Abstract
Chemical investigation of the fern Pityrogramma trifoliata (L.) R. M. Tryon (Pteridaceae) yielded a new complex flavonoid, here named trifoliatol, and 4 known complex flavonoids in addition to known chalcones and flavanones. The absolute configuration of the complex flavonoids is proposed, based on circular dichroism (ECD), crystal x-ray diffraction, and [α]D measurements. White and yellow farinas, as well as 10 flavonoids were tested for antifungal activity against a panel of clinically important fungi, including yeast and dermatophytes. Both farinas and 2 of the flavonoids were notably active against Trichophyton rubrum and T. mentagrophytes (MICs and MFCs between 7.8 and 31.2 μg/mL). The localization of flavonoids in the plant trichomes was accomplished by use of scanning electron microscopy and histochemical methods.
Introduction
Pityrogramma (Pteridaceae) is a fern genus distributed in humid regions of North and South America. Some species exhibit farinose exudates “farina” on the underside frond surface. White and yellow exudates are mainly constituted by mixtures of simple and complex flavonoids. 1
P trifoliata (L.) R. M. Tryon is a tropical American fern 2 distributed from Mexico to South America from sea level up to 2100 masl. It grows in roadside ditches, river banks and open fields, fully exposed to sunlight. The plant resists long periods of desiccation and shows no sign of predatory activity. 3 Previously, in Argentina, this species had been taxonomically described under the genus Trismeria, 4 widely spread in subtropical regions of the country.
P. trifoliata produces both gold and white farina, and the color is related to the chemical structure of the farina's flavonoids. 5 Ethnomedical uses for eye infections have been reported in Peru for this species. 6
Fungal infections in humans are increasing considerably, particularly those involving skin and mucosal surfaces, with dermatophytes and species of Candida being the most frequent pathogens. 7 Although there are several medicines for the treatment of mycoses, 8 side effects have been detected during treatments and quick development of resistance occurs due, in part, to intensive prophylactic use. 9 There is an urgent need for new antifungal agents with natural metabolites being promising alternatives. 10 Species of Pityrogramma are used for different ethnomedical purposes, particularly as topical antifungals and leishmanicides. 11
As part of our ongoing natural product investigation of Argentine ferns, 12 the isolation and structural characterization of 1 new and 4 known complex flavonoids, together with other common flavonoids from the white and yellow exudates of P trifoliata, are described herein. The absolute configuration of the complex flavonoids is proposed, based on circular dichroism (ECD), crystal x-ray diffraction, and [α]D measurements. The localization of flavonoids in the plant trichomes was accomplished by use of scanning electron microscopy and histochemical methods. The antifungal activity was determined of the white and yellow farina, as well as that of 10 flavonoids from P. trifoliata.
Results and Discussion
Structure Elucidation
Fresh fronds containing white and yellow farinas were rinsed separately with EtOAc and the farinas were completely dissolved. After solvent removal under reduced pressure, both white and yellow residues were separately processed by chromatographic methods (a residue portion was reserved for antifungal activity assays). Silica gel chromatography, followed by HPLC on normal and reverse phase of the yellow farina yielded the new complex flavonoid, here named trifoliatol (
Trifoliatol (

Structures of compounds
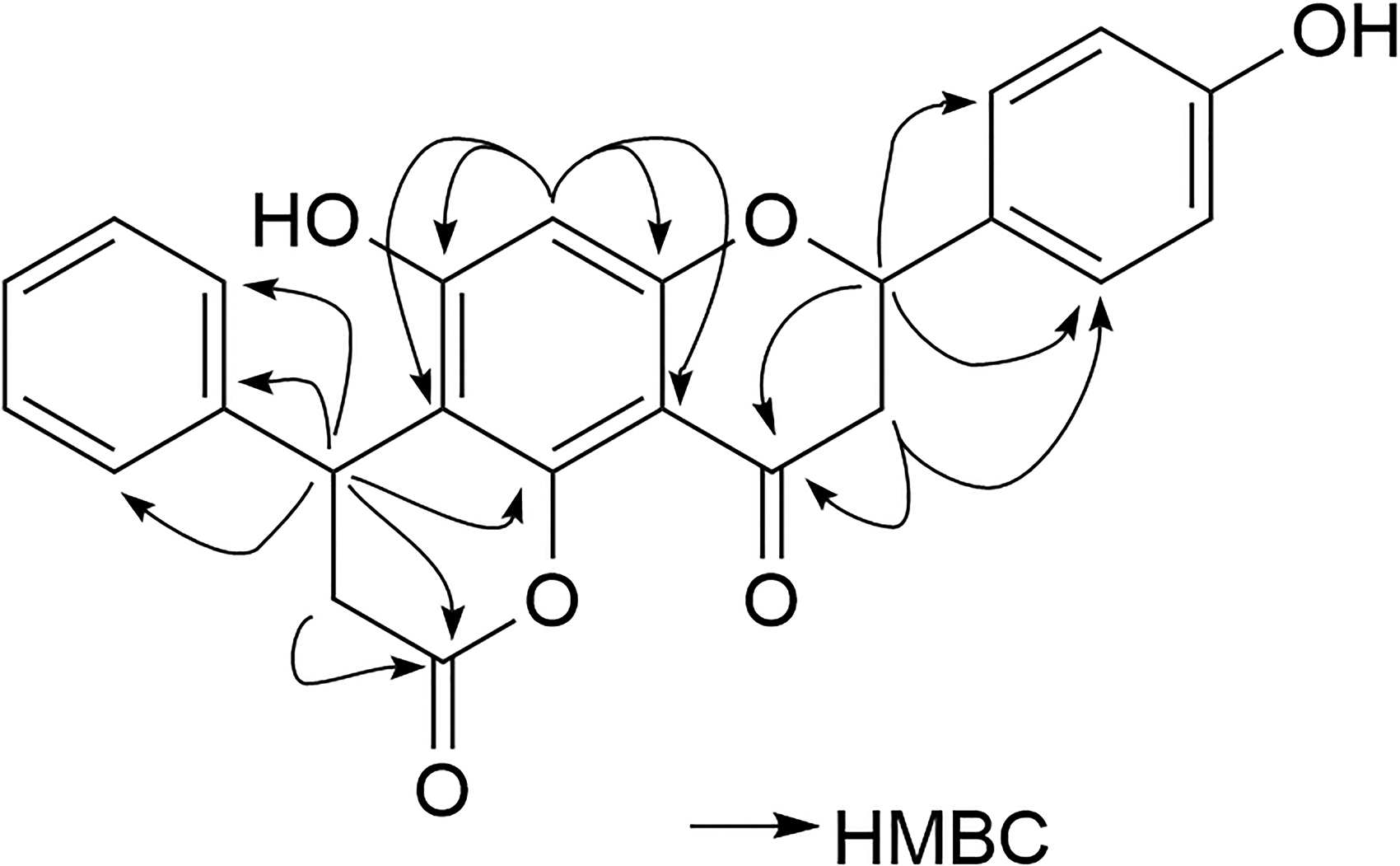
The key HMBC (arrows) data of
1H-, 13C-, and HMBC NMR Data for Compound
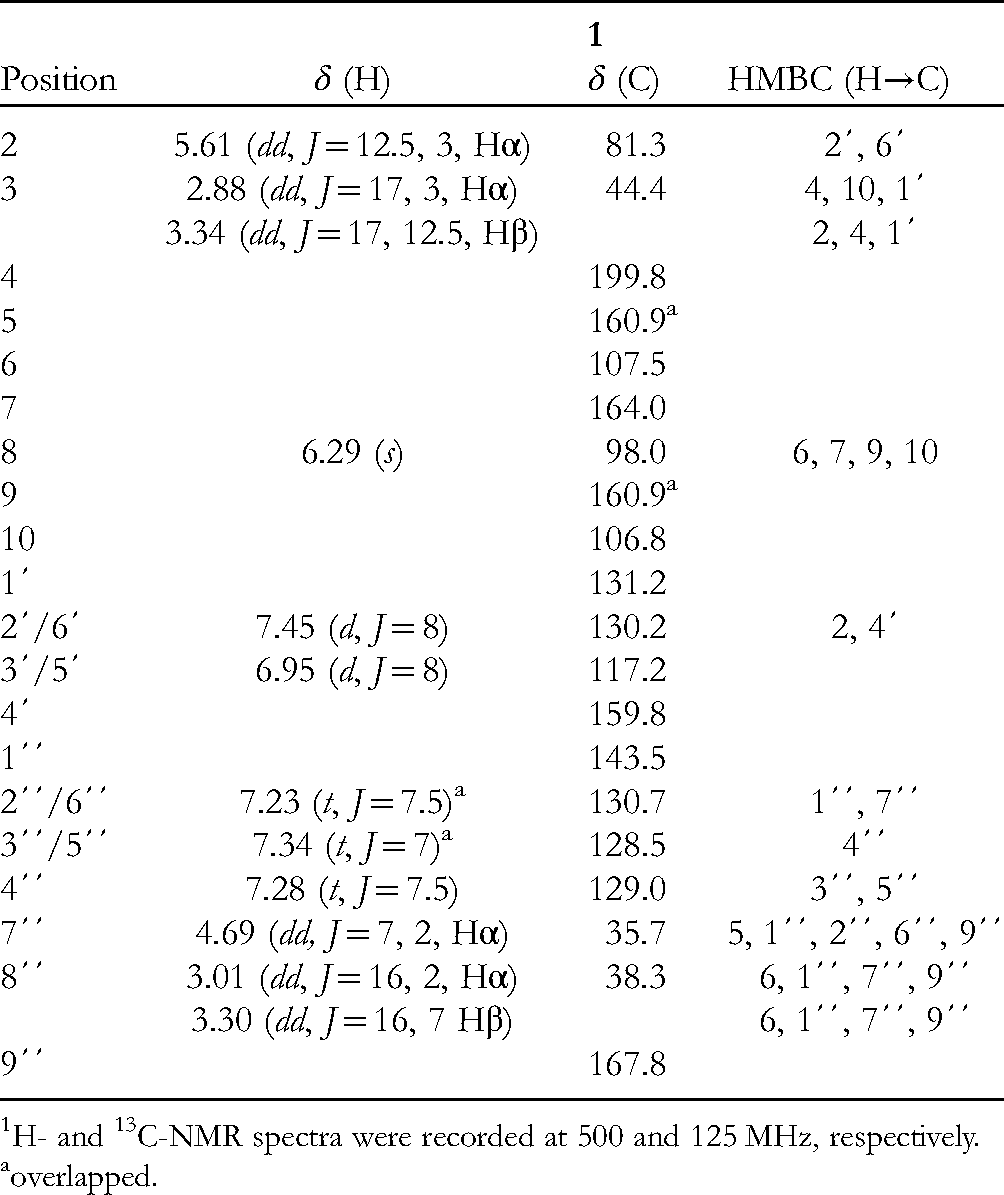
H- and 13C-NMR spectra were recorded at 500 and 125 MHz, respectively.
overlapped.
In order to determine the absolute configuration of the 2 chiral centers of compound
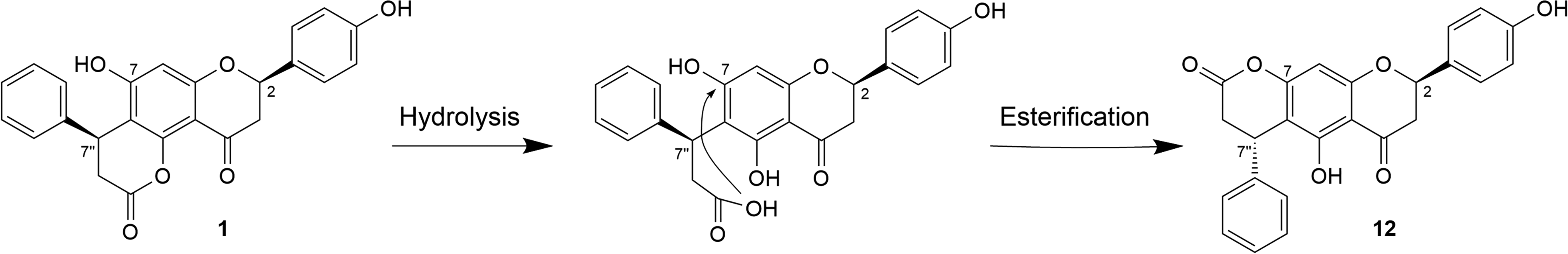
Hydrolysis followed by esterification of

ORTEP diagram for compound
The circular dichroism (CD) spectrum of compound

Experimental ECD spectrum of
The absolute configuration of the only chiral carbon of compound
Antifungal Activity
The yellow and white farina, as well as compounds
Antifungal Activities of Pityrogramma trifoliata Farinas and Isolated Compounds Against a Panel of Human Opportunistic and Pathogenic Fungi.

Abbreviations: MIC, minimun inhibitory concentration (µg/mL); MFC, minimum fungicidal concentration (µg/mL); Mg, Microsporum gypseum C 115-2000; Tr, Trichophyton rubrum C113-2000; Tm, Trichophyton mentagrophytes ATCC 9972; Ca, Candida albicans ATCC 10231; Cn, Cryptococcus neoformans ATCC 32264; ATCC, American Type Culture Collection; C, CEREMIC; Amph B, Amphotericin B; YFE, Yellow Farina Extract; WFE, White Farina Extract; I, inactive (MIC or MFC > 250 µg/mL).
Histochemical Localization of Flavonoids in the Fern
Farinose exudates of ferns of the big family Pteridaceae are considered to play an important role in plant protection.25, 26 In P. trifoliata fronds, farina (Figure 6) is secreted by glandular trichomes located in the abaxial surface, associated with the development of sporangia (Figure 7A). This is the reason why only fertile pinnae have farina, as previously reported for Onychium siliculosum (Desv.) C. Chr. (Pteridaceae). 25

Pityrogramma trifoliata frond abaxial view (A, B). Aspect of the abaxial frond with farina and sporangia covering the surface, observed in a stereoscopic microscope (C,D).

Scanning electron micrographs showing distribution pattern and trichome types in Pityrogramma trifoliata. (A): Frond abaxial surface of P trifoliata yellow farina and (B): white farina. (C): Glandular trichome with a long 2 celled stalk. (D): Glandular trichome with a single cell basal short stalk. s: Sporangia; et: Eglandular trichomes; gt: Glandular trichomes. Scale bar (A, B) = 100 μm; (C, D) = 10 μm.
P trifoliata shows 3 types of trichomes, 1 eglandular (100-500 μm) (Figure 7B), a glandular trichome with a single cell basal short stalk and a spherical secreting head (60-70 μm) (Figure 7C), and a glandular trichome with a long 2-celled stalk and oval head (150-240 μm) (Figure 7D). The gland surface is smooth in both types of glandular trichomes, due to the detachment of the cuticle from the gland cell wall. It forms a small subcuticular chamber where secretory substances accumulate before their release. 26
Histochemical tests were useful to localize in situ the flavonoids present in trichome secretions. Results show (Figure 8) that in plants with yellow farina both glandular trichome types contain flavonoids. The fluorochromes for flavonoid detection, such as the Benedict reagent (Figure 8A) and aluminum trichloride (Figure 8B) induced an intense green and light yellow fluorescence in the whole secreting head of glandular trichomes. Staining with vanillin/HCl, cherry-red vanillin-flavonoid condensates were formed immediately in both the head and neck cells (Figure 8C). In plants with white farina only, long glandular trichomes secrete flavonoids. With Benedict reagent, these trichomes showed intense yellow fluorescence in the head and neck cells (Figure 8D), while with aluminum trichloride they gave florescence mainly in the head cell (Figure 8E). Red drops were observed in head and neck cells and in the stalk when vanillin/HCl was applied (Figure 8F). Similar results were reported for Notholaena sulphurea. 26

Micrographs of glandular trichomes (A-F). Fluorescence micrographs under UV light (A, B, D, E) showing the response of glandular trichomes of Pityrogramma trifoliata fronds with yellow farina (A-C) and with white farina (D-E) to histochemical tests. (A, D) Fluorescence with Benedict reagent for flavonoids. (B, E) Intense green fluorescence of the head cell stained by aluminum trichloride. Light micrographs (C, F) Vainillin/HCl test showing cherry-red staining of flavonoids.
Conclusions
One new and 10 known flavonoids have been isolated from an Argentinian collection of P. trifoliata. The absolute stereochemistry for all quiral centers of the isolated complex flavonoids is proposed. Antifungal activities of white and yellow farinas, and those of 10 flavonoids isolated from them, were determined against a panel of dermatophytes and yeasts. Farinas and some flavonoids displayed antifungal effects with MIC and MFC values lower than 20 µg/mL. The activity is relevant when compared with other natural compounds cited in the literature. Histochemical studies demonstrated that flavonoids accumulate in glandular trichomes located in the abaxial surface of fronds and play a defensive role in the plant. Complex flavonoids are solid compounds which have been isolated from many other species of ferns, as shown in the literature cited, though they have not been investigated by x-ray crystallographic diffraction methods to confirm their structures and stereochemistry. Determination of ECD spectra has not been reported for them either, and, therefore, no absolute configurations have been proposed using this method. Based on the mentioned aspects, we consider that the present investigations open new perspectives in these fields of research.
Experimental
Plant Material
Fronds of P trifoliata with white and yellow farina were collected separately in Tucumán, Argentina, in March 2011 (GPS data: 902 masl; 27°08′39′′ South; 65°45′10′′ West). The species has been identified by Dr Marcela A. Hernández and a voucher specimen (LIL 612885 yellow and 612863 white) was deposited at the Herbarium of the Fundación Miguel Lillo, Tucumán, Argentina.
Extraction and Isolation
Fresh fronds with yellow farina (980 g) were rinsed with EtOAc and the extract was then filtered and evaporated to dryness (6.93 g). Part of the extract (4 g) was subjected to CC (SiO2; hexane and increasing amounts of EtOAc) to afford 12 fractions. Fr. 2 (107.5 mg) was chromatographed by HPLC (SiO2; hexane/EtOAc/HAc 88:12:1, 2.0 mL/min) to yield 50.5 mg of
Structural Characterization of Trifoliatol
General Experimental Procedures
Thin layer chromatography: pre-coated silica gel plates (Kiesegel 60 F254; Merck); visualized by UV light (254 and 365 nm) and by spraying with Godin reagent followed by heating at 120 °C. Column chromatography (CC): silica gel (230-400 mesh). HPLC: Knauer System, pump S1000 and RI detector S-2400, Luna C8 and C18 columns (10 × 250 mm i.d., 5 µm) and Develosil 60-5 column (10 × 250 mm i.d., 5 µm ChemcoPak). IR spectra: Bruker Vektor 22 FTIR spectrometer in an ATR mode. NMR spectra: Bruker Avance 500 spectrometer at 500 MHz (1H) and 125.6 MHz (13C) in acetone-d6; δ in ppm with the solvent signal as internal reference, J in Hz. High-resolution mass spectra were measured using a Finnigan MAT 95 spectrometer. ECD spectra were registered on a JASCO J-815 spectropolarimeter, using EtOH as solvent.
Single-crystal x-ray Diffraction
Single crystals of
Pd/C-Catalyzed Chemoselective Hydrogenation
Compound
Antifungal Activity Assays
For the antifungal evaluation, strains from the American Type Culture Collection (ATCC) and from Centro de Referencia en Micología, CEREMIC (C), were used: Candida albicans ATCC10231, Cryptococcus neoformans ATCC32264, Trichophyton rubrum C110, Trichophyton mentagrophytes ATCC9972, and Microsporum gypseum C115. Strains were grown on Sabouraud-Chloramphenicol-Agar slants for 48 h at 30 °C, maintained on slopes of Sabouraud-Dextrose-Agar (SDA, Oxoid) and sub-cultured every 15 days to prevent pleomorphic transformations. Inocula of cell or spore suspensions were obtained and quantified following reported procedures (CLSI).23, 24 Amphotericin B (Sigma), Terbinafine (Novartis), and Ketoconazole (Sigma) were used as positive controls. MICs and MFCs were determined as previously described. 31 MIC represents fungistatic activity of the sample tested and MFC indicates its fungicide capacity.
Scanning Electron Microscopy and Histochemical Assays
Measurements of trichomes were calculated for n = 100 of each type of trichome. Electron microscopy studies were performed as previously described. 26
For histochemical assays, fresh frond material was free hand sectioned and stained using the following reagents for flavonoids: Benedict reagent, aluminum trichloride, and Vanillin/HCl. 32 Observations were made as previously reported. 26
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221114755 - Supplemental material for Complex Flavonoids of Pityrogramma trifoliata: Absolute Configuration, Antifungal Effects, and Localization in the Plant
Supplemental material, sj-docx-1-npx-10.1177_1934578X221114755 for Complex Flavonoids of Pityrogramma trifoliata: Absolute Configuration, Antifungal Effects, and Localization in the Plant by Ana M. Rodriguez, Marcos Derita, Elena Cartagena, Marcela A. Hernández and Alicia Bardón in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
