Abstract
Introduction
Acute bronchitis (AB) is very common in clinical practice and is 1 of the top 10 medical diseases, 1 which is an acute inflammation of the tracheobronchial mucosa caused by infections, physical or chemical irritation, or allergic factors, with clinical manifestations dominated by coughing, which may be accompanied by symptoms of upper respiratory tract infections, such as nasal congestion and runny nose. Without timely and effective treatment, it may lead to pneumonia and acute respiratory failure, threatening patients’ lives. 2 Currently, there are fewer animal models for the study of AB, and generally smoke is used, 3 and some of them will use smoke combined with Streptococcus pneumoniae 4 or smoke combined with cold stimulation 2 to model the animals. While there are more clinical applications of research drugs for the treatment of AB, for example, Albrecht et al. 5 revealed the healing-promoting effects of drug components by designing a randomized, 2-armed, placebo-controlled, double-blind, phase IV study that scored the severity of bronchiolitis in patients and compared them with each other. Li et al. 6 assessed the efficacy and safety of the drugs based on the clinical symptoms and adverse events of the patients by using a randomized, double-blind, placebo-controlled trial. Clinically, AB is often treated with antibiotics, but due to the strong drug dependence and side effects of antibiotics on the organism, as many as 60% of antibiotics for AB are prescribed irrationally, and there is an urgent need for more drugs with low side effects for the treatment of AB. Therefore, traditional Chinese medicine (TCM) is gaining more and more attention in the treatment of AB. 7 Maqin Xiaoke Granule (MQ) has unique advantages in the treatment of this disease, which can regulate the body’s constitution and fundamentally reduce the occurrence and recurrence of the disease.
MQ is derived from the clinical prescription of traditional Chinese medicine. It is based on the addition and subtraction of “Maxing Shigan Decoction” included in “Treatise on Febrile Diseases.” It is composed of 15 kinds of traditional Chinese medicine, such as Ephedra (honey-fried), Mori Cortex, Lonicerae Japonicae Flos, and Scutellariae Badix. It has the effects of clearing lung and resolving phlegm to alleviate cough and asthma. Although it has been reported that MQ is commonly used in the treatment of AB and acute exacerbation of chronic bronchitis, which belongs to phlegm-heat stagnation syndrome, it has an inhibitory effect on a variety of bacteria and effectively reduces the toxic and side effects of western medicine. 8 However, few reports have elucidated the mechanism of MQ in the treatment of AB.
Metabolomics is an important part of systems biology,9,10 which mainly studies the changing law of its metabolites after an organism is perturbed, 11 including targeted and nontargeted metabolomics, and nontargeted metabolomics can provide metabolite information through high-throughput screening, which is more suitable for the study of the mechanism of action of traditional Chinese medicines and other multicomponent drugs. 12 The aim of this study was to evaluate the efficacy of MQ in the treatment of AB through the observation of pathological changes in lung tissue and bronchus by hematoxylin-eosin (HE) staining, the analysis of the expression levels of inflammatory factors by enzyme-linked immunosorbent assay (ELISA), and the analysis of the expression levels of relevant mRNAs by real-time fluorescence quantitative polymerase chain reaction (RT-qPCR), and then to investigate the mechanism of action of MQ in the treatment of AB in rats through the screening and characterization of the differential metabolites by the nontargeted metabolomics approach based on ultra high-performance liquid chromatography-quadrupole-time of flight-mass spectrometry/mass spectrometry (UHPLC-Q-TOF-MS/MS), as well as the analysis of the possible metabolic pathways to provide a basis for the further treatment of AB.
Materials and Methods
Animal Experiment
Fifty-six SPF Sprague-Dawley (SD) rats, half male and half female, each weighing 200 to 220 g, were purchased from Chengdu Dashuo Experimental Animal Co., Ltd, with production license number SCXK (Chuan) 2022-030. Animals were free to eat and drink under the conditions of temperature of 18 to 24 °C, humidity of 45% to 55%, 12 h light/dark alternation, and air circulation.
Sixteen rats were randomly divided into blank control group (C) and blank Maqin group (B-M; MQs, 2.4 g/kg, crude drug/weight; Tianchang Yifan Pharmaceutical Co., Ltd, 221207), with 8 rats in each group, and the remaining rats were subjected to modeling of AB. The modeled rats were placed in a 38 × 28 × 22 cm glass box, and 2.3 g of tobacco (Chengdu Tobacco Company) and 0.20 g of dried chili pepper (Chengdu) were lit on fire, and smoked with unburned smoke once a day in the morning and once in the evening, each time for 30 min, for 21 days. In addition, rats were treated intranasally with Streptococcus pneumoniae (BNCC, Beina Chuanglian Biological Technology Co., Ltd.) 0.10 mL, 4 twice a week, for a total of 6 times. When the model rats showed cough, runny nose, squint, shortness of breath, piled up, listlessness, weight loss, and other symptoms, indicating that the AB model was successfully established. 2
From the 11th day, all rats with successful modeling were randomly divided into model group (M) and high-dose Maqin group (MQ-H; 4.8 g/kg, crude drug/weight), medium-dose Maqin group (MQ-M; 2.4 g/kg, crude drug/weight), low-dose Maqin group (MQ-L; 1.2 g/kg, crude drug/weight), positive drug group (P; Amoxicillin Capsules, H34023532; Anhui Anke Hengyi Pharmaceutical Co., Ltd), 8 rats in each group. The blank control group and the model group were given distilled water 1.0 mL/100 g every day, and the other groups were given the same volume of corresponding drugs orally every day under the same conditions, once a day for 10 consecutive days.
Sample Collection and Processing
The rats were fasted overnight, but free to drink water. On the 22nd day, the rats were anesthetized and blood was taken from the abdominal aorta and placed at room temperature for 1 h. Serum was collected after centrifugation at 12 000 r/min for 10 min at 4 °C. A portion was taken for ELISA test; in the other part, 100 μL of serum was taken and placed in a 1.5 mL EP tube, and 400 μL of acetonitrile: methanol (1:1) was added. The protein was precipitated by vortex for 30 s. After ultrasonication in an ice water bath for 10 min, it was placed in a refrigerator at−20 °C for 1 h, and centrifuged at 12 000 r/min and 4 °C for 15 min. The supernatant was transferred and blown dry with N2 and placed in a refrigerator at −80 °C for testing.
Bronchial and lung tissues were collected. The fourth lobe of the right lung was taken to prepare lung homogenate in an ice water bath, and the supernatant was collected after centrifugation at 4 °C and 12 000 r/min for 10 min. The remaining right lung tissue was stored in a refrigerator at −80 °C for subsequent RT-qPCR analysis. The left lung and bronchus of the rats were collected and fixed with 4% paraformaldehyde (Biosharp BL539A) for more than 24 h for histopathological examination.
Histopathological Analysis
Fixed lung tissues and bronchial tubes of 6 rats in each group were randomly selected, dehydrated and embedded in paraffin wax and cut into 3 µm thick slices, dewaxed and dehydrated, the slices were stained with HE, and the dehydrated and transparent sealing slices were examined for histopathological changes under a light microscope. The lung histopathologic damage score was calculated using a method developed by Peng13,14: 0, no damage; 1, less than 25% damage; 2, between 25% and 50% damage; 3, between 55% and 75% damage; and 4, more than 75% damage. Bronchial pathologic damage score: 0, bronchial epithelium without hyperplasia and inflammation, normal bronchus; 1, bronchial epithelium with mild segmental hyperplasia, insignificant inflammation, and structurally normal; 2, bronchial epithelium hyperplasia surrounded by inflammation and vascular congestion; and 3, bronchial epithelium markedly hyperplasia surrounded by diffuse inflammation and vascular congestion. The lung tissue score plus the bronchial score was the final score.
Determination of Inflammatory Factors
The levels of tumor necrosis factor-α (TNF-α, EK382/3-96, MULTISCIENCES), interleukin-6 (IL-6, EK306/3-96, MULTISCIENCES), interleukin-1β (IL-1β, EK301B/3-96, MULTISCIENCES), and interleukin-10 (IL-10, EK310/2-96, MULTISCIENCES) in serum and lung homogenate of rats in each group were detected according to the instructions of ELISA kit.
RT-qPCR Detection of mRNA Levels of NOD-Like Receptor Thermal Protein Domain Associated Protein 3 and p38 Mitogen-Activated Protein Kinase in Lung Tissue
Three samples were randomly selected from each group for RT-qPCR detection, and 100 mg of rat lung tissue was taken from each sample. Trizol was used to extract total RNA from the lung tissue of each group and detect the concentration. RNA was reverse transcribed into cDNA and amplified. Reaction system: 20 μL, cDNA 2 μL, iTaqTM universal SYBR Green supermix (2×) 5 μL, upstream and downstream primers (Table S1) each 1 μL, H2O 10 μL. Reaction conditions: Predenaturation at 95 °C for 5 min; denaturation at 95 °C for 10 s, annealing at 60 °C for 30 s, and extension at 72 °C for 30 s, for a total of 40 cycles. The relative expression of target genes was calculated by 2−ΔΔCt method using GAPDH as an internal reference.
Nontargeted Metabolomics Analysis
Pretreatment of serum Samples
The serum residue was redissolved with 100 μL of methanol, vortexed for 60 s, centrifuged at 12 000 r/min for 10 min, and the supernatant was transferred to a vial for detection. An equal amount of 10 μL of each serum sample in each group was taken and mixed as a quality control sample.
UHPLC-Q-TOF-MS/MS Analysis
LC-MS/MS analysis was performed using an ultra high performance liquid chromatography (UHPLC) LC-30A system (Shimadzu) equipped with a TripleTOF 6600 mass spectrometer (Sciex). Xbridge BEH amide column (100 mm × 2.1 mm, 1.7 mm inner diameter) (Waters) was used to separate samples. The chromatographic conditions were as follows: Water-acetonitrile-ammonium acetate-ammonium hydroxide (solvent A, 100%/0%/25 mM/10 mM; solvent B, 0%/100%/0 mM/0 mM) gradient elution (0-2 min, 95% B; 2-18 min, 95%-65% B; 18-20 min, 65%-40% B; 20-22 min, 40% B; 20-20. 1 min, 40%-95% B; 22.1-30 min, 95% B). The flow rate was 0.3 mL/min, the column temperature was 30 °C, and the injection volume was 1 μL. The mass spectrometry conditions were electrospray ionization source (ESI) with positive-negative ion mode detection; ion voltage, 4500 V; curtain gas, 35 lb/in2; 55 lb/in2 for both the nebulizer gas and the heated gas; ion source temperature, 550 °C; de-clustering potential, 80 V; and each scanning cycle consisted of 1 time-of-flight mass spectrometry (MS) survey scan and 15 MS/MS scans. The detection mode was the information-dependent data acquisition mode, with multiple mass deficit filtering and dynamic background subtraction as conditions that triggered the second level, and compliance with which was prioritized for the second level scan. Convert wiff format to mgf format via MS Convert. 15
Metabolite Identification and Analysis
According to the method of 2.6.2, the total ion current diagram of serum in positive and negative ion modes was obtained (Figure S1). The related compound information such as compound name was obtained by matching and analyzing the cleavage rule with MetDNA platform (Metabolite identification and Dysregulated Network Analysis, Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, CAS), and the processed data matrix was imported into SIMCA 14.1 software. Principal component analysis (PCA) in unsupervised mode and orthogonal projections to latent structures discriminant analysis (OPLS-DA) in supervised mode were performed. Combined with fold change (FC) > 1.5, variable important in projection (VIP) > 1, t test P < .05, the differential metabolites between the blank control group and the model group, the model group and the positive group, the positive group and the MQ-H group were screened. The model was verified by 200 response permutation tests. Then, based on the OPLS-DA model, the volcano plot was used to visualize the FC and P values. Clustering heat mapping was performed using Origin 9.5.1 software, followed by correlation analysis and enrichment of in vivo metabolic pathways involved in differential metabolites using MetaboAnalyst 5.0.
Statistical Analysis
GraphPad Prism 8.0 software was used for statistical analysis and plotting, and the measurement data were expressed as mean ± standard deviation (mean ± SD). Normality was determined using the Shapiro-Wilk test, and the t’ test was used to analyze when variance was not uniform. One-way analysis of variance was used for between-group analysis, with P < .05 indicating that the difference was statistically significant.
Result
the Effect of MQ on Bronchus and Lung in Acute Bronchitis rat Model
The lung tissue structure of the rats in the blank control group was clear, no inflammatory cell infiltration, no congestion, and the bronchi were normal. In the model group, the mucosal epithelial cells in the tissue were necrotic and exfoliated, and a large number of cell structures were seen in the cavity; the interstitial and alveolar consolidation around the bronchi were obvious, the alveolar structure disappeared, and different sizes of cavity structures and focal nodules were formed, and lymphocytes were seen in some cases. The interstitium around the blood vessels and bronchi were widened, accompanied by diffuse infiltration of inflammatory cells and diffuse hemorrhage in the alveolar cavity. Compared with the model group, the necrosis and shedding of epithelial cells in the MQ-L group were reduced, the infiltration of inflammatory cells was reduced, the interstitial broadening and damage were weakened (P < .05), but some cells were shed. In the positive group, MQ-H and MQ-M groups, the necrosis and shedding of epithelial cells were reduced, and the lumen was obvious. Alveoli rarely seen honeycomb structure, consolidation decreased significantly; the repair is better, but a few visible smooth muscle hyperplasia, vascular wall thickening. It can be seen that medium and high doses of MQ and positive control drugs reduced tissue damage caused by smoke combined with Streptococcus pneumoniae (P < .01, .001, .0001) (Figure 1).

He staining image ( × 400) and score histogram (mean ± SD, n = 6) of bronchus and lung tissue of rats in each group, scale = 25 μm; red arrows represent red blood cell exudation; black arrows represent interstitial widening; green represents lymphocytes; yellow represents vascular wall thickening, smooth muscle hyperplasia; blue represents bronchial epithelial cell deformation; orange represents cell shedding; compared with the blank control group, ####P < .0001 ; compared with the model group, *P < .05, **P < .01, ***P < .001, ****P < .001.
Regulation of Inflammatory Factor Expression by MQ in a rat Model of Acute Bronchitis
The levels of inflammatory factors were determined by animal experiments. The results showed that compared with the blank control group, the levels of TNF-α, IL-6, and IL-1β in the serum of the model group were significantly increased (P < .05, P < .05, P < .0001), and the levels of IL-10 were significantly decreased (P < .0001). Compared with the model group, the levels of TNF-α, IL-6, and IL-1β in the serum of each administration group were significantly decreased (P < .05, P < .01, P < .001), and the level of IL-10 was significantly increased (P < .01, P < .0001) (Figure 2A).

(A) is the histogram of serum inflammatory factors (mean ± SD, n = 7), and (B) is the histogram of lung homogenate inflammatory factors (mean ± SD, n = 6). One-way analysis of variance was used for statistical analysis. Compared with the blank control group, #P < .05, ##P < .01, ###P < .001, ####P < .0001; compared with the model group, *P < .05, **P < .01, ***P < .001, ****P < .001.
Compared with the blank control group, the levels of TNF-α, IL-6 and IL-1β in the lung homogenate of the model group were significantly increased (P < .001, P < .0001), and the level of IL-10 was significantly decreased (P < .01). Compared with the model group, the lung homogenate levels of MQ-H in each administration group and the levels of TNF-α, IL-6, and IL-1β in the positive group were significantly decreased (P < .05, P < .01, P < .001, P < .0001) (Figure 2B).
RT-qPCR Analysis
The results of RT-qPCR showed that compared with the blank control group, the mRNA expression of NOD-like receptor thermal protein domain associated protein 3 (NLRP3) and p38 mitogen-activated protein kinase (p38 MAPK) in the lung tissue of the model group was significantly increased (P < .05). Compared with the model group, the mRNA expression of NLRP3 and p38 MAPK in the lung tissue of rats in the positive group, MQ-H group and B-M group was significantly decreased (P < .05, P < .01) (Figure 3).

The mRNA expression levels of NLRP3 and p38-MAPK in lung tissues of rats in each group (mean ± SD, n = 3). One-way analysis of variance was used for statistical analysis. Compared with the blank control group, #P < .05; Compared with the model group, *P < .05, **P < .01. Abbreviations: NLRP3, NOD-like receptor thermal protein domain associated protein 3; p38 MAPK, p38 mitogen-activated protein kinase.
Multivariate Statistical Analysis
In the PCA model, the metabolites of the model group and the blank control group were aggregated in a certain area in the positive ion mode and the negative ion mode, with good differentiation, indicating that the metabolites of the model group and the blank control group were significantly different (Figure 4A). Further between-group comparisons using OPLS-DA statistics revealed that the groups could also be significantly separated. The OPLS-DA model was tested with 200 permutations, and the intercepts of the vertical coordinates of Q2 were all less than 0. The results showed that the model was not overfitted and had good stability and predictive ability (Figure 4B and C). The volcano plot identifies the differential metabolite screening results with dot colors, where significantly upregulated metabolites are identified with red dots, significantly down-regulated metabolites with green dots, and nonsignificantly different metabolites with gray dots (Figure 4D).

Multivariate statistical analysis. PCA is shown in (A), OPLS-DA in (B), 200 permutation test in (C), and volcano diagram in (D). Abbreviations: PCA: principal component analysis; OPLS-DA, orthogonal projections to latent structures discriminant analysis.
Differential Metabolite Analysis
The significant parameters (VIP, FC, P) obtained by univariate statistical analysis and multivariate analysis were used to set the differential metabolite screening conditions of VIP > 1, FC > 1.5, and P < .05, and the serum metabolites were screened in the positive and negative ion modes in each group of rats, and the differential metabolites that were statistically different and biologically significant between the groups were obtained (Table S2). By comparing the blank control and model groups, 18 differential metabolites were finally screened from the serum. Among them, 7 metabolites were up-regulated and 11 metabolites were down-regulated. By comparing the model and positive groups, 7 metabolites were up-regulated and 5 metabolites were down-regulated; among them, Car (18:1) was significantly regressed after amoxicillin treatment. In order to more visually represent the levels of changes in rat serum differential metabolites in the blank control and model groups, and in the model and administered groups, the results of the obtained analyses were presented in the form of heat maps. The differential metabolites in each group were subjected to Pearson correlation analysis to obtain the correlation between differential metabolites between groups (Figure 5).
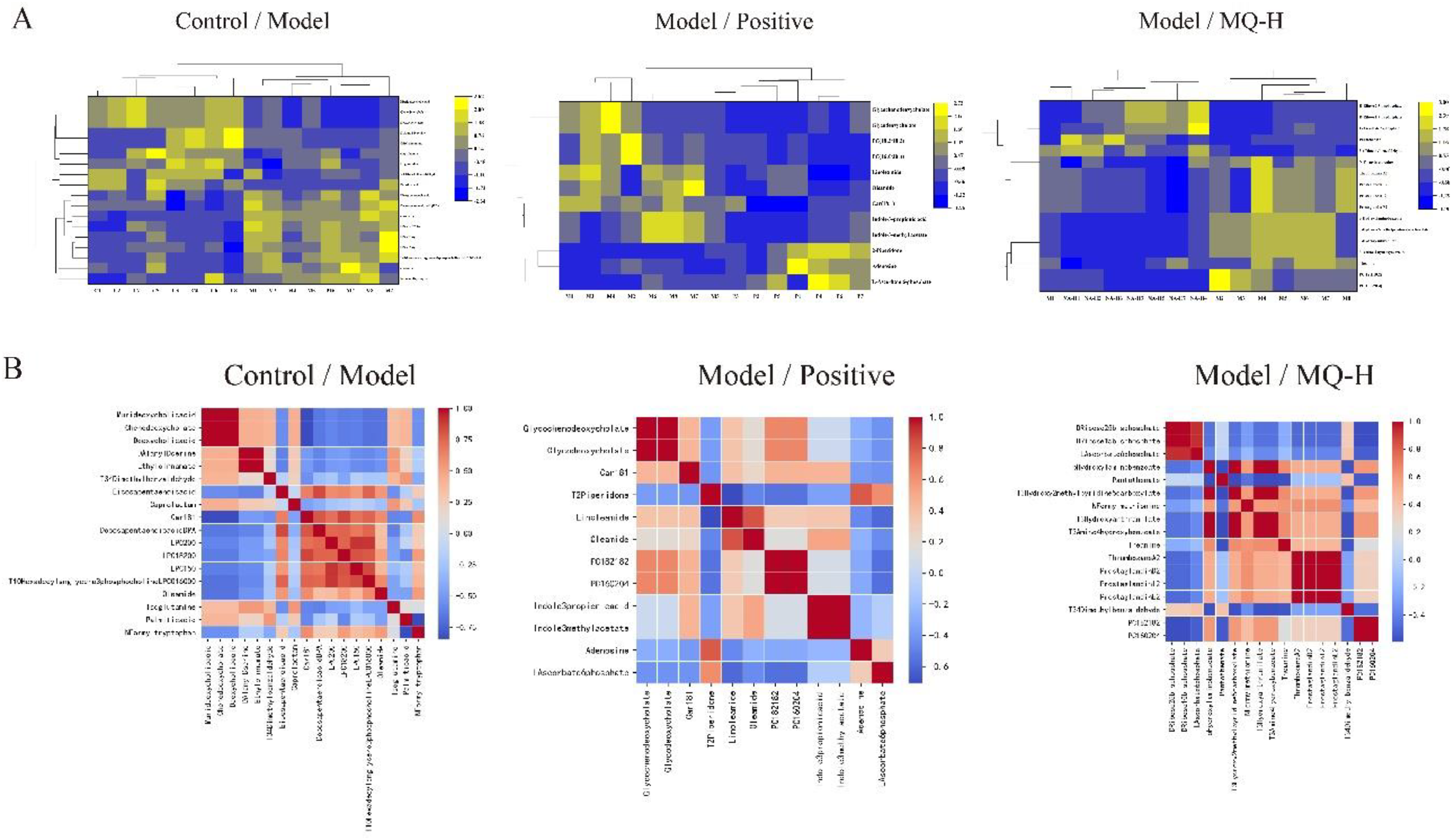
(A) is a heat map, blue indicates that the substance is relatively down-regulated, and yellow indicates that the substance is relatively up-regulated; (B) is a correlation analysis, where blue color indicates a poor correlation between substances and red color indicates a good correlation between substances.
Pathway Enrichment
The screened differential metabolites were subjected to metabolic pathway analysis, and the results are shown in Figure 6. Five metabolic pathways were obtained from enrichment analysis in the blank control group and the model group, and 6 metabolic pathways were obtained from the other 2 groups, respectively. MQ can regulate glycerophospholipid metabolism, tryptophan metabolism, arachidonic acid metabolism, linoleic acid metabolism, alpha-linolenic acid metabolism, Pantothenate and CoA biosynthesis pathways to reduce inflammatory response. Among them, glycerophospholipid metabolism was involved in all 3 groups and had the highest impact factor, suggesting that it is the predominant metabolic pathway.

From left to right, there were comparison between blank control group and model group, comparison between model group and positive group, comparison between model group and high-dose Maqin group (MQ-H) group.
Discussion
Clinical treatment for AB will use antibiotics, 16 but antibiotics have a certain degree of resistance, 17 and some studies have shown that the beneficial effect of antibiotics on AB is minimal, 1 TCM and other alternative therapies can be used in the treatment of AB in order to reduce or delay the use of antibiotics, 18 in contrast, Chinese herbal medicines have fewer side-effects and are willing to be taken by people, which is being increasingly recognized and accepted by many people. 19
Inflammation is a physiological response of the immune system required to clear pathogenic bacteria, and its basic pathological changes include localized tissue degeneration, exudation, and proliferation. The 3 are interrelated, with degeneration being the damaging process and exudation and proliferation being the antidamaging and repairing process. Under the stimulation of inflammatory factors or certain physicochemical factors, the macrophages in the inflammatory localization may undergo proliferation. On the one hand, inflammation is activated by pro-inflammatory factors, such as TNF-α, interleukin-18 (IL-18). On the other hand, anti-inflammatory factors inhibit inflammation, such as interleukin-4 (IL-4), IL-10. The outcome of inflammatory diseases is determined by a combination of pro-inflammatory and anti-inflammatory factors. Studies have shown that IL-6 is secreted mainly by macrophages and is a marker of systemic inflammation, 20 and IL-10 prevents macrophages from producing IL-6. 21 In this study, the content of IL-10 in the administration group was significantly increased, while the content of IL-6 and TNF-α was significantly decreased. It may be that IL-10 prevents macrophages from producing pro-inflammatory factors, thereby inhibiting the body's inflammatory response. The NLRP3 inflammasome is a multiprotein intracellular innate immune sensor composed of NLRP3, apoptosis-associated speck-like protein (ASC), and pro-caspase-1, with NLRP3 predominating. NLRP3 inflammatory vesicles are activated in respiratory infections and lung diseases and subsequently activate Caspase-1 by recruiting ASC, and activated Caspase-1 further activates IL-1β and IL-18. 22 In this study, compared with the blank control group, the level of pro-inflammatory factor IL-1β in the model group was significantly increased, and the results of RT-qPCR showed that the mRNA expression level of NLRP3 in the model group was also significantly increased. It may be that the NLRP3 inflammasome is activated, and then mature IL-1β is produced to cause inflammation in the body. p38 MAPK is one of the MAPK family members, cell differentiation, proliferation, inflammation, and death related, 23 which can mediate the inflammatory response by regulating the expression of a variety of inflammatory cytokines such as IL-1β, IL-6, and TNF-α. 24 In this study, histopathological results showed that a large number of inflammatory cells infiltrated in the bronchi and alveoli after modeling, and several diffuse necrosis foci were observed. The alveolar wall was thickened, diffuse edema was obvious, and interstitial congestion was enhanced. The levels of pro-inflammatory factors TNF-α, IL-1β, and IL-6 were significantly increased by smoke and Streptococcus pneumoniae stimulation. This coincided with the pathological observations of lung tissue and bronchitis, proving the success of this model establishment. And after treatment with amoxicillin and MQ in the AB model in this study, the degree of cellular damage was reduced and the inflammatory cells became significantly less. It is proved that the intervention with MQ can significantly reduce the degree of pathologic damage of bronchial and lung tissues caused by smoke combined with Streptococcus pneumoniae in rats.
The Maxing Shigan decoction is a famous prescription of traditional Chinese medicine, which is contained in the famous Chinese medicine book “Treatise on Febrile Diseases” written by Zhang Zhongjing 1800 years ago, and it has good safety and effectiveness in clinical practice, and is commonly used to treat colds, upper respiratory tract infections, AB, pneumonia and so on.25,26 Glycyrrhizin in Maxing Shigan Decoction can regulate IL-6, NF-κB, and other transcription factors and anti-inflammatory mechanism. 27 MQ is based on the addition and subtraction of “Maxing Shigan Decoction,” which mainly uses Ephedra and Mori Cortex as the monarchs, and the combination of these 2 herbs can promote the lungs, relieve coughs, and stabilize wheezing. Ephedra is one of the oldest Chinese herbs, belonging to the Ephedra family, and is usually used to treat asthma, bronchitis, cough, etc It is also bacteriostatic and antipyretic. 28 A study validated the inhibitory effect of Ephedra on pro-inflammatory factors by molecular docking of 5 major human anti-inflammatory cytokine receptors (IL-6, hybrid TLR4, TNF-α, IL-1β, and extracted TLR4). 21 Mori Cortex is an edible traditional Chinese medicine with antibacterial, anti-inflammatory, and treatment of asthma. Studies have shown that after treatment with Mori Cortex, IL-1β inflammatory factors in rats were significantly reduced, and the activation of NLRP3 was attenuated. 29 Therefore, the mechanism of action of MQs may be to inhibit proinflammatory factors through Ephedra, or to inhibit the activation of NLRP3 inflammasome by Mori Cortex, and then inhibit the production of mature IL-1β, thereby reducing the body's inflammation. Currently, some studies have utilized Western blot assay to investigate the protein expression related to MQ treatment of chronic bronchitis, and speculated that the mechanism of action of MQs on smoke-induced chronic bronchitis may be related to the down-regulation of the TLR4/NF-κB signaling pathway as well as the upregulation of the Nrf2/HO-1 signaling pathway. 8 No one has analysed the mechanism of action of MQ in the treatment of AB, so this study has some practical significance.
Glycerophospholipid, one of the major classes of lipids, is the main component of cell membranes and directly maintains the physiological functions of cells. Studies have shown that glycerophospholipid (PC (18:0/18:00), PC (16:0/16:0), etc) is altered in PM 2.5-exposed rats, with PC (16:0/16:0) significantly elevated. 30 As can be seen in the metabolomics results of this study, both PC (16:0/20:4) and PC (18:2/18:2) were significantly upregulated in the positive group, MQ-H, after administration of the treatment, suggesting that MQ treatment of AB is closely related to glycerophospholipid metabolism pathway. 6-Benzylaminopurine can regulate lipid metabolism by altering genes associated with fatty acid and glycerophospholipid metabolism 31 ; subacute cadmium disrupts intestinal microecological balance and glycerophospholipid metabolic homeostasis. 32 In addition, there is growing evidence that inflammation is related to glycerophospholipid metabolism. Allergic asthma involves a variety of inflammatory cells PD00 can modulate glycerophospholipid and purine metabolic pathways to reduce allergic asthma inflammation, thereby improving the pathophysiological features of asthma 33 ; Dietary β-CG induced enterocolitis in different intestinal segments and altered glycerophospholipid metabolism and arginine biosynthesis pathways 34 ; OGPC down-regulated TNF-α induced IL-6 secretion in vitro, and OGPC associated with glycerophospholipid metabolism pathway was significantly reduced, and glycerophospholipid metabolism was the main metabolic pathway for IL-6 differential expression in rheumatoid arthritis patients 35 ; Sepsis is a severe inflammatory disease, and STV-Na significantly alters the synthesis of glycerophospholipid metabolism, among others thereby modulating inflammation in sepsis 36 ; by altering glycerophospholipid metabolism pathways, Herba patriniae significantly modulates lipid metabolism and inflammation levels, demonstrating the potential for antiatherosclerotic development. 37 At present, there is no report that AB is related to glycerophospholipid metabolism. Therefore, this study can provide some reference for the related research of AB.
In this paper, metabolomics and RT-qPCR were combined to start from endogenous metabolites and validation of genes related to AB, respectively, and then to elaborate the pathway of MQ treatment of AB, which can more comprehensively respond to the process and mechanism of drug treatment of the disease. However, due to limited funding, this study only did nontargeted metabolomics and validated at the gene level, which has some limitations. Nontargeted metabolomics is a comprehensive and systematic analysis of endogenous metabolites in organisms, which is an unbiased metabolomics analysis, and it can detect a large number of metabolite signals at the same time, but the sensitivity and qualitative and quantitative accuracy are poorer than that of targeted metabolomics. Subsequently, the proteins related to AB can be verified at the protein level and the glycerophospholipid pathway can be further studied and analyzed on the basis of targeted metabolomics, in order to provide further reference for drug treatment of AB. In addition, in this study, 1 rat in the MQ-H group died on day 17, and 1 rat in each of the positive and MQ-M groups died on day 19, and the cause of death was confirmed anatomically to be due to improper gavage. However, since the model had been completed, it was not suitable to replace the rats, the results of PCA and OPLS-DA in metabolomics would be less than 1 rat each.
Conclusion
In summary, in this study, a rat model of AB induced by mixed smoke combined with Streptococcus pneumoniae was successfully established. MQ can reduce the inflammatory response of the body by inhibiting the levels of proinflammatory factors (TNF-a, IL-6, IL-1β), increasing the levels of anti-inflammatory factors (IL-10), reducing the mRNA expression of NLRP3 and p38 MAPK, and improving glycerophospholipid metabolism.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241247988 - Supplemental material for Maqin Xiaoke Granule Alleviates Acute Bronchitis Induced by Smoking Combined With Streptococcus pneumoniae by Regulating NLRP3/p38 MAPK/Glycerophospholipid Pathway
Supplemental material, sj-docx-1-npx-10.1177_1934578X241247988 for Maqin Xiaoke Granule Alleviates Acute Bronchitis Induced by Smoking Combined With Streptococcus pneumoniae by Regulating NLRP3/p38 MAPK/Glycerophospholipid Pathway by Wanxia Men, Ruipeng Liu, Min Yang, Yang Xu, Yanli Lei and Tao Liu in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X241247988 - Supplemental material for Maqin Xiaoke Granule Alleviates Acute Bronchitis Induced by Smoking Combined With Streptococcus pneumoniae by Regulating NLRP3/p38 MAPK/Glycerophospholipid Pathway
Supplemental material, sj-docx-2-npx-10.1177_1934578X241247988 for Maqin Xiaoke Granule Alleviates Acute Bronchitis Induced by Smoking Combined With Streptococcus pneumoniae by Regulating NLRP3/p38 MAPK/Glycerophospholipid Pathway by Wanxia Men, Ruipeng Liu, Min Yang, Yang Xu, Yanli Lei and Tao Liu in Natural Product Communications
Supplemental Material
sj-docx-3-npx-10.1177_1934578X241247988 - Supplemental material for Maqin Xiaoke Granule Alleviates Acute Bronchitis Induced by Smoking Combined With Streptococcus pneumoniae by Regulating NLRP3/p38 MAPK/Glycerophospholipid Pathway
Supplemental material, sj-docx-3-npx-10.1177_1934578X241247988 for Maqin Xiaoke Granule Alleviates Acute Bronchitis Induced by Smoking Combined With Streptococcus pneumoniae by Regulating NLRP3/p38 MAPK/Glycerophospholipid Pathway by Wanxia Men, Ruipeng Liu, Min Yang, Yang Xu, Yanli Lei and Tao Liu in Natural Product Communications
Footnotes
Acknowledgments
The authors thank the editor and the reviewers for their useful feedback that improved this paper.
Authors’ Contributions
WXM and RPL contributed to design, conceptualization, formal analysis, data curation, and writing—original draft; MY contributed to the investigation; YX contributed to visualization; YLL contributed to resources; TL contributed to supervision, validation, and writing—review and editing.
Data Availability
Data will be provided by the corresponding author upon a reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was reviewed by the Ethics Committee of Chengdu University and considered to meet the relevant requirements of animal experimental ethics. The approval number was FLD202301-03.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Sichuan Province Central Guide Local Development Fund project (grant number: 23ZYZYTS0039); Sichuan Scientific and Technological Achievements Transfer and Transformation Demonstration Project (grant number: 2023ZHCG0071).
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
