Abstract
Tetrastigma hemsleyanum Diels et Gilg (T hemsleyanum), known as “San ye qing” in China, is a well-known rare folk medicinal plant belonging to the Vitaceae family and to the same genus as Tetrastigma obtectum. 1 Its tubers or whole herbs are used medicinally, having the traditional Chinese medicine functions of dispelling wind and phlegm, activating blood circulation, relieving pain, clearing heat and detoxification, etc.2,3 With few toxic side effects, it has been widely used in clinical practice because of its anti-inflammatory, antitumor, and antiviral properties.4–7
In recent years, the demand for T hemsleyanum has gradually increased, and the price of the herb has risen, which has led to the overexploitation of its wild populations, 8 and the loss of wild T hemsleyanum resources has been indirectly aggravated by artificial afforestation and land reclamation; thus, some species are now on the verge of extinction. 9 Although T hemsleyanum has been widely cultivated as a cash crop, its slow growth, difficulty with root formation and low active ingredient content directly affect the economic prospects in artificial cultivation. 10 Therefore, an in-depth molecular biology study on the T hemsleyanum growth and development regulatory mechanism to elucidate the in vivo active ingredient metabolic process and mechanism of action will provide important information for artificial T hemsleyanum cultivation and breeding and will improve the utilization of its medicinal properties. 11
Terpenoids are the main active components of T hemsleyanum and are synthesized mainly in the cytoplasm via the mevalonate pathway (MVA). 12 3-Hydroxy-3-methylglutaryl-CoA reductase (HMGR) catalyzes the formation of MVA from HMG-CoA, so HMGR is considered a key enzyme in the MVA pathway. 13 In this study, we used transcriptome sequencing technology to sequence the transcriptomes of the roots, stems, and leaves of whole 3-year-old mature T hemsleyanum plants and then screened and analyzed the DEGs related to metabolic pathways. The gene expression for 3-hydroxy-3-methylglutaryl-CoA reductase (Thhg) in different T hemsleyanum tissue parts was analyzed by qPCR, and the active components of the roots, stems, and leaves were extracted to reveal the antitumor activity of different plant parts to provide a molecular and experimental basis for the development of effective T hemsleyanum medicinal components, a reference for artificial T hemsleyanum breeding and cultivation, and a theoretical basis for the comprehensive development and utilization of T hemsleyanum resources.
Materials and Methods
Materials
3-Year-old mature T hemsleyanum plants were harvested from Pan’an, Zhejiang Province, China. Three parts, namely, the root, stem, and leaf, were taken as the test materials, which were recorded as the root, stem, and leaf, respectively, and three biological replicates were used for each sample: TRIzol Reagent (Invitrogen), a fluorescent quantitative PCR kit (Thermo Fisher), an iScript cDNA Synthesis kit (Bio-Rad), RPMI 1640 medium (Gibco), fetal bovine serum (FBS; HyClone), and a Cell Counting Kit-8 (MCE).
Total RNA Extraction
Total RNA was extracted using the RNA Nano 6000 Assay Kit of the Bioanalyzer 2100 system (Agilent Technologies), and 1 μg of RNA per sample was used as input material for RNA sample preparation.
Sequencing
The samples that passed the test were enriched in mRNA, and the cDNA second strand was synthesized and purified. PCR amplification was performed after connecting sequencing junctions and selecting fragment sizes, and AMPure XP beads were used to purify the PCR products to obtain the final library. The library was quality checked and accurately quantified, and second-generation high-throughput sequencing was performed after the library was checked.
Gene Expression Level Analysis
The transcriptome obtained by Trinity splicing was used as the reference sequence (ref), and the clean reads from each sample were mapped to the ref using RSEM software (Bowtie2 parameter mismatch 0). 14 The results were also compared and counted to further determine the number of reads for each sample compared to each gene, and FPKM conversion was performed. The number of read counts for each sample was further compared to the number of reads for each gene and converted to FPKM to analyze the gene expression level.
Gene Differential Expression Analysis
TMM was used to normalize the read count data, and DEGseq was subsequently used for differential expression analysis. The q-value (q-value < 0.05) combined with fold change (|log2FoldChange|>1) screening was used to control the false-positive rate. 15
GO Functional Enrichment Analysis
The Gene Ontology (GO) database (http://www.geneontology.org/) was used to map all differentially expressed genes to individual terms. The number of genes per term was calculated, and the genes that were significantly enriched among the DEGs compared to the whole genomic background were identified. 16
Pathway Enrichment Analysis
KEGG enrichment analysis was performed using Kobas (2.0) software for the results of the comparative difference analysis in each group. The parameter fdr was set to BH (ie, using BH correction), and pathways with FDR ≤ 0.05 were defined as pathways significantly enriched in DEGs. 17
Analysis of Thhg Expression
Total RNA was obtained from the T hemsleyanum roots, stems, and leaves, and cDNA first-strand cDNA was synthesized according to the iScript cDNA Synthesis Kit. qPCR primers were designed according to the successfully sequenced Thhg gene sequence (Table 1). qPCR was performed to detect Thhg expression in different tissue parts of T hemsleyanum according to SYBR Premix Ex Taq™ II instructions, and the relative expression of the gene was calculated by the 2−ΔΔCt method with actin as an internal reference. 18
Primer Sequences for RT-qPCR.

Determination of Triterpenes Content
Total triterpenes from the T hemsleyanum roots, stems, and leaves were aspirated and added to a stoppered test tube. Then, vanillic acid and perchloric acid were added to the sample, which was mixed well and kept warm in a water bath at 60 °C. Afterward, the sample was removed and placed in cold water, glacial acetic acid was added, mixed well, and the absorbance of the sample was measured and recorded at A245 nm. The total triterpene content of the sample was then obtained using the regression equation of the standard curve with oleanic acid as the standard. 19
Preparation of Ethanol Extracts
The roots, stems, and leaves of T hemsleyanum were dried, crushed, sieved through a 60-mesh sieve, spun and extracted with anhydrous ethanol for 4 hours at a 1:10 mass-to-volume ratio of material to liquid (1:10). The sample was subsequently filtered, and the filtrate was centrifuged at 3000 g/min. The supernatant was the crude extract. The precipitate was extracted again by the same method and centrifuged, after which the two crude extracts were combined. The precipitate was extracted again by the same method and then centrifuged, and the dry extract resulting from freeze-drying was dissolved in DMSO to a mass concentration of 0.1 mg/mL for the biological assay.
Cell Culture
Human HCC cell lines Huh-7 and Hep3B were purchased from the Chinese Academy of Medical Sciences and were cultured in RPMI 1640 medium supplemented with 10% FBS and incubated at 37 °C in an incubator containing 5% CO2.
Antitumor Activity Studies
Huh-7 and Hep3B cells were seeded separately at 5000 cells per well in 96-well plates with fresh medium. The plates were preincubated for 24 hours (37 °C, 5% CO2) after the addition of 0.1 mg/mL raw extract at each site. After incubation for different time periods, 10 μL of CCK-8 solution was added to each well and incubated at 37 °C for 2 hours. The absorbance at 450 nm was measured. 19
Results and Discussion
Gene Expression Levels Analysis
The total numbers of high-quality sequencing tags obtained by filtering sequences from the roots, stems, and leaves of T hemsleyanum were 43176464, 42219270, and 50084798, respectively. The numbers of sequences that could be localized to the reference sequences were 28707348, 28952326, and 34598330. All high-quality sequencing tags were compared with the reference sequences. There were 28707348, 28952326, and 34598330 sequences within the reference sequences, accounting for 66.49%, 68.58%, and 69.08% of the number of high-quality tag species, respectively (Table 2).
Comparison of Reads With Reference Sequences.

Analysis of Differential Expression Levels Between Root and Other Tissues
The number of DEGs identified by comparing roots with stems and leaves was counted. Compared with those in the stems, the number of upregulated genes in the roots was 3604, and the number of downregulated genes was 2783 (Figure 1B). Compared with the number of upregulated genes in the leaves, those in the roots was 10796, and the number of downregulated genes was 3989 (Figure 1C).

Volcano plot of differently expressed genes between root and stem, root and leaf: (A) root, stem, leaf of Tetrastigma hemsleyanum; (B) comparison of different genes in root and stem; (C) comparison of different genes in root and leaf.
To understand the root-specific expression of genes, genes that were significantly differentially expressed in roots compared to stems and leaves were analyzed. Genes with exact functions identified by homology comparison (Blastnr) are listed in Table 3 (showing the top 8 genes significantly upregulated in the roots compared to the stems) and Table 4 (showing the top 8 genes significantly upregulated in the roots compared to the leaves). Compared with the genes in the stem, those whose expression was upregulated in the root were mainly involved in metabolic pathways, including the glutathione S-transferase, serine carboxypeptidase, germacrene D synthase, aminocyclopropane carboxyl oxidase, and serine/threonine kinase pathways (Table 3). Compared with those in the leaf, the genes whose expression was upregulated in the root were mainly those encoding various catalytic enzymes, such as β-glucosidase, α-dioxygenase, α-carbonic anhydrase, peroxidase, and bZIP transcription factors and cytochrome P450 (Table 4). These DEGs may play a key role in the accumulation of active ingredients in the roots of T hemsleyanum.
The Results of Blastnr Comparison of Genes Differently Expressed Between Root and Stem.
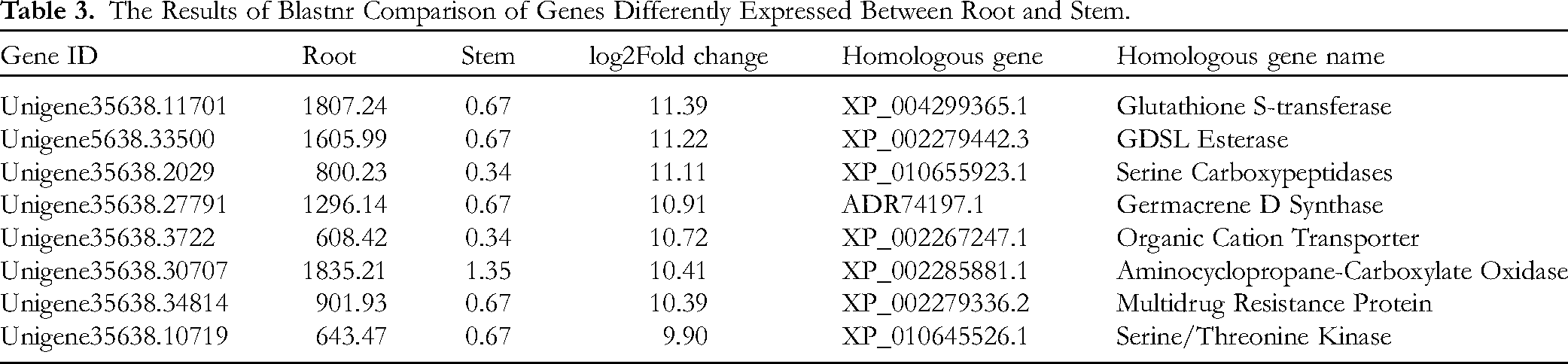
The Results of Blastnr Comparison of Genes Differently Expressed Between Root and Leaf.

GO Function Enrichment Analysis
The biological process category (root and stem, root and leaf) included a total of 2158 and 2615 GO terms, respectively. The cellular component (root and stem, root and leaf) category consisted of 505 and 602 GO terms, respectively. The molecular function category (root and stem, root and leaf) consisted of 1032 and 1311 GO terms, respectively. Among the significantly different GO terms enriched between the roots and stems of T hemsleyanum, 20 were significantly enriched in the biological process ontology, 20 were significantly enriched in the molecular function ontology, and 3 were enriched in the cellular component ontology. Among them, the differential expression of genes related to metabolic processes was the most obvious in the biological process ontology, while the differential expression of genes related to catalytic activity was the most obvious in the molecular function ontology, followed by genes related to ion-binding activity and metal ion-binding genes (Figure 2A).

Significantly enriched Go terms of differently expressed genes between root, stem, and root, leaf (showing highly expressed genes in root): (A) GO analysis of different genes in root and stem; (B) GO analysis of different genes in root and leaf.
Among the significantly different GO terms enriched between the roots and leaves of T hemsleyanum, 11 were significantly enriched in ontologies involved in biological processes, 18 were significantly enriched in ontologies involved in molecular functions, and only 3 were significantly enriched in ontologies involved in cellular components. Among them, the differentially expressed genes related to catalytic activity, ion binding, and hydrolase activity in the molecular function ontology were the most obvious (Figure 2B).
Pathway Significant Enrichment Analysis
The significant enrichment analysis of DEGs between the root and stem samples of T hemsleyanum is shown in Figure 3A. Among the top 20 significantly enriched pathways, the most enriched pathways were associated with zeatin biosynthesis, ribosome, and phenylpropanoid biosynthesis. Among them, the pathway with the most obvious differences was associated with zeatin biosynthesis and diterpene biosynthesis.

Scatter diagram of pathway enrichment analysis between root and stem and root and leaf: (A) KEGG analysis of different genes in root and stem; (B) KEGG analysis of different genes in root and leaf.
The results of the DEG enrichment analysis between the T hemsleyanum root and leaf samples are shown in Figure 3B. Among the top 20 pathways with significant differences, the most enriched pathways were associated with phenylpropanoid biosynthesis, phenylalanine biosynthesis, and flavonoid biosynthesis. Among them, the differentially expressed pathways that were most significant were associated with diterpene biosynthesis and flavonoid biosynthesis. The pathway with the most DEGs was the phenylpropanoid biosynthesis pathway.
Comparison of Thhg Expression and Triterpenes Contents
Thhg plays a key role in the synthesis of triterpenes, and we investigated its expression in different tissues via transcriptome sequencing. The results showed that Thhg was expressed in each tissue, among which the expression of Thhg in roots was greater than that in stems and leaves, indicating that Thhg was induced by certain growth and development pathways and reached its highest level in the roots of T hemsleyanum (Figure 4A).

Thhg expression and total triterpenes contents in different tissues of T hemsleyanum: (A) expression profile of Thhg in different tissues of T hemsleyanum; (B) total triterpenes contents in different tissues of T hemsleyanum.
The medicinal parts of T hemsleyanum are roots, fruits or whole herbs, and different triterpene contents may cause different medicinal effects on T hemsleyanum. Our results revealed that the triterpene content in the roots was significantly greater than that in the stems and leaves (Figure 4B), which was positively correlated with the expression of Thhg in T hemsleyanum, indicating that Thhg plays an important role in triterpene accumulation.
Antitumor Activity of Different Parts of T hemsleyanum Analysis
The results showed that the antitumor activity of the root alcoholic extract of T hemsleyanum was the highest, and that of the stem alcoholic extract was second only to that of the root (Figure 5A and B). The leaf alcoholic extract also showed some antitumor activity with some utility, although it did not have a stronger inhibitory effect on liver cancer cells than did the roots or stems (Figure 5C).

Identification of antitumor activity of the alcoholic extracts of the root, stem, and leaf of T hemsleyanum: (A) morphological changes of Huh-7 and Hep3B cells after 48 hours of treatment with alcoholic extract of each part of T hemsleyanum; (B) survival analysis of Huh-7 and Hep3B cells after 48 hours of treatment with alcoholic extract of each part of T hemsleyanum; (C) proliferation analysis of Huh-7 and Hep3B treated with alcoholic extract of each part of T hemsleyanum; CK: PBS treated group, n = 3. **p < 0.01.
Discussion
The root tuber is the main T hemsleyanum part used in Chinese medicine, and our study has shown that it has a relatively good medicinal effect. However, the amount of T hemsleyanum tuber harvested per plant was not much. The aboveground parts of T hemsleyanum, including the stem and leaf, can be harvested every year; these parts are larger than those of the root tuber, but they are rarely used medicinally, and there is a large waste of resources. 8
Comparative transcriptomic studies can be used to analyze the different developmental stages of medicinal plants at the molecular and biological levels. 20 The transcriptomic study of the DEGs in the roots, stems, and leaves of T hemsleyanum can be used to understand the molecular regulatory pathways involved in its development, which can help us to grasp the temporal and spatial effects of the accumulation of its active medicinal ingredients. 21 This approach can be used not only to make a case for the use of the aboveground parts of T hemsleyanum and greatly reduce the waste of resources but also to efficiently screen the main active parts of T hemsleyanum to determine the effective material basis for its antipyretic and antitumor effects and to lay the foundation for subsequent development and research. 22 Our study elucidated the DEGs in the transcriptome among the roots, stems, and leaves of T hemsleyanum under normal culture conditions and through KEGG analysis explored the pathways that were most enriched in DEGs, perhaps revealing the main reason for the differences in active ingredients in different parts of T hemsleyanum. However, there are some limitations in this work. Although we demonstrated the antiproliferative effectiveness of alcohol extracts of T hemsleyanum against tumor cells at the cytological level, in vivo evidence was lacking, which is the next step of our research.
Terpenoids have been found to stimulate a broad range of activities like oxidative stress suppression, inflammation reduction, apoptosis induction, cell cycle regulation, and cell proliferation inhibition. 23 Some of the terpenoids exert their anti-cancer effects largely through modulation of the immune system and have been successfully appraised in preclinical animal models of liver cancer. 24 Seeing the utility of these botanical active ingredients, terpenoids may be used in combination with chemotherapeutic drugs in the development of cancer treatments. 25
Understanding the function of the HMGR gene and its regulatory mechanism plays an important role in elucidating the biosynthesis of terpenoids in T hemsleyanum. In the future, it will be necessary to further investigate the molecular mechanism of terpenoid synthesis in T hemsleyanum and utilize this regulatory mechanism as a reference for molecular breeding, as well as for mass production of active terpenoids. This will increase the economic value of rare or low-utility medicinal plants, which can greatly assist in the development of traditional Chinese medicine.
Conclusions
In conclusion, the expression of Thhg had an important influence on triterpene content in T hemsleyanum, and the triterpene content was positively correlated with the expression of Thhg. The root alcohol extract of T hemsleyanum had the strongest antitumor activity, and the aboveground parts of T hemsleyanum also have some medicinal value. Our study will not only help to explore the mechanism underlying the formation of effective medicinal components of T hemsleyanum but will also lay the foundation for future work on T hemsleyanum molecular breeding.
Footnotes
Authors’ Contributions
Methodology was done by J. L. Validation was done by Y. L. and L. L. G. C. has written the original draft preparations. J. C. and L. L. performed the supervision. Project administration was done by L. L. All authors have read and agreed to the published version of the manuscript.
Data Availability Statement
The data presented in this study can be obtained by contacting the corresponding author and requesting access to the data.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Ethics Committee of Tongde Hospital of Zhejiang Province, Zhejiang Province, China.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Project of Administration of Traditional Chinese Medicine of Zhejiang Province of China, Zhejiang Province Public Welfare Technology Application Research Project (grant numbers 2024ZF049, 2022ZB076, No. LTGD23C040004).
Statement of Human and Animal Rights
All of the experimental procedures involving animals were con-ducted in accordance with the Institutional Animal Care guidelines of Zhejiang Academy of Traditional Chinese Medicine, China and approved by the Ethics Committee of Tongde Hospital of Zhejiang Province, Zhejiang Province, China.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Statement of Institutional Review Board
Not applicable.
