Abstract
Graphical Abstract
Keywords
Introduction
Studies on the molecular recognition of carbohydrates, which act as information carriers in natural systems and thus play a key role in many vital processes, are of great importance and the subject of current research. The design of carbohydrate-binding agents (artificial carbohydrate receptors; for examples of reviews, see references1‐11) which enable selective and effective biomimetic recognition via noncovalent interactions is aimed either at a deeper understanding of natural recognition phenomena or at various potential applications. In addition to their function as model systems for a better understanding of the molecular details of carbohydrate-mediated natural recognition processes, such compounds have the potential to serve as a the basis for the development of new therapeutic agents (anti-infective and anticancer agents)12‐16 or diagnostics.
Although very interesting binding preferences of the artificial receptors have already been determined, the exact prediction of receptor selectivity remains a challenge.
X-ray diffraction studies of protein-carbohydrate complexes revealed that the selectivity and efficiency of carbohydrate-binding proteins is achieved by a combination of different noncovalent interactions17‐25 such as neutral and charge-enhanced hydrogen bonding to the sugar hydroxy groups, CH···π interactions between the CH units of the sugar and the aromatic amino acid residues (see Figure 1a) as well as van der Waals contacts. The involvement of the sugar substrate in different types of noncovalent interactions, including CH···π interactions, has also been shown by crystallographic studies with artificial receptors,26‐29 as illustrated exemplarily in Figure 1b-d. In addition to the crystallographic findings, various studies with artificial carbohydrate receptors30‐56 (see notes in references30,39,45 ) and other model systems57‐62 in solution, especially NMR investigations, clearly showed the tendency of sugar molecules to participate in CH···π interactions with aromatic group(s) of the binding partners.
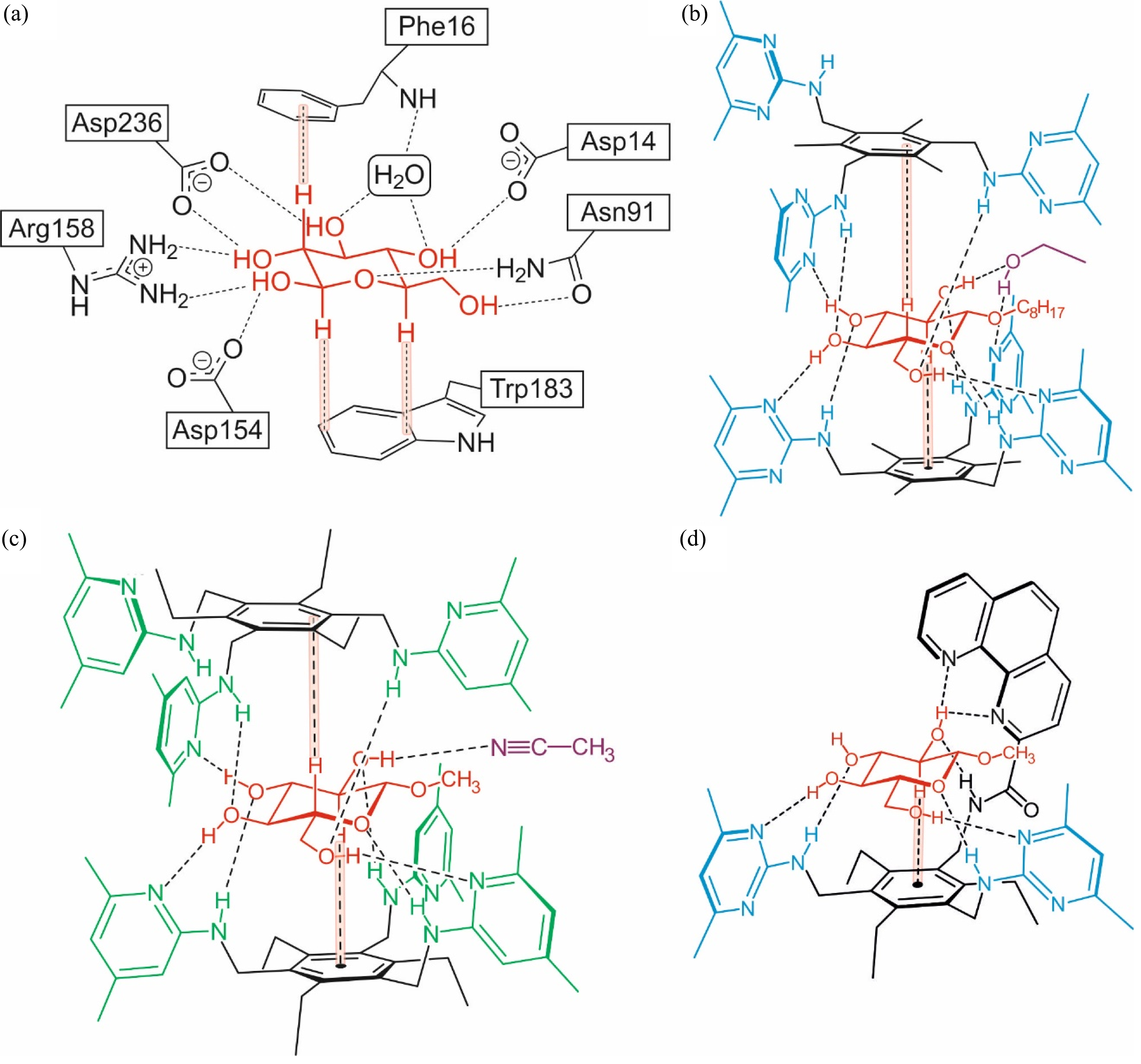
(a) Examples of hydrogen bonds and CH···π interactions observed in the crystalline complex formed between D-galactose-binding protein (GBP) and D-glucose. 22 (b) and (c) Schematic representation of hydrogen bonds and CH···π interactions in two crystalline 2:1 receptor-carbohydrate complexes formed between a trimethyl- 26 or triethylbenzene-based receptor 27 and β-D-glucoside (octyl and methyl β-D-glucoside, respectively); the pyrimidine- (blue) and pyridine-based (green) recognition units of the receptor molecules are highlighted in color. (d) Hydrogen bonding and CH···π interactions observed in the crystalline 1:1 complex of a triethylbenzene-based receptor with methyl β-D-glucoside. 27
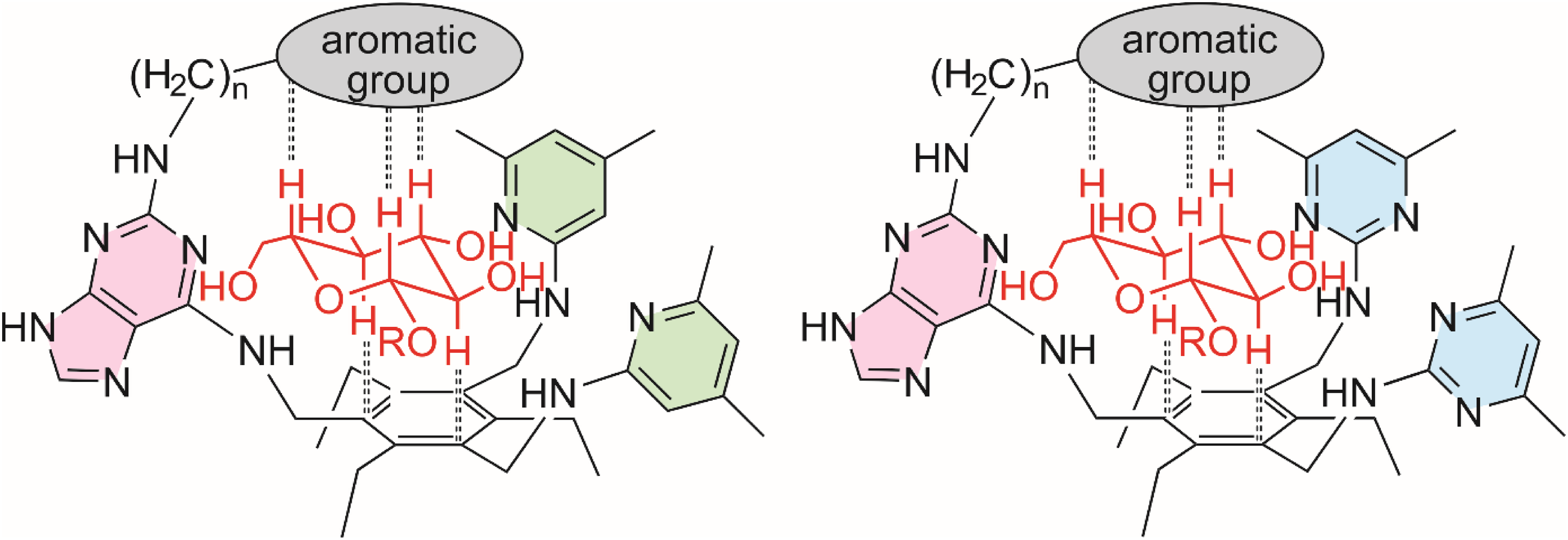
In the natural complex shown in Figure 1a, the protein uses different aromatic groups (indole and benzene ring of tryptophan and phenylalanine, respectively) for the formation of CH···π interactions with the carbohydrate substrate. In the complexes formed by artificial receptors shown in Figure 1b and c, both sides of the glucoside (α- and β-face) are involved in CH···π interactions with the same type of aromatic group (here benzene ring). The sandwich-like complexation of the glucoside is realized in this case by the formation of 2:1 receptor-carbohydrate complexes, and the first crystal structure of such a complex 26 (Figure 1b) acted as a source of ideas for the design of a new macrocyclic receptor architecture with flexible side-arms created by bridging the two receptor molecules.39,41 By combining a macrocyclic backbone with two flexible side-arms (Figure 2), receptor molecules have been developed that predictably form 1:1 complexes with suitable carbohydrates, such as glucosides.39,41‐44

(Left) A representative 41 of the class of macrocyclic compounds bearing two flexible side-arms [here side-arms with pyrimidinyl groups (blue)]. (Right) Schematic representation of the hydrogen bonds and CH···π interactions stabilizing the complex of this macrocyclic compound with octyl β-D-glucopyranoside (the macrocyclic backbone is not completely depicted). Shown are interactions indicated by molecular modeling and confirmed by NMR spectroscopy).
In addition to macrocyclic compounds, well-designed acyclic receptor molecules also enable such sandwich-like complexation within a 1:1 complex. This was shown, for example, by initial studies with purine-containing receptor molecules, 32 whose binding mode is schematically shown in Figure 3. This type of compounds has the ability to bind suitable carbohydrates through combined noncovalent interactions, where the CH···π interactions can be formed with both faces of the carbohydrate substrate. Compared to the macrocyclic system, the structural variation of the acyclic architecture is easier to perform. As a result, compounds with two identical or different types of aromatic groups, which can be involved in the formation of the abovementioned CH···π interactions, are synthetically more easily accessible (e.g., a combination phenyl/indolyl groups, like in the case of the natural complexes shown in Figure 1a).
Schematic representation of the structures and mode of action of purine-containing receptors. The pyridine- (green) and pyrimidine-based (blue) recognition units and the exchangeable aromatic unit (gray) on the purine ring (magenta) are highlighted in color.
The aim of the current studies was to synthesize a series of compounds (potential carbohydrate receptors), the structure of which is shown schematically in Figure 3, and to analyze in detail the influence of the nature of the structural subunits on the binding properties. In particular, the influence of the character of the aromatic group and the nature of its linkage to the purine ring was analyzed. In addition, it was investigated whether the replacement of the aminopyridine-based by aminopyrimidine-based recognition units would lead to more powerful (more efficient) carbohydrate receptors, as has been repeatedly observed by us for previously examined receptor systems. Such systematic studies of the structure-binding activity relationships provide useful information for the development of effective carbohydrate receptors with predictable binding properties.
Structures of the Target Compounds
Recently we have reported the synthesis of new 1,3,5-substituted 2,4,6-triethylbenzenes consisting of both purine unit(s) and pyridine-/pyrimidine-based recognition groups
32
(for a review on 1,3,5-substituted 2,4,6-triethylbenzene derivatives, see reference
63
). Compounds bearing 2-chloro-substituted purine unit(s) [2-chloro-9
Initial binding studies with compounds prepared in this way indicated that their binding properties can be fine-tuned by the variation of the substituent attached to C2-position of the purine ring. Depending on the nature of the aromatic group and on the type and length of the linker unit, which connects this aromatic group with the purine ring, carbohydrate receptors with different binding properties can be developed.
In this study, we focused on the compounds shown in Figure 4, which contain phenyl, 1-naphthyl, 3-indolyl or 5-methoxy-3-indolyl group as an additional aromatic moiety. The unit –NH(CH2)n– with n = 1-3 was chosen as the linker, as illustrated in Figure 3. The binding properties of compounds containing aminopyridine-based recognition units (
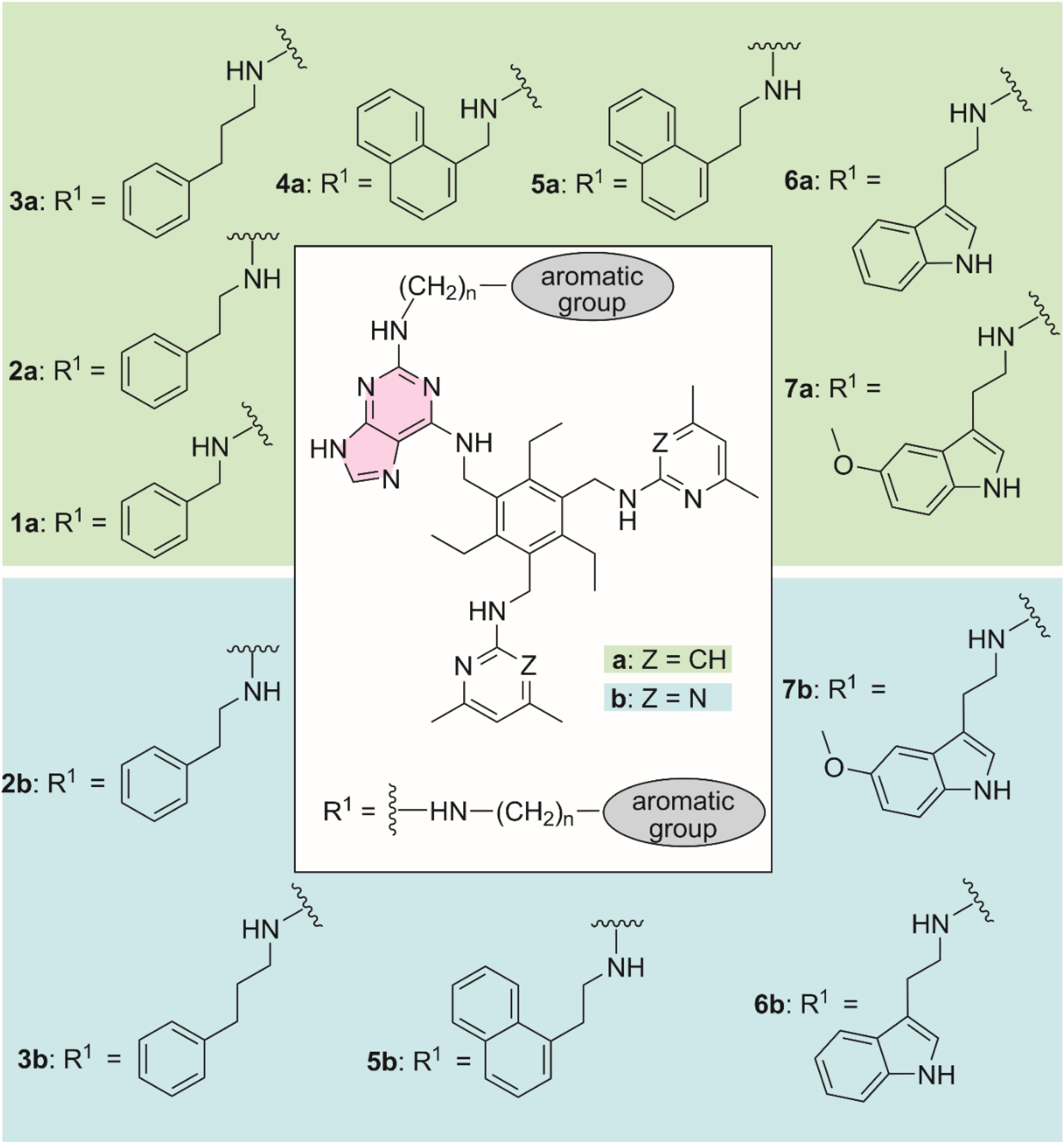
Structures of target compounds considered in this study.
Results and Discussion
Synthesis of the Target Compounds 1a -7a and 2b , 3b , 5b -7b
The syntheses of the target compounds

Synthesis of compounds
The 1,3,5-substituted 2,4,6-triethylbenzene derivatives
The linkage of the amino group of
As shown in our previous study,
32
the nucleophilic displacement of the chlorine atom in the 2-position of the purine unit of
Binding Properties of Compounds 1a -7a , 2b , 3b , and 5b -7b : 1H NMR Spectroscopic and Microcalorimetric Titrations
The compounds described in this work were developed as artificial receptors especially for carbohydrates with equatorial substitution pattern, such as β-glucosides. As a substrate for the binding studies was selected octyl β-D-glucoside (
In addition to the 1H NMR spectroscopic titrations in which the concentration of the tested compound was kept constant and that of the carbohydrate was varied, inverse titrations were also carried out in which the concentration of the sugar remained unchanged and that of the binding partner was varied. The extensive titration experiments thus enabled a detailed analysis of the complexation-induced shifts of the receptor signals and those of the carbohydrate substrate. The titration experiments were performed in CDCl3 at 20 °C. The 1H NMR titration data were analyzed using the WinEQNMR 68 and SupraFit 69 programs; the binding constants are summarized in Table 1. The complex stoichiometry was analyzed on the basis of the mole ratio method 70 (for an example, see Figure S1 in Supporting Information).
Association Constantsa-c for Compounds

aAverage
1H NMR titrations in CDCl.
bErrors were estimated at ≤ 5%.
c
It should be noted that although the molecular recognition of carbohydrates in water is of great importance, the binding studies in organic media should not be neglected. Analysis of the binding mode of carbohydrate-binding proteins has shown that the hydrogen bonds formed between these proteins and the sugar substrate are shielded from the bulk solvent, meaning that they exist in a lower dielectric environment.21,23 Thus, binding studies in organic media such as chloroform provide valuable information about the factors that contribute to the affinity between receptors and sugars and can make an important contribution to our understanding of the complex carbohydrate-mediated processes in nature. Such studies are also an important tool in the search for new structural elements that are useful for the construction of artificial receptors with both a selective and effective mode of action.
The addition of

Excerpts from 1H NMR spectra of compound
This was also confirmed by the analysis of the data obtained on the basis of the inverse titrations. It should be noted that in the case of the last-mentioned titrations, the CH signals of
The detailed analysis of the titration data revealed that in the case of the compounds bearing pyrimidinyl groups (
For the purine-containing molecules described here, the aminopyridine- or aminopyrimidine-based recognition units are not the only hydrogen bonding sites, but an important role is also played by the purine residue. The latter also acts as a bridging unit, allowing the introduction of a well-positioned second aromatic group (in addition to the central benzene ring), which can participate in additional CH···π interactions contributing to the stabilization of the receptor-sugar complex. Thus, the importance of the combination of the different receptor subunits involved in the formation of various (combined) noncovalent interactions should be emphasized. The significant role of combined noncovalent interactions has also been identified in many other processes of molecular recognition (see, for example, reference 72 ; for examples of further discussions on supramolecular complexations, see references73‐79).
The complexation properties of all tested compounds (
In accordance with the aim pursued with the design of this type of carbohydrate receptors, the presence of a suitable and well-positioned aromatic moiety at C2 of the purine ring is favorable for the targeted formation of 1:1 receptor-carbohydrate complexes. At this point it should be mentioned that the tendency to form complexes with higher stoichiometry represents a disadvantage of some acyclic compounds.
When considering the influence of the type of aromatic unit (phenyl/naphthyl
In the case of the phenyl group-bearing compounds
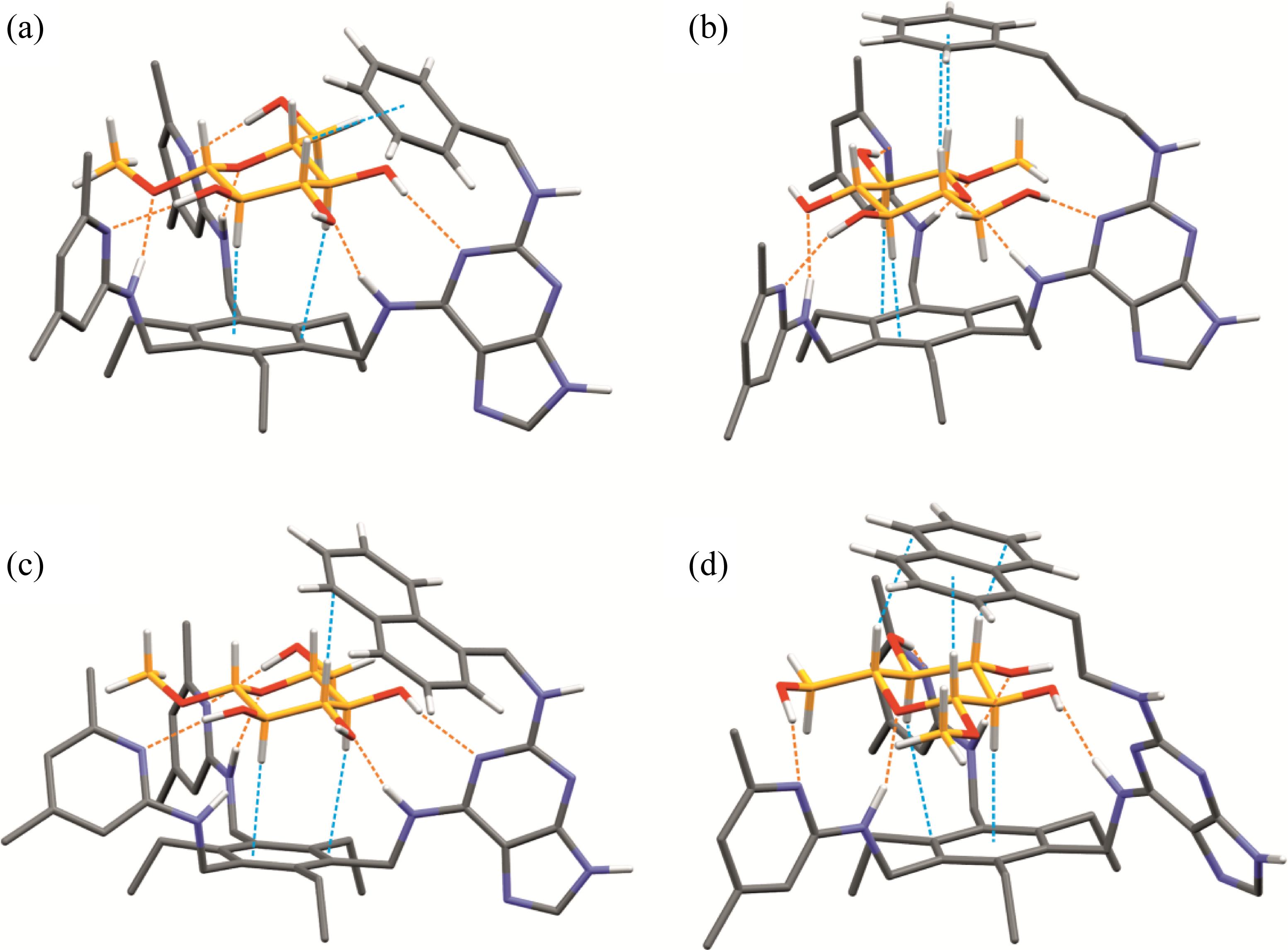
Energy-minimized structures of the 1:1 complexes formed between methyl β-glucopyranoside and compounds
The presence of an indolyl group, which is also used by carbohydrate-binding proteins for the formation of CH···π contacts, should have a positive effect on the binding strength, but compounds
As indicated by molecular modeling calculations, intramolecular hydrogen bonds may be responsible for this. For example, the energy-minimized structure of
Among the compounds bearing a 3-indolyl or 5-methoxy-3-indolyl group (compounds

Binding efficiency of compounds
Compounds with an unsuitable linker or aromatic group represent weaker receptor molecules. In these cases, in addition to the 1:1 complexes, the formation of weaker 2:1 receptor-substrate complexes was also observed (for example in the case of compound
The binding properties of compound

Exemplary ITC thermogram (left) and titration curve-fitting (right) for the titration of
Results of Microcalorimetric Titrations of Compound

aIn dry CHCl3 at 293 K; calorimeter:
bStandard deviations for a minimum of three replicated ITC titrations: about 0.1 kJ/mol.
Abbreviation: ITC, isothermal titration calorimetry.
The results of these investigations are now being used to further improve the design of receptor molecules and to develop systems with accurately predictable binding properties in different solvents, including aqueous media (for a discussion of solvent effects, see reference 86 ). It should be noted that the recognition of carbohydrates in water is particularly challenging because a receptor must select its target molecule from a large excess of competing water molecules.
Conclusion
1,3,5-Substituted 2,4,6-triethylbenzene derivatives equipped with two aminopyridine- or aminopyrimidine-based recognition units and a purine moiety, which serves both as a hydrogen bonding site and as a bridging component for the incorporation of additional substituents, have the potential to act as effective carbohydrate receptors and to form predictable 1:1 complexes with a suitable substrate. The use of the purine moiety allows the construction of compounds with two aromatic groups (central benzene ring and the additional aromatic group connected to the C2 position of the purine ring via a linker) positioned to interact with the sugar CHs in a sandwich-like manner. The effective complexation depends on the nature of this aromatic group and the length of the –(CH2)n– unit linking this group to the C2 position of the purine ring. Compared to the macrocyclic system shown in Figure 2, the acyclic architecture enables a simpler construction of compounds containing either two identical or different aromatic groups that can participate in the interactions with the sugar CH units. For example, it was found that the incorporation of a well-positioned phenyl or naphthyl group predictably leads to the formation of strong 1:1 complexes in which the substrate is sandwiched between two benzene rings or between a benzene and a naphthalene moiety.
The use of the purine moiety in the construction of carbohydrate receptors with a 1,3,5-substituted 2,4,6-triethylbenzene backbone has proved to be a very promising approach, and it should be emphasized that the possibilities for structural variations of this molecular architecture are manifold. Thus, a whole range of compounds can be synthesized to perform extensive studies on the relationships between structure and binding efficiency.
Materials and Methods
Experimental Section
Analytical TLC was carried out on silica gel 60 F254 plates, column chromatography was carried out on silica gel. Melting points are uncorrected. Benzylamine, 2-phenylethylamine, 3-phenyl-1-propylamine, 2-(1-naphthyl)ethylamine, tryptamine and 5-methoxytryptamine are commercially available; the synthesis of 1-naphthylmethylamine was performed according to the procedure described in reference
64
. The preparation of
General Procedure for the Preparation of Compounds 1a -7a , 2b , 3b , and 5b -7b
Route A (Sealed Tube)
1-[(2-Chloro-9
Route B (Microwave Conditions)
Compound
1-[(2-Benzylamino-9H-purin-6-yl)aminomethyl]-3,5-bis-[(4,6-dimethylpyridin-2-yl)aminomethyl]-2,4,6-triethylbenzene (1a ).
32
Route A
Compound
Route B
Compound
1-[(2-Phenylethylamino-9H-purin-6-yl)aminomethyl]-3,5-bis-[(4,6-dimethylpyridin-2-yl)aminomethyl]-2,4,6-triethylbenzene (2a )
Route A
Compound
Route B1 (Preparative HPLC)
Compound
Route B2 (Column Chromatography)
Compound
1-{[2-(Phenylpropylamino)-9H-purin-6-yl]aminomethyl}-3,5-bis[4,6-dimethylpyridin-2-yl)aminomethyl]-2,4,6-triethylbenzene (3a )
Route B
Compound
1-{2-[(Naphth-1-yl)methylamino]-9H-purin-6-yl}aminomethyl}-3,5-bis[4,6-dimethylpyridin-2-yl)aminomethyl]-2,4,6-triethylbenzene (4a )
Route B (Preparative HPLC)
Compound
1-{{2-[(Naphth-1-yl)ethylamino]-9H-purin-6-yl}aminomethyl}-3,5-bis[4,6-dimethylpyridin-2-yl)aminomethyl]-2,4,6-triethylbenzene (5a )
1-{{2-[(Indol-3-yl)ethylamino]-9H-purin-6-yl}aminomethyl}-3,5-bis-[(4,6-dimethylpyridin-2-yl)aminomethyl]-2,4,6-triethylbenzene (6a )
Route A
Compound
Route B
Compound
1-2-[(5-Methoxyindol-3-yl)ethylamino]-9H-purin-6-yl}aminomethyl}-3,5-bis[4,6-dimethylpyridin-2-yl)aminomethyl]-2,4,6-triethylbenzene (7a )
Route A
Compound
Route B
Compound
1-[(2-Phenylethylamino-9H-purin-6-yl)aminomethyl]-3,5-bis-[(4,6-dimethylpyrimidin-2-yl)aminomethyl]-2,4,6-triethylbenzene (2b )
Route B
Compound
1-[(2-Phenylpropylamino-9H-purin-6-yl)aminomethyl]-3,5-bis-[(4,6-dimethylpyrimidin-2-yl)aminomethyl]-2,4,6-triethylbenzene (3b )
Route B
Compound
1-[(2-Naphthylethylamino-9H-purin-6-yl)aminomethyl]-3,5-bis-[(4,6-dimethylpyrimidin-2-yl)aminomethyl]-2,4,6-triethylbenzene (5b )
Route B
Compound
1-{{2-[(Indol-3-yl)ethylamino]-9H-purin-6-yl}aminomethyl}-3,5-bis-[(4,6-dimethyl-pyrimidin-2-yl)aminomethyl]-2,4,6-triethylbenzene (6b )
27
Route B
Compound
1-{{2-[(5-Methoxyindol-3-yl)ethylamino]-9H-purin-6-yl}aminomethyl}-3,5-bis-[(4,6-dimethyl-pyrimidin-2-yl)aminomethyl]-2,4,6-triethylbenzene (7b )
Route B
Compound
Description of the 1H NMR and Microcalorimetric Titrations
The 1H NMR titrations were carried out in CDCl3 at 20 °C [CDCl3 was deacidified over basic aluminium oxide (Brockmann I) and stored over molecular sieve]. Stock solutions in CDCl3 were prepared and homogenized for the respective receptor and sugar. These solutions and CDCl3 were added together in a manner that the concentration of the receptor was kept constant and that of the sugar was varied. The receptor concentration was adjusted to about 1 mM to avoid self-aggregation. For the inverse titrations, the concentration of the sugar was kept constant and that of the receptor was varied accordingly. For each titration, 15-20 samples were prepared, thoroughly homogenized and the 1H NMR spectra were recorded. For each system at least three titrations were carried out.
ITC was carried out in CHCl3 [deacified over basic aluminium oxide (Brockmann I) and stored over molecular sieve] at 20 °C on
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241258352 - Supplemental material for Sandwich-Like Complexation of Carbohydrates by Hydrogen Bonding and CH-π Interactions
Supplemental material, sj-docx-1-npx-10.1177_1934578X241258352 for Sandwich-Like Complexation of Carbohydrates by Hydrogen Bonding and CH-π Interactions by Linda Köhler, Stefan Kaiser and Monika Mazik in Natural Product Communications
Supplemental Material
sj-pdf-2-npx-10.1177_1934578X241258352 - Supplemental material for Sandwich-Like Complexation of Carbohydrates by Hydrogen Bonding and CH-π Interactions
Supplemental material, sj-pdf-2-npx-10.1177_1934578X241258352 for Sandwich-Like Complexation of Carbohydrates by Hydrogen Bonding and CH-π Interactions by Linda Köhler, Stefan Kaiser and Monika Mazik in Natural Product Communications
Footnotes
Acknowledgment
Open Access Funding by the Publication Fund of the Technische Universität Bergakademie Freiberg is gratefully acknowledged.
Data Availability
Data will be provided by the corresponding author upon a reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable for this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Deutsche Forschungsgemeinschaft.
Statement of Human and Animal Rights
No animal or human studies have been conducted.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
