Abstract
Introduction
Acute lung injury (ALI), the fatal pulmonary inflammatory syndrome boasting a death rate of up to 40%,1–3 develops into acute respiratory distress syndrome (ARDS) without rapid and effective treatment. ALI/ARDS encompasses diffuse interstitial and alveolar edema, stemming from the impairment of pulmonary capillary endothelial and alveolar epithelial cells, as well as inflammatory factor storms, characterized by severe hypoxemia.4,5 However, the pathological mechanism of ALI/ARDS has not been determined. At present, glucocorticoids are used to treat ALI/ARDS, and many adverse reactions could be occurred. Therefore, it is very important for us to find novel drugs. An emerging body of research posits that traditional Chinese medicine holds promise as a potent therapeutic modality for ALI. Lin et al 6 have found that traditional Chinese medicine could inhibit immune cell inflammation and has become a promising strategy for inhibiting the transformation of mild to moderate diseases into critical and critical diseases.
Rhodiola rosea L. (RL) was an important natural plant, known as “plateau ginseng.” According to the Great “Dictionary of Chinese Medicine,” 7 the “Shennong Ben Cao Jing” 8 and “Jingzhu Bencao,” 9 RL has the effect of treating pneumonia and clearing the lung to relieve cough. Previous studies have shown that RL could improve immunity, resist fatigue, and enhance cell vitality.10,11 Our previous study showed that the active ingredient of RL could reduce the expression of inflammatory factors and alleviate various organ damage caused by inflammation. However, the mechanisms of RL for the treatment of ALI was still be investigated.12,13
Network pharmacology, used to explain the process of disease occurrence and development from the perspectives of systems biology and biological networks, could be used to elucidate the action of traditional Chinese medicine.14,15 Molecular docking was a method to calculate the binding affinity between small molecules and specific receptors by calculating and screening active ingredients. 16 Consequently, the investigatory prowess of network pharmacology and molecular docking was harnessed to unravel the efficacious constituents and mechanistic pathways underlying the therapeutic impact of RL in addressing ALI. Finally, the effects and mechanism of RL in treating ALI were further validated at the animal level.
Materials and Methods
Network Pharmacology Analysis
RL Active Ingredients and Target Genes Acquisition
The bioactive constituents of RL were curated from the Organchem database (https://organchem.csdb.cn/), and their chemical composition was elucidated through interrogation of the PubChem database (https://pubchem.ncbi.nlm.nih.gov/). Employing the Swiss Target Forecast and Swiss ADME online databases (http://www.swisstargetprediction.ch/), meticulous screening of compounds was conducted, and potential targets were extracted, employing a filter condition set at a probability threshold * > .02. Subsequently, target acquisition and standardization were executed through the UniProt database 17 (https://www.uniprot.org/).
Prediction and Collection of Disease Target
ALI-related targets were identified by keyword “acute lung injury” from OMIM (http://OMIM.org/), GeneCards (http://www.gengcards.org/), and DisGeNET (https://www.disgenet.org/) databases. The Venny 2.1.0 tool facilitated the identification of intersecting genes between RL and ALI, which were subsequently standardized. Visualization was achieved through Wayne plots.
Construction of Ingredient-Target Network
Utilizing Cytoscape 3.8.2 (http://www.cytoscape.org/), the active ingredients of RL and ALI targets were input, allowing for the creation of an intricate network. Subsequent analysis focused on discerning RL's active ingredients implicated in the prevention and treatment of ALI.
PPI Network Construction and Filtering
The STRING database 18 (https://string-db.org) facilitated the analysis of RL-ALI intersection genes, with species restricted to “Homo sapiens” and confidence scores set at ≥0.9. Results were visualized through network diagrams, and drug-target protein interactions were incorporated into Cytoscape 3.8.2. We used the cytoHubba plugin to assign values to each gene through topological network algorithms and sequence and mine core genes. In addition, a bar plot of the core genes was drawn.
Enrichment Analysis of GO and KEGG Pathways
Intersection genes were input into the Metascape 19 (Version v3.5.20230501) database (https://metascape.org) for comprehensive Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analyses. Simultaneously, an “ingredient-target-pathway” network diagram was constructed using Cytoscape 3.8.2 to elucidate the intricate relationships.
Molecular Docking
Core small molecule structures were acquired from the PubChem database (https://PubChem.ncbi.nlm.nih.gov/), converted into three-dimensional structures, and saved in *MOL format. AutoDock (Version 1.5.6) processed and saved them as *PDBQT files. Crystal structures of Estrogen Receptor 1 (ESR1), Epidermal Growth Factor Receptor (EGFR), nonreceptor tyrosine kinase (SRC), and serine/threonine kinase 1 (AKT1) (PDB ID: 1SJ0, 1h10, 1fmk, and 1xkk) were obtained from the PDB database (http://www.RLsb.org). PyMol and AutoDdock software were utilized to process target protein molecules and saved in *PDBQT format. Auto Ddock Vina performed molecular docking with ESR1, EGFR, SRC, and AKT1, and results were visualized in Discovery (Version 4.5) Studio and PyMol (Version 1.7.x).
Animal Experimental Validation
Reagents
Lipopolysaccharide (LPS) (Lot: L2880) was purchased from Shanghai Payi Biotechnology Co., Ltd. Kae (CAS: 520-18-3; HY-14590; purity≧99.67%) was purchased from MedChemExpress Company. Dexamethasone (Lot: 1022070762) injection was from Shiyao Yinhu Pharmaceutical Co., Ltd. Rat Phosphorylated Protein Kinase B (p-AKT) (Lot: FH0183193) and Phosphorylated Epidermal Growth Factor Receptor (p-EGFR) (Lot: FH0183127) Enzyme-linked Immunosorbent Assay (ELISA) kits got procured from Hunan Fenghui Biology Co., Ltd. Tumor necrosis factor-alpha (TNF-α) (Lot: KRC3011) and Interleukin-6 (IL-6) (Lot: BMS625) ELISA kits got procured from Thermo Fisher Scientific China Co., Ltd.
Animal and Drug Handling
Sprague Dawley (SD) (Lot: SCXK (Jing) 2019-0010) rats (n = 36, weight = 190 ± 10 g, 6-8 weeks old) originated from Beijing Sibeifu Experimental Animal Co., Ltd. Animal experiments met the requirements and were conducted in accordance with National Institutes of Health Laboratory Animal Care and Use Guidelines.
Following a 1-week acclimatization period, all rats were subjected to random allocation into 6 distinct groups, each comprising six rats: (1) PBS control (Control); (2) 8 mg/kg LPS challenge (LPS); (3) positive control group with 5 mg/kg dexamethasone + 8 mg/kg LPS (Dex); (4) 25 mg/kg Kaempferol + 8 mg/kg LPS (L-Kae); (5) 50 mg/kg Kaempferol + 8 mg/kg LPS (M-Kae); (6) 100 mg/kg Kaempferol + 8 mg/kg LPS (H-Kae). Rats underwent a 12-h fasting period with ad libitum access to water. Before inducing ALI, rats in the Dex and Kae groups (5 mg/kg) and Kae (100 mg/kg, 50 mg/kg, 25 mg/kg) received intraperitoneal injections 2 h prior. The control group received an equivalent volume of PBS. Subsequently, after 2 h, groups 2, 3, 4, 5, and 6 received intraperitoneal injections of LPS (8 mg/kg), while the control group was administered an equivalent amount of PBS. Following a 6-h interval, the rats were euthanized, and lung tissue was extracted for subsequent analysis (Figure 1).

Technology roadmap and animal experiment design.
Lung Tissue Wet/Dry Ratio
The left lower lobe of the lung was meticulously isolated to measure its wet weight. Subsequently, the left lower lobe underwent incubation at 80 °C for 48 h to facilitate the removal of all water, and its resultant dry weight was quantified. The ratio of the wet weight to the dry weight (W/D) was then calculated.
Histological Analysis
The excised tissue was promptly dissected and subjected to fixation in 4% paraformaldehyde for 24 h. Following fixation, the tissue was embedded in paraffin, sectioned, and stained using Hematoxylin and Eosin (H&E). Moreover, the sections were meticulously examined for pathological alterations utilizing an optical microscope (Nikon). The histological characteristics of organ damage were assessed by scoring bleeding and edema in 6 visual fields per group. The scoring system ranged from 0 to 4 points, with values representing: 0 (no damage), 1 (<25% of the damaged area), 2 (25%-50% of the damaged area), 3 (50%-75% of the damaged area), and 4 (>75% of the damaged area).
Measurement of Inflammatory Factors, p-EGFR, and p-AKT Levels
Collected lung tissue homogenate supernatant from each group and used ELISA kit to determine p-EGFR and p-AKT (according to the instructions of Hunan Fenghui Biological Co., Ltd) TNF-α And IL-6 (according to the method described in the instructions of Thermo Fisher Scientific China Co., Ltd).
Western Blot Analysis
The lung tissue from each group was homogenized by protein lysis buffer on ice, and the protein concentration was determined using a BCA protein assay kit (PC0020, Solarbio, China). Equal amounts of protein were separated by SDS-polyacrylamide gel and transferred to PVDF membranes. The membranes were blocked with 5% skimmed milk for 1 h at room temperature in TBST, followed by incubation with specific primary antibodies overnight at 4 °C. The antibodies against AKT, P-AKT, EGFR, P-EGFR, GAPDH were obtained from Cell Signaling Technology (USA). Subsequently, the membranes were incubated with secondary antibody at the room temperature for 1 h. Protein blots were analyzed with a BeyoECL Plus kit (P0018S, Beyotime, China), and imaged by the Tanon 4800 imaging system (Tanon, China).
Statistical Method
Network pharmacology used Cytoscape Version 3.8.2 for data analyses. Go and KEGG analyses got completed via Metascape as well as Weisheng software. Molecular docking used AutoDock Vina Version 1.5.6 and Pymol Version 1.7 software. Animal experiments were used One-way ANOVA was performed using GraphPadPrism Version 7.0 (GraphPad Software Inc., San Diego, CA, USA) to compare two groups. Data were in the form of means ± SD (x ± SD). Statistical significance: P < .05.
Results
Screening and Collection of Active Ingredients and Targets of RL
According to our research hypothesis, we obtained a Technology roadmap (Figure 1). The RL active ingredients obtained from the Organchem database were screened through the Swiss ADME database to obtain eligible active ingredients and corresponding targets. A total of 18 chemical components and 413 targets were selected.
Disease and Drug-Disease Intersection Targets
In the quest for targets associated with ALI, Genecards, DisGeNET, and OMIM databases were meticulously mined, yielding a total of 2703 targets postduplicate removal. Employing Venny 2.1.0 (https://bioinfogp.cnb.csic.es/tools/venny/), an intersection analysis was conducted, revealing 306 common targets shared between the component targets of RL and those implicated in ALI prevention and treatment (Figure 2).
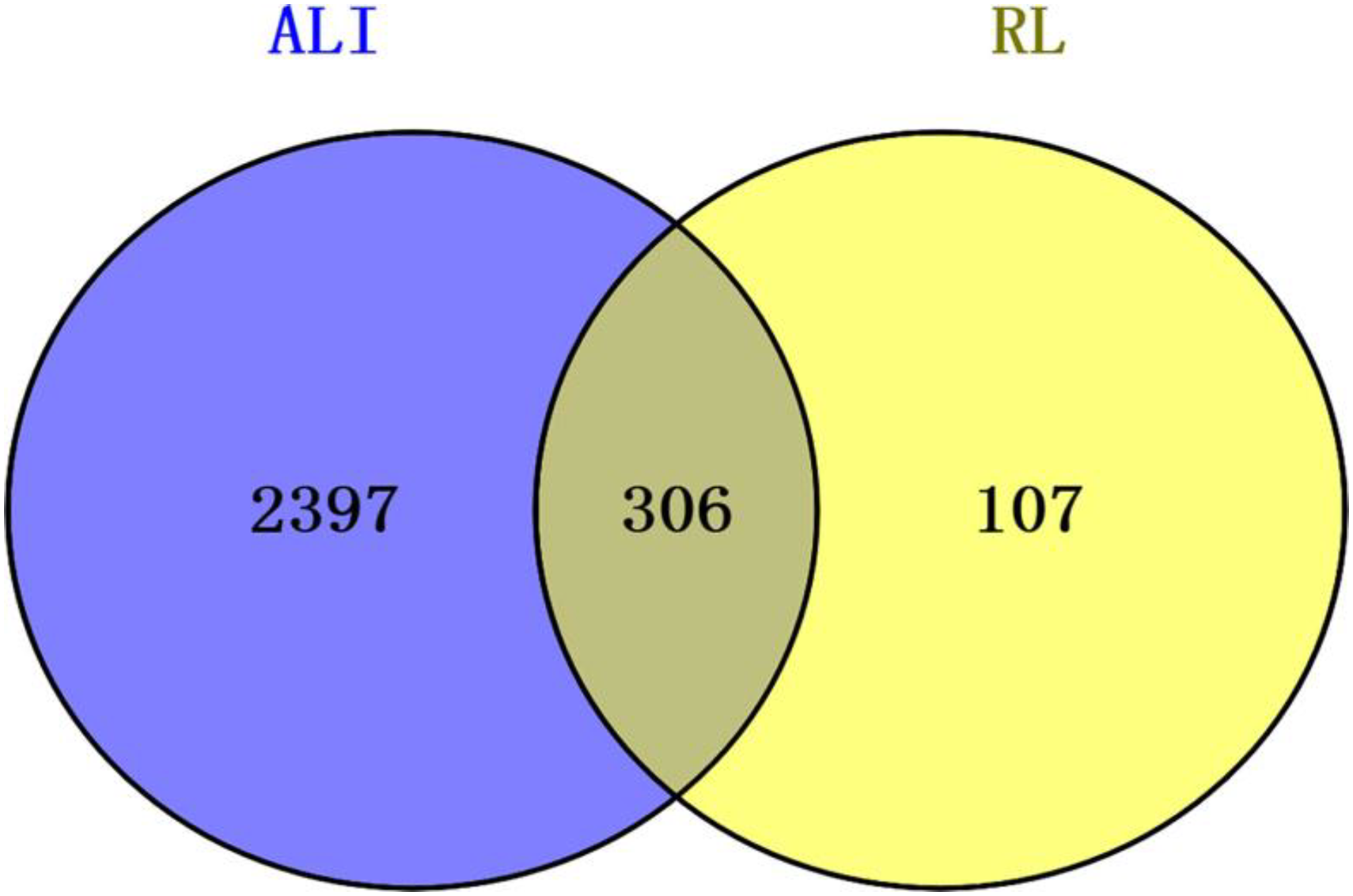
Venny diagram of RL therapeutic targets for ALI.
Ingredients-Targets Network Diagram
The acquired bioactive ingredients and their corresponding targets for RL were input into Cytoscape 3.8.2, facilitating the construction of a visually insightful network diagram. Hexagons within the diagram denoted components, while circles represented genes. The size and numerical values of the entities correlated positively with their degree, signifying a higher likelihood of a component's efficacy in disease treatment (Figure 3). Noteworthy findings identified Dibutyl phthalate (RL14), Kaempferol (RL16), Ethyl octanoate (RL1), Linoleic acid (RL12), and Oleic acid (RL4) as pivotal bioactive ingredients within ALI prevention and treatment in RL (Table 1).

RL ingredient-target network diagram. Eighteen RL ingredients (hexagon, pink), four hundred and thirteen targets (circle, green).
Active Ingredient and Degree in RL.

RL, Rhodiola rosea L.
Construction of PPI Network Diagram and Core Targets
The PPI network included 305 nodes and 647 edges (Figure 4A). Then, it was input into Cytoscape 3.8.2 to obtain a protein–protein visualization network diagram, which included 647 nodes and 632 edges. Analyzed it using the CytoHubba plugin to construct a hub network, including and drawing a bar chart of the core target points (Figure 4B). EGFR, SRC, Mitogen-Activated Protein Kinase 3 (MAPK3), Mitogen-Activated Protein Kinase 1 (MAPK1), Signal Transducer and Activator of Transcription 3 (STAT3), AKT1, and Phosphoinositide-3-Kinase Regulatory Subunit 3 (PIK3R) were closely related to ALI.

PPI network diagram and core targets. (A) Protein-protein network diagram for STRING. (B) Protein-protein network diagram for cytoscape.
Enrichment Analysis of GO and KEGG Pathways
GO enrichment analysis demonstrated that the BP was related to signal transmission and protein phosphorylation, etc. CC were related to the plasma membrane, cytoplasm and nucleus; MF were related to protein binding and ATP binding (Figure 5). Enrichment analysis of the KEGG pathway showed that the pathway of RL in the prevention and treatment of ALI mainly involved PI3K-Akt, MAPK, EGFR tyrosine kinase inhibitor resistance pathway, etc meanwhile (Figure 6). The top 10 pathways with targets were integrated to construct a “component-target-pathway” network diagram (Figure 7).
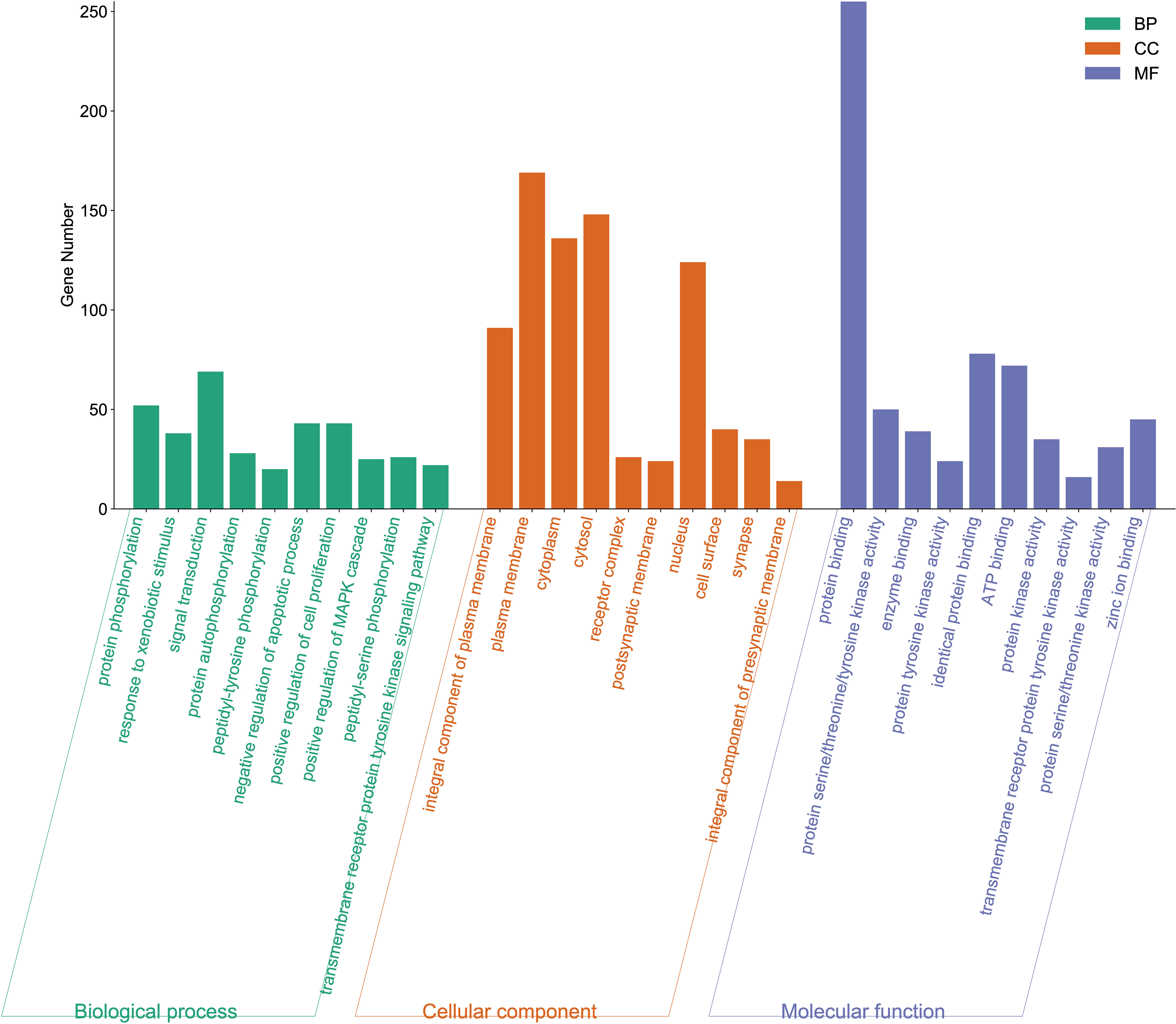
Bar chart of GO functional enrichment analysis.
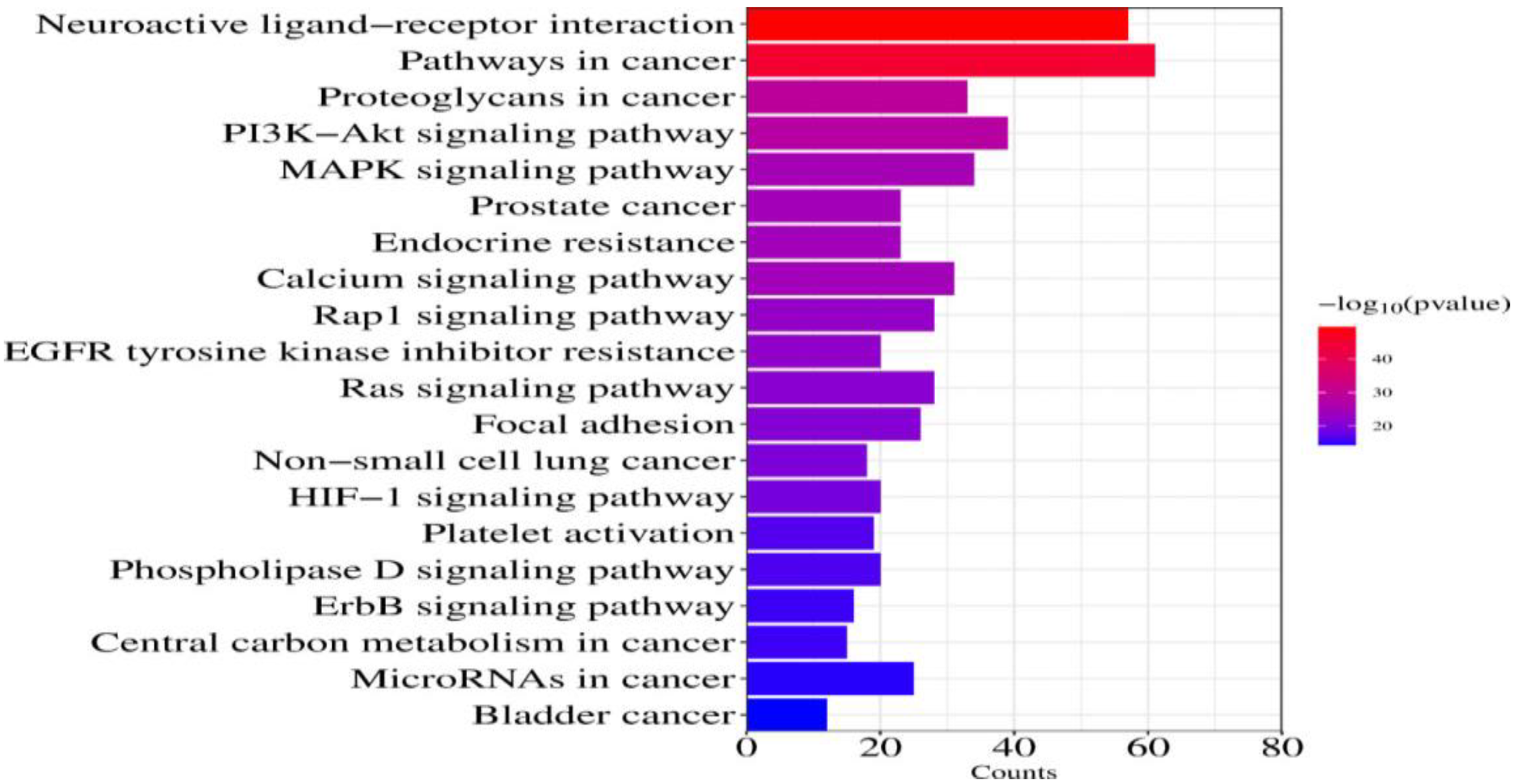
Bar chart of KEGG enrichment analysis.

RL-ALI interaction network diagram of ingredients-targets-pathways.
Core Components With ESR1, EGFR, SRL, and AKT1: Molecular Docking Results
Macromolecular docking results show that the lower the Binding energy, the greater the affinity between the receptor and ligand, and the greater the possibility of binding. The molecular docking energies of kaempferol with ESR1, EGFR, SRC, and AKT1 were −6.93 kJ/mol, −5.29 kJ/mol, −6.99 kJ/mol, and −6.93 kJ/mol. Its Binding energy is less than −5 KJ/mol, indicating that kaempferol is stable in combination with ESR1, EGFR, SRC, and AKT1. (Table 2 and Figure 8).
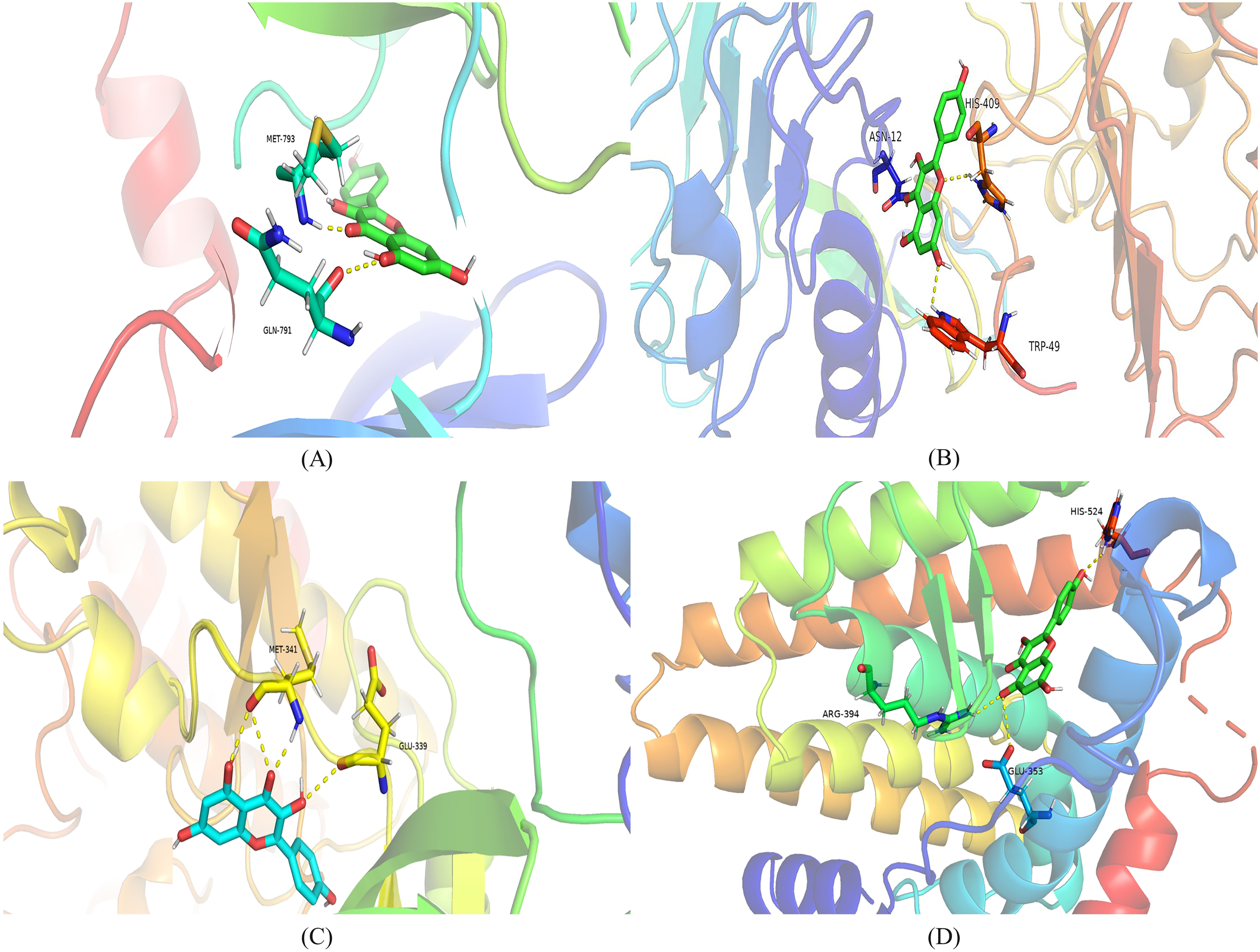
Molecular docking. (A) The combination between Kae and AKT1. (B) The combination between Kae and EGFR. (C) The combination between Kae and SRC. (D) The combination between Kae and ESR1. The binding affinities are all less than −5 kcal/moL.
Verification Results of Molecular Docking.

ESR1, Estrogen Receptor 1; AKT1, serine/threonine kinase 1; EGFR, Epidermal Growth Factor Receptor; SRC, nonreceptor tyrosine kinase.
Validation of Animal-Level Research
Morphologic analysis. As shown in Figure 9A, the surface of the control group's lung tissue was dry, without inflammatory cells and red blood cell exudation, and without congestion; In comparison to the normal control, the LPS group lung tissue got congested and red fluid exudated. In comparison to the LPS cohort, Kae treatment exhibited a notable reduction in bleeding, reduced inflammatory cell infiltration, and relief of lung damage.

(A) morphologic analysis; (B) histochemistry analysis, magnification: 400×; scale = 100 µm. (n = 6).
Histochemistry analysis. H&E staining exhibited pathological changes in each group of lung samples. As depicted in Figure 9B, in comparison to normal control, LPS cohort exhibited significant bleeding and inflammatory infiltration. Results of Kae and Dex groups indicate that compared to the LPS group, Kae has varying degrees of protective effects on LPS-induced ALI. Combined with the lung injury score (Figure 10A), it was shown that Kae has a significant protective effect on lung tissue.
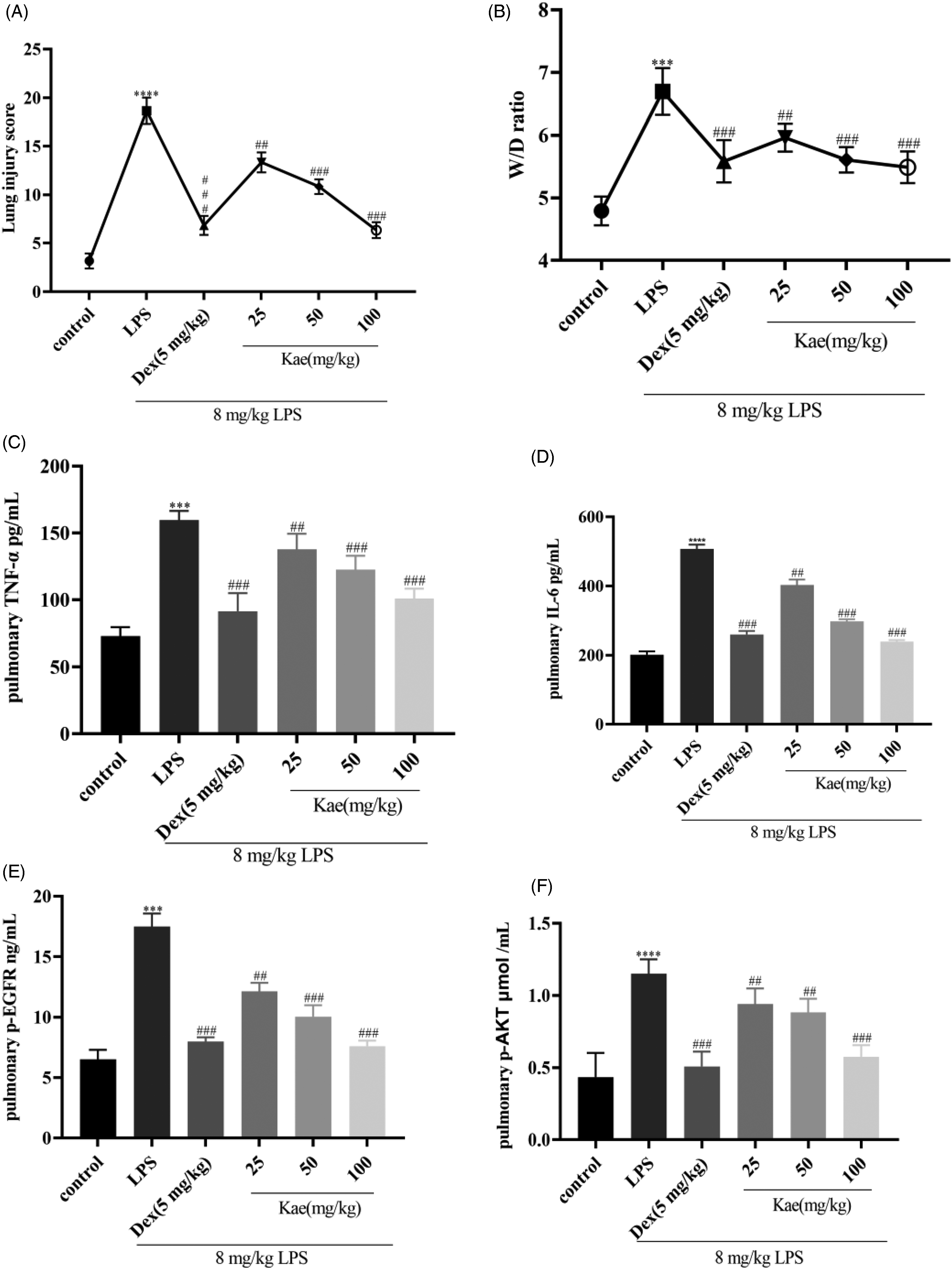
(A) Lung injury score; (B) The ratio of Wet/Dry; (C) Detection of TNF-α by ELISA; (D) Detection of IL-6 by ELISA; (E) Detection of p-EGFR by ELISA; (F)Detection of p-AKT by ELISA.*P < 0.05 versus control group and ##P < 0.01 versus LPS group. Data was mean ± SD (n = 6).
Reduced W/D lung ratio. The W/D lung ratio serves as a direct metric for pulmonary edema and stands as a sensitive gauge of the severity of lung injury. Our observations revealed a significantly elevated W/D ratio in the LPS treatment group compared to the control group. Intriguingly, various Kae treatment groups exhibited a discernible reduction in the W/D ratio, indicative of a potential ameliorative effect on pulmonary edema (Figure 10B).
Effects of kaempferol on p-AKT, p-EGFR, IL-6, and TNF-α. Robust alterations in cytokines, p-AKT, and p-EGFR were evident in both serum and lung tissue homogenates across the experimental groups. Relative to the control group, TNF-α and IL-6 exhibited significant escalation in the LPS group. Notably, different Kae treatment groups demonstrated a pronounced attenuation of TNF-α and IL-6 compared to the LPS groups (Figure 10C and D). Simultaneously, measurements of p-EGFR and p-AKT content unveiled a substantial surge in the LPS group compared to the control group. Conversely, varied Kae treatment groups exhibited a noteworthy reduction in p-EGFR and p-AKT content relative to the LPS group (Figure 10E and 10F). These findings suggest that Kaempferol may exert its anti-inflammatory effects in ALI rats by modulating the EGFR-AKT pathway.
Western blot Analysis. The expressions of p-AKT and p-EGFR were significantly changed in the experimental groups. Compared with the control group, the expression of p-AKT was significantly downregulated in the LPS group. Conversely, p-AKT levels in different Kae treatment groups showed a dose-dependent increase, compared to the LPS groups (Figure 11 and Figure 12A). However, for p-EGFR, there was the opposite changing trend to that of p-AKT expression. p-EGFR level was significantly upregulated in the LPS group. As the concentration of Kae increased, the expression of p-EGFR decreased (Figures 11 and 12C). The grayscale value of the strips is shown in Table 3.

The protein expression of p-AKT, AKT, p-EGFR, and EGFR in lung tissue homogenate by western blot. (n = 3).

The protein expression of p-AKT, AKT, p-EGFR, and EGFR in lung tissue homogenate by western blot.*P < 0.05 versus control group and ##P < 0.01 versus LPS group. Data was mean ± SD (Grayscale value, n = 3).
The Grayscale Values of Western Blot.

LPS, lipopolysaccharide; p-EGFR, Phosphorylated Epidermal Growth Factor Receptor; EGFR, Epidermal Growth Factor Receptor; LPS, lipopolysaccharide; p-AKT, Phosphorylated Protein Kinase B; AKT, serine/threonine kinase.
Discussion
ALI delineates the acute hypoxic respiratory insufficiency or respiratory failure stemming from diffuse pulmonary interstitial and alveolar edema, induced by the injury to pulmonary capillary endothelial cells and alveolar epithelial cells in noncardiac diseases such as severe infection, shock, trauma, and burns. The progression to ARDS20–22 is feasible when the inflicted injury reaches a critical threshold. Notably, a specific pharmacological intervention for treating ALI remains elusive, and prevailing therapeutics in clinical practice exhibit drawbacks, encompassing proclivity for adverse reactions such as coagulation disorders, gastric ulcers, and osteoporosis. Traditional Chinese medicine mainly adopts the method of syndrome differentiation and treatment, using compound or single traditional Chinese medicine to prevent and treat ALI/ARDS, and has achieved certain results. Rhodiola rosea L., indigenous to alpine environments and entrenched as a traditional folk remedy for plateau-related maladies, has piqued modern pharmacological interest due to its discerned anti-inflammatory properties. These properties manifest in the significant reduction of inflammatory factors TNF-α, IL-6, and Interleukin-1β (IL-1β), 23 concomitant with the alleviation of pulmonary edema, thereby impeding the onset of ALI. Nonetheless, the precise mechanistic underpinnings necessitate further exploration. In our study, we employed network pharmacology and molecular docking methodologies to unravel the active ingredients and the mechanism of action of RL in ALI treatment, subsequently substantiating our findings through animal experiments. We found that (A) Kae was a key component in the treatment and prevention of ALI with RL, executed through multiple targets and signaling pathways. (B) Kae can effectively inhibit the TNF-α and IL-6 levels in ALI, while the level of p-EGFR was decreased and p-AKT was increased.
In the ingredient-target-disease network diagram, it can be found that the components closely related to ALI were Oleic acid, Linoleic acid, and Kae. Kim et al 4 found that oleic acid can inhibit allergic inflammation of IL-17 secretion in asthma mice. Aslani et al 25 showcase that conjugated linoleic acid in Chronic Obstructive Pulmonary Disease patients effectively reduces IL-6 levels. Linoleic acid demonstrates efficacy in diminishing, IL-6, Interleukin 8, and IL-1β content in allergic inflammation. 26 Kae, a natural flavonoid, displays remarkable antiinflammatory activity. It reduces Interleukin 4 and Interleukin 13 expression, inhibiting inflammation in specific dermatitis mouse models. 27 In gouty arthritis models, 28 Kae inhibits Transforming Growth Factor-β1, IL-1β, IL-6, and TNF-α, reducing ankle edema and inflammation. Kae also mitigates neurotoxic effects induced by bupivacaine in dorsal root ganglia, deactivating TNF Receptor Related Factor 6 (TRAF6)/NF-κB signaling and reducing IL-6, IL-1β, and TNF-α levels. 29 In LPS-induced cytokine storm models, Kae modulates proinflammatory and antiinflammatory factors, protecting organs from damage. 13 Qian et al 30 found that Kae mitigates LPS-induced lung injury by regulating TRAF6 and Interleukin-1 Receptor Related Kinase 1. Rabha et al 31 proposed that Kae regulates iNOS and ICAM-1 pathway, reducing proinflammatory factors (IL-6, IL-1β, TNF-α) and alleviating sepsis-induced ALI in mice. In our study, Kae effectively reduces pro-inflammatory factor secretion (IL-6, TNF-α) in LPS-induced ALI rats, exerting a protective influence on lung tissue. These findings align with existing literature, providing a foundation for future research and a theoretical framework for subsequent animal-level inquiries.
The outcomes of network pharmacology, enrichment analysis, and molecular docking unveil pivotal targets implicated in the prevention of ALI by RL. The identified core targets encompass SRC, MAPK1, MAPK3, STAT3, AKT1, HSP90AA1, EGFR, and PIK3R1. Previous studies 32 have shown that within the LPS-induced ALI mouse model, MAPK phosphorylation levels are inhibited by Kae, which reduces lung inflammation. Zhang X et al 33 found that Kae can inhibit the phosphorylation level of AKT in a rat pulmonary hypertension model, thereby improving pulmonary hypertension symptoms.
The GO functional enrichment analysis delineated that the biological processes associated with ALI encompass signal transmission and protein phosphorylation. Regarding cellular components, RL exhibits efficacy in impacting the plasma membrane, cytoplasm, and nucleus. Moreover, ALI appears to influence protein binding and ATP binding. The concurrent KEGG pathway enrichment analysis elucidates that the ALI treatment process involves pivotal pathways, including the PI3K-Akt signaling pathway, MAPK signaling pathway, and EGFR pathway. These pathways intricately connect with cell proliferation, differentiation, apoptosis, and immune inflammation, positioning RL as a promising candidate for ALI prevention and treatment. Prior research 34 has confirmed that the PI3 K/Akt signaling pathway was involved in lung inflammation and was associated with neutrophil apoptosis in the lungs. Salidroside, a component of RL, has demonstrated the ability to activate the PI3 K/Akt signaling pathway, mitigating proinflammatory factor secretion and safeguarding against LPS-induced ALI in rats. 35 In the rat model of ALI, the MAPK signaling pathway was also involved, thus limiting the development of inflammation and downregulating IL-6 and TNF-α secretion. 36 Research has confirmed that in the sepsis-induced ALI rats, inhibiting EGFR phosphorylation converts the M1 phenotype of macrophages into the M2 phenotype, which reduces ALI, 37 Meanwhile, EGFR was significantly activated. When EGFR is inhibited, it can limit the permeability of pulmonary blood vessels and neutrophils. 38 The PI3 K/Akt pathway assumes a pivotal role in cellular defense against inflammatory stimuli, intricately participating in the orchestration of cell growth, cell cycle entry, migration, and survival. Activation of Akt transpires via the PI3 K pathway, wherein PI3 K orchestrates the production of Phosphatidylinositol 3,4,5-triphosphate, culminating in Akt recruitment to the cell membrane. Subsequently, Akt phosphorylation is initiated through phosphoinositol-dependent kinase 1, facilitating a cascade of cellular responses. 39 It was found that Naringenin pretreatment significantly reduced the phosphorylation of PI3 K and Akt induced by ALI, which indicates that Naringenin pretreatment can protect lung tissue by inhibiting the PI3 K/Akt pathway. 40 Tao et al 41 showed that phosphorylated EGFR was significantly increased in mouse ALI, and p-EGFR was significantly reduced after administration of inhibitors, thereby protecting lung tissue.
These investigations have underscored that the activation of signaling pathways, including PI3 K/Akt, MAPK, and EGFR, plays a pivotal role in mediating the onset and progression of ALI, presenting themselves as promising therapeutic targets for Kae. In our study, Kae demonstrated a significant ability to reduce p-EGFR and promote p-AKT secretion. The level of p-EGFR in the treatment group was significantly lower than that in the LPS group, while the level of p-AKT was higher than that in the LPS group. This observation strongly suggests that Kae attenuates the release of proinflammatory factors by blocking EGFR and activating the AKT pathway, thereby implanting protective effects on lung tissue and mitigating lung injury. Our research agrees with theirs.
When ALI occurs, acute systemic inflammatory reaction can cause microvascular damage, increase the permeability of pulmonary blood vessels and epithelium, and protein and fluid rich in inflammatory cells flow into the alveolar cavity, leading to noncardiogenic Pulmonary edema, reduction of Gas exchange alveolar capillaries, and then lead to hypoxic respiratory failure and ARDS, thus endangering life. Ricard et al 42 found that proinflammatory factors TNF-α, IL-6, and IL-1β notably increased in ALI rat and mouse models. Previous studies 43 have shown that neutrophils infiltrate in the early stages of ALI, and J Gromes et al 44 have pointed out that removing neutrophils may reduce the severity of ALI. After ALI occurs, lung tissue may experience edema, bleeding, and other phenomena. 45 Of course, there are some limitations in this article. (1). Animal experimental studies are sometimes incomplete and the results obtained through animal models are not reliable to extrapolate to other species. (2). Animal experiments are sometimes misleading many abnormal phenomena that are obviously different from those seen in human beings in animal tests may be the normal biological characteristics of the tested animal species itself, or changes caused by unnatural means, or due to stress reactions in the laboratory environment. (3). Not validated at the cellular level.
Conclusions
In summary, Kae was regarded as an effective component within ALI treatment with RL via network pharmacology and molecular docking technology, preliminarily verified through animal experiments that Kae pretreatment protected lung tissue by regulating the EGFR-AKT signaling pathway.
Supplemental Material
sj-doc-1-npx-10.1177_1934578X241245550 - Supplemental material for Mechanism and Experimental Verification of Rhodiola rosea L. for the Treatment of Acute Lung Injury Through Network Pharmacology and Molecular Docking
Supplemental material, sj-doc-1-npx-10.1177_1934578X241245550 for Mechanism and Experimental Verification of Rhodiola rosea L. for the Treatment of Acute Lung Injury Through Network Pharmacology and Molecular Docking by Min Zhao, Guanping Lv, Xi Huang, Yong Niu, Qunhui Zhang and Wenhua Li in Natural Product Communications
Footnotes
Authorship Contribution Statement
Conceptualization: M.Z., Q. Z., and W.L. crafted the overarching framework; methodology: M.Z., G.L., X.H., and Y.N. devised the intricate approach; validation: M.Z., X.H., and Y.N. scrutinized and corroborated the results; formal analysis: M.Z., G.L., and X.H. conducted meticulous data analysis; investigation: M.Z. delved into the intricacies of the research; data curation: M.Z., G.L., Q.Z., and W.L. systematically organized and curated the data; writing—original draft preparation: M.Z. authored the initial manuscript; writing—review and editing: Q.Z. and W.L. refined and enhanced the manuscript; visualization: M.Z. employed innovative methods for data representation; project administration: Q.Z. and W.L. oversaw the project dynamics; funding acquisition: M.Z., X.H., and W.L. secured the necessary financial support. All authors have critically reviewed and unanimously endorsed the final version of the manuscript for publication.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China project (No. 81760332), the Yunnan Provincial Department of Education research project (No.2023J1460), the key discipline of Lijiang University of Culture and Tourism—Pharmacy (No. 2021xk04), the Lijiang University of Culture and Tourism Innovation Project (No. XC202319), Health Research Project of Hunan Provincial Health Commission (No. W20243099), Comparative study on the effects and mechanisms of Rhodiola glycoside and Kaempferol in Tibetan medicine Rhodiola grandiflora against cytokine storm induced myocardial injury (No. XJ2023001101) and the commissioned by the Key Laboratory of Molecular Mechanism and Intervention Research of High Altitude Related Diseases in Tibet (No. KF202008)..
Ethical Approval
All plans have been approved by the Ethics Committee of the Medical Department of Xizang University for Nationalities. Ethics Approval No. 2022.
Statement of Human and Animal Rights
All experimental procedures involving animals were carried out in accordance with the guidelines of the Helsinki Declaration and approved by the Ethics Committee of the Medical Department of Xizang University for Nationalities.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
