĩnh” and “Tuệ Tĩnh toàn tập” are the most famous books about traditional Vietnamese medicine. However, the original versions of these books were written in ancient Vietnamese in the 14th and 18th centuries, respectively. The versions that circulate in Vietnam today were translated into modern Vietnamese so that we could choose these books for our research.
The Chosen Traditional Vietnamese Books.

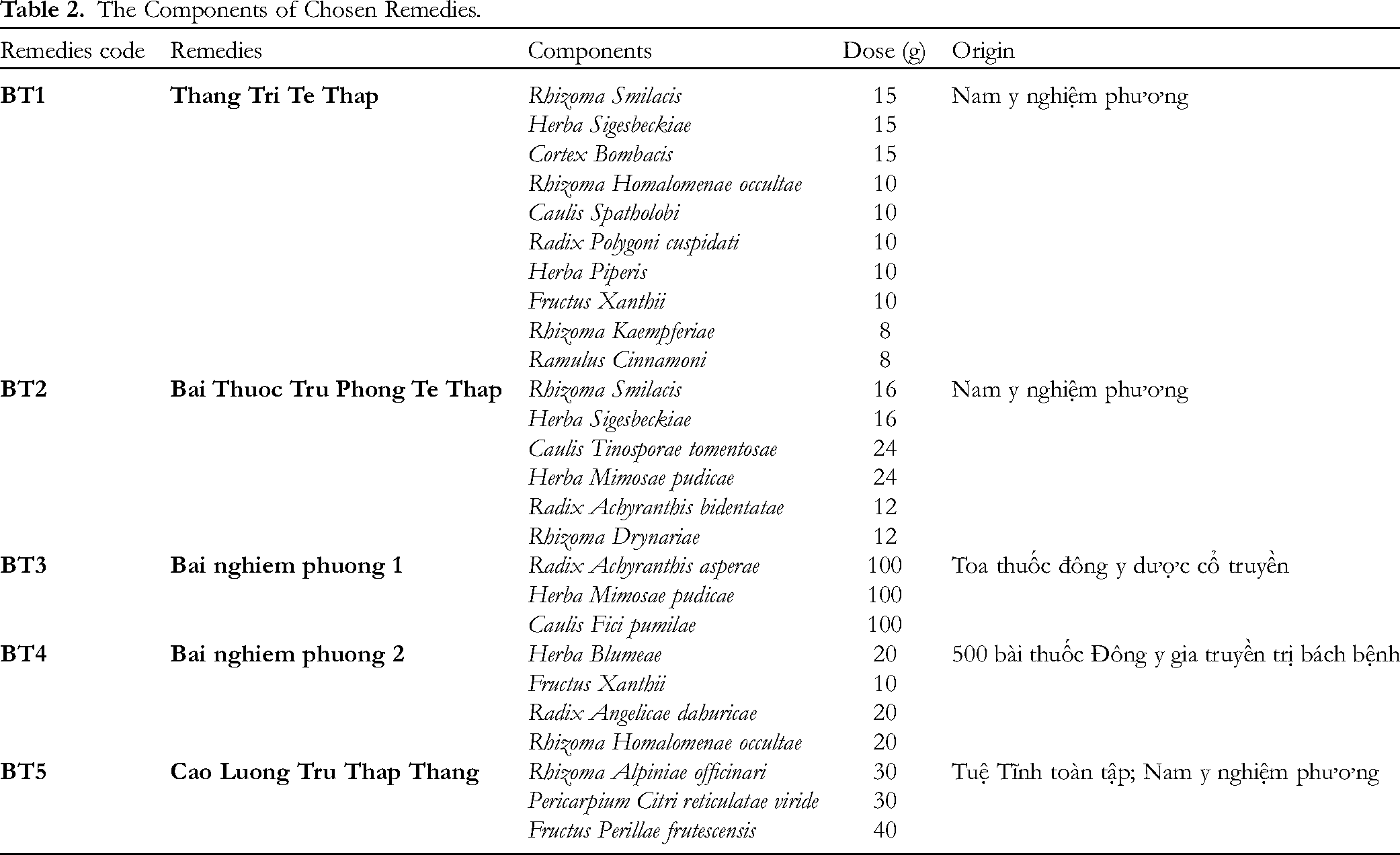
From the selected books, five remedies met the criteria were chosen. The ingredients, doses, and parts used for each drug in those five remedies are shown in Table 2. The book Những bài thuốc kinh nghiệm bí truyền của các ông lang bà mế miền núi did not give the exact dose of each drug in the remedies, so these remedies were eliminated. All the remedies recorded in the books of Việt Cúc did not show the exact part used of the drugs; therefore, they cannot be chosen for our study. The two books Hải thượng Y tông tâm lĩnh and Tuệ tĩnh toàn tập face both of the above problems, so all the remedies recorded in these books were excluded. Cao Luong Tru Thap Thang, which was first recorded in Tuệ tĩnh toàn tập without the dosage, was recorded in Nam y nghiệm phương with the exact dosage as shown in Table 2.
The Components of Chosen Remedies.
Preliminary Screening of the Chemical Composition of the Extracts
Phytochemical screening of all the extracts is shown in Table 3. In general, all the extracts contained flavonoids, saponins, alkaloids, tannins, and carbohydrates. Additionally, extract BT1 (Thang Tri Te Thap) contains anthranoids, as indicated by the presence of Radix Polygoni cuspidati in this extract. 8 Moreover, the BT4 extract (Bai nghiem phuong 2) contains coumarins because the extract contains Radix Angelicae dahuricae. 9
Preliminary Investigation of the Chemical Composition of the Extracts.

In Vitro Xanthine Oxidase Inhibitory Activity
Using the procedure of Noro et al with some modifications, the XO inhibition activity of the extracts was tested at multiple concentrations (20, 40, 60, 80, and 100 µg/mL). Meanwhile, the positive control, allopurinol, was tested at concentrations of 13, 25, 38, 50, and 75 µM. The inhibition rates of the extracts are shown in Table 4. In general, all the extracts (except for the BT4 extract) inhibited XO in a dose-dependent manner. At a concentration of 100 µg/mL, the BT4 extract inhibited the enzyme activity by 8.73 ± 4.69%. Therefore, the extract was not tested at lower concentrations to determine the IC50 value.
Inhibition Rates and IC50 Values of the Extracts and Allopurinol.

From the XO inhibition rate at multiple concentrations (as shown in Table 4 and Figure 1), standard curve equations for samples BT1, BT2, BT3, BT5, and allopurinol were established, and the IC50 values of those samples were calculated. As shown in Table 4, BT1 Thang Tri Te Thap was the most potent at inhibiting XO, with the lowest IC50 value. Therefore, the Thang Tri Te Thap extract was used for subsequent experiments.

Inhibition rates of extracts and allopurinol.
Acute Toxicology of “Thang tri te thap”
The maximum dose that can be injected through a blunt needle for oral administration was 31.76 g/kg. After 24 h of administration at this dose, no abnormal symptoms of behavior or movement appeared in the mice. After 72 h of treatment, no mice died, and the mice did not show any unusual symptoms. During the next 14 days, no abnormalities in the behavior or movement of the mice were noted, and no mouse deaths were recorded.
Therefore, the LD50 of the Thang Tri Te Thap extract could not be determined, and the Dmax of the extract was 31.76 g/kg. From the Dmax value, the two doses used in the TN1 and TN2 groups in the next experiment were calculated as 1.5 g/kg and 3 g/kg, respectively.
Hypouricemic Effect of the Thang Tri Te Thap Extract
The hypouricemic effect of the Thang Tri Te Thap extract is shown in Table 5 and Figure 2.
Hypouricemic Effect of the Thang Tri Te Thap Extract.

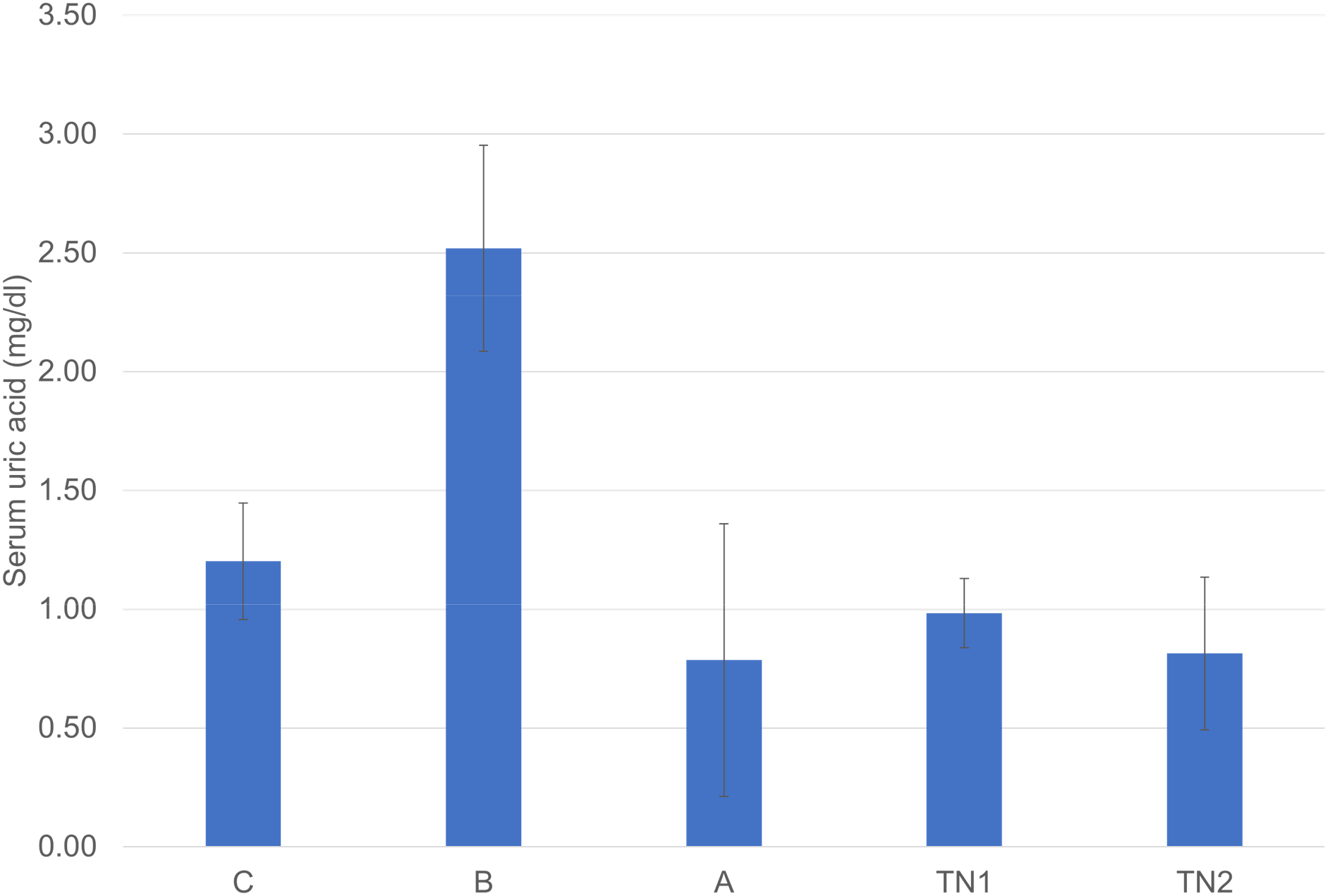
Serum uric acid levels of the mice in each group.
The results from Table 5 show the following:
The serum uric acid concentration of the mice in the control group (intraperitoneally injected with 300 mg/kg potassium oxonate) increased by 109.51 ± 36.08%, which was significantly different from that of the mice in the normal group (t test, t = 1.761, p < 0.001). In the allopurinol-treated group, the concentration of uric acid decreased by 66.70 ± 22.80%, which was significantly different from that in the control group (t test, t = 1.761, p < 0.001). The results showed that the potassium oxonate-induced hyperuricemic mouse model was successfully established. This model could be used to evaluate the hypouricemic effect of the Thang Tri Te Thap extract.
Mice in the two groups treated with the Thang Tri Te Thap extract extract exhibited a significant decrease in the serum uric acid concentration (p < 0.05).
Thang Tri Te Thap extract at a dose of 1.5 g/kg reduced the serum uric acid concentration by 59.84 ± 5.79%, which was significantly greater than that of the control group (t test, t = 1.761, p < 0.001), and the hypouricemic activity of the extract was similar to that of the allopurinol-treated group (t test, t = 1.895, p = 0.152).
Thang Tri Te Thap extract at a dose of 3 g/kg significantly reduced the serum uric acid concentration by 65.01 ± 12.73%, compared to that of the control group (t test, t = 1.761, p < 0.001), and the hypouricemic activity of the extract was similar to that of the allopurinol-treated group (t test, t = 1.895, p = 0.417).
There was a significant difference in the incidence of hypouricemia between the two groups using Thang Tri Te Thap at doses of 1.5 g/kg and 3 g/kg (t test, t = 1.895, p = 0.035). It was suggested that Thang Tri Te Thap reduced serum the uric acid concentration in a dose-dependent manner.
Discussion
According to traditional medicine theories, Bi syndrome is caused by the evasion of exogenous factors including wind, cold, and dampness. The basic foundation treatment for Bi syndrome is to expel wind, cold, and dampness. Among the chosen remedies, the four former remedies contain herbal drugs that expel wind, cold, and dampness as the main components. The Thang Tri Te Thap and Bai Thuoc Tru Phong Te Thap contain drugs that promote qi and blood, nourish the liver and kidney to enhance the effect of nourishing the bone marrow, and relieve pain. Although Cao Luong Tru Thap Thang is commonly used in treating Bi syndrome, this remedy contains drugs that circulate qi from the spleen and nourish the spleen to remove dampness. 10
In vitro tests revealed that extracts from Thang Tri Te Thap, Bai Thuoc Tru Phong Te Thap, Bai nghiem phuong 1, and Cao Luong Tru Thap Thang inhibited the enzyme XO at multiple concentrations in a dose-dependent manner. Many ingredients in these remedies such as Herba Piperis, Herba Mimosae pudicae, Radix Achyranthis asperae, and Fructus Perillae frutescensis inhibited XO in vitro. 7 Additionally, the linear relationships between the extract concentration and inhibition rate of these four extracts had high R2 values (over 0.9), suggesting a strong correlation between the extract concentration and the XO inhibition rate. Interestingly, Cao Luong Tru Thap Thang – a remedy used to treat endogenous dampness syndrome – showed XO inhibition activity similar to that of allopurinol, and had a lower IC50 than the other exogenous-dampness treatment extracts (48.73 ± 8.99 µg/mL). This result once again confirmed the correlation between uric acid in modern medicine and dampness syndrome in traditional medicine.
In previous studies, methanol extracts from Herba Blumeae and Rhizoma Homalomenae occultae inhibited XO in vitro, with IC50 values of 6.0 µg/mL and 58.1 µg/mL, respectively.7,11 Although Bai nghiem phuong 2 contains these two drugs, it does not inhibit the enzyme XO. One hypothesis is that the interaction between the bioactive components in the four drugs when combined, created a suppressive effect that led to a reduction in the ability to inhibit the enzyme of the remedy. Additionally, Herba Blumeae possesses a large amount of chlorophyll, so we cannot rule out the possibility that when extracted with 70% ethanol, the chlorophyll is dissolved with other active ingredients, affecting the biological activity of the extract.
To date, there has been no research on the acute toxicology of Thang Tri Te Thap. The experimental results showed that at a dose of 31.76 g/kg, which is the highest dose that can be injected through a blunt needle for oral administration, no deaths were recorded. This dose is 20 times greater than the effective dose in mice calculated from the human dose. The results suggested that the use of Thang Tri Te Thap extract in the treatment of hyperuricemia does not cause acute toxicity. However, three of the ingredients in the remedy, which are Herba Sigesbeckiae, Caulis Spatholobi, and Fructus Xanthii were reported to be slightly toxic.12–14 One hypothesis is that the combination of these ingredients with others in Thang Tri Te Thap could reduce the toxicity of the above three ingredients, which is an advantage of using a remedy instead of using one herb. Therefore, further research is needed to determine the safety of this remedy when it is used on humans.
At both 1.5 g/kg and 3 g/kg, the Thang Tri Te Thap extract had a hypouricemic effect similar to that of allopurinol at a dose of 10 mg/dl, and the difference was not statistically significant (t test, p > 0.05). The results suggested that Thang Tri Te Thap could have great potential for replacing allopurinol in the treatment of hyperuricemia. However, further research is needed because this study did not determine the long-term hypouricemic effect of the extract.
According to recent hyperuricemia treatment guidelines, the management of hyperuricemia patients includes lifestyle modification and drug therapy. Allopurinol, a first-line drug, has many side effects, and the risk of these side effects increases in the Vietnamese population, where the frequency of the HLA-B5801 allele is high. 5 Therefore, there is a need for research investigating alternative drugs with fewer side effects.
The results of this study showed that the Thang Tri Te Thap extract is safe and effective for treating hyperuricemia in mice. The herbs in Thang Tri Te Thap are all available in Vietnam and have abundant reserves. These medicinal herbs all grow and develop well in our country's tropical climate and have been used for a long time in traditional Vietnamese medicine. In addition to their ability to lower blood uric acid, the drugs in this remedy also treat several metabolic disorders that often accompany hyperuricemia, such as dyslipidemia and hyperglycemia, and have pharmacological effects related to gouty arthritis and urate kidney stones, such as anti-inflammatory, analgesic, diuretic, and anti-kidney fibrosis effects. 15 Therefore, this is a remedy with great potential for researching drugs and preparations to treat hyperuricemia and gouty arthritis clinically.
This study has several limitations. First, the traditional Vietnamese medicine books in Vietnam are very dispersed, and some of them were devastated during the Vietnam War, so we can assess only some documents that recently circulated in Vietnam. In addition, flavonoids possess XO inhibitory activity. In our study, we used 70% ethanol as the solvent for the remedies because flavonoids are well dissolved in 70% ethanol. Moreover, the most commonly used form of these remedies is water decoction. This study neither investigated the XO inhibitory effect and the hypouricemic effect of the water extract nor compared the XO inhibitory effect of the water extract to that of the ethanol extract. Finally, ULTs require long-term use, but we only evaluated the effects of these remedies on an acute hyperuricemia mouse model. We propose to test the effect of the Thang tri te thap on hypouricemia in a prolonged hyperuricemia mouse model.
Conclusion
The 70% ethanol extract from the Bai nghiem phuong 2 did not show xanthine oxidase inhibition activity in vitro. The 70% ethanol extracts from the Thang Tri Te Thap, Bai Thuoc Tru Phong Te Thap, Bai nghiem phuong 1, and Cao Luong Tru Thap Thang had XO inhibitory effects, and the IC50 values of the Thang Tri Te Thap extract were the lowest among those of the other extracts.
The 70% ethanol extract from the Thang Tri Te Thap at a dose of 31.76 g/kg caused no death and did not cause abnormal manifestations in mice within 14 days after oral administration.
Treatment with 70% ethanol extract of Thang Tri Te Thap at doses of 1.5 g/kg and 3 g/kg caused hypouricemia in potassium oxonate-induced hyperuricemic mice. The hypouricemic effect of the extract was the same as allopurinol at a dose of 10 mg/kg.
Materials and Methods
Materials and Chemicals
All herbal medicines were purchased from the Department of Pharmacy – University Medical Center Branch No. 3, Ho Chi Minh City, Vietnam. The materials met in-house specifications or Vietnamese Pharmacopoeia V. The materials in each remedy were pulverized into a fine powder.
Xanthine, xanthine oxidase, and allopurinol were obtained from Aldrich-Sigma, USA. Sodium phosphate buffer, acid hydrochloric, and sodium carboxymethyl cellulose (CMC) were obtained from Xilong, China.
Animals
Male mice (Swiss albino, 6 to 8 weeks old, weighting 20-22 g) were provided by the Pasteur Institute in Ho Chi Minh City. Mice were raised in an air-conditioned room at 22 °C to 24 °C under a constant day-night cycle for more than 1 week with food and water provided freely. All experiments were performed under the Guidelines for the Care and Use of Laboratory Animals approved by the Unit of Traditional Medicine and Pharmacy – University of Medicine and Pharmacy at Ho Chi Minh City.
Criteria for Choosing Books and Remedies
The remedies chosen in the books met the following conditions: (1) they were remedies used to treat Bi syndrome, and (2) they were published by a reputable publisher in Vietnam.
All remedies that meet these conditions will be chosen: (1) used to treat Bi syndrome with the symptom of pain in the toes and knees joints; (2) used orally; (3) given a detailed dose; (4) in which part of each medicinal plant used in the remedies is described clearly; and (5) possess at least one ingredient that has been proven to have XO inhibition activity. The selected remedies will be eliminated if they meet at least one of the following criteria: (1) possess XO inhibition activity and/or hypouricemia effects; (2) contain at least one drug that originated from animals; or (3) are toxic to humans.
Extraction of Selected Remedies
The remedy powder was extracted using percolation extraction method. 16 The remedy powder was soaked in the solvent (70% ethanol) for 2 to 4 h to obtain an adequate moisture mixture. The soaked powder was transferred to a percolator, and solvent was added until the medicinal powder was completely submerged. The powder was soaked for 24 h, after which the extract was removed at a rate of 5 drops/minute. The remaining solvent was added when the amount of solvent in the percolator was almost empty so that the total amount of solvent used/medicinal powder was (10:1). The extract was condensed by a rotary evaporator (Buchi Rotavapor R-300 – Switzerland). The extract was then steamed until it became a concentrated extract (the moisture content of the extract was no more than 20%).
Primary Phytochemical Screening of the Crude Extracts
A phytochemical screening test was qualitatively performed for flavonoids, saponins, anthranoids, coumarins, alkaloids, tannins, and carbohydrates by using specific reactions. 16
Xanthine Oxidase Inhibitory Assay
The xanthine oxidase inhibitory activity of the extracts was determined using the procedure of Noro et al 17 Briefly, in a 96-well plate, the initial mixture comprised 100 µL of the test solution, 300 µL of 50 mM phosphate buffer (pH = 7.5), 100 µL of XO solution (0.2 U/mL) mixed in the buffer, and 100 µL of distilled water. The mixture was incubated at 37 oC for 15 min, 200 µL of 0.15 mM xanthine dissolved in buffer was added, and the mixture was incubated for 30 min. The reaction was stopped by adding 200 µL of 0.5 M HCl. The absorbance of the mixture was then measured using a spectrometer at 295 nm. A similar procedure was used for the control sample, but the test solution was replaced with a buffer solution. Allopurinol was used as the positive control. The experiment was performed in triplicate. The test solution was screened at concentrations of 20, 40, 60, 80, and 100 µg/mL. Moreover, allopurinol was screened at concentrations of 13, 25, 38, 50, and 75 µM.
The XO inhibition rate (%I) was calculated as follows: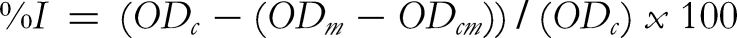
From the calculated results, a standard curve equation relating the inhibition rate and sample concentration was established, and the IC50 values of the samples were calculated. The IC50 values of the extract(s) that had inhibition rates lower than 50% at the concentration of 100 µg/mL were not determined. The extract showing the most potent inhibitory enzyme XO (with the lowest IC50 value) was used for the subsequent experiments.
Acute Toxicology Experiment
The extract was dissolved in distilled water. Healthy male mice (Swiss albino, 6 to 8 weeks old, weighting 20-22 g) were kept stable for one week before the test. Twelve hours before the treatment, food was withdrawn from the case. Mice were orally administered 0.2 mL/10 g of weight only once at 8:00–9:00 AM. The experiment was performed in two phases as follows: 18
In the exploration phase (each group contained 6 mice), the highest dose that can be injected through a blunt needle for oral administration was started to determine the LD0 (maximum dose that does not cause death) and LD100 (minimum dose that causes 100% death) of the mice.
In the determination phase, the mice were divided into multiple groups, and the drugs were administered at doses between the LD0 and LD100, which were divided exponentially.
The occurrence of any behavioral or motor abnormalities in the mice within the first 72 h after drug administration and the time from the initiation of treatment until the appearance of unusual symptoms were recorded. The number of dead mice in each group was recorded. The Dmax or LD50 was determined according to the Karber-Behrens method (if any) as a basis for calculating the dose for pharmacological testing.
The two orally administered doses to mice in the pharmacological test will be determined to be 1/10 and 1/20 LD50 or 1/10 and 1/20 Dmax, respectively (in cases where the LD50 could not be determined).
Potassium Oxonate-Induced Hyperuricemia Mice Experiment
The mice were divided randomly into 5 groups, and each group contained 8 mice. Mice were orally administered 0.1 mL/10 g of weight. Each group was treated with distilled water or drugs as follows:
- Group 1 (normal group) (group C): distilled water. - Group 2 (control group) (group B): distilled water. - Group 3 (group A): allopurinol at a dose of 10 mg/kg. - Group 4 (group TN1): remedy extract at a dose of 1/20 LD50 (if any) or 1/20 Dmax. - Group 5 (group TN2): extract at a dose of 1/10 LD50 (if any) or 1/10 Dmax.
All drugs were dissolved in distilled water before being orally administered. A hyperuricemic mouse model was generated using the procedure of Huo et al
19
with some modifications. Briefly, the mice were orally administered distilled water or drugs once daily at 8:00–9:00 AM for 7 consecutive days. One and a half hours before the treatment, food was withdrawn from the case, and water was provided ad libitum. On the seventh day, all the mice (except for the mice in group 1) were intraperitoneally injected with potassium oxonate (dissolved in CMC 5%) at a dose of 300 mg/kg one hour before treatment with the final dose of distilled water or drugs. Blood samples were collected from the tail veins of the mice 1 h after the final dose of distilled water or drugs. Blood was stored in EDTA tubes, clothed for approximately 1 h at room temperature, and then centrifuged at 3000x g for 10 min to collect the serum. The serum was kept at −20 oC until use. Serum uric acid was measured using the biochemistry analyzer Screen Master 3000 (Italia).
Statistical Analysis
All the data are presented as the means ± SDs. Statistical analysis was performed using MS.EXCEL 2016. The statistical significance of differences between two groups was tested by Student's t test. A pvalue < 0.05 was considered to indicate statistical significance at the 95% confidence level. All charts were drawn using MS.EXCEL 2016.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241269432 - Supplemental material for Xanthine Oxidase Inhibitory Activity of Some Vietnamese Remedies Used to Treat Bi Syndrome in Traditional Medicine
Supplemental material, sj-docx-1-npx-10.1177_1934578X241269432 for Xanthine Oxidase Inhibitory Activity of Some Vietnamese Remedies Used to Treat Bi Syndrome in Traditional Medicine by Duc Phan Nguyen Duong, Chen Van Tran, My-Duyen Thi Chung, Bay Thi Nguyen and Triet Thanh Nguyen in Natural Product Communications
Footnotes
Acknowledgment
This study was funded by the University of Medicine and Pharmacy at Ho Chi Minh City, Vietnam under grant number 60/2023/HĐ-ĐHYD on March 20, 2023. The authors would like to thank Associate Professor Bancha Yingngam from Ubon Ratchathani University (Thailand) for correcting the English errors in the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University of Medicine and Pharmacy at Ho Chi Minh City, Vietnam [grant number 60/2023/HĐ-ĐHYD on March 20, 2023].
Ethical Approval
This article does not contain any studies involving human subjects. The animal experiments in this study were approved by the Animal Experimental Ethics Committee of the University of Medicine and Pharmacy at Ho Chi Minh City (Approval no. 424/GCN-HĐĐĐNCTĐV) on March 30th, 2023.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the protocols approved by the Animal Experimental Ethics Committee of the University of Medicine and Pharmacy at Ho Chi Minh City (Approval no. 424/GCN-HĐĐĐNCTĐV).
Statement of Informed Consent
There were no human subjects included in this study, and informed consent was not available.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
