Abstract
Objective
The aim of this study was to assess the ability of polyphenolic extracts from different kiwifruit genotypes to affect tyrosinase enzyme activity and determine potential relationships between tyrosinase inhibition and polyphenolic composition. To date, research on the tyrosinase inhibitory bioactivity of kiwifruit have focused on a single A. chinensis cultivar. Given the numerous genotypes of kiwifruit commercially grown have varying compositions, and a variety of polyphenolic compounds present within each of these cultivars, the full potential of the tyrosinase inhibitory of kiwifruit has not been fully evaluated.
Methods
The polyphenolic composition of ethanol extracts from the cortical flesh of 10 kiwifruit genotypes was characterized using high-resolution accurate mass liquid chromatography mass spectrometry. The tyrosinase activity of each extract was measured spectrophotometrically in vitro. Multivariate analysis was subsequently conducted to identify kiwifruit compounds that may correlate with tyrosinase enzyme inhibition.
Results
The kiwifruit extracts displayed a broad range of tyrosinase inhibitory activity with some extracts having minimal or no inhibitory effect while the most potent extract showed 33.54 ± 1.59% inhibition (P < 0.01) at a dose of 125 µg/mL. Multivariate analysis of polyphenolic and tyrosinase inhibition data identified that polymeric procyanidins likely explain the tyrosinase inhibitory ability and the differences between genotypes. This was confirmed by the high relative concentrations of these procyanidins in extracts that exhibited tyrosinase inhibitory properties, while being absent or at low concentrations in extracts that did not.
Conclusion
Our findings demonstrate that some polyphenol kiwifruit extracts inhibit tyrosinase activity. The variability in the tyrosinase inhibitory activity of polyphenol extracts from 10 kiwifruit genotypes is attributed to the differences in the relative concentrations of polymeric procyanidins in these extracts. These results highlight that the tyrosinase inhibitory properties of kiwifruit genotypes could be examined further to support the development of kiwifruit-based cosmetic products.
Introduction
Maintaining healthy skin is a major goal of the cosmetic and nutraceutical industry, with an emphasis on mitigation of blemished and discolored regions to promote a youthful appearance. Pigmentation from melanin is central to the uniform color of perceived healthy, undamaged skin. Melanin overexpression and hyperpigmentation causes freckles and senile lentigines, which are regarded by some as esthetic issues to be avoided. Skin can be damaged through injuries and by the damaging effects of ultraviolet (UV) light. The desire to have a smooth and youthful complexion and to avoid damage to skin has contributed to the aspiration for a lighter skin tone, which has emerged as a major aspect of female consumer beauty culture, notably in Asia. 1 Some commonly used skin-whitening chemicals that contain hydroquinone have been associated with long-term side effects, and there is some concern about hydroquinone because it is a derivative of benzene. 2 Therefore, the identification of novel and safe bioactive compounds that can improve complexion and/or are able to help skin lightening is a major goal in cosmeceutical research.
Melanin is the key pigment in mammalian pigmentation and provides protection from UV radiation. Melanocytes, located in the stratum basale, produce melanin in the form of melanoblasts, which are then transferred to adjacent keratinocytes. Melanocyte tyrosinase is a rate-limiting enzyme in melanogenesis, catalyzing the hydroxylation of tyrosine to L-3,4-dihydroxyphenylalanine (L-DOPA) and the oxidation of L-DOPA to dopaquinone. Dopaquinone is then converted to melanin through a series of reactions. Excessive activity of melanocyte tyrosinase leads to various pigment disorders, including lentigo, photoallergic/phototoxic reactions, and Addison's disease. 3 Some polyphenol oxidases in plants have tyrosinase activity 4 which is partly responsible for the unwanted browning of fruits and vegetables during storage. 5
Phytochemicals are widely distributed in plants and their fruits. They are a major part of the human diet and the flavan-3-ols have been reported to possess beneficial health effects including antioxidant, cardioprotective, and neuroprotective properties. 6 Kiwifruit (genus Actinidia) are nutrient-rich fruit that confer numerous health benefits, including digestive and metabolic health, 4 which has been partly attributed to the polyphenolic compounds present in this fruit. There are two distinct commercial kiwifruit cultivars that are promoted for their attributes; green and gold-fleshed kiwifruit—both cultivars of A. chinensis. Wild-type Actinidia display great diversity and potentially harbor novel bioactive polyphenolic compounds that could be used for supporting human health and can be useful targets for breeding programs. 7
A small number of studies have demonstrated that kiwifruit may have tyrosinase inhibitory bioactivity.8–10 Skin melanocyte tyrosinase has been shown to be inhibited by kiwifruit seed and pericarp extracts in a dose-dependent manner.8,10 Kiwifruit extracts also significantly reduce melanin synthesis and transfer in normal human melanocytes and recipient keratinocytes. 11 The tyrosinase inhibitory efficacy of kiwifruit has been attributed to their procyanidin content with the degree of polymerization of these compounds associated related with their tyrosinase inhibitory properties. 8 To date, research on the tyrosinase inhibitory bioactivity of kiwifruit have focused on a single A. chinensis cultivar. Given that numerous genotypes of kiwifruit commercially grown have varying composition, and the variety of polyphenolic compounds present within each of these cultivars, the full potential and knowledge of the tyrosinase inhibitory of kiwifruit has not been fully evaluated.
Our aim in this study, therefore, was to gain a more comprehensive understanding of the ability of kiwifruit to inhibit tyrosinase activity in vitro, by examining the potential variation in bioactivity from a range of kiwifruit genotypes and seeking to identify the relationships between bioactivity and polyphenolic content.
Materials and Methods
Chemicals and Reagents
Kojic acid (2-hydroxymethyl-5-hydroxy-γ-pyrone; Sigma Aldrich, Cat No K3125), L-DOPA (L-3,4-dihydroxy-L-phenylalanine; Sigma Aldrich, Cat No D9628), β-Arbutin (4-hydroxyphenyl-β-D-glucopyranoside; Sigma Aldrich, Cat No A4256), Tyrosinase from mushroom (Sigma Aldrich, Cat No, T3824-50KU), DMSO (Sigma Aldrich, Cat No D4540), and Amberlite XAD-7 (Sigma Aldrich, Cat No, XAD7HP) were purchased from Sigma-Aldrich (Castle Hill, Australia). Potassium dihydrogen phosphate (KH2PO4; Riedel-de Haën, Cat No 04243) and di-Potassium hydrogen phosphate (K2HPO4; Riedel-de Haën, Cat No 04248) were purchased from Honeywell (North Ryde, Australia). All other general reagents were of Analar grade.
Preparation of Kiwifruit Polyphenolic Extracts
Ten different genotypes of kiwifruit were sourced from The New Zealand Institute for Plant & Food Research Ltd breeding program. Kiwifruit polyphenolic extracts from these different cultivars were prepared as previously described. 12 Briefly, for each kiwifruit genotype, 400 g of cortical flesh (cortex) was extracted in ethanol/water/formic acid (80/20/1) as follows. The cortex was homogenized (GLH homogenizer, Omni International, Auckland, New Zealand) and placed in solvent overnight at 1 °C. Coarse material was removed by filtration (no. 3 Whatman) and the extracts concentrated to approximately 100 mL by rotary evaporation at 40 °C. Column chromatography with XAD-7 resin was used to prepare a polyphenol-enriched fraction and remove sugars. Polyphenols bound to the XAD-7 were eluted with 100% ethanol and dried using a rotovapor and stored at –20 °C until used in experiments.
Polyphenol Identification and Semi-quantification
High-resolution accurate mass liquid chromatography mass spectrometry (HRAM-LC-MS) was used to identify and quantify kiwifruit extract compounds. Each of the 10 kiwifruit extracts was dissolved in solvent (1% formic acid in methanol) and diluted to approximately 20 mg/mL (total extract weight/volume), for an injection of 1 µL.
The HRAM-LC-MS system used was composed of a Dionex Ultimate® 3000 Rapid Separation LC and a micrOTOF QII high-resolution mass spectrometer (Bruker Daltonics, Bremen, Germany) fitted with an electrospray ion source. The LC column was a Luna Omega C18 200 × 2.1 i.d. mm, 1.6 µm (Phenomenex, Auckland, New Zealand) and was maintained at 40 °C. The flow was 400 µL/min. The solvents were A = 0.2% formic acid and B = 100% acetonitrile. The solvent gradient was 5% A:95% B, 0-0.5 min; linear gradient to 15% A:85% B, 0.5-7 min; linear gradient to 40% A:60% B, 7-12 min; linear gradient to 5% A:95% B, 12-16 min; composition held at 5% A:95% B, 16-18 min; linear gradient to 5% A:95% B, 18-18.2 min; to return to the initial conditions before another sample injection at 20 min. The injection volume for samples and standards was 1 μL. The micrOTOF QII parameters for these analyses were temperature 225 °C; drying N2 flow 6 L/min; nebulizer N2 1.5 bar, endplate offset −500 V, mass range 100-1500 Da, acquired were acquired at 2 scans/s. Negative ion electrospray was used with a capillary voltage of +3500 V. Post-acquisition internal mass calibration used sodium formate clusters with the sodium formate delivered by a syringe pump at the start of each chromatographic analysis. Mass spectral data were analyzed using DataAnalysis v4.1 (Bruker Daltonics, Bremen, Germany) software. Metabolite concentrations were calculated from the raw LCMS data using TASQ (Bruker Daltonics, Bremen, Germany) software. Exact ion chromatograms (EICs) were generated for each target compound using the exact mass for the [M-H]- ion. Target compounds identification was based on the accurate mass, retention time, and isotope ratio, and the concentration calculated by reference to an analytical standard. Where possible authentic standards were used for compound quantitation, however, standards were not available for all compounds. In these cases, concentrations were calculated using a reference standard that was chemically similar to the analyte.
Procyanidins are polymeric compounds and require a specific quantitation method. The dimeric procyanidins (B1, B2, B7) were quantified as procyanidin B2 equivalents. The mass spectra of procyanidin polymers contain prominent [M-H]2- or [M-H]3- ions, and weak or absent [M-H]- ions, depending on the size of the polymer. The most appropriate ion for each polymer size was selected based on intensity and specificity. Thus, procyanidin trimers were quantified using [M-H]- EICs, procyanidin tetramers using [M-H]2-, procyanidin pentamers using [M-H]2-, and procyanidin hexamers using [M-H]2-(13C).
Assessment of Tyrosinase Activity
The effect of kiwifruit extracts prepared from ten kiwifruit genotypes in inhibiting tyrosinase enzyme activity at a range of concentrations (15.6, 31.25, 62.5, 125, 250, and 500 µg/mL) was measured spectrophotometrically according to the method of Uchida et al, 13 with slight modifications. Briefly, kiwifruit extracts were dissolved in DMSO (5 mg/mL) and working stocks from this were used in the assay. A 10 µL sample of each kiwifruit extract was added to a 96-well plate and mixed with 60 µL of 50 mmol/L phosphate buffer (pH 6.8), and 20 µL of 0.9 mg/mL L-DOPA in phosphate buffer. Mushroom tyrosinase (10 µL 500 U/mL in phosphate buffer) was added to make a final working volume of 100 µL which was mixed for 1 min and incubated at 27 °C for 10 min. Absorbance was measured using a BMG PolarStar Omega plate reader (Alphatech, Auckland) at 493 nm to capture dopachrome production. Kojic acid (0.39, 0.78, 1.56, 3.125, 6.25, 12.5, 25, 50, and 100 µg/mL) and arbutin (62.5, 125, 250, 500, and 1000 µg/mL) were used as positive inhibitory controls for comparison. For experiments optimizing the substrate dose in the tyrosinase assay, 10 µL phosphate buffer was used instead of known inhibitors or kiwifruit extracts. Tyrosinase enzyme activities of all samples and controls at each of the doses assessed were analyzed in triplicate (n = 3), and tyrosinase activity is presented as absorbance units or as a percentage of the no treatment control. The dose effect on tyrosinase enzyme activity of known tyrosinase enzyme inhibitors was measured and expressed as the concentration necessary to achieve 50% tyrosinase enzyme inhibition (IC50).
Statistical Analysis
A one-way ANOVA with Tukey's post-tests was used for statistical comparisons of single sets of data. For the comparison of a series of data, a two-way ANOVA was performed with Bonferroni post-tests. Correlations were performed using the R statistical language, and the ggplot2 and tidyR packages. Statistical significance was set at a confidence threshold of P < 0.05. Each experiment was carried out at least three times. Data are presented as means ± SEM unless otherwise stated.
Principal component analysis (PCA) was performed using the LC-MS chemical composition data of the 10 kiwifruit genotype extracts and the tyrosinase inhibition results (expressed as % control) at 125 µg/mL to define groups. The data were log10 transformed, pareto scaled, analyzed by PCA and the heat map prepared (Figure 3) using the MetaboAnalyst 5.0 platform (www.metaboanalyst.ca). 14 IC50 calculations were performed using GraphPad Prism version 5.0.3 for Windows, GraphPad Software, San Diego, California USA, www.graphpad.com.
Results
Polyphenolic Composition of Kiwifruit Extracts
A total of 21 polyphenolics were identified through molecular ion identification (M+, or M-1-) using HRAM-LC-MS (Supplementary table). A large amount of variability in the presence and levels of these polyphenolic compounds extracted from the 10 kiwifruit genotypes examined was observed.
Optimization of Tyrosinase Activity Assay
To maximize the ability to detect an effect on tyrosinase enzyme activity (tyrosinase inhibition), we initially examined a range of doses of the substrate L-DOPA (0.01 to 0.72 mg/mL, which is oxidized to dopachrome detected at 493 nm), to determine the most suitable dose required. A dose-dependent and rapid linear increase in absorbance (indicating dopachrome formation) was observed up to a concentration of 0.18 mg/mL L-DOPA (0.51 ± 0.03 absorbance), with increasing the concentrations of L-DOPA, after which the absorbance values began to plateau (Figure 1a). Increases in the concentrations of L-DOPA had minimal effect on absorbance (0.57 ± 0.03 and 0.59 ± 0.03, 0.36 and 0.72 ng/mL L-DOPA, respectively). A substrate dose of 0.18 mg/mL of L-DOPA was chosen for future experiments in this study as it was the minimal dose of L-DOPA required to mediate threshold dopachrome production.

Optimization of the tyrosinase activity assay. Tyrosinase activity at a range of L-DOPA substrate concentrations (a), a range of doses of inhibitor kojic acid (b), and a range of doses of inhibitor arbutin (c). Tyrosinase activity was assessed using L-DOPA as substrate in phosphate buffer with mushroom tyrosinase and dopachrome production (10 min incubation at 27 °C) measured at 493 nm absorbance. Data are shown as means ± SEM from three experiments. Broken line signifies IC50. ** = P < 0.01, *** = P < 0.001, versus control (0 μg/mL inhibitor). L-DOPA, L-3,4-dihydroxyphenylalanine.
To characterize our tyrosinase enzyme inhibition assay, we evaluated known inhibitors of tyrosinase, kojic acid, and arbutin, at a range of concentrations (Figure 1b and c). In the absence of arbutin or kojic acid, tyrosinase activity was maximal and gave absorbance values of 0.61 ± 0.02 (n = 3). As concentrations of kojic acid increased (especially above 6.25 µg/mL), a significant inhibition of tyrosinase activity was observed compared to the negative control (0 µg/mL; P < 0.01). From 6.25 to 100 µg/mL kojic acid, a dose-dependent inhibition of tyrosinase was observed. At 100 µg/mL kojic acid, absorbance values were (0.04 ± 0.01) compared to the control (0.61 ± 0.02) (P < 0.01), representing a 96% inhibition of tyrosinase activity. Our data revealed an IC50 for kojic acid-mediated inhibition of tyrosinase activity of 9.2 µg/mL (Figure 1b).
Arbutin, a known tyrosinase inhibitor found naturally in some plants, is a glycosylated hydroquinone and commonly used in cosmetics. 15 Inhibition of tyrosinase activity by arbutin was not as marked as that of kojic acid. Lower concentrations of arbutin (up to 250 µg/mL) demonstrated only minor (insignificant) inhibition of tyrosinase activity (Figure 1c). Only at arbutin concentrations of 500 µg/mL and higher was significant tyrosinase inhibition observed compared with the control (0.61 ± 0.02 vs 0.47 ± 0.02 absorbance values, respectively, P < 0.01). Absorbance values at 1000 µg/mL arbutin were 0.42 ± 0.02 versus the control 0.61 ± 0.02 (P < 0.01), representing a 60% inhibition of tyrosinase activity. Our data revealed an IC50 for arbutin-mediated inhibition of tyrosinase is 384.6 µg/mL (Figure 1c).
Kiwifruit Extract Mediated Inhibition of Tyrosinase Activity
Polyphenolic extracts derived from 10 commercial and proprietary kiwifruit varieties were assessed at a range of concentrations (0-500 µg/mL) for their efficacy in inhibiting tyrosinase activity. Tyrosinase enzyme inhibition varied greatly between kiwifruit polyphenol extracts as presented in Table 1.
Tyrosinase Enzyme Inhibition of Kiwifruit Polyphenolic Extracts.
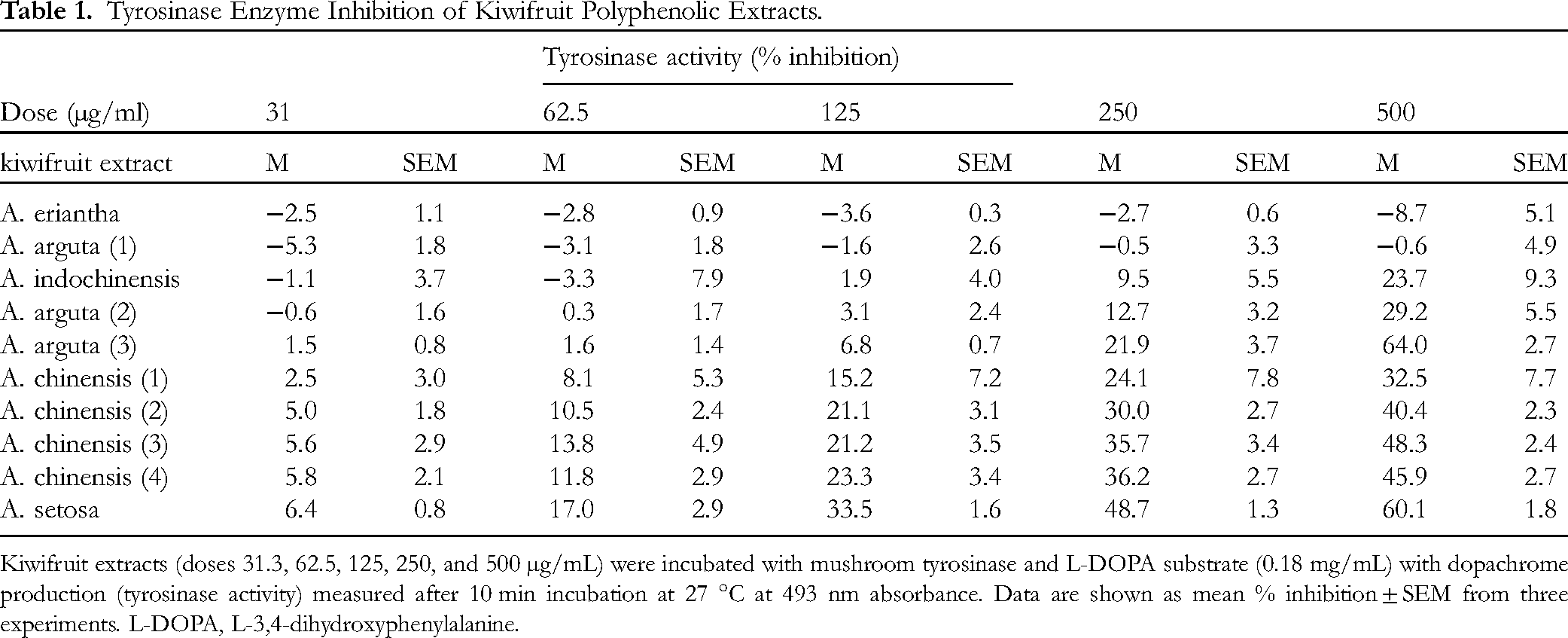
Kiwifruit extracts (doses 31.3, 62.5, 125, 250, and 500 µg/mL) were incubated with mushroom tyrosinase and L-DOPA substrate (0.18 mg/mL) with dopachrome production (tyrosinase activity) measured after 10 min incubation at 27 °C at 493 nm absorbance. Data are shown as mean % inhibition ± SEM from three experiments. L-DOPA, L-3,4-dihydroxyphenylalanine.
The efficacy of the 10 kiwifruit polyphenolic extracts in inhibiting tyrosinase activity at a defined dose (125 µg/mL) was measured to further examine the variable potency of the extracts prepared from different kiwifruit genotypes in inhibiting tyrosinase (Figure 2). Arbutin control (500 µg/mL) significantly inhibited tyrosinase 18.88% ± 0.05 (P < 0.001). The greatest inhibition (at 125 µg/mL of extract) of tyrosinase activity was 33.54% ± 1.59 (P < 0.001) by A. Setosa. Polyphenolic extracts prepared from A. chinensis (4), A. chinensis (3), A. chinensis (2) and A. chinensis (1) modestly inhibited tyrosinase activity (15.18% ± 7.17, 21.20% ± 3.54, 21.05% ± 3.06 and 15.18% ± 7.17, respectively). In contrast, polyphenol extracts prepared from A. eriantha (−3.55% ± 0.32), A. indochinensis (1.91% ± 3.96) A. arguta (1) (−1.57% ± 2.58), A. arguta (2) (3.01% ± 2.4), and A. arguta (3) (6.79% ± 0.65) had no significant effect on tyrosinase enzyme activity.

Tyrosinase inhibitory effects of polyphenolic compound enriched extracts from 10 genotypes of kiwifruit. Kiwifruit extracts (125 μg/mL) and arbutin control (500 μg/mL) were incubated with mushroom tyrosinase and L-DOPA substrate (0.18 mg/mL) with dopachrome production (tyrosinase activity) measured after 10 min incubation at 27 °C at 493 nm absorbance. Data are shown as means ± SEM from three experiments. ** = P < 0.01, *** = P < 0.001, versus control (0 μg/mL inhibitor). L-DOPA, L-3,4-dihydroxyphenylalanine.
Kiwifruit Polyphenolics and Tyrosinase Inhibition
We evaluated the correlation between the amounts of each polyphenolic compound present in each kiwifruit extract to tyrosinase inhibition and assessed statistical significance (Table 2). This identified several compounds that were correlated to tyrosinase inhibition. At the 99% confidence level procyanidin B2, procyanidin hexamers, procyanidin pentamers, procyanidin tetramers, and procyanidin trimers were correlated to tyrosinase inhibition. At the 95% confidence level, epicatechin and procyanidin B7 were also correlated to the inhibition of tyrosinase activity.
Correlation Coefficient Analysis of Inhibition of Tyrosinase Activity and Kiwifruit Extract Polyphenolic Composition of the Different Genotypes a .

*=P < 0.05, *** = P < 0.01
PCA was performed on tyrosinase inhibitory activities at a dose of 125 µg/mL and polyphenolic content for the 10 genotypes (Figure 3a). The first component represented 66.2% of this total variation and allowed for the discrimination of two distinct groups. This unsupervised model neatly separated kiwifruit polyphenolic extracts into those that inhibited tyrosinase activity from those with no inhibitory effect on the activity of the enzyme.
The HRAM-LC-MS intensity of each of the 21 identified compounds of the 10 kiwifruit polyphenolic extracts is shown as a heat map (Figure 3b). The two groups highlighted by the PCA analysis had different polyphenolic compositions. The kiwifruit polyphenolic extracts that demonstrated an inhibitory effect on tyrosinase activity contained high proportions of procyanidins (B2, B7, trimers, tetramers, pentamers, hexamers), epicatechin, (E)-caffeoyl-4-glucoside, and quercetin-3-rhamnoside. These procyanidin compounds were scarce in the kiwifruit polyphenol extracts that exhibited minimal or no tyrosinase enzyme inhibition. Further, the two kiwifruit extracts which showed the greatest tyrosinase inhibition contained procyanidin hexamers, tetramers, and pentamers at higher relative concentrations as identified by HRAM-LC-MS. Interestingly, four of the five kiwifruit extracts that inhibited tyrosinase activity contained fraxin, 2-O-caffeoylthreonine, chlorogenic acid, and neochlorogenic acid. These compounds are rich in samples that inhibited tyrosinase, but are absent in those that showed limited or no tyrosinase inhibition.

PCA score plot (a) and heat map (b) obtained from HRAM-LC-MS profiles of 21 polyphenolic compounds identified in 10 different kiwifruit extracts. In score plots, green crosses represent polyphenolic extracts inhibitors of tyrosinase activity, red triangles represent polyphenolic extracts non-inhibitors of tyrosinase activity. Tyrosinase inhibition by the different polyphenol kiwifruit extracts is displayed in the heat map as colors ranging from red to blue as displayed in the key. HRAM-LC-MS, high-resolution accurate mass liquid chromatography mass spectrometry.
Discussion
The desire for an effective and natural treatment for skin damage from UV radiation and hyperpigmentation has emerged as a major aspect of female beauty culture, especially in Asia, where a lighter skin tone is cosmetically valued. 1 Hence in recent years, there has been a growing interest and search for natural, safe bioactive compounds capable of inhibiting tyrosinase to help skin lightening and complexion. Tyrosinase is a rate-limiting enzyme in the biosynthesis of melanin—a natural skin pigment, catalyzing the hydroxylation of tyrosine to L-DOPA and the oxidation of L-DOPA to dopaquinone. Through a series of reactions, dopaquinone is then converted to melanin. Here we have evaluated 10 different polyphenolic extracts from wild-type, commercial, and proprietary genotypes of kiwifruit sourced from our organization's breeding program for their tyrosinase inhibitory properties in vitro. The polyphenolic compositions of the extracts were quantified to determine correlations to bioactivity.
We tested known inhibitors of tyrosinase (kojic acid, arbutin) at a range of doses and confirmed their reported tyrosinase inhibitory ability in our assay. Kojic acid, a fungal metabolite, is a well-studied inhibitor of tyrosinase, and is currently used as a cosmetic skin-whitening agent and as a food additive for preventing enzymatic browning. 16 However, the use of kojic acid in cosmetics has been limited because it has been reported to be carcinogenic. 17 Arbutin, a prodrug of hydroquinone found in some plants, has also been reported to reduce or inhibit melanin synthesis by inhibiting tyrosinase activity. However, natural forms of arbutin are chemically unstable and can release hydroquinone which is catabolized to benzene metabolites with potential toxicity for bone marrow. 18 For these reasons, natural and safe alternative inhibitors of tyrosinase are being sought for their potential use in the cosmetic and food processing industries.
Some flavonoids are known to inhibit tyrosinase due to their ability to chelate copper at the enzymes’ active site, 19 with (+)-catechin-aldehyde polycondensates inhibiting both L-DOPA oxidation and L-tyrosinase hydroxylation. 20 Procyanidins are competitive inhibitors of tyrosinase monophenolase and diphenolase. 8 Kiwifruit have previously been identified as having tyrosinase inhibitory properties and the underlying compounds were identified as likely due to procyanidins. 8 Chai et al showed that the amount of inhibition was also related to the degree of proanthocyanidin polymerization. Specifically, the authors identified an increase in monophenolase and diphenolase activity with an increase in the degree of proanthocyanidin polymerization. However, it is difficult to know whether this tyrosinase inhibitory property of kiwifruit is exclusively due to procyanidins given that kiwifruit extracts tested for tyrosinase activity by Chai et al 8 focused on a single variety. A study by Park et al 9 reported differences in tyrosinase enzyme inhibition from four commercial kiwifruit cultivars with the most potent extract from the “Jecygold” cultivar. However, as the phytochemical composition of the extracts was not reported, the variation in the inhibitory effect of the varieties could not be associated to differences in their composition. In the present study, we sought to characterize the tyrosinase modulatory properties of polyphenolic extracts prepared from 10 kiwifruit cultivars. Our results show that not all kiwifruit polyphenolic extracts inhibited tyrosinase enzyme inhibition even at the highest concentration evaluated of 500 µg/mL. When the tyrosinase inhibitory ability of each extract was analyzed at a dose of 125 µg/mL, a clear differentiation in extracts that inhibited tyrosinase from those that had minimal, or no effect was observed. Given that all extracts were prepared under the same conditions, these findings highlight that the intrinsic tyrosinase inhibitory properties of kiwifruit may be genotype dependent.
To gain further insight on how kiwifruit genotype might underpin kiwifruit bioactivity modulating tyrosinase enzyme activity, the polyphenolic composition of the extracts from the 10 kiwifruit genotypes was characterized. Using the UHPLC-MS, 21 polyphenolic compounds were identified and semi-quantified for each of the extracts. A PCA and PLS-DA evaluation of the compositional and tyrosinase inhibition data was then conducted to reveal interactions between kiwifruit genotype and their tyrosinase bioactivity. Using these models allowed for the discrimination of extracts from the 10 kiwifruit genotypes into two distinct groups based on their polyphenolic composition. The strength of tyrosinase inhibitory bioactivity of kiwifruit extracts within each group was then correlated with each of the identified polyphenolic compounds present in the extracts.
Interestingly, multivariate analysis of our results shows that the polyphenolic compositions of kiwifruit extracts that inhibited tyrosinase activity were distinctly different from those that exhibited no activity. The polyphenolic extracts from kiwifruit genotypes with tyrosinase inhibitory activity commonly contained significant concentrations of dimeric and polymeric procyanidin compounds. In contrast, these compounds were largely absent in extracts that were categorized as having no tyrosinase inhibitory activity. These results confirm and extend the findings of Chai et al 8 who first reported the tyrosinase inhibiting properties of kiwifruit. Our results also further confirm that the procyanidin compounds with the greater polymer length were found at higher levels in the kiwifruit extracts with the greater inhibitory effect. 8 Correlation analysis between tyrosinase inhibition and the polyphenolic compounds present in the 10 kiwifruit extracts identified several compounds as being positively (epicatechin, procyanidin B2, procyanidin B7, procyanidin hexamer, procyanidin pentamer, procyanidin tetramer and procyanidin trimer) and negatively (quercetin-3-galactoside) correlated to tyrosinase inhibition. These findings further support the role of procyanidins and their various polymeric forms or degree of polymerization in variably mediating tyrosinase activity in kiwifruit. Furthermore, quercetin glycosides were only found in the extracts exhibiting no tyrosinase inhibitory activity (A. eriantha, A. indochinensis, A. arguta (1), A. arguta (2) and A. arguta (3)), with quercetin glycosides being negatively correlated to tyrosinase inhibitory activity. These findings support those of Ye et al 21 who reported the efficacy of quercetin-3-galactoside in increasing tyrosinase activity and inducing an increase in intracellular melanin production in murine melanocytes.
This work highlights the variety of complex polymeric procyanidins that are associated with tyrosinase inhibition, beyond the relatively simple compounds such as epicatechin and procyanidin B2. Future work could study the molecular binding of known structures to tyrosinase, if it becomes possible to isolate and purify polymeric compounds from kiwifruit, specifically the tetramers, hexamers and pentamers.
Study Limitations
Gąsowska-Bajger & Wojtasek 22 identified a few compounds, including quercetin and kaempferol, that are not only oxidized by tyrosinase, but also react with o-quinones generated from catechols by oxidation, resulting in an apparent inhibitory effect. Therefore, in the search for tyrosinase inhibitors using the standard assay, compounds with strong reducing properties will not give reliable results 23 and could be a limitation of our study. Our results show that kiwifruit genotypes rich in quercetin glycosides had no tyrosinase inhibitory effects. However, the data suggests that the assay is still sensitive to differentiate between genotypes with strong versus weak tyrosinase inhibitory effects.
Secondly, in our study, we used mushroom tyrosinase instead of human tyrosinase. This assay relies on mushroom tyrosinase extracted from Agaricus bisporus, which is widely used in the search for inhibitors of tyrosinase in relation to skin-whitening agents for humans, due to the low cost of sourcing. 24 It should be noted that the sequences of human and Agaricus bisporus tyrosinase are only 23% identical; 25 therefore, mushroom and human tyrosinases carry structural differences which might affect substrate or inhibitor binding. However, the amino acids of the central copper-binding domain between tyrosinases are conserved.26,27 Interestingly, previous work shows that kojic acid was a potent inhibitor of Agaricus bisporus tyrosinase (IC50 53.70 μM). However, Kojic acid is much less effective against human tyrosinase (IC50 571.17 μM), as seen by the 10.6-fold difference in inhibitory effects. 28 Another limitation of this study is that we did not use human tyrosinase, therefore, it is unknown how well the kiwifruit extracts from this study will translate to human tyrosinase inhibitory activity. Therefore, additional studies will need to be conducted to confirm the effectiveness of kiwifruit extracts for their effectiveness on human tyrosinase, as a few of the procyanidins that appear effective in tyrosinase inhibition may not be bioavailable or might even have unforeseen side effects, including cytotoxicity, within human cell culture models.
Conclusions
Results reported in this study support and extend previous research demonstrating the inherent tyrosinase enzyme activity properties of kiwifruit. Furthermore, the enzyme inhibitory activity of the 10 kiwifruit polyphenolic extracts that were assessed varied greatly, indicating that some kiwifruit genotypes were more potent inhibitors than others. Multivariate analysis identified that the variation in tyrosinase inhibitory ability between genotypes is likely explained by the presence of procyanidin compounds and the degree of their polymerization. Our study highlights the value of examining a genetically diverse variety of fruits from the same species in the search for tyrosinase inhibitors. This study's main limitation is that it is in vitro only, and whether the variation in tyrosinase inhibitory activity displayed in kiwifruit genotypes reported here translates to differences in melanin production in biological systems remains to be confirmed. Additional in vitro and in vivo investigations are therefore warranted. Taken together, however, our findings confirmed the tyrosinase inhibitory properties from a variety of kiwifruit genotypes. These can now be examined as targets for breeding programs and utilized by innovative industries to support the development of new natural kiwifruit-based skin beauty and complexion products.
Supplemental Material
sj-xlsx-1-npx-10.1177_1934578X241267063 - Supplemental material for The Effect of Polyphenolic Extracts from 10 New Zealand Kiwifruit Cultivars in Inhibiting Tyrosinase Enzyme Activity In vitro
Supplemental material, sj-xlsx-1-npx-10.1177_1934578X241267063 for The Effect of Polyphenolic Extracts from 10 New Zealand Kiwifruit Cultivars in Inhibiting Tyrosinase Enzyme Activity In vitro by Andrew M. Carroll, Gregory M. Sawyer, Laurie Favre, Tony K. McGhie, Dominic Lomiwes and Roger D. Hurst in Natural Product Communications
Footnotes
Acknowledgements
The authors would like to acknowledge Duncan Hedderley (Biometrician, Plant & Food Research) for statistical analysis advice.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by The New Zealand Institute for Plant & Food Research Limited, a New Zealand Crown Research Institute.
Ethical Approval
Ethical approval is not applicable for this article.
Informed Consent/Patient Consent
There are no human subjects in this study and informed consent is not applicable.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Supplemental Material
Supplemental material for this article is available online.
Trial Registration Number/Date
Trial registration number is not applicable for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
