Abstract
Introduction
Among known plants, the genus Buxus has attracted significant attention from scientists. Over 200 compounds have been isolated from this genus since the 1990s, including alkaloids, triterpenoids, steroids, flavonoids, coumarins, lignans, and other types.1,2 While Buxus species are widely recognized for their ornamental significance, a number of scientific reports have emphasized their ethnomedicinal properties. Additionally, the biological activities of compounds isolated from this genus have been proven to be highly diverse, such as acetyl- and butyrylcholinesterase inhibitory, antimicrobial, antifungal, antiemetic, antispasmodic, cytotoxic activities, and so on. 3
Vietnam is a tropical monsoon country that is home to more than 5,000 known medicinal plants. Among those, Buxus latistyla Gagnep., also known as “Cà mà vòi to” in Vietnamese, is found in central Vietnam and has been traditionally utilized by local communities to treat various illnesses such as malaria, hemoptysis, amebiasis, and edema. 4 However, its chemical constituents and biological activities remain incomplete. Therefore, in the pursuit of discovering new compounds with biological potential, particularly in cancer treatment, we report herein the isolation and structural elucidation of new compounds as well as their cytotoxicity toward three cancer cell lines, including MCF-7, SK-LU-1, and SW480, along with a normal cell line, HEK-293A, utilizing the sulforhodamine B assay.
Results and Discussion
The chromatographic separation resulted in the isolation of seven compounds (

Chemical structures of compounds
The 1H NMR spectrum of
1H (500 MHz) and 13C (125 MHz) NMR data of compounds

Assignments were accomplished by HSQC, HMBC, COSY, and NOESY experiments.
aOverlapping signals.
Analysis of the 13C NMR and HSQC spectra of
Indeed, three 1H-1H COSY spin systems [C(5)H-C(6)H2-C(7)H2; C(9)H-C(11)H2-C(12)H2-C(13)H-C(18)H-C(19)H-C(21)H2-C(22)H2; C(15)H2-C(16)H2] and important HMBC correlations, including H3-23 (δH 1.00)/H3-24 (δH 1.03) with C-4 (δC 42.7)/C-5 (δC 54.0), H3-25 (δH 1.07) with C-5/C-9 (δC 44.2)/C-10 (δC 48.5), H3-26 (δH 1.07) with C-7 (δC 34.3)/C-8 (δC 42.8)/C-9/C-14 (δC 42.9), H3-27 (δH 0.98) with C-8/C-13 (δC 38.1)/C-14/C-15 (δC 27.8), H3-28 (δH 0.80) with C-16 (δC 35.6)/C-17 (δC 43.1)/C-18 (δC 48.3)/C-22 (δC 40.0), and H2-29 (δH 4.56 and 4.68) with C-19 (δC 47.9)/C-30 (δC 19.3) confirmed the 1-norlupane-type skeleton in
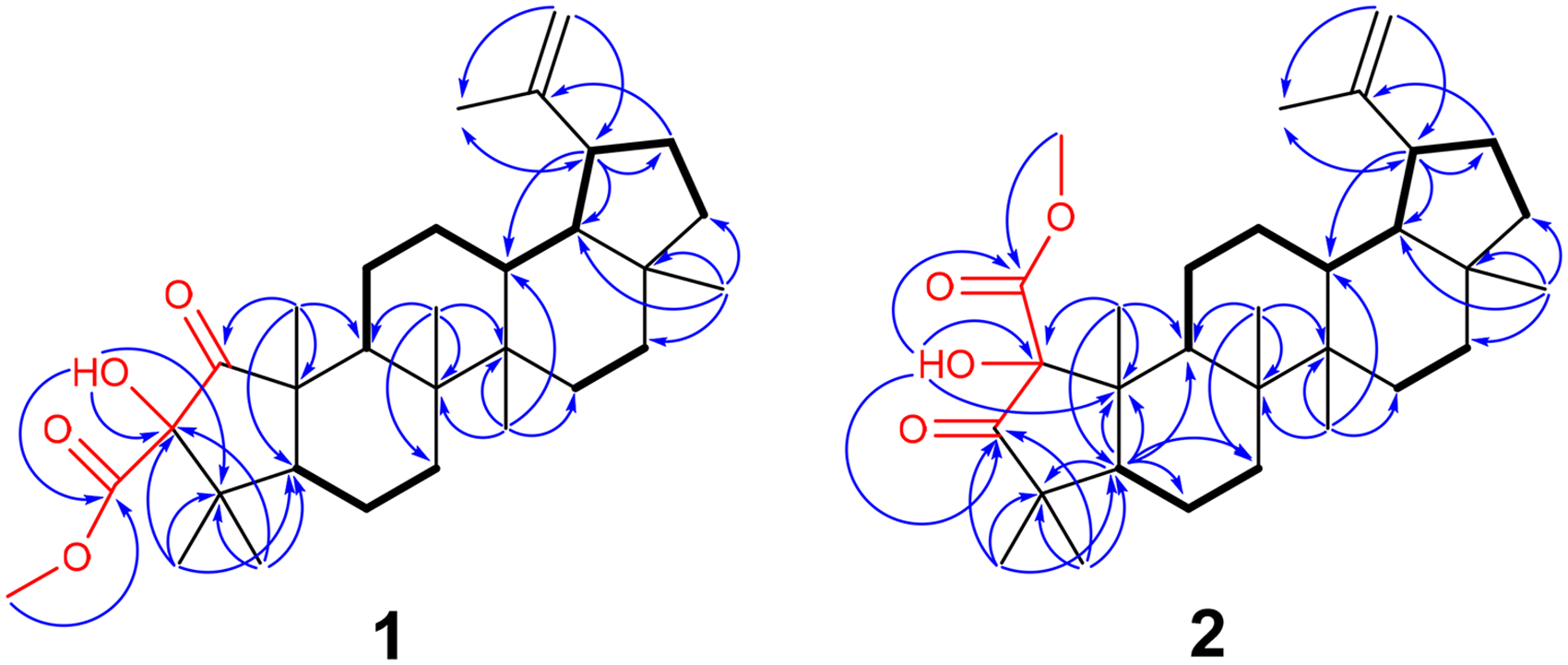
Key HMBC (1H→13C, arrows) and COSY (bold lines) correlations of compounds
The stereochemistry of

Key NOESY correlations (dashed arrows) of compounds
Compound
The cross-peak between 1-OH (δH 3.36) and H3-25 (δH 0.90) was detected in the NOESY spectrum (Figure 3). Therefore, the β-orientation was assigned for both 1-OH and Me-25. Meanwhile, the key NOESY correlation of 1-COOMe (δH 3.81) to H3-27 (δH 0.95) allowed the unambiguous assignment of α-orientation for carbomethoxy and Me-27 groups. The stereo-structure of the A-ring was supported by the good agreement of chemical shift values of C-1 (δC 89.9), C-2 (δC 171.1), C-3 (δC 220.5), C-4 (δC 44.3), C-5 (δC 50.7), C-6 (δC 17.5), C-10 (δC 49.5), C-23 (δC 26.8), C-24 (δC 22.3), C-25 (δC 13.9) (in
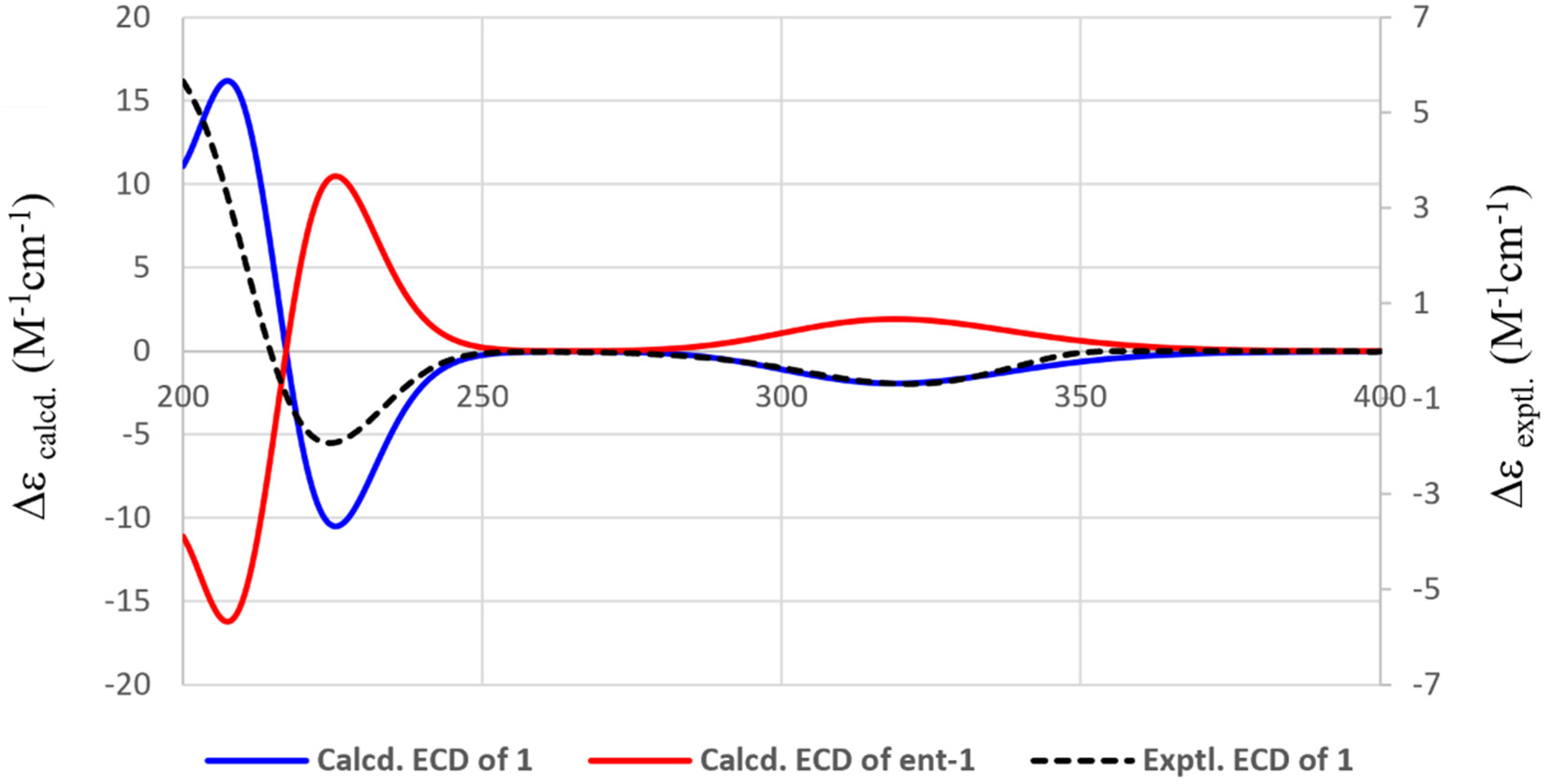
Experimental and calculated ECD spectra of compound
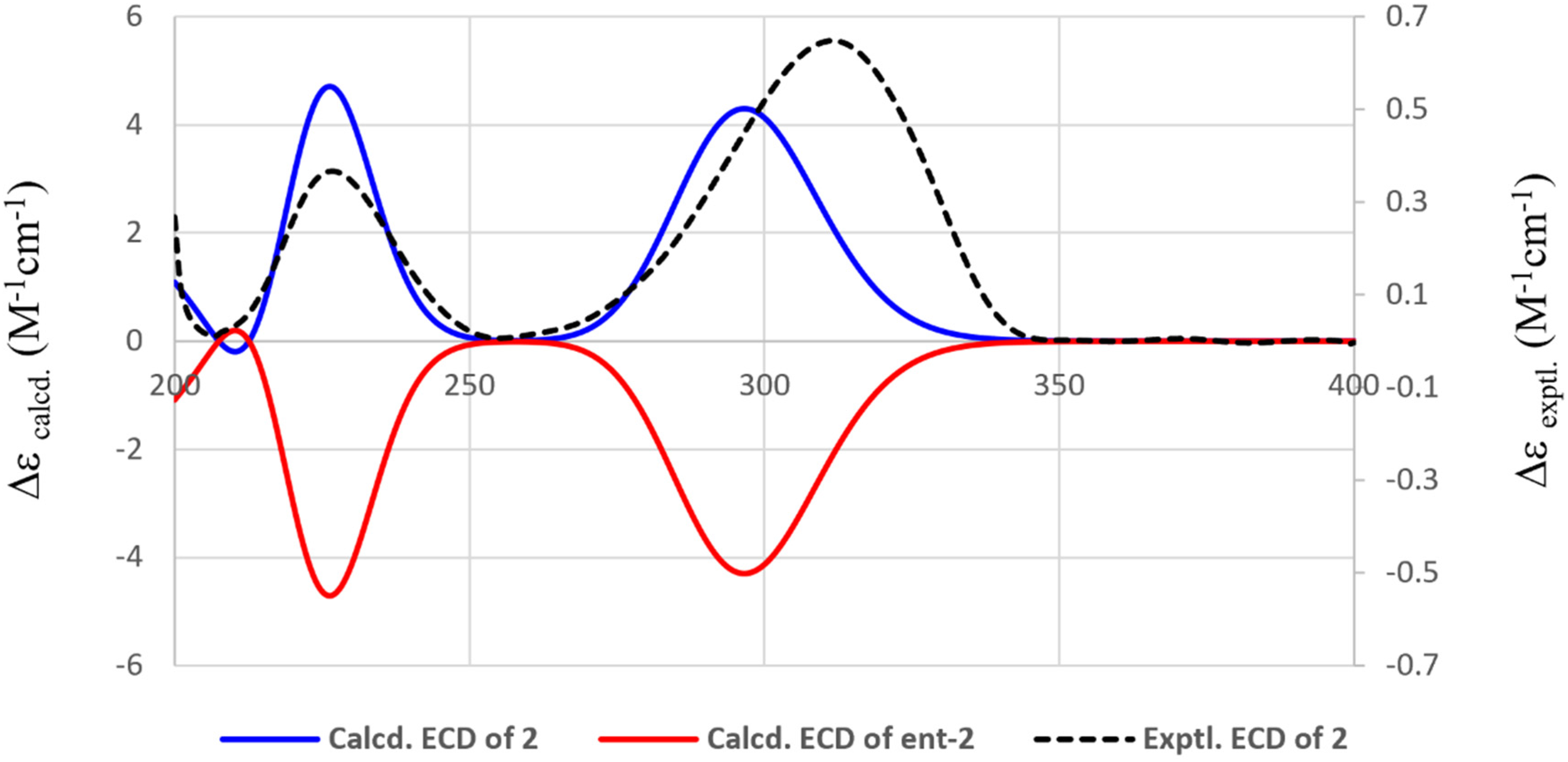
Experimental and calculated ECD spectra of compound
Finally, based on NMR spectroscopic data and the comparison with published literature, the remaining compounds were identified as lupeol (
The isolated compounds were evaluated for their cytotoxicity toward human cells, including MCF-7 (breast carcinoma), SK-LU-1 (lung carcinoma), SW480 (colon carcinoma) cancer cells, and a normal cell line (HEK-293A, human embryonic kidney cells) (Table 2). Among all tested cell lines, compound
Cytotoxic Effect of Compounds
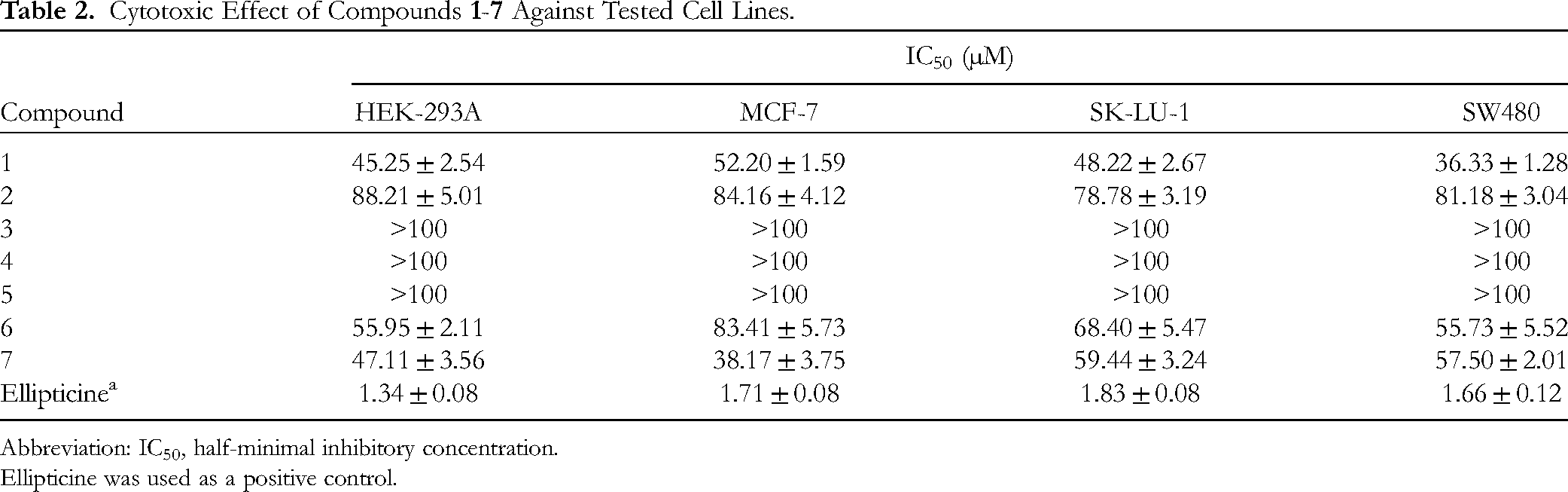
Abbreviation: IC50, half-minimal inhibitory concentration.
Ellipticine was used as a positive control.
Furthermore, compounds
The proposed biosynthetic pathways of compounds

Plausible biosynthetic pathways of compounds
Conclusion
Phytochemical investigation of the stems and leaves of B. latistyla Gagnep. led to the isolation of two new lupane-type derivatives (
The results of the cytotoxic assay might suggest the relatively selective effect of compound
Materials and Methods
General Experimental Procedures
Column chromatography was carried out using silica gel (60 N, spherical, neutral, 40–50 µm, Kanto Chemical Co., Inc.), Cosmosil 75C18-OPN (Nacalai Tesque Inc.), YMC RP-18 (Fuji Silysia Chemical Ltd.), Sephadex LH-20 (Dowex® 50WX2-100, Sigma-Aldrich), and Diaion HP-20 (Mitsubishi Chem. Co., Tokyo, Japan). Precoated silica gel 60F254 and RP-18 F254 plates (0.25 or 0.50 mm thickness, Merck KGaA) were used for analytical TLC. NMR spectra were recorded employing a Bruker Avance 500 spectrometer (Bruker), with TMS as an internal reference. HRESIMS data were measured on Agilent 6500 series Q-TOF B.06.01 (B6172 SP1) Accurate-Mass Sytems (Agilent). UV spectra were measured with a Shimadzu UV-1800 spectrophotometer (Shimadzu). CD spectra were determined using a ChirascanTM CD spectrometer (Applied Photophysics Ltd.). IR spectra were recorded with KBr pellets on an IR Prestige-21 spectrometer (Shimadzu).
Plant Materials
The stems and leaves of B. latistyla were collected from Quang Tri province, Vietnam (N16°38ʹ18.8ʺ E106°48ʹ09.8ʺ) in August 2020. The scientific name of the plant was determined by one of the authors (Dr. Anh Tuan Le). A voucher specimen (coded DHYD-QT-01) has been preserved at the Faculty of Pharmacy, Hue University of Medicine and Pharmacy, Hue University, Vietnam.
Extraction and Isolation
Stems and leaves of B. latistyla were washed, cut into small pieces, and dried at 50 °C. They are ground into a powder (3 kg) and subsequently soaked in methanol (MeOH) at room temperature (8 L × 2 times). The combined extract was evaporated under reduced pressure to obtain a MeOH extract (540 g). This crude extract was suspended in water, successively partitioned with n-hexane (Hex), dichloromethane (Di), and ethyl acetate (EtAc) (1 L × 7 times for each solvent), yielding BLH (80 g), BLD (90 g), BLE (20 g), and the remaining water-soluble portion BLW (350 g), respectively. The BLH extract (80 g) was subjected to a normal-phase (NP) silica gel column, eluted with a gradient solvent system of n-hexane/acetone (Hex/Ac) (100/0, 60/1 to 0/100, v/v, 1 L each), and finally with MeOH to afford nine fractions (BLH1–9). Fraction BLH1 (23.2 g) was applied to an NP column using a step-gradient elution of Hex/Ac (100/0, 40/1, 30/1, v/v) to obtain nine fractions (BLH1.1–9). Fraction BLH1.5 (305 mg) was further purified through a reversed-phase (RP-18) column eluted using Ac/MeOH/water (1/1/0.1, v/v) to yield compound
The BLD extract (90 g) underwent solid-phase extraction using NP silica gel with a step-gradient elution of a DiMeOH solvent system (100/0 to 0/100, v/v, 1 L each), yielding eight fractions (BLD1–8). BLD1 (16.2 g) was loaded onto a Sephadex LH-20 column eluted with Di/MeOH (1/1, v/v) to remove chlorophyll, followed by purification on NP silica gel with a gradient of Hex/Ac (100/0 to 0/100, v/v, 500 mL each), resulting in eight subfractions (BLD1A-H). Crystals obtained from BLD1E (2.9 g) were subsequently purified on an NP column eluted with Di/EtAc (30/1, v/v), yielding compound
Similarly, the BLE extract (20 g) underwent solid-phase extraction on NP silica gel with a gradient solvent system of Di/MeOH (100/0 to 0/100, v/v, 1 L each), yielding eight fractions (BLE1–8). BLE1 (14.5 g) was loaded onto a Sephadex LH-20 column eluted with Di/MeOH (1/1, v/v), resulting in seven subfractions (BLE1A-J). The purification of BLE1F (2.9 g) was conducted similarly to BLE1 to obtain three subfractions (BLE1F1-3). BLE1F2 (750 mg) was then chromatographed on an NP column with Hex/EtAc/MeOH as a mobile phase (6/1/0.1, v/v/v), providing nine subfractions (BLE1F2A-I). Lastly, compound
Cytotoxic assay. Refer to Supplemental Material.
Theoretical calculation of the ECD spectra of compounds
Statistical Analysis
The data, expressed as M ± SD from three independent experiments, were analyzed using TableCurve 2D software (v4.0, Systat Software Inc.) to calculate the half-maximal inhibitory concentration (IC50). Further analysis was conducted with Prism 9 software (v9.5.1, GraphPad Software, CA, USA). One-way analysis of variance (ANOVA) is utilized to compare IC50 values among different cell lines for a compound and vice versa. Meanwhile, a two-way ANOVA followed by Tukey's post hoc test was employed to compare the IC50 values of the isolates across cell lines. Differences were considered significant at a P-value < .05.
Supplemental Material
sj-doc-1-npx-10.1177_1934578X241246689 - Supplemental material for Two New Lupane-Type Triterpenes From the Stems and Leaves of Buxus latistyla Gagnep. and Their Cytotoxic Activity
Supplemental material, sj-doc-1-npx-10.1177_1934578X241246689 for Two New Lupane-Type Triterpenes From the Stems and Leaves of Buxus latistyla Gagnep. and Their Cytotoxic Activity by Hung Quoc Vo, Ty Viet Pham, Anh Tuan Le and Hanh Nhu Thi Hoang, Phu Quynh Dinh Nguyen, Le Nhat Thi Doan, Hoai Thi Nguyen, Duc Viet Ho in Natural Product Communications
Footnotes
Author Contributions
Duc Viet Ho, Hoai Thi Nguyen, and Hung Quoc Vo conceived and designed the research. Ty Viet Pham, Anh Tuan Le, Hanh Nhu Thi Hoang, Phu Quynh Dinh Nguyen, Le Nhat Thi Doan, Hoai Thi Nguyen, Duc Viet Ho, and Hung Quoc Vo conducted experiments and analyzed data. Duc Viet Ho, Ty Viet Pham, and Hung Quoc Vo wrote the manuscript. All authors read and approved the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Hue University, Vietnam (ID No.DHH-2021-04-152).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
