Abstract
Introduction
Synthetic chemical drug treatments have drawbacks including adverse effects and toxicity. Natural alternatives are sought after. Chemotherapy can be toxic. Medicinal plants offer a suggested alternative. This study examines P. harmala plant extracts for phenolic and flavonoid content, antimicrobial, antioxidant, and anticancer activities.
Methods
Two samples of P. harmala plant extract were prepared using ultrasonication, with ethanol concentrations of 100% and 50%. The total phenolic and flavonoid contents were determined using a chemical assay method. The antimicrobial activity of the extracts was evaluated against gram-positive bacteria (Staphylococcus aureus and Streptococcus) and gram-negative bacteria (Escherichia coli) using the diffusion procedure. The antioxidant impact of the extracts was assessed using the DPPH procedure. P. harmala extracts showed inhibitory effects on MCF7 breast cancer and HT29 colon cancer cell lines.
Results
The results of the study indicated that the P. harmala plant extracts were rich in phenolic compounds (with total phenolic content of 215.8 ± 3.5 and 155.8 ± 2.9 mg gallic acid per g extract for 50% and 100% ethanol extracts, respectively) and flavonoids (with total flavonoids content of 112.1 ± 3.1 and 92.3 ± 1.8 mg catechin per g extract) and had a high rate of antioxidant activity. The 50% ethanol extract yielded 411.8 ± 3.5 μmol trolox/g, while the 100% ethanol extract yielded 312.9 ± 8.2 μmol trolox per g extract. P. harmala extracts exhibited potent antimicrobial activity against E. coli, S. aureus, and Streptococcus. They also demonstrated strong anticancer activity, causing significant cell death in breast and colon cancer cell lines within 48 h of culturing.
Conclusion
P. harmala ethanolic extracts are rich in polyphenolic compounds and flavonoids, displaying high antioxidant activity. They also exhibit strong inhibitory effects against gram-positive and gram-negative bacteria and demonstrate potent anticancer activity against breast and colon cancer cell lines.
Introduction
The worldwide interest in ethnopharmacy as an alternative source of pharmacologically impact substances has grown, especially in the search for novel drugs to treat a variety of diseases.1-5 However, within developed nations, the widespread application of chemicals and synthetic pharmaceuticals gained significant traction, resulting in the rise of harmful multidrug-resistant microorganisms, thereby giving rise to grave health concerns.6-8 As a consequence, there has been a global movement toward choosing natural-source treatment options.9-11 Multiple studies had shown that medicinal as well as aromatic plants are highest in phytochemicals, which have a variety of biological effects like antioxidant, antineoplastic, antibacterial, as well as anti-inflammatory characteristics.12-17 The Mediterranean environment supports the development of many plant types, many which have been different ecological, biological, nutritional, as well as therapeutic properties.18-25 Peganum harmala L. (P. harmala), native to Central Asia, Middle East, and North Africa, is a traditional treatment that has long been used in folk medicine, due to its long-standing antibacterial characteristics. Many studies have shown that P. harmala has various activities that involve antioxidant, antidiabetic, analgesic, anti-inflammatory, antiviral, antimicrobial, and antineoplastic. 26 Many biological activities involved in P. harmala have been proposed to be mediated by phytochemicals like polyphenols, saponins, tannins, flavonoids, as well as alkaloids. 27
P. harmala contains many bioactive compounds like harmine, harmaline, and tetrahydroharmine which are the most well-known alkaloids. These compounds are responsible for many of the plant's pharmacological effects. Other phytochemicals found in P. harmala include quinazoline alkaloids, coumarins, flavonoids, and tannins. These compounds contribute to the plant's medicinal and psychoactive properties.28,29 The current study has demonstrated measurement of the anticancer, antimicrobial, as well as antioxidant activities of the P. harmala (Zygophyllaceae) plant hydroethanolic extract. The two cancer cell lines (MCF7 breast as well as HT29 colon cancer cell lines) were selected as they are commonly used to test the anticancer activity of plant extracts. Regarding the two bacterial cell types, two gram-positive bacteria (S. aureus and Streptococcus) and one gram-negative bacteria (E. coli) have been selected for this study.
Materials and Methods
Plant Collection and Extraction
The plant materials (leaves of P. harmala) were obtained from the Palestinian soil (from Hebron in West Bank) in March 2022 with voucher number (Pharm-PCT-260), and it had been dried and then ground into small parts. One sample of ground plant materials weight three grams was added to a 50 mL ethanol solvent with a concentration of 50%, and a second sample, weight three grams, added to a 100 mL ethanolic solvent with a concentration of 100%. Both mixtures transferred into water bath to optimize the extraction process, then the extracts have filtered via filter paper, and the filtrate had evaporated by water bath for 2 hours at a constant temperature of 53 °C. Then, the resulting mixture was pipetted into small containers for evaporation of the solvent, and the crude extracts were acquired as well as stored at 4 °C. The concentration of the extracts which used for all analyses and testing antimicrobial and anticancer activities is 0.1 mg/mL.
Evaluation of Total Phenolic Content
For the assessment of phenolic content within the P. harmala extract, a 0.2 mL of the extract (0.5 mg/mL) was combined with 1 mL of diluted Folin-Ciocalteu oxidizing agent in 50 mL of deionized water. This mixture was enabled to interact with the phenolic compounds for five minutes. Subsequently, 0.8 mL of 7.5% Na2CO3 (sodium carbonate) solution was then added, as well as the mixture was left for a duration of 120 min. The occurrence of phenolic compounds was signaled by an alteration in the color of the Folin-Ciocalteu reagent. To ascertain the phenolic content, the specimens were subjected to analysis using a UV-visible spectrophotometer for absorbance measurement at 760 nm. Moreover, the results were quantified in milligrams (mg) of gallic acid per each gram of extract, serving as a standard phenolic compound.24,29
Evaluation of Total Flavonoid Content
For the evaluation of flavonoid content within the P. harmala extract, a mixture was dressed by combining 1 mL of the plant extract with 4 mL of deionized water, 0.3 mL of 10% AlCl3·6H2O (a reagent that reacts with flavonoids in the ortho position), and 0.3 mL of 5% NaNO2. Subsequently, after a 6-min interval, 2 mL of 1N NaOH as well as 2.5 mL of deionized water were introduced to the final mixture. After that, the absorbance was identified at 510 nm utilizing a UV-visible spectrophotometer. Quantitative expression was carried out in milligrams of catechin per gram, serving as a standard flavonoid. A calibration curve was constructed employing catechin concentrations spanning from 50 to 100 ppm. Following the determination of catechin flavonoid concentration within the extract through UV-visible spectrophotometry and graphical representation of values, the resultant curve exhibited linearity.17,29
Testing for Antioxidant Activity
The investigation into the antioxidative activity of the P. harmala extract was conducted employing the methodology outlined according previous study, 30 utilizing DPPH (2,2-diphenyl-1-picrylhydrazyl) assay. Furthermore, DPPH serves as a solid free radical, exhibiting an inherent coloration. When the extract is introduced to the DPPH, a reduction in color intensity occurs if the sample exhibits positive antioxidant activity due to the ensuing reaction. The measurement of this reaction is carried out through absorbance analysis. The DPPH radical assay, known for its stability, is rooted in the assessment and computation of antioxidant scavenging potential against the enduring DPPH radical. The procedure entailed the addition of a 4 mL portion of 0.0634 mM of the DPPH solution with methanol (95% ethanol) to the 0.1 mL of the plant extract, followed by thorough agitation. Following a 30-min incubation period, absorbance was identified at 515 nm. Quantitative presentation was shown in micromoles of Trolox per each gram, with Trolox being an antioxidant and a derivative of vitamin E. A calibration curve was established, encompassing concentrations spanning from 20 to 180 ppm of Trolox.
HPLC Analysis
The constituents present within the P. harmala extracts were described using HPLC analysis, employing an ODS column from Waters (XBridge, 5 μm, 4.6 ID×150 mm). Then, the mobile phase encompassed a blend of 0.5% water and acetic acid (as solvent A) with acetonitrile (as solvent B). The gradient elution profile involved a reduction of solvent A from 100% to 70% within 40 min, followed by subsequent decreases to 40% after an additional 20 min and 10% over a 2-min interval. This solvent composition was maintained for 6 min, before being restored to its initial state within 2 min. Prior to the injection of the subsequent sample, the HPLC system was stabilized with solvent A for 7 min. All samples underwent filtration via a 0.45 μm PTFE filter. Furthermore, the photodiode array detector was configured to scan wavelengths within the range of 210-500 nm. The flow rate was 1 mL/min, whereas the injection volume was 20 microliters. The temperature of the column was strictly monitored at 25 °C throughout the analysis.
Testing for Antimicrobial Activity
To evaluate the antimicrobial efficacy of P. harmala, the bacterial strains of E. coli, S. aureus, and Streptococcus were employed as test organisms. Preparation of Mueller Hinton Agar media involved measuring 19 grams of the powder in a weighing dish and combining it with 500 mL of deionized water within an Erlenmeyer flask. Following heating to the boiling point, the media underwent autoclaving at 121 °C for 15 min. Upon completion of autoclaving and subsequent cooling, the media was dispensed into six culture plates for solidification. Upon adequate solidification of the agar, two plates each were subjected to inoculation with S. aureus, Streptococcus, and E. coli bacterial strains. The preparation of the bacterial inoculum adhered to the McFarland turbidity standard, a universally accepted criterion for adjusting bacterial suspension densities. The turbidity of the inoculum was assessed intermittently using a McFarland card, a card featuring contrasting black lines that reach the McFarland standard when their distortion matches the standard's turbidity. 31 Subsequently, the inoculum was swabbed onto the agar media's surface utilizing inoculating loops. To investigate the antimicrobial potential of P. harmala extracts, the well diffusion procedure was adopted. Wells had fashioned in the agar media by utilizing the back of a 100 µL of pipette tip, which had inserted and extracted to create voids. The wells were subsequently filled with 100% ethanol extracts of P. harmala. The bacterial strains under scrutiny were S. aureus, Streptococcus, and E. coli. The culture plates were then placed within an incubator set at 37 °C, and observations were carried out 24 h later.
Testing for Inhibitory Effects to Cancer Cell Line
The experimental study used two different cancer cell lines, the first being MCF7 breast cancer cells as well as the second being HT29 colon cancer cells. Additionally, the extracts were tested against LX-2, (normal human hepatic stellate cells). These cells were cultivated within RPMI media and subjected to a 48-h incubation period prior to initiating the experiments. To establish a reference baseline, negative control dishes were assembled containing solely MCF7 or HT29 cells. To address the potential cytotoxicity of dimethyl sulfoxide (DMSO), an additional control was prepared involving the introduction of 50 µL of DMSO to each cell line. The experimentation employed a total of eight culture plates, where the DMSO concentrations were manipulated within a range from 300 µL to 50 µL, decreasing incrementally by 100 µL to gage its toxicity.
For the 50% ethanol concentration of the P. harmala extract, four plates were set up, encompassing DMSO volumes of 300 µL, 200 µg/mL, 100 µL, and 50 µL. This procedure was repeated for the 100% ethanol concentration of the P. harmala extract. Both cancer cell lines were subjected to this experimental design. Following the preparation and introduction of test substances, the cells were subjected to a subsequent 48-h incubation period. Subsequent observations were conducted to detect any observable alterations within the cellular environment.
Statistical Analysis
Results (total phenolic and flavonoids contents and antioxidant activities as well as antimicrobial activity) were expressed as average ± standard deviation of three samples (n = 3).
Results
Extraction Process of the Plant
Ultrasonic extraction also known as sonication was used in this study for extraction of plant compounds due to several advantages compared to traditional extraction methods. Ultrasonic extraction enhances mass transfer by generating acoustic cavitation, which creates microbubbles in the solvent. These microbubbles collapse near the plant material, leading to the formation of microjets and shockwaves. This phenomenon disrupts the cell walls and increases the surface area available for extraction, thereby improving extraction efficiency. 32 Ultrasonic extraction is also a rapid process compared to conventional extraction methods. It can significantly reduce extraction times, allowing for high-throughput processing of plant samples and increasing productivity in the laboratory or industrial settings. 33 Overall, ultrasonic extraction offers numerous advantages including improved efficiency, speed, selectivity, and environmental friendliness, making it a valuable technique for extracting bioactive compounds from plants for various applications in food, pharmaceutical, and cosmetic industries.
Total Phenolic Content
The quantification of the overall phenolic content present within the P. harmala extracts was conducted through utilization of the Folin-Ciocalteu reagent. The outcomes of this analysis were subsequently articulated in the terms of gallic acid milligrams per each gram (mg /g) after establishment of a calibration curve. The findings prominently showcased the substantial phenolic richness of the plant's ethanolic extract. Specifically, the 50% ethanol extract exhibited a substantial phenolic content of gallic acid 215.8 ± 3.5 (mg per gram of extract), while the 100% ethanol extract displayed a noteworthy phenolic content of gallic acid 155.8 ± 2.9 (mg per gram of extract), as shown in Table 1.
Total Phenolic Content as Gallic Acid (mg per Gram of Extract), Total Flavonoids Content as Catechin (mg per Gram of Extract) and Antioxidant Activity (µmol Trolox per Gram of Extract).

Total Flavonoid Component
The quantification of the complete flavonoid content within the P. harmala extracts was performed employing the aluminum chloride reagent, followed by the expression of results in terms of milligrams of catechin per gram (mg /g) subsequent to establishment of a calibration curve. The results prominently highlighted the substantive flavonoid abundance within the plant's ethanolic extract. Specifically, the 50% ethanol extract exhibited a considerable flavonoid content of catechin 112.1 ± 3.1 (mg per gram of extract), whereas the 100% ethanol extract displayed a noteworthy flavonoid content of catechin 92.3 ± 1.8 (mg per gram of extract), as illustrated in Table 1.
Antioxidant Impact
The antioxidant capability of a plant signifies the effectiveness of its oxygen radical scavenging components, notably phenolic compounds. The antioxidative potential of phenols emanates from their redox attributes, enabling them to operate as reducing actors, hydrogen donors, as well as suppressors of singlet oxygen. 34 DPPH is a rapid and sensitive way to determine the antioxidant activity of plant extracts. 35 The findings indicated robust antioxidant potency within the P. harmala plant, which was demonstrated through the DPPH assay outcomes presented in terms of micromoles of trolox (μmol /g), as delineated in Table 1. The 50% ethanol extract exhibited a substantial antioxidant potential, yielding Trolox 411.8 ± 3.5μmol trolox per each gram of extract, while the 100% ethanol extract displayed a value of 312.9 ± 8.2 μmol trolox per each gram of extract. Discrepancies in the efficacy of DPPH assessments across different locales arise from variations in the extracting solvent, coupled with geographic elements like location, extraction methodologies, and seed diversities. 36
HPLC Profiles of the Plant Extracts
In Figure 1, the tested chromatogram presents the ethanolic raw extract of the P. harmala plant leaves at a wavelength of 280 nm. This specific wavelength was chosen due to its correspondence with the point of maximal absorption exhibited by the principal peaks. The resolved constituents were discerned within the timeframe of 19.7 to 37.4 min for the 50% ethanolic extract and 24.4 to 38.5 min for the 100% ethanolic extract. These ranges imply the presence of comparatively nonpolar compounds.
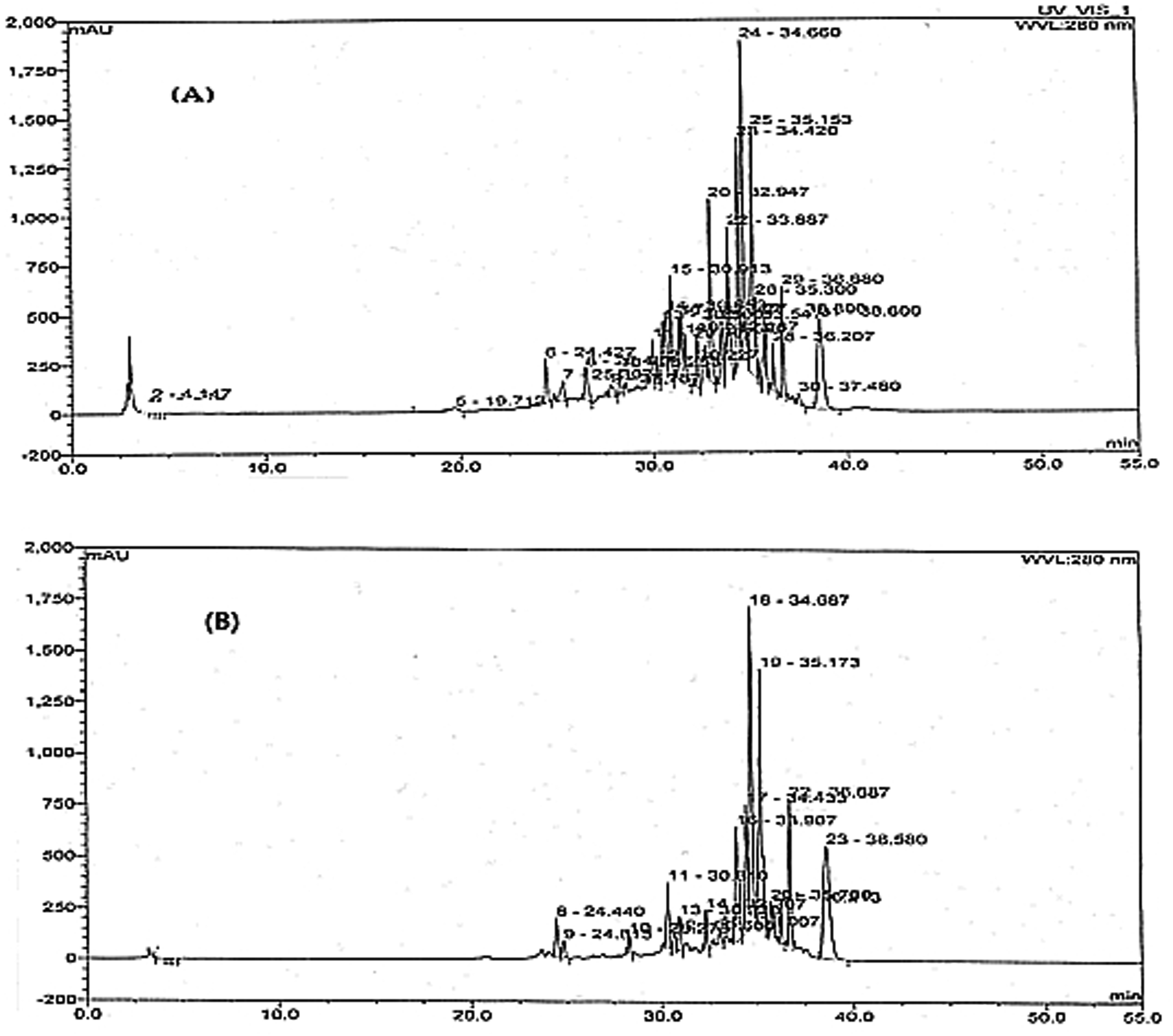
HPLC chromatogram of 50% ethanolic plant extract (A) and 100% ethanolic extract (B) of P. harmala plant at 280 nm.
Antimicrobial Activity
After performing the well diffusion assay, the results demonstrated that the P. harmala extracts exhibited notable antimicrobial activity towards three bacterial strains (Figure 2): the gram-negative like E. coli, as well as the gram positive like S. aureus and Streptococcus (as shown in Table 2). However, the P. harmala extracts showed one of the most notable antimicrobial activities among all the samples tested. The E. coli, S. aureus, and Streptococcus all showed a zone of inhibition (ZOI) for the P. harmala extracts, ranging from 0.9 cm to 2 cm in diameter.
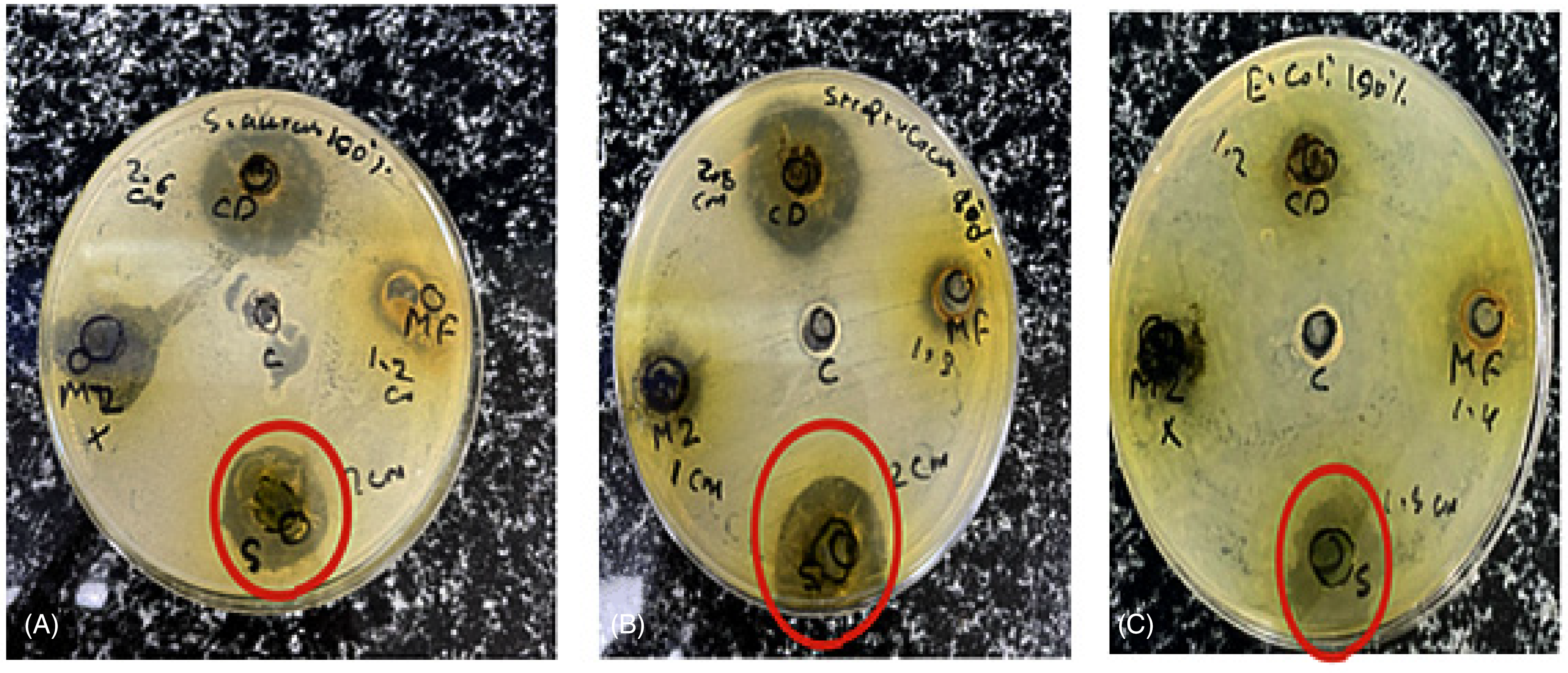
Antibacterial activity of P. harmala extracts against three bacterium types. Zone of inhibition is indicated by the red circles. (A) Staphylococcus aureus; (B) Streptococcus; (C) E.coli.
Antimicrobial Activity of the Plant Extracts Against Different Bacteria Cell Lines.

Inhibitory Effects to Cancer Cell Line
At first, the P. harmala plant extracts were tested against LX-2, (normal human hepatic stellate cells) and results showed that the extracts have no activity against these normal human cell lines. The anticancer effects of P. harmala plant extracts have been investigated on MCF7 breast as well as HT29 colon cancer cells. The MCF7 control, prepared without DMSO, exhibited all living cells 48 h later upon observation. The plate containing 300 μL of DMSO displayed dead cells, indicating DMSO toxicity. Cells in the plate containing 200 μL of DMSO were also deceased, while those in 100 μL of DMSO were partially adherent, and those in 50 μL were mostly adherent (as shown in Figure 3). These outcomes were consistent with the results obtained for the HT29 cells, which demonstrated increased survival as the concentration of DMSO decreased.

Anticancer activities of control samples of the MCF7 and HT29 cell lines 48 h after culturing with DMSO. (A) Untreated MCF7 cells; (B) MCF7 DMSO control 300μL; (C) MCF7 DMSO control 200μL; (d) MCF7 DMSO control 100μL; (E) MCF7 DMSO control 50μL; (F) Untreated HT29 cells; (G) HT29 DMSO control 150μL; (H) HT29 DMSO control 50μL. DMSO, dimethyl sulfoxide.
The MCF7 cells containing the 50% ethanol extract of the P. harmala plant, which were in 100 µL DMSO, showed complete cell death 48 h post-culture (100% cell death), while those in 50 µL DMSO showed 90% cell death (Table 3). Figure 4 shows % cell death of the plant extracts against MCF7 and HT29 cancer cell lines. The MCF7 cells containing the 100% ethanol extract of the P. harmala plant, which were in 100 µL DMSO, also showed complete cell death within 48 h (95% cell death), as did those in 50 µL DMSO (as shown in Figure 5). The HT29 cells containing the 50% of ethanol extract as well as those containing the 100% of ethanol extract in both 50 µL and 100 µL DMSO also resulted in cell death (90-95% cell death), Table 3. The fact that the decreasing concentration of DMSO did not influence the experiment and did not result in less cytotoxicity shows that most of the cell death resulted from the plant extracts. This phenomenon has been corroborated by earlier studies in which DMSO alone does not have significant effects.

Percentage cell death of the plant extracts against MCF7 (series 1) and HT29 (series 2) cancer cell lines.
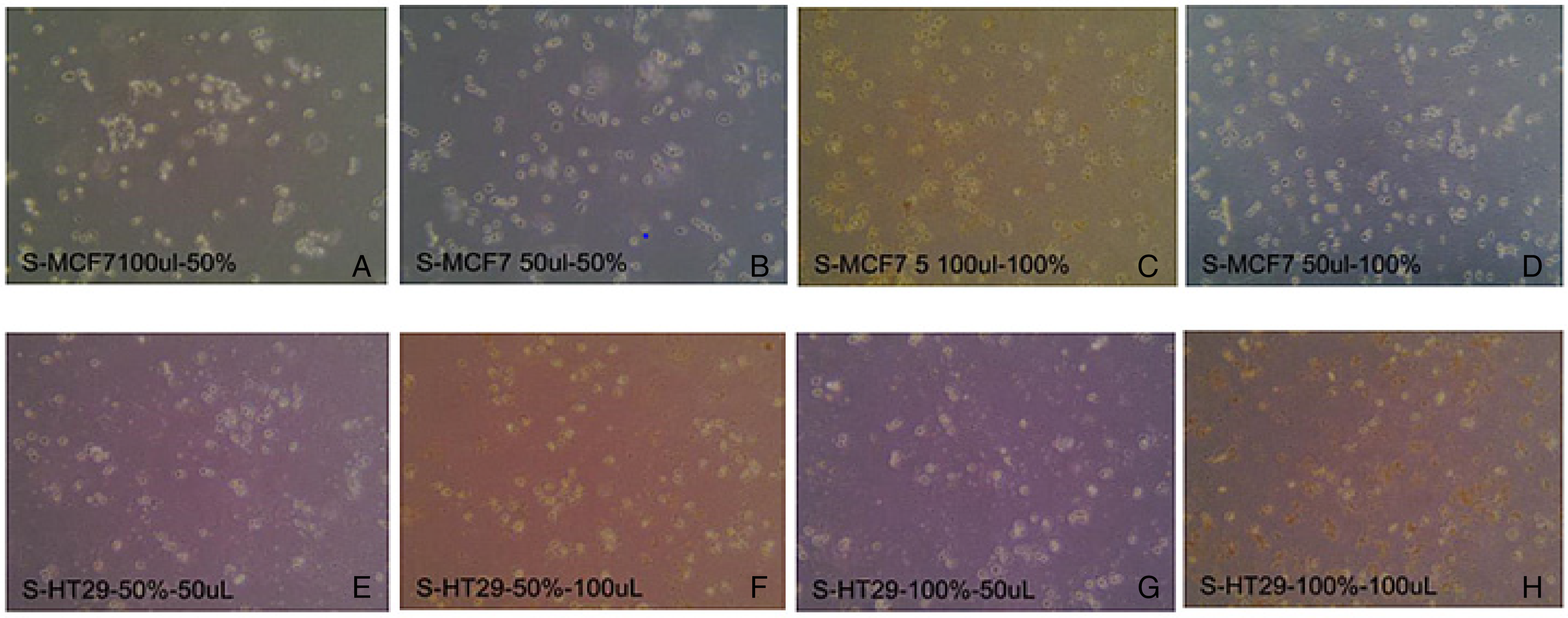
Anticancer activities of P. harmala plant extracts treated with 50μL and 100μL DMSO 48 h after culturing. (A) MCF7 cells in 100μL DMSO and 50% ethanol extract; (B) MCF7 cells in 50μL DMSO and 50% ethanol extract; (C) MCF7 cells in 100μL DMSO and 100% ethanol extract; (D) MCF7 cells in 50μL DMSO and 100% ethanol extract; (E) HT29 cells in 50μL DMSO and 50% ethanol extract; (F) HT29 cells in 100μL DMSO and 50% ethanol extract; (G) HT29 cells in 50μL DMSO and 100% ethanol extract; (H) HT29 cells in 100μL DMSO and 100% ethanol extract. DMSO, dimethyl sulfoxide.
Anticancer Activities (Percentage Cell Death and the IC50) of the Plant Extract Against MCF7 and HT29 Cancer Cell Lines.

DMSO, dimethyl sulfoxide.
Discussion
First, the plant leaves were air dried under shade at room temperature to preserve the bioactive compounds present in the leaves as temperature may affect the content of these phytochemical. Then, the dried leaves were ground to get fine powder so that extraction efficiency of phytochemicals is increased. Ethanol is a good extraction solvent of flavonoids and polyphenolic compounds where 50% and 100% ethanol were used for extraction process.17,24
The testing for total phenolic as well as flavonoid component, and antioxidant effects of the P. harmala extracts, confirmed the presence of bioactive compounds within the extracts. However, the results varied based on the concentration of ethanol utilized in the extraction process. The plant extract with the 50% ethanol mixture yielded higher results than the 100% ethanol extract. This could be due to the water increasing the extraction efficiency or solubilizing activity. These findings are consistent with a previous study that found the methanolic plant extract to have the greatest overall phenolic content (371.4 mg GAE/g) as well as flavonoid content (1.3 mg QE/g), followed by DCM as well as hydro-alcoholic plant extracts.28,29
The phenolic and flavonoid composition of the extracts was reflective of the antioxidant capabilities of the P. harmala plant. The results were promising, as the antioxidant effects of the plant were evaluated to be quite high, suggesting that it has potential as a candidate for the development of antioxidant drugs.25,29
The antimicrobial activity testing using ethanol extracts of the P. harmala plant was successful, as all three bacterial strains tested, including the gram-positive S. aureus and Streptococcus, as well as the gram-negative E. coli, showed zones of inhibition upon culturing with the P. harmala extracts. 29 The fact that the plant extracts were efficient against both gram-positive and gram-negative bacteria is particularly intriguing due to the notorious resistance of gram-negative bacteria resulting from differences in their cell walls compared to those of gram-positive bacteria. The resulting ZOIs of the P. harmala wells had notable diameters ranging from 0.9 cm to 2 cm. P. harmala is one of the few plants that is able to yield such effects against gram-negative bacteria, and some studies have shown that its effectiveness towards gram-negative bacteria is greater than that with gram-positive bacteria.
The inhibitory effects to cancer cell line activity of P. harmala were as hypothesized, as the 50% and 100% ethanol extracts of the plant showed activity with the MCF7 breast as well as the HT29 colon cancer cell lines, resulting in cell death in all samples. The death percentage ranged from 90% to 100% within 48 h of culturing. The differences in color and turbidity observed in the figures corresponding to the anticancer results were due to differing concentrations of ethanol in the extracts. It is important to note that these results are based on in vitro testing, and that the in vivo results may differ depending on the interaction of the extracts with the internal conditions of the organism in question.
The choice of 48-h incubation period in anticancer activity studies is often based on several factors, including the growth kinetics of cancer cells, the need to allow sufficient time for the tested compounds to exert their effects, and practical considerations for experimental design. Cancer cells typically have a faster proliferation rate compared to normal cells. A 48-h incubation period allows for multiple cell cycles to occur, ensuring that a significant number of cells are actively dividing and thus susceptible to the effects of the tested compounds. 37 A 48-h incubation period provides a relatively short-term exposure to the tested compounds, allowing researchers to assess their immediate effects on cancer cell viability, proliferation, apoptosis, or other relevant endpoints. 38 Longer incubation periods may not be feasible due to limitations in cell culture maintenance, resource availability, or experimental duration. A 48-h timeframe strikes a balance between allowing sufficient time for cellular responses to occur and keeping experimental timelines manageable. 39 Many published studies investigating the anticancer activity of compounds have used a 48-h incubation period. Maintaining consistency with established protocols facilitates the comparison of results across different studies.37-40
P. harmala is rich in various bioactive constituents eg, alkaloids, coumarins, flavonoids, and tannins, with different pharmacological activities. There are many traditional uses of P. harmala eg, psychoactive properties in spiritual rituals. Also, it has many therapeutic effects, including analgesic, anti-inflammatory, and anti-parasitic properties. However, it is important to note that the utilization of P. harmala should be approached cautiously due to its psychoactive nature and potential toxicity. A comprehensive understanding of its active constituents and traditional uses provides a basis for exploring its potential applications in contemporary medicine. 29
Research on P. harmala plant extract has revealed correlations between its phytochemical constituents analyzed from HPLC and GC, and various biological activities. Harmine and harmaline have shown cytotoxic effects against various cancer cell lines, including breast, prostate, and colon cancer cells. They induce cell cycle arrest and apoptosis in cancer cells. Harmine and harmaline exhibit antioxidant activity by scavenging free radicals and reducing oxidative stress. 41 β-carboline alkaloids possess antimicrobial properties against bacteria, fungi, and parasites. 42 Flavonoids exhibit anti-inflammatory effects by inhibiting inflammatory mediators and pathways. These compounds scavenge free radicals and protect cells from oxidative damage. 43
While the study on the inhibitory effects of P. harmala on cancer cell lines, as well as its antimicrobial and antioxidant activities, provides valuable insights, it has certain limitations. Firstly, the scope of the research may be limited by the specific cancer cell lines used for assessment. Different cell lines may exhibit varying responses, and the findings might not fully represent the complexity of in vivo conditions. Additionally, the antimicrobial and antioxidant activities were likely evaluated in vitro, and translating these findings to actual clinical applications requires further investigation in more complex biological systems. Furthermore, the study may benefit from a more extensive exploration of the specific bioactive compounds responsible for the observed effects. Finally, considering the multifaceted nature of P. harmala, future studies might explore potential side effects or toxicity profiles associated with its use. Despite these limitations, the study contributes valuable insights into the potential therapeutic applications of P. harmala in cancer treatment and antimicrobial and antioxidant interventions.
Conclusion
P. harmala, a plant with an extensive history of traditional use, exhibits a diverse range of medicinal properties. These encompass anti-tumor, angiogenesis inhibition, anti-inflammatory, anti-parasitic, antibacterial, antioxidant, anthelmintic, analgesic, hallucinogenic, antipyretic, emetic, antispasmodic, narcotic, nauseant, uterine stimulant effects, and diuretic, among others. This investigation concluded that P. harmala as a promising source of antioxidants due to its rich phenolic and flavonoid composition. Moreover, it demonstrates the plant's efficacy as an antimicrobial agent, displaying effects against both gram-positive bacteria such as S. aureus and Streptococcus, and gram-negative bacteria like E. coli. Finally, it was found to be a successful anticancer agent due to the observed cell death in the MCF7 breast as well as HT29 colon cancer cell lines. P. harmala should be further studied and integrated into pharmaceutical formulations to better understand and fully benefit from its vast medical capabilities.
Footnotes
Acknowledgements
The authors would like to extend their sincere gratitude and acknowledge the unyielding support of the Deanship of Scientific Research at their respective universities throughout this research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable to this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
