Abstract
Flavone is a very simple and unsubstituted flavonoid of restricted distribution in the plant kingdom. Historically it is known for a long time as major constituent of many Primula and Dionysia exudates produced by glandular hairs in unusually large amounts, often as a farinose (mealy) deposit on aerial plant parts. The ecological function of this unique farinose deposit and its main compound flavone is still not really understood. Concerning flavone, a knowledge gap exists about its biosynthetic origin, when compared to all of the flavonoid biosynthetic routes known to date. Thus, this review addresses not only the history of this compound and its occurrence in plants and in their respective compartments, but also discusses ideas on its putative biosynthesis. Furthermore, we provide up-to-date analytical data on this compound from a natural source, rather than from chemosynthesis, in a comprehensive way. Eco-functional aspects complement this study that is intended to be highly stimulating for future research in ecology and evolution.
Introduction
Unsubstituted flavone is a pretty simple flavonoid, bare of any substitution as is common in flavonoids, except the typical keto group (Figure 1). One might assume that much information should be available about this compound, and that it should be easily detected when present in plant tissues. Its structure is well elucidated, albeit mostly from synthetic flavone, 1 but apparently it is not as common in the plant kingdom as the simple structure suggests. The history of natural flavone is tightly associated with its presence as a major compound in the conspicuous, mealy (farinose) exudates produced by glandular hairs, typical of many Primula species (Primulaceae), and frequently accompanied by smaller amounts of flavones of unusual structure and of unknown biosynthetic origin, termed as Primula-type flavones. 2 As for flavone, only very few reports exist on its occurrence in other species outside this family, mostly without indication of respective storage compartments. Apart from information about the history of flavone and the farinose depositions typical of Primula species, its distribution in Primula and related genera, as well as outside Primulaceae is reviewed. As most analytical data refer to synthetic flavone, a comprehensive analytical study was carried out on natural flavone from Primula by state-of-the-art methods. Another central focus is the question of how flavone is biosynthesized in planta, an aspect discussed at length with known data, and ideas and hypotheses based upon them. In this context, the site of storage and production, such as the glandular hairs in case of Primula, deserve major attention. Finally, ecological and functional aspects complement this review, with the aim to stimulate future research in ecology and evolution.

Structure of flavone with numbering of the positions according to the NMR spectroscopic data.
History of Flavone
As early as in 1895, Kostanecki and Tambor 3 introduced the terms ‘flavone’ and ‘flavonol’ in a paper dealing with the structural analysis of a flavonoid named chrysin, thus coining those standard terms still in use today. Not much later, unsubstituted flavone (Figure 1) itself was first synthesized by Feuerstein and Kostanecki. 4 The natural occurrence of this compound was established by Müller, 5 who isolated flavone out of mealy exudates that he had brushed off the aerial parts of several Primula species. Some years later, flavone was shown to be the major constituent in Primula denticulata Sm. exudates, 6 amounting to 75% of the farina. Since then, the genus Primula with its more than 450 species and its unique farina was studied more intensively for flavonoids and other constituents, and the widespread presence of flavone both in mealy and oily exudates confirmed.2,7–10
Exudates and waxy coatings of plants were in focus also quite early. Plant waxes are cuticular products, mostly consisting of aliphatic or cyclic (triterpene) compounds, often arranged in showy crystalline structures that might be species specific. Exudate production requires specific structures such as glandular hairs or tissues for secretion of often volatile terpenoids and lipophilic flavonoids. In both cases, compounds are deposited on the aerial surfaces of plant tissues.11,12 These waxy or mealy layers (farina) attracted plant physiologists quite early, 13 but the chemical composition was hard to assess. From anatomy of epidermal structures, it was considered that mealy exudates of gymnogrammoid ferns and of Primula should be different from the frequently observed waxy coatings found in many higher plants.13,14 They described yellow or white exudates on gymnogrammoid ferns, that much later were shown to consist inter alia of flavonoid aglycones, as also is the case in Primula spp. 12 So far, farina is known only from Primula and some fern genera. 11 Sublimation techniques were used for determination of the skin irritant derived from Primula obconica Hance, and mealy excretions of other Primula spp. came slowly into focus as well. 15 In a vial, dry farinose exudate reassembles into long threads as observed for Primula florindae Ward (see Figure S1), resembling the in vivo observed extrusion from Dionysia glandular hairs. 16
Methodology and Analytical Data
(a) Extracting Exudate Compounds
For obtaining exudate compounds, some care is needed. One way is to remove the glandular hairs mechanically and subject them to extraction with a lipophilic solvent; the other possibility is a careful short rinsing of the plant material with a nondestructive lipophilic solvent such as acetone. Solvents like chloroform are not recommended as this might destruct tissue cells and thus compounds of tissues would be extracted as well. After careful rinsing, compounds present in the tissue such as flavonoid glycosides might be extracted by methanol or comparable solvents. This procedure is advantageous over the general extraction with eg methanol and further separation by liquid-liquid extraction, which does not give any indication in which part of the plant the compounds would be located. Separation and compound isolation follow the general procedures as denoted in previous studies.9,10
(b) Elucidating the Molecular Structure Of Flavone
The molecular structure of flavone (Figure 1), often from synthesis, has been described in a series of scientific papers. However, the corresponding analytical studies in the various publications do generally not base on comprehensive analyses using several different methods. Rather, analytical data have often been compared with corresponding earlier reported data, thus confirming the identity of the flavone by comparison. Even, in a recent study on Dionysia tapetodes Bunge, 16 analytical data were given on natural flavone, but not completely discussed. Therefore, we have made a comprehensive structural analysis, using several spectroscopic and spectrometric methods. For this purpose, solid flavone, obtained from the reference sample collection of E. Wollenweber (Darmstadt) and previously isolated from Primula species, was used for spectroscopic, spectrometric, and chemical analysis. All data recorded by us fully confirm the structure of flavone.1,17 Thus, a comprehensive summary of structural analysis data of flavone, obtained by the commonly used structural analytical methods is now available. Experimental and analytical data are are summarized in the supplentary, together with relevant spectra (Figures S2. to S11., Table S1.). In the following, relevant MS, IR, UV and NMR data as well as a CHNS-O analysis are discussed below.
Flavone shows in HR ESI TOF MS (positive ionization) a deprotonated molecular ion [M + H]+ of m/z 223.0752, and a [M + Na]+ of m/z 245.0543. These correlate well with the calculated [M + H]+ of m/z 223.0753 and [M + Na]+ of m/z 245.0573 of the molecular formula C15H10O2. The adducts [2M + Na]+ (m/z 467.1256) and [3M + Na]+ (m/z 689.1943) indicate a tendency towards dimer and trimer formation, which is likely caused by π-π stacking. The molecular formula C15H10O2 is confirmed by elemental analysis with 81.26% C, 14.97% O, and 4.45% H. Slight deviations from the theoretical percentage values for the proportions of carbon, oxygen, and hydrogen can be explained by the fact that this flavone sample originates from natural plant sources and still contains small amounts of impurities as well as some residual water.
NMR spectroscopy confirms the presence of 10 methine groups and 5 quaternary carbon atoms. Likewise, the spin system of the protons in positions In total, 5 to 8 shows that the A-ring does not carry any substituent in these positions. The C-ring is annelated at positions 4a and 8a, whereby the chemical shift of these carbon atoms indicates the oxygen in pos. 1 and the carbonyl group in pos. 4. Furthermore, the double bond between C-2 and C-3 is confirmed, with the proton in position 3 (6.86 ppm) indicating that no substituent is bound there. The 1H and 13C chemical shifts of the B-ring show it to be symmetrical and not bearing any substituent.
The IR spectrum confirms these observations by the sole presence of C—H stretching vibrations on sp 2 hybridized carbon atoms (3075-3041 cm−1). The C = O stretching vibration (1639 cm−1) indicates the ketone in pos. 4, the C = C stretching vibrations (1604 to 1569 cm−1) confirm the double bond as well as the aromatic moieties and the C—O stretching vibration (1226 cm−1) indicate the presence of oxygen in pos. 1. Additionally, the UV spectrum (Figure S1) showing absorption maxima at 308 (shoulder), 296, 252 and 214 nm (shoulder) confirms the presence of a chromophore with a weak auxochromic as well as a weak antiauxochromic group.
Numbering of proton and carbon positions is shown in Figure 1. 1D and 2D NMR spectra are shown in Figures S2-S8, HR ESI TOF MS spectrum (pos. mode) is shown in Figure S9, IR spectrum is shown in Figure S10, UV spectrum is shown in Figure S1 and data of elemental analysis are shown in Table S1.
Occurrence of Flavone in Higher Plants
As mentioned, the genus Primula and associates (= Primula assemblage) are the main source of this exudate compound (Table 1) that is produced by glandular hairs in quite large amounts, an unusual feature of flavonoid exudation. Diversifications of the glandular flavonoid composition appear to be related to the phylogeny of the Primula assemblage, and flavone content may change in populations depending on habitat conditions (altitude, climate) and seasonal changes as shown for Primula auricula L. of the European Alps. 18 Members of Dodecatheon are understudied and only 1 record exists on small amount of flavone in the exudate of D. meadia L. (syn. of Primula meadia (L.) A:R.Mast & Reveal) (Priemer, pers. comm.; Table 2). Some of the newer evolutionary lineages show reduced amounts of flavone, as eg Dionysia spp., and some of the European alpine taxa.19,20
Frequency of Exudate Flavone Occurrence within the Primula Assemblage*.

* Comparative studies were considered only. Generic names and nomenclature as in 24 and refs. cited here.
Flavone Reported from Other Genera and Putative Storage Organs.

Huge amounts of plant material, little amounts of flavone in relation.
Leaves highest flavone content.
Flavone dominant.
Literature search for further sources is hindered by the common use of the term ‘flavone’ not necessarily denoting the unsubstituted compound. Careful search in SciFinder, 1 however, revealed only scattered reports on a few unrelated families and some species (Table 2). These include also reports on fruits, such as in Feijoa sellowiana (O.Berg) O.Berg (syn. Acca sellowiana (O.Berg) Burret) (Myrtaceae). Upon reanalysis, we could confirm the presence of flavone in this species. 22 To destroy doubts about the other named sources (Table 2), available analytical data were checked, and the presence of flavone thus confirmed with current data (see section Methodology and Analytics). As flavone is not expected to occur in cell vacuoles of tissues, search for mention of secretory or corresponding structures was conducted, and respective information is included in Table 2. However, there is so far no proof for these anatomical structures as either sites of biosynthesis and/or accumulation. More detailed studies would be helpful in general when it comes to accumulation sites of unique structures such as flavone.
Accumulation and Secretion
Comparatively little is known about the processes behind the accumulation of flavone in both the Primula assemblage as well as in the other mentioned sources (Tables 1 and 2). As for the Primula assemblage, early studies on the ultrastructure of glandular hairs indicated that the lipophilic exudate is excreted through the cuticle and not stored in a subcuticular space, both in oily and in farinose Primula spp. No indication to a specific storage organelle responsible for the exudation process could be given in this study. 38 Recently, a detailed analysis of the glandular structures of Dionysia tapetodes revealed micron-size holes in the glandular trichomes, through which the woolly fibers, consisting almost of flavone only, extrude. 16 A vacuole-like structure was present in the glandular hairs, and it was suggested to play a role in the exudation process. This raises the question, how a lipophilic compound like flavone is stored in a compartment that normally serves for storage of hydrophilic compounds. This issue could not be resolved so far. 16
Maleci et al. 39 analysed the development of glandular hairs in Primula obconica towards the secretion process. An increase in size of the vacuole of the neck cell is accompanied by the formation of a number of vacuoles in the head cell. However, this species produces neither flavone nor a farinose exudate. The role of the vacuole in the context of secretion remains elusive. In previous ultrastructural studies, no aggregation of exudates was detected in cytoplasm, vacuoles or in other compartments. 38 The end cells of glandular hairs of studied Primula spp. were found to be rich in endoplasmic reticulum, mitochondria, leucoplasts, and microbodies, but elements such as dictyosomes and lipoid droplets are rare. 38 Some of these intracellular compartments may be involved in the biosynthesis of flavonoids.
Biosynthetic Origin of Flavone
The issue of biosynthesis should not be seen as separated from tissues and organs that may be involved in this process. As for Primula, previous studies indicated that flavone is present in glandular hairs only, but not in underlying leaf tissues, where flavonoid glycosides are predominant.9,40 Despite the fact, that flavonoid biosynthesis is generally well understood, 41 practically nothing is known about the biosynthesis of unsubstituted flavone, neither in Primula, nor in the other mentioned sources (Tables 1 and 2). In the following, related aspects and hypotheses will be discussed.
(a) Biosynthetic Machinery Operating in Glandular Hairs in Primula Classical biosynthetic pathways leading inter alia to flavones and flavonols with 5,7,4ʹ trihydroxylated structure, as based upon the respective starter molecules. These starter molecules are not feasible to generate an unsubstituted flavone structure, and chalcone dehydroxylase is known for acting on position 5 of Ring A only.
42
Abbreviations: PAL, phenylalanine-ammonia lyase; C4H, cinnamate-4-hydroxylase; 4CL, 4-coumaroyl CoA-ligase; CHS, chalcone synthase; CHI, chalcone isomerase.
Key enzymes in flavonoid biosynthesis are phenylalanine ammonia-lyase (PAL) and chalcone synthase (CHS). PAL catalyses the deamination of the aromatic amino acid phenylalanine, and it exists in isoforms that lead to different end products via metabolic channeling.
42
CHSs are a superfamily of enzymes belonging to the type III polyketide synthases, known to be widespread in plants, but they were also described from bacteria and fungi. As part of the flavonoid biosynthetic pathway, CHS catalyses generation of the chalcone backbone, being the key compound for biosynthesis of further flavonoid groups (see Figure 2). Within one plant, they may be expressed in different tissues at varying developmental stages, and they are involved inter alia in defensive reactions to biotic and abiotic stresses. Localization studies indicated their presence in epidermal and mesophyll cells of leaves, further in the nucleus, the plastids, and the vacuole, as well as in cytoplasm and endoplasmic reticulum, depending on species studied. More information may be obtained from a comprehensive review on this subject.
43
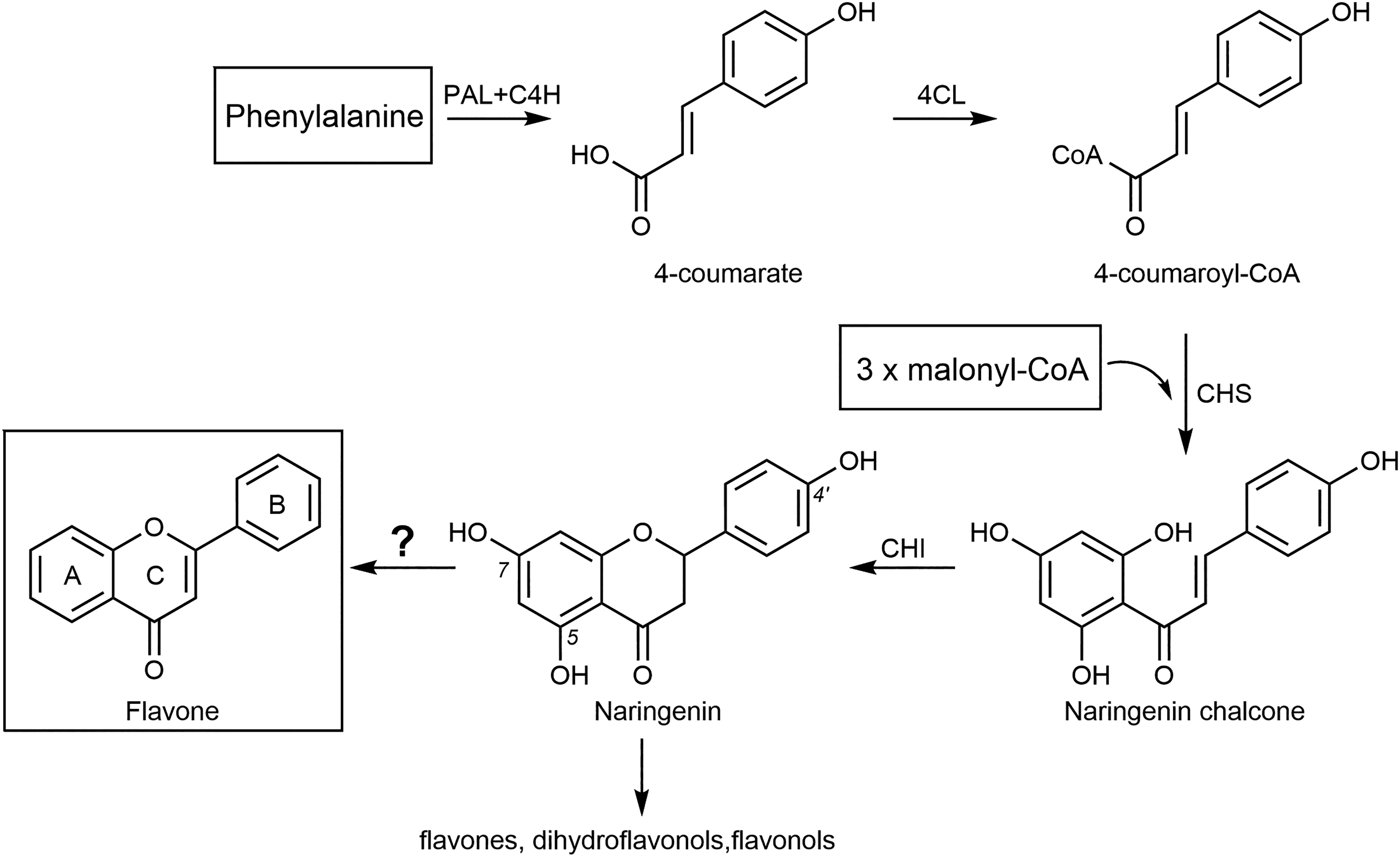
There are no recent studies to address the enzymology of flavone production in glandular hairs of Primula. Earlier, Schöpker et al. 44 analysed isolated glands of Primula × kewensis W.Watson by immunohistochemical techniques to localize key enzymes of flavonoid biosynthesis. Thus, PAL and CHS could be localized exclusively in the flavonoid-producing head cells of the glandular hairs, while those enzymes could not be detected in flavonoid-deficient mutants. PAL was located in regions of the dense tubular endoplasmic reticulum and occasionally found in more or less spherical organelles yet unidentified. An appreciable proportion of the enzyme protein was associated with the plasma membrane and the cell wall of the head cells. CHS, however, was restricted to spherical unidentified cell components. It looks that in this case leucoplasts are not involved in the biosynthesis of flavonoids. Recent publications indicate that besides chloroplasts (arogenate pathway) also the cytosol participates via the phenylpyruvate pathway in the biosynthesis of the aromatic amino acid phenylalanine, which would be precursor in the phenylpropanoid and flavonoid pathways.45,46 It is suggestive to assume that flavonoid biosynthesis is associated with the cytosol in Primula glands.
(b) Search for Precursors as Feasible Starting Points in Flavone Biosynthesis in Primula
In Figure 2, classical biosynthetic pathway steps are denoted in brief, leading to the majority of flavonoids known to date. Specific pathways leading either to the 5-deoxyflavones typical of legumes,
42
or to B-ring unsubstituted flavones as in Scutellaria baicalensis Georgi (Lamiaceae)
47
are rare exceptions. The latter pathways, however, do not account for the formation of unsubstituted flavone. This raises the question about possible precursors for generation of flavone, a compound that is produced in unusual large quantities. It is unlikely that flavone represents a degradation product from regularly substituted flavones, also in view of the lack of evidence of degrading enzymes. It is suggestive that other than the classical starter molecules give rise to unsubstituted flavone. However, both, the starter molecules and the enzymes involved, are still obscure (see Figure 2). Unsubstituted cinnamic acid could be one candidate, and 4-coumaroyl CoA-ligase (4CL) would not be operative in the case of flavone. A specific CHS would have to link with another phenolic ring that possibly derives from a fatty acid-like precursor.
Distribution of Chalcones and Flavanones in the Primula Assemblage.

*Isolated from extracts of aerial parts in minute amounts
Finally, there are some interesting phenylpropanoids reported from Primula exudates, such as acetophenone or salicylic acid derivatives, found in essential oils derived from the glandular hairs of Primula, 50 or Dionysia, 51 and thus co-occurring in the same compartment as flavone. It would be tempting to see them as starting point of the biosynthesis of flavone, but so far, there are only chemosynthetic approaches known leading to flavone or the precursor chalcone. 52 It should be mentioned that Primula veris L. epicuticular waxes contain phenylalkane derivatives, being new compounds for the genus Primula. 53 So far, their presence in glandular hairs could not be verified, and their phenolic residue could be derived from a parallel pathway related to the wax production.
(c) Searching for Endophyte Assistance
Endophytic organisms of medicinal plants producing desired plant drugs are quite a hot topic today. Despite existing doubts when it comes to biosynthesis of eg the anticancer drug taxol (Paclitaxel) by endophytic fungi instead of their host yew plant,
54
one cannot completely neglect this aspect when it comes to production of flavone or other unusual structures in Primula (Bhutia et al.
8
and refs. cited therein). So far, no endophytic fungi have been isolated from Primula species, but endophytic bacteria have been reported to be present in Primula sinensis Lour., without specification of the bacterial genus.
55
Within Primulaceae, the genus Ardisia harbors a leafnodule symbiotic bacteria, Burkholderia, which is involved in specialized metabolism, producing a derivative of the flavonoid epicatechin.
56
This bacterial genus could, however, not be detected in the studied Primula species (Herpell, J.B., pers. comm.).
In a few cases, endophytic bacteria were proven to occur in glandular hairs, such as in Humulus lupulus L. (Cannabaeceae), among others, but those were involved in nitrogen metabolism. 57 In general, bacteria are quite capable of biosynthesizing secondary metabolites, a feature used in biotechnology. 58 Although no specific bacterial strains have been explicitly described from Primula leaves or glands, their presence and involvement in biosynthesis of unusual flavones cannot be excluded.
Function of Farina and Flavone
The discussion of flavone function cannot be separated from consideration about possible function of farina. The amount of farina produced eg in Primula auricula is tightly correlated with the amount of flavone present. It was shown that there is a strong influence of climatic and seasonal factors on the amount and composition of farina, indicating an ecological significance. 18 Ferns are the only other producers of farina known. Their farina, albeit containing other flavonoids than in Primula, is assumed to protect the plant by reflexing excess light 12 or water loss. 59 Farina of the fern Hemionitis sulphurea (Cav.) Christenh. (syn. Notholaena sulphurea (Cav.) J.Sm. (Pteridaceae) exhibited antifungal and molluscicidal activity. 60 The farina may also protect against herbivores, acting as a physical barrier.
Flavone itself might be involved in abiotic interactions, in fight against excessive cold, by enhancing freezing tolerance. 61 Concomitantly with amounts of farina produced, amounts of flavone contained were shown to be habitat and climate correlated, as well as ontology and season dependant, 18 thus representing an ecological character. There are also indications about involvement in biotic interactions. Farina could be washed off by heavy rain and the compounds might get to the soil where they could act as phytotoxic agents. Experiments with garden cress (Lepidium sativum L.; Brassicaceae) showed that flavone does not inhibit germination, but the growth of the seedlings is disturbed. 62 Little is known about the role of flavone in insect-plant relationships. Flavone was found to effectively enhance the larval tolerance of Spodoptera litura against synthetic insecticides by eliciting ROS bursts followed by enhanced antioxidant enzyme activities. At the same time, it shows antifeedant effect by downregulating many protease and lipase genes in Spodoptera. 63 Antifungal activity was assessed eg by Weidenbörner & Jha, 64 and in a preliminary experiment with the fungus Trichoderma, showing effective growth inhibition in a disk diffusion assay (Hinterdobler, pers. comm.).
Finally, flavone might be useful as medical agent. Studies on Feijoa revealed its antibacterial activity. 65 Flavone of Primula macrocalyx Bunge (syn. of Primula veris subsp. macrocalyx) is a remarkable inhibitor of the drug transporter OAT1/3, relating to renal function and blood pressure, 66 while the same compound isolated from Primula boveana Decne. Ex Duby (syn. of Evotrochis involucrata Raf.) exhibited antibacterial activity and it was inhibiting acetyl/butyrylcholinesterase. 67 Further bioactivities include antileishmanial and cytotoxic (cited in Mahran et al. 67 ). It is also known for its anticancer activity, 68 and more explicitly, as tankyrase inhibitor, 69 and for inhibition of tumor growth by inducing apoptosis. 70
Conclusions and Outlook
Despite the amount of literature available on flavone, there are still gaps in our understanding about the accumulation and function of this compound in planta. It is shown here that the ultrastructure of the compartment, in which the compound is stored and/or biosynthesized, is of great importance when it comes to functional aspects. Thus, it is generally recommended to consider the organ- and compartment-specific accumulation of specialized plant metabolites in future studies. Especially in Primula, where flavone contributes in large amounts to the exudate produced from glandular hairs, functional studies need to be extended. Exudates and isolated compounds should also be tested in various ecological bioassays, especially regarding antimicrobial/antifungal and cytotoxic activities. Such studies could link to the role of flavone in natural environments, complemented by common garden experiments. Finally, we have quite a gap of understanding the biosynthesis of flavone. Here, molecular and genetic studies would be helpful, such as mining for biosynthetic enzymes, ideally on isolated glandular hairs.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241258290 - Supplemental material for Flavone From Plants—Uncommonly Common? A Comprehensive Review
Supplemental material, sj-docx-1-npx-10.1177_1934578X241258290 for Flavone From Plants—Uncommonly Common? A Comprehensive Review by Karin Valant-Vetschera, Lothar Brecker and Johann Schinnerl in Natural Product Communications
Footnotes
Acknowledgments
The authors acknowledge NMR Center and MS Center, Faculty of Chemistry, University of Vienna for measuring NMR and MS spectra, respectively. Johannes Theiner, Faculty of Chemistry, University of Vienna is acknowledged for recording the IR spectrum and performing the elemental analysis.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Open access publication was funded by the University of Vienna, Austria.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
