Abstract
Humans are often exposed to a variety of toxic substances (including some drugs, herbs, environmental pollutants); all these heterologous substances can result in metabolic changes in organism. Clarifying their metabolic details will help to understand the beneficial and toxic effects of those heterologous substances. Traditional toxicology research tends to focus on toxic doses and time rather than on mechanisms and detoxification methods. Compared with other omics methods, metabolomics has unique, dynamic, and phenotypic characteristics. Similarly, metabolomics can link the interaction between genes and the environment; it represents both the downstream output of the genome and the upstream input of the environment, and reflects the interactions between biological system and environmental stimulation. It highlighting not only biological mechanisms but also closely related to the phenotype of biological events, which is widely employed in toxicology studies presently. Therefore, strengthening toxicological research based on metabolomics can help better understand the mechanisms of action and toxicological effects of toxic substances. Thus, present review systematically summarized and discussed the current application status of metabolomics approaches in toxicology, which might contribute to provide a deeper understanding of the processes and mechanisms of toxicity caused by related chemicals and herbal medicines, thereby reducing the occurrence of damage and providing technical reference for toxicity research.
Introduction
Toxicology research patterns are currently changing, and traditional toxicity testing and evaluation rely extremely on the observations of pathological tissues, many of which predict 50% of the lethal concentration (LC50) and a 50% lethal dose (LD50) of an external substance on animal carriers.1,2 Although this is a necessary step in toxicity evaluation and the first line of defense for rapid identification of acutely toxic chemicals, while the sources and processes of adverse reactions, as well as predicting the occurrence of such phenomena in advance, are also important aspects of toxicology research. It is best to determine the sub-accumulation dose of interference factors and related biological information. 3 Also, the transient characteristics of new psychoactive substances (NPSs) that cannot be directly detected and the uncertainties of the huge internal components of herbal medicines (HMs) and their natural compounds are problems that traditional toxicology cannot solve temporarily.4–7
Metabolomics research strategies may be flexible and sufficient to deal with the above thorny problems. Compared with traditional toxicology research methods, metabolomics can reveal the essence and laws of life phenomena at a holistic level with more holistic and systematic. Its high-throughput screening characteristics and highly sensitive detection capabilities make it great advantages in the study of toxic pathways and mechanisms of action. Moreover, metabolomics has become increasingly important in toxicological studies because it represents a collection of intermediates and end products of cellular processes and is the reporter closest to biological exposure to environmental and pathological processes (Figure 1).8–10 By analyzing the metabolites profiling in poisoning biological fluids/tissues that interact with toxic substances, as well as the variation patterns with exposure time and dose of poisoning, the target organs/tissues, toxic processes, and biomarkers of toxic effects can be determined. Thus, conducting toxicological mechanism research or toxicity evaluation, metabolomics has become an indispensable tool in toxicology research. 11 In short, future toxicology research may develop into predictive science based on biological observations of mechanism.

Genomics, transcriptomics, proteomics, and metabolomics are important components of systems biology, and metabolomics is the closest to phenotype.
In view of the advantages of metabolomics and the future trends of toxicological research, this article reviews the application of metabolomics in toxicology, including metabolomics techniques and methods, the application of metabolomics in evaluating adverse drug reactions (ADRs), elucidating toxic mechanisms, predicting drug addiction, and monitoring chemical residues and pollution.
Analytical Techniques in Metabolomics
GC-MS
GC-MS is a reproducible analysis method, especially for the detection of thermally stable volatile compounds that require stability and time-consuming sample preparation. Some highly polar substances can be derivatized to increase their volatility to obtain the requirements of GC analysis, but the process will be more complicated and may introduce errors to quantitative analysis.12–14 Even so, GC-MS still plays an important role in toxicology-related research.15,16 Joanne Taylor et al conducted a GC-MS study of e-cigarette or vaping, product use-associated lung injury (EVALI): 17 12 patients had been exposed to 46 e-cigarette or vaping products (EVPs) containing tetrahydrocannabinol, and Vitamin E acetate has been detected in lung fluid samples. When more vitamin E acetate is inhaled continuously, phosphatidylcholines could transition from gel to liquid crystalline phase, which will cause a lose of the ability to maintain surface tension that is necessary to maintain lung respiration. 18 Therefore, vitamin E acetate may cause EVALI by causing respiratory disorders, but the research on this mechanism is not comprehensive, so health authorities, producers, and users need to be aware that vitamin E acetic acid should not be added to EVPs until the relationship between inhaled vitamin E acetate, and the lungs is better determined. In addition, with the popularity of e-cigarettes, various types of chemicals were added to improve evaporation efficiency, provide aroma, or have a dilution effect, for instance, oil-like terpenes such as squalene (SQE) and squalane (SQA). However, little was known about the inhalational toxicity of SQE and SQA, Cowan et al developed a GC-MS method for detecting the content of SQE and SQA in EVPs, and applied the method to measure SQE and SQA in 153 EVALI case-related products. 19 Among them, SQA was detected in four samples and SQE was detected in 71 samples, indicating the universality of SQE in EVPs. However, the health effects of long-term inhalation of SQE are still unclear. Therefore, establishing sensitive and stable detection methods is the main means of monitoring the above additives, and GC-MS technology provides strong support in toxicity research.
LC-MS
LC-MS can be used to analyze biochemical, organic, and inorganic compounds commonly found in complex samples of environmental and biological origin, and widely used in biotechnology, pharmaceutical industry, safety, and effectiveness research.20,21 Recently, it was reported that chondroitin/dermosin sulfate derived from the xyloside of human breast fibroblast cell line Human breast ductal carcinoma 70 (HCC70) was cytotoxic. 22 However, the structure of xyloside-primed chondroitin/dermatan sulfate was different from the previously discovered chondroitin/dermatan sulfate. In order to investigate the structural requirements for the cytotoxic effect, a novel LC-MS/MS approach was developed to characterize the chondroitin/dermosin sulfate, which is essential to determine the structure-function relationship between chondroitin/dermosin sulfate and the biomolecules they interact with and to understand the biosynthesis of chondroitin/dermosin sulfate. Based on the reversed phase dibutylamine ion-pairing chromatography and negative mode higher energy collision dissociation method, the linkage regions, internal oligosaccharides, and nonreducing ends can be characterized, not only revealed differences from previously discovered chondroitin/dermatan sulfate, but also revealed the undescribed methylation and sulfation before. This result further understands the chemical structure of cytotoxic chondroitin/dermosin sulfate produced by xylosides-induced HCC70 cells and proves the applicability of the LC-MS/MS method for structural characterization of xyloside-primed chondroitin/dermatan sulfate.
CE-MS
Capillary electrophoresis (CE) uses high voltages to generate an electroosmotic flow to separate ions, and the analyte migrates from one end of the capillary to the other according to its charge, viscosity, and size. It provides high separation efficiency with high resolution and sensitivity, relatively small volume, and high-speed analysis. Thus, capillary electrophoresis mass spectrometry (CE-MS) has higher column efficiency than GC and LC. 23 It is especially suitable for charged ions or polar drugs. Atsuko Miyagi applied CE-MS to explore the response of ion beam to oxalate metabolism in R. obtusifolius of Rumex species, of which the soluble oxalate in Rumex species causes severe kidney damage. Ion beam is ionizing radiations that have high linear energy transfer, and can cause mutational variation and novel mutants. 24 Irradiated R. obtusifolius seeds with ion beams might be an effective way to develop and reduce oxalate accumulation. The results showed that the ion beam fluctuates the carbon flux of the isocitrate pathway and indirectly increases the oxalate content. 25 The results of this study suggest that the isocitrate metabolic pathway can be controlled to reduce the production of oxalate, which is beneficial to protect the kidney injury response. Although CE-MS is currently less used in metabolomics research, its advantages should not be ignored and deserve further development and utilize.
MSI
MSI is a technique for rapid analysis of the compound composition, spatial distribution, and relative abundance in samples.26,27 Multiple ionization methods can be employed for MSI, the diversity of ion sources, unique advantages, and broad application prospects further promote MSI widespread application in the field of toxicology research.
Matrix-assisted laser desorption ionization is currently a popular and highly commercialized ion source. Tissue slices were sprayed with appropriate matrix, and laser beam irradiated the tissue surface and triggered ablation and desorption of the molecules. The matrix molecules absorbed laser energy and transferred to the analyte, and then ionized and entered the mass analyzer for detection.28,29 Although pesticides play an important role in the agricultural industry, they are also the important component of organic pollutants. Some pesticides can even enter the aquatic environment through surface runoff or rainwater erosion, thereby affecting human health. 30 Ma et al employed MSI method to reveal the chronic toxicity of indoxacarb on the liver of adult zebrafish, six representative ions showcasing the tissues and organs of zebrafish, including the eyes, swim bladder, intestine, brain, gills, and liver. 7 Moreover, MSI revealed the spatial distribution of key metabolites in the liver of zebrafish that affect the tricarboxylic acid (TCA) cycle and amino acid metabolism pathways by indoxacarb, including lysoPC (18:1) was mainly located in the eyes and gills, whereas diglyceride (DG) (34:2), DG(36:4), DG(36:2), triglyceride (TG)(48:1), TG(48:2), and TG (50:2) were distributed in the body of zebrafish, which provided new insights for the assessment of the impact of pesticides on aquatic environments.
Desorption electrospray ionization (DESI) is an ambient ionization technique with no vacuum requirements, no matrix and little to no sample preparation. A charged solvent spray was applied to impact the tissue surface and generated ions from sample and the extracted analytes were transferred to MS inlet and accelerated into the mass analyzer for detection. 31 Perez conducted a metabolomics experiment based on DESI-MSI to detect the toxic effects of the surfactant AMMOENG 130 in personal care products on zebrafish. 32 After scanning the whole zebrafish, AMMOENG 130 can penetrate the blood-brain barrier and cause neurotoxicity. This environmental ionization MS technology provides broad prospects for directly analyzing biological tissues to monitor toxic and persistent environmental pollutants in aquatic organisms.
Secondary ion mass spectrometry (SIMS) employed a finely focused primary ion beam to bombard the surface of the sample and generated characteristic secondary ions from sample surface, and subsequently entered the quality analyzer for analyzing. 33 CCl4 is a common chemical substance that causes liver damage, and Otrubova et al utilized biochemical, histopathological analyze, and SIMS to characterize the liver tissue in CCl4-induced liver cirrhosis rats. The damaged liver tissue can be distinguished according to the difference of lipid contents, among them, the content of linoleic acid, oleic acid, and cholesterol increases exponentially, especially diacylglycerol, which indicating that it can serve as an indicator of CCl4-induced liver injury. 34
NMR
NMR-based technology has been successfully used to the study of identification and screening toxic biomarkers, and disease mechanisms. In particular, it is not only non-destructive, but also provides comprehensive metabolic information and is highly reproducible. 35 The safety of NMR-based metabolomics assessment has increasingly attracted the attention of researchers. Metalaxyl and metalaxyl-M are widely applied fungicides, and there are few reports on their subacute toxicity studies. An 1H-NMR-based untargeted metabolomics method reveals the toxic metabolic mechanism of the above pesticides, and the study found that the TCA cycle and urea cycle are the main metabolic pathways affected by toxic reactions; moreover, the urea cycle is the key metabolic pathway for detoxification. 36 This case reflects the important role of metabolomics in assessing the risk of pesticide residues and corresponding detoxification effects. Tris (2-chloroethyl) phosphate (TCEP) is one of the most commonly detected organic pollutants, and it has shown that TCEP is an endocrine disruptor and carcinogen. 37 Yang et al used an NMR-based metabolomics technology to evaluate changes in brain neurochemistry in female rats after 60 days of exposure to TCEP, and it was displayed TCEP exposure mainly interfered with amino acid and neurotransmitter metabolism, energy metabolism, and cell membrane function integrity by changing the concentrations of glutamate, γ-aminobutyric acid, N-acetyl-D-aspartate, creatine, and lactic acid metabolites. 38 Besides, taurine, myoinositol, creatine, and choline metabolites might be a neuroprotection mechanism to prevent the neurotoxicity induced by TCEP, which broadened the understanding of TCEP neurotoxicity and prevention.
Taken together, there is no single analytical tool to accurately identify and quantify thousands of target small molecules, choosing the most appropriate analytical tool is usually a compromise between sensitivity and selectivity. NMR has good selectivity but the sensitivity is relatively low and is limited to the analysis of concentrated metabolites. GC-MS is more sensitive than NMR but less selective. The analysis of certain nonvolatile/polar metabolites requires additional chemical derivation steps. Besides, due to differences in the efficiency of metabolite derivatization, the overall reproducibility of the analysis might be affected. 39 LC-MS also provides high sensitivity and selectivity, which can be used to analyze non-polar and polar metabolites. Moreover, like GC-MS, LC-MS can also use derivatization to improve the properties of compounds for analysis. 40 By improving the CE interface, the performance of CE-MS can be significantly improved, and CE-MS can also be used for single-cell metabolomics research. 41 However, the flow rate, capillary diameters, capillary coating and electric field strength should be considered to obtain stable spray and maximum ionization efficiency. Although MSI can display in situ information, in the low molecular weight range, ion suppression phenomenon still needs to be addressed and new matrix need to be developed. In addition, the identification of isomers is also a challenge for MSI. Therefore, a combination of different analytical techniques is needed to better cover metabolites with a wide range of polarity and molecular weight.42–44
Metabolomics Approaches
Untargeted Metabolomics
The main outstanding feature of untargeted metabolomics is the discovery of novel metabolites related to the research background. It provides better methods for detecting new metabolites through comprehensive and unbiased analysis of global metabolites, and untargeted analysis is considered the most promising resource for identifying prior unknown metabolites and pathways. 45 Although this methods cover a wide range of compounds, they are particularly insensitive to low abundance metabolites.46,47 Naringenin is a flavonoid compound found isolated from Citrus sinensis (var. Valencia) peels, which can improve CCl4-induced liver toxicity. 15 It can attenuate pathological changes in liver tissue by lowering glutamic-pyruvic transaminase and glutamic oxaloacetic transaminase and can also improve kidney tissue architecture. Its protective effect on the liver and kidney may be related to its regulation on fatty acids, amino acids, and energy metabolism. These pathways may also be the effective targets for regulating liver and kidney toxicity.
Targeted Metabolomics
Targeted metabolomics focuses on a set of predefined metabolites, which are typically highly correlated with the studied disease/environment, and aim towards their determination, often to the point of absolute quantification. 48 The main disadvantage is that the target metabolite must be understood before a given compound can be used as an absolute quantitative standard.49,50 Irinotecan (CPT-11) is a derivative of camptothecin (a natural product of anti-tumor drug), but its clinical application is usually limited by the induction of severe gastrointestinal (GI) toxicity (especially delayed diarrhea), and it can create disorders of bile acid metabolism. Interestingly, the Chinese medicine prescription of Huang Qin Decoction (HQD) can regulate abnormal levels of bile acids and protect the GI tract from damage. 51 Thus, HQD can be considered as a protective measure while applying CPT-11. This result also fully demonstrates the great potential of targeted metabolomics to further elaborate on the toxicity mechanism.
In any case, untargeted metabolomics has no bias and comprehensively and systematically reflects the characteristics of the life-body metabolome, but the repeatability is relatively poor and the linear range is limited; while the repeatability and sensitivity of targeted metabolomics have been significantly improved, the metabolite corroboration is simple and the linear range is wide, but a preknowledge background is required. Therefore, it is suggested that the two strategies can be used together to facilitate toxicological research.
Application of Metabolomics in Toxicology Research
Metabolic Markers and Pathway Analysis Databases and Platforms
Biomarker identification and metabolic pathway enrichment analysis are crucial parts of the metabolomics workflow (Figure 2), online and offline electronic databases are embedded in instrumental analysis software, which promotes the development of commercialization while understanding metabolic knowledge. Human metabolome database (HMDB) (https://www.hmdb.ca) is a free database initiated by the Canadian human metabolome project. 52 At present, the latest version of HMDB5.0 contains 217920 metabolites. HMDB supports multiple search methods, including compound name search, molecular weight search, molecular structure search, and experimental spectrum matching of known metabolites, which is significantly beneficial for targeted metabolomics research. However, the library currently does not support batch search, which is limited to a single metabolite search, and the search efficiency is low. 53 Even so, HMDB is still the most complete and comprehensive collection of human metabolites and human metabolism data in the world. The Kyoto Encyclopedia of Genes and Genomes (KEGG) (https://www.kegg.jp/) database project was initiated by Minoru Kanehisa, 54 a professor at the Institute of Chemistry, Kyoto University, Japan in 1995. It is one of the largest and most complete bioinformatics databases in the world. It contains some metabolomics information, which mainly focuses on metabolic pathways and integrated metabolic, genetic, and protein pathway information. 55 Researchers who identify unknown metabolites usually prefer databases such as METLIN (Scripps Center for Metabolomics) (http://enigma.lbl.gov/metlin/) and MassBank (http://www.massbank.jp/) because they contain rich spectral information of primary and secondary MS56,57 and assist in the identification of metabolites and chemical entities by publicly accessing their comprehensive MS/MS metabolite data repository. Also, Golm Metabolome Database (GMD) (http://GMD.mpimp-golm.mpg.de/) and National Institute of Standards and Technology (NIST) (https://www.nist.gov/) are widely employed in the field of plant metabolomics,58,59 but GMD is limited to GC-MS data retrieval. In a word, to benefit from this rapidly developing field, it is expected that multiple complementary databases emerge will appear, which can not only improve the identification of known metabolites but also enrich chromatographic and spectral information to accurately identify unknown compounds with low sensitivity to penetrate the subtle features of metabolic changes.

The workflow of metabolomics analysis. Including sample preparation, data collection, data preprocessing, multivariate and univariate statistical analysis, and the interpretation of biological significance.
Evaluation of Adverse Drug Reactions
The causes of ADRs are diverse and unstable, including the use of anesthetics and therapeutic drugs for diseases. Mild symptoms can be recovered by themselves, and severe conditions may damage organs or even die. Both doctors and patients should focus on them, which not only affects the quality of life of patients, confidence in the medical system and hospital stay, but also fail to identify ADR may lead to unnecessary examinations, may be adverse to patients and increase the financial burden.60,61 The metabolomics has the potential to provide early diagnosis and predict ADR biomarkers.
CPT-11 as an anticancer drug has potential toxicity to other organ tissues, which limits its clinical application. A study combined metabolomics with multivariate statistical analysis to identify liver, ileum, and jejunum as CPT-11 toxic target tissues. Also, the Krebs cycle and amino acid metabolism of target tissues were disturbed. 62 The above work suggests that metabolomics approaches can not only as powerful tools for identifying toxic target tissues but can be used to dissect specific biochemical responses to target tissues. It is believed that the anatomical workflow of target toxic tissue based on metabolomics and multivariate statistical analysis can be extended to other drug tissue toxicity studies. It is also well known that the anti-tumor drug doxorubicin is cardiotoxic at cumulative doses, increasing reactive oxygen species in the heart cells. In the absence of alternatives, people are accustomed to choosing a combination of drugs to reduce ADR. Interestingly, a metabolomics study based on 1H-NMR found that spinochrome D protects cardiomyocytes without affecting the specificity of doxorubicin in cancer cells. 63 The mechanism mainly regulates glutathione metabolism and increases adenosine triphosphate production and oxygen consumption rate.
In addition, we also listed the work on ADR with the help of metabolomics methods in recent 10 years (Table 1), which providing new insights for the occurrence and development of ADR, allowing people to predict and diagnose ADR in advance and take corresponding measures.
Summary of Metabolomic Approaches to Elucidate the Mechanism of Adverse Drug Reactions.

TCA, tricarboxylic acid; ATP, adenosine triphosphate.
Exploration of Mechanism of Toxic Effects Caused by HMs and Their Natural Products
HMs has a long history of clinical application and rich experience in the treatment and healthcare of diseases in China. They have their own diagnostic and treatment standards, and flexibly combine HMs according to the different syndrome subtypes of patients, playing an important role in the treatment of diseases.73,74 With the promotion and application of HMs, problems that must be solved are exposed. First, HMs contain complex and vast information about ingredients, how do they work therapeutically? Besides, the content of compounds is susceptible to external factors, at the same time, some components that play a therapeutic role also have a potentially toxic effect. However, HMs seems to be a treasure trove, HMs and their natural products are one of the important ways to develop new drugs, and researchers need to eliminate these risk factors, clarify the efficacy and toxic substances, minimize harmful effects, and develop and utilize more drugs from HMs that are beneficial to human medical health. The application of metabolomics in the toxicity study of HMs and their natural products should pay attention to the correlation between adverse compounds and biomarkers, develop an analytical strategy, identify the form of toxic HMs in organisms, apply high throughput and sensitive instruments to establish statistical analysis association with toxic biomarkers, and interpret the toxic forms of HMs and their natural products (Figure 3).
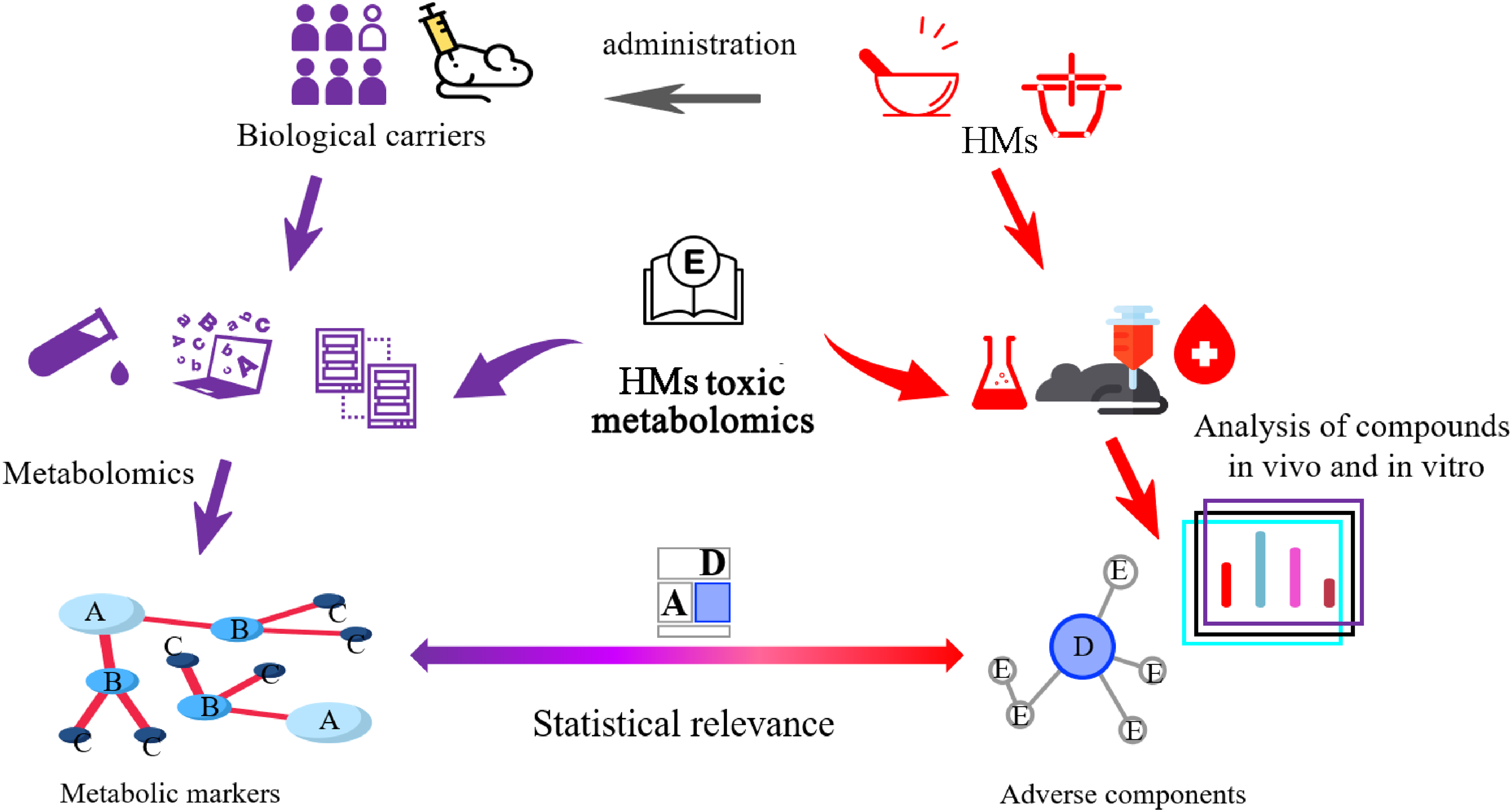
Metabolomics works in HMs toxicology, identifying toxic compounds and biomarkers, and establishing their relevance. HM, herbal medicine.
The processing and compatibility of HMs play a crucial role in ensuring the safe use of potentially toxic herbs. 75 The compatibility of HMs is the essence of prescriptions, which means that two or more HMs are mixed for use according to their properties and clinical evaluation. Compared with herbs or compounds alone, the combined herbs will have better therapeutic effects and fewer adverse effects. A study explores the mechanism of Polygoni Multiflori Radix Praeparata (PM)-induced liver injury in rat models. PM can cause pathological changes in liver tissue and metabolic background abnormalities; 76 it is worth noting that the combination of PM and Poria cocos (Schw.) Wolf at a ratio of 2:1 minimizes liver toxicity. It regulates arginine and proline metabolism, primary bile acid biosynthesis, and sphingolipid metabolism.
In addition to applying herbs in combination, the processing of herbs can also reduce toxic reactions. Pinellia ternata (Thunb.) Breit (PR) is a commonly used HM, which is reported to be cardiotoxic, while, PR treated with aluminum and freshly squeezed ginger juice can be formed to Pinelliae Rhizoma Praeparatum cum Zingibere et Alumine (PRZA), a processed product with less toxicity. Su et al conducted untargeted metabolomics to elucidate the biological mechanism. 77 The results show that abnormal levels of 10 metabolites, including proline, dihydrouracil, saccharopine, etc, inhibit mTOR signaling and activate the TGF-β signaling pathway, which contributed to PR-induced cardiotoxicity, and free radical scavenging might be responsible for the toxicity-reducing effect of PRZA. Metabolomics can contribute to predict and understand the mechanism of toxic effects caused by drugs and herbs. It is interesting that in the face of non-therapeutic effects, HMs can indirectly reduce the pain of patients. Also, more case studies of HMs-related toxicity using metabolomics in recent 10 years are summarized in Table 2.
Summary of Metabolomic Approaches to Elucidate the HM-Related Toxicity Mechanism.

HM, herbal medicine; TCA, tricarboxylic acid.
Applications of Metabolomics in Forensic Toxicology
Drug abuse is an increasingly serious problem worldwide, especially as progressively NPSs enter the drug market. The direct detection and identification of NPS is an analytical challenge due to their ephemerality on the drug scene.4,101,102 The application rate of ketamine in entertainment places ranges from 6.7% to 67.8%.103,104 There is evidence that long-term or repeated use of ketamine can cause nerve and psychological damage, and the mechanism of the effect of ketamine on the nervous system is still unclear. The untargeted metabolomics explains the effect of ketamine on the nervous system of rats. The researchers used LC-MS analysis of three brain tissues of prefrontal cortex, hippocampus, and striatum and found different metabolic markers, but they all affected purine metabolism and glycerophospholipid metabolism. Relative to the hippocampus, and the striatum area, prefrontal cortex may be the long-term location of ketamine. Heroin is one of the most harmful drugs of abuse. It can lead to addiction very quickly and cause great harm to individuals physically and psychologically. Metabolomics-based study have found that hair can serve as a suitable sample for studying heroin addiction, helpful for further understanding of heroin addiction and clinical diagnosis. 105 Besides, effects of heroin on metabolites persist after prolonged withdrawal, although majority of the metabolite changes could recover after months of withdrawal, the levels of alpha-aminobutyric acid, alloisoleucine, ketoleucine, and oxalic acid do not recover. 106 From this, it can be seen that metabolomics not only contributes to the study of the mechanisms of action of addictive drugs, but also provides a therapeutic strategy for the intervention of addictive drugs during withdrawal.
Without direct attention to NPS and its related compounds, the application of metabolomics may detect biomarker information and mechanisms related to drug abuse (Table 3), provide a new method for indirect detection of NPS, and benefit the practical work of forensic toxicology.
Summary of Studies Applying Metabolomics for Forensic Toxicology.

MALDI, matrix-assisted laser desorption ionization; TCA, tricarboxylic acid.
Monitoring Chemical Residues and Pollution
Economic development and industrial progress have boosted the production of many chemical products while benefiting people, and at this time, access to these items, including their degradation products, has increased. In addition, because industrial and environmental pollution in the production or use process is slowly endangering the environment and people's health, they may accumulate in the human body through water, crops, and air. Metabolomic analysis of affected vectors and bioflows was employed to find sensitive and specific physicochemical disruptor biomarkers and improve the quality of life of social groups. Microplastics have been found in oceans, rivers, sediments, sewage, soil, and even salt, which can accumulate in the body's tissues and organs, affecting health (Figure 4). Metabolomic experiments based on 1H-NMR have shown that microplastics accumulate in the liver, kidneys, and intestines of mice, disturbed creatine, 2-oxygendic acid, citric acid, taurine, and suline metabolites cause energy and lipid metabolism disruption, induce oxidative stress, and include neurotoxic reactions. 124 The application of a metabolomic plan can identify biological metabolic changes and assess hazards, but it is difficult to solve the problem of biochemical pollution and sub-accumulation unless the characteristics of chemical materials are fundamentally changed, weighing the pros and cons, environmental protection has been a worldwide initiative. To prove the strong support of the metabolome in chemical residues and pollution, we also listed the research application of this technology in recent 10 years (Table 4).

Chemical contamination and agricultural residue accumulation interfere with metabolism and cause organ damage.
Summary of Studies Applying Metabolomics for Chemical Residues and Pollution Research.
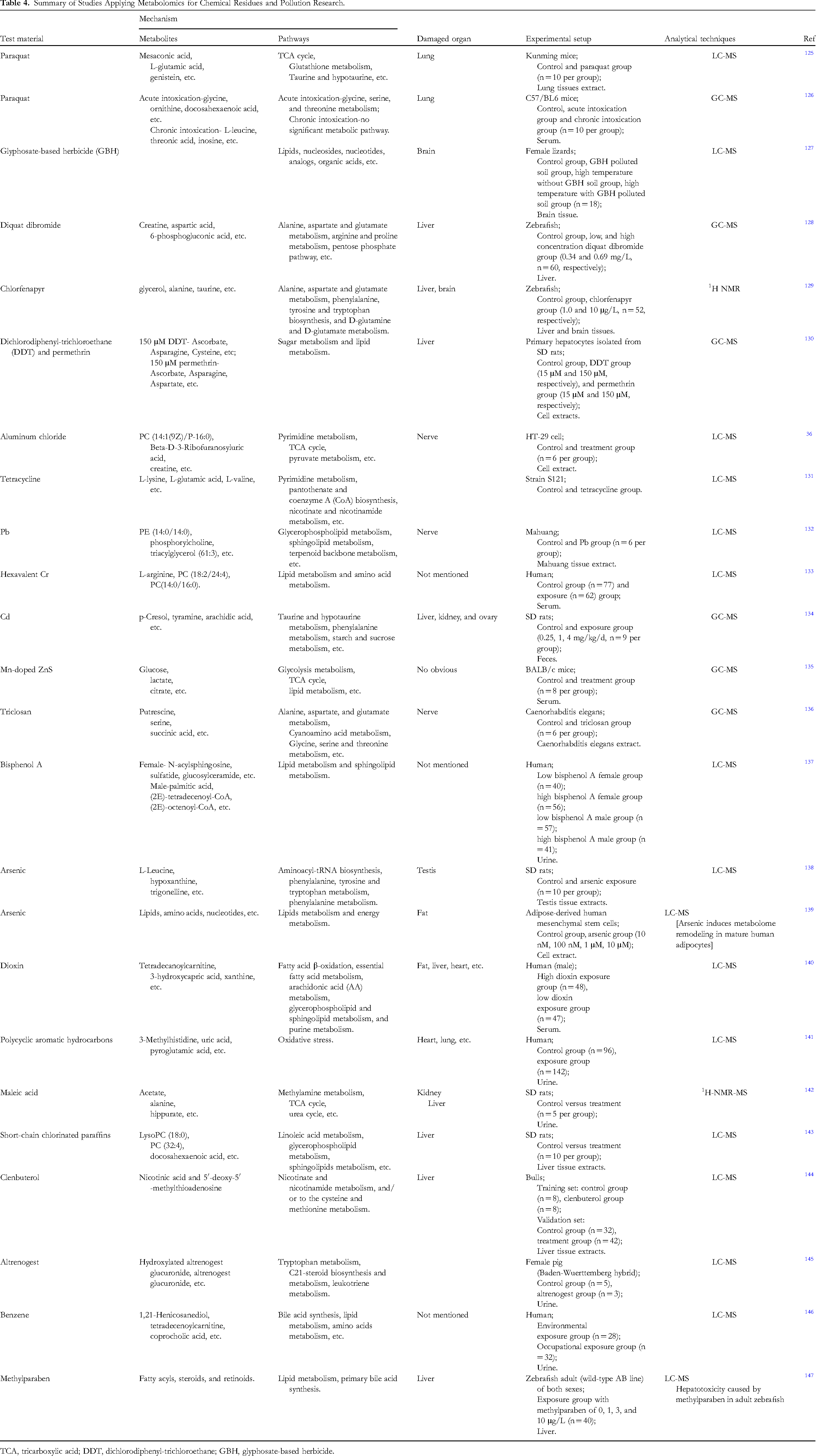
TCA, tricarboxylic acid; DDT, dichlorodiphenyl-trichloroethane; GBH, glyphosate-based herbicide.
Discussion and Outlook
The analytical strategies and ideas of metabolomics are increasingly widely used in toxicology. The application of metabolomics in toxicology pays more attention to the reaction and influence of the external environment on organisms, including environmental pollutants, pesticide residues, psychoactive drugs or herbs, etc. There is no doubt that the toxicology research objects of applied metabolomics are not only limited to the relationship between dose and time, the performance of clinical biochemical pathology but invest more resources to analyze the biological substance, because the ideal state when small-molecule events are prevented or interference may change the final result. This means to monitor disease status and recovery after treatment identifies and evaluates biomarkers of systemic/organ-specific toxicity. At the same time, the method characterized and identified the source molecules causing metabolic disorders, and finally performed other biological validation and structural modifications to reduce toxicity or develop lead compounds.
The application of high throughput and sensitive analytical instruments in metabolomics provides insight into changes in endogenous markers and metabolic mechanisms of organisms. In the study of toxicology, metabolomics has played its advantages, promoted the development process of toxicology research, and provided new insights and approaches for the generation and detoxification of toxicity. In general, toxicology work in the future will not rely solely on physiological and biophysical indicators, and histopathology observation, combined with advanced biological information analysis to comprehensively judge the adverse physiological state, of course, can be prejudged and diagnosed in advance, to avoid unnecessary economic losses. Even so, metabolomics own shortcomings are exposed in toxicology studies because of its high sensitivity and its workflow, which is susceptible to interference to form difficult to repeat conclusions. In toxicology experiments, biological models, analytical instruments, databases, analytical methods, etc. have a direct impact on the results.148,149 For example, two studies on methamphetamine have conflicting results. Both studies found abnormal energy metabolism (TCA cycle), one study found that the levels of fumarate, malate, and succinate associated with the TCA cycle were reduced, 150 while the other result showed that the above marker levels were compared with the control group increase. 151 These differences are likely related to different research settings. At present, metabolomics is still in the field of fundamental research with most studies that are still in the stage of discovery of biomarkers and metabolic pathways, lacking validation of potential biomarkers and exploration of metabolic pathways. Moreover, if this technology and its data conclusions are to be recognized by regulations, the entire metabolomics methodology still needs to undergo unified workflow and standardized validation. It is believed that with the formulation of relevant rules, metabolomics as a technical support can better assist toxicity research.
In addition, the majority of the current research on the application of metabolomics in toxicology research only focuses on phenotypes and lack of indepth research on the mechanism of action and validation. It is worth noting that no single technology can cover all metabolites, and multi-pathway metabolism can also affect the omics identification of specific metabolites. If necessary, isotope-assisted metabolic flux analysis should be used. Moreover, it is necessary to integrate genomics, transcriptomics, and proteomics to provide a comprehensive and broad perspective for toxicological evaluation, systematically reveal the toxic mechanisms of chemicals and HMs, and comprehensively interpret the effects of toxic substances on the organism. Additionally, on the basis of metabolomics research, advanced technologies such as spatial omics and organoids can be introduced in future research to explain the occurrence of toxicity mechanisms from multiple dimensions, accurately predict toxic reactions, and develop precise detoxification drugs to benefiting human health.
Conclusion
In this review, we systematically introduced and discussed the application of metabolomics in toxicological research, including ADR, HMs adverse reactions, drug abuse, chemical residues, and pollution, etc. In general, present review emphasized the broad potential of metabolomics in promoting toxicity assessment and mechanism of action research. With the rapid development of various technologies, metabolomics has indeed accelerated the process of toxicology research and brought new development directions to toxicology research, its application potential in toxicology research is worthy of being explored indepth. We also look forward to the acceptance of metabolomics data as part of regulatory toxicology research with the improvement of relevant regulations, thereby benefiting more people.
Footnotes
Acknowledgements
The authors would like to thank the editor and anonymous reviewers for their thoughtful comments and efforts toward improving our manuscript.
Author Contributions
X.W. have involved in conceptualization and designed the review topic; X.L., J.R., and T.L. wrote the manuscript; T.L. prepared the figures; Y.H., H.D., H.S., G.Y., and X.W. revised the manuscript. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (Grant numbers 81830110, 81903818, 82274199), Key Research and Development Program of Heilongjiang (grant number 2022ZX02C04), University Nursing Program for Young Scholars with Creative Talents in Heilongjiang Province (grant number UNPYSCT2020224), and the Heilongjiang University of Chinese Medicine Foundation (grant number 2018bs02).
