Abstract
Objective/background
A previous study showed that geranium essential oil predominantly affects the parasympathetic nervous system. This article examined the effects on the autonomic nervous system of linalool, one of the main components of geranium essential oil.
Methods
Mice were placed on a narrow elevated open platform and pulse wave measurements were taken while they were awake, with behaviors restricted. Heart rate and heart rate variability were analyzed from the pulse waves obtained and the effects on the parasympathetic and sympathetic nerves were examined.
Results
The results showed that linalool (5 μL/L air, inhaled) resulted in parasympathetic dominance before and after administration. Conversely, a tendency toward sympathetic dominance was observed with fluoxetine (1 mg/kg, intraperitoneal) as the positive control in the elevated open-platform test.
Conclusion
These results suggest that linalool and essential oils containing linalool regulate the autonomic nervous system by promoting dominance of the parasympathetic nervous system.
Introduction
Linalool is one of the major volatile components in the lavender and geranium essential oils used in aromatherapy. For this reason, many studies have investigated linalool, revealing sedative,1,2 anxiolytic,3,4 vasorelaxant, 5 analgesic, 6 and neuroprotective 7 effects. Geranium essential oil is also said to regulate the autonomic nervous system. In a previous study, we investigated the effects of geranium essential oil on awake mice and reported that geranium essential oil regulates the autonomic nervous system by promoting dominance of the parasympathetic nervous system.8,9 The geranium essential oil used in that previous study contained 6.3% linalool. If the effect of linalool on the autonomic nervous system becomes clear, it is thought that essential oils containing linalool may also have an effect on the autonomic nervous system. Ultimately, essential oils containing linalool are thought to be effective for patients with autonomic nervous system imbalance. The present study focused on linalool, one of the major components of geranium essential oil, and investigated the effects of linalool on the autonomic nervous system.
In this study, we adopted an elevated open-platform to measure the pulse waves of awake mice. Elevated open-platforms can restrict mouse behavior and create stress loading in mice. Furthermore, the pulse oximeters used to measure pulse waves are noninvasive devices. Pulse oximetry thus reduces the burden on mice compared to telemetry systems that require surgical interventions. In particular, when measuring the potentially subtle effects of essential oil components, such as in this study, a pulse oximeter system that minimizes the burden on mice is considered suitable.
Results and Discussion
The simple flow of the experiment was as follows. After the first measurement (“Before”) was performed for 10 min, inhalation administration (i.h.) was performed for 90 min, intraperitoneal administration (i.p.) was continued for 30 min, and the second measurement (“After”) was performed for 10 min. Results Before and After were compared to eliminate the influences of individual differences in mice.
As shown in Figure 1, the control group (water i.h., saline i.p.) showed significantly decreased heart rate After (719.3 ± 9.3 beats/min to 654.5 ± 11.3 beats/min). Similarly, linalool (5 μL/L air, i.h.) tended to decrease heart rate After (697.6 ± 25.9 beats/min to 653.1 ± 16.4 beats/min). On the other hand, fluoxetine, a serotonin reuptake inhibitor and positive control in the elevated open-platform test, resulted in little change to heart rate between before and after administration (702.1 ± 26.6 beats/min to 709.3 ± 11.9 beats/min).

Heart rates before and after administration of control (A), linalool (B: 5 μL/L air, i.h.), or fluoxetine (C: 1 mg/kg, i.p.). Values represent the mean ± standard error of heart rate for 10 min (n = 5-8).
The next result was the effect on the autonomic nervous system. In Figure 2, controls showed an increasing trend in HF as an index of parasympathetic system activity (63.4 ± 0.5% to 66.3 ± 2.1%), and a decreasing trend in LF/HF as an index of sympathetic system activity (0.37 ± 0.02 to 0.35 ± 0.03).

Autonomic changes before and after control administration. (A) Percentage (%) of high frequency (HF) as a measure of parasympathetic nervous system activity. (B) Low frequency/high frequency (LF/HF) as a measure of sympathetic nervous system activity. Values in the graph represent the mean ± standard error of 10-min measurements before and after control administration (n = 7).
Linalool administration (5 μL/L air, i.h.) showed a significant increase in HF (61.1 ± 1.4% to 65.6 ± 0.9%, p < .01) and a decreasing trend in LF/HF (0.43 ± 0.02 to 0.42 ± 0.01) (Figure 3). Intraperitoneal administration of saline was also used during inhalation of linalool.
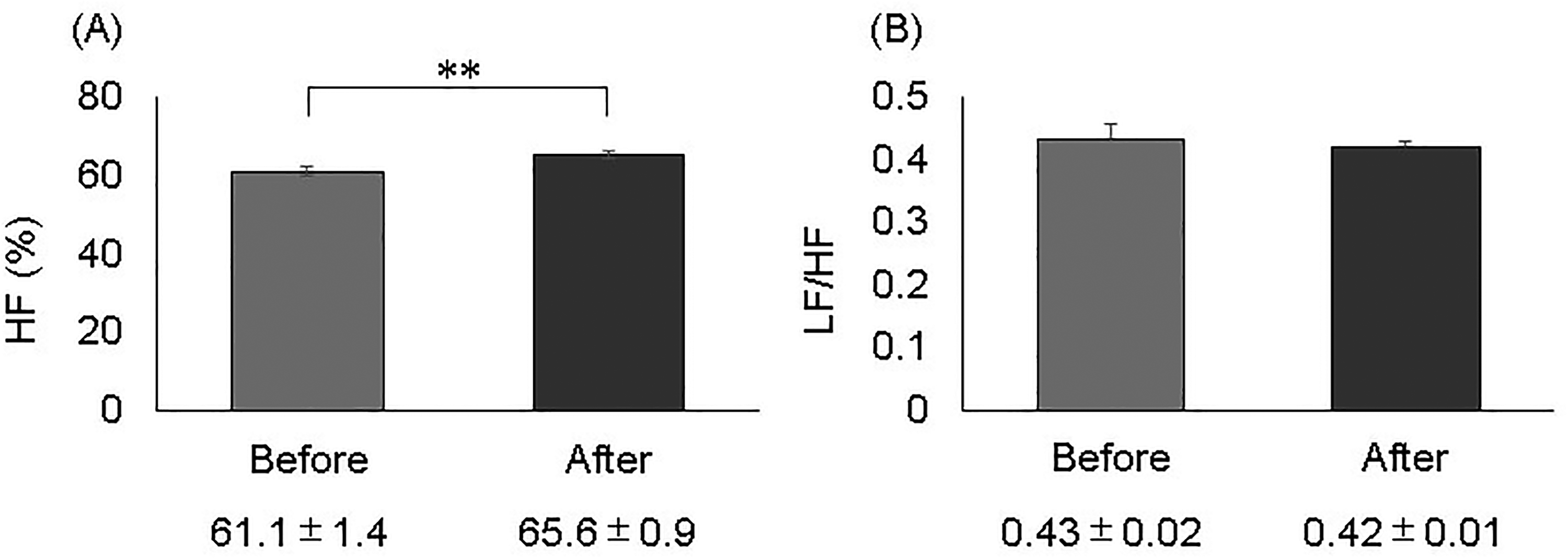
Autonomic changes before and after linalool (5 μL/L air, i.h.) administration. (A) Percentage (%) of high frequency (HF), as a measure of parasympathetic nervous system activity. (B) Low-frequency/high-frequency (LF/HF), as a measure of sympathetic nervous system activity. Values represent the mean ± standard error of 10-min measurements before and after linalool administration (n = 8). **p < .01.
Fluoxetine administration (1 mg/kg, i.p.) resulted in little change in HF (62.2 ± 1.6% to 61.8 ± 1.5%), but a trend toward increasing LF/HF (0.43 ± 0.01 to 0.45 ± 0.03) (Figure 4). In addition, the fluoxetine group was also administered water inhalation.

Autonomic changes before and after fluoxetine (1 mg/kg i.p.) administration. (A) Percentage (%) of high frequency (HF) as a measure of parasympathetic nervous system activity. (B) Low frequency/high frequency (LF/HF) as a measure of sympathetic nervous system activity. Values in the graph represent the mean ± standard error of 10-min measurements before and after fluoxetine administration (n = 5).

Experimental procedure of control (A), linalool (B: 5 μL/L air, i.h.), or fluoxetine (C: 1 mg/kg, i.p.).
Heart rate is thought to fluctuate primarily under the influence of the autonomic nervous system. Measurements in this study showed a significant decrease in heart rate after the control was administered, possibly due to the mice becoming accustomed to the device (elevated open-platform) after two measurements. From that perspective, the fact that the administration of fluoxetine, as the positive control in the elevated open-platform test, resulted in no decrease in heart rate can be considered to indicate that it objectively resulted in an increased heart rate. The increase in heart rate with fluoxetine can also be considered to correlate with the results of sympathetic dominance. Fluoxetine is a serotonin reuptake inhibitor and may decrease height-related stress by promoting dominance of the sympathetic nervous system. Currently, there are no drugs that can specifically regulate the autonomic nervous system. Additionally, fluoxetine is not a drug that clearly exhibits autonomic nerve-modulating effects. The positive control fluoxetine used this time is a positive control for elevated open-platform. 10
The sedative and vasodilatory effects of linalool have already been reported.1,5 We believe that our findings that linalool inhalation promotes parasympathetic nervous system dominance support previous findings for the sedative and other effects of linalool. The present results also support the findings of previous reports that inhalation of geranium essential oil during arousal causes parasympathetic dominance. Linalool is one of the major volatile components of geranium essential oil, so the parasympathomimetic effects of geranium essential oil are likely to involve linalool. Furthermore, we believe that the mechanism of action of linalool on the autonomic nervous system is a future issue.
The effects of aromatherapy are thought to represent a combination of weak pharmacological effects mediated through pharmacological transmission pathways and the effects of neurotransmitters mediated through olfactory stimulation. Volatile components such as linalool have already been reported to be transferred to the brain and body through inhalation (pharmacological transfer pathway by blood flow). 11 Olfactory stimulation by fragrances is thought to exert strong placebo effects in humans (neurological transmission pathway by olfactory stimulation). Although the present study did not seek to eliminate or account for olfactory sensation in mice, linalool was the first scent perceived by the captive mice. The psychological placebo effect is linked to past memories. 12
Therefore, inhaled therapies in mice, unlike in humans, are not expected to exert placebo effects. On the other hand, olfactory stimulation by linalool itself reportedly has effects on the autonomic nervous system. 13 Despite the demonstration of only weak pharmacological effects in the present results, humans may be more prone to experiencing stronger placebo effects if they are able to recognize even these weak pharmacological effects. A clearer pharmacological effect would be expected to induce a correspondingly clearer placebo effect.
Conclusions
This research revealed that linalool, a component of essential oils, has the effect of making the parasympathetic nervous system dominant in experiments on mice. Although this effect is very weak, it is a scientifically clear result. We also believe that if humans recognize this fact, the placebo effect of aromatherapy will be enhanced.
Experimental
Samples
Linalool was purchased from Tokyo Chemical Industry Co. Fluoxetine hydrochloride was purchased from Sigma-Aldrich Japan K.K.
Experimental Animals
In this experiment, 5-week-old male ICR mice (body weight approximately 30 g) were used. Mice were purchased from Japan SLC Co., Ltd. The mice had free access to food (Labo MR Stock; Nosan Co., Ltd) and water. The mouse breeding environment was set at a room temperature of 24 ± 5 °C and had a 12-h light/dark schedule (light from 7:00 to 19:00, dark from 19:00 to 7:00). This experiment was conducted with the approval of the International University of Health and Welfare Laboratory Animal Committee from the perspective of protecting the welfare of laboratory animals (animal experiment plan approval number 20015).
Measurement of Heart Rate From Pulse Wave
Mice's heart rates were measured from the pulse wave using a pulse oximeter. The mice's necks were shaved the previous day to facilitate measurement. To control mouse behavior, we used a modified elevated platform reported by Miyata et al 10 The elevated platform measured 40 cm in height and 5 cm in diameter. The elevated platform was surrounded by a wall with a diameter of 30 cm and a height of 55 cm.
A MouseOx® PLUS system (Starr Life Sciences Corp.) was used to measure pulse waves, and a PowerLab 2/26 system (AD Instruments Japan) and LabChart 8 (AD Instruments Japan) were used to analyze the data.
On the test day, individual mice were left on the elevated platform for 10 min, and the pulse wave under normal conditions was measured as the “Before” condition. Mice then inhaled 5 μL water/L air or 5 μL linalool/L air in a 10 L sealed container for 90 min. Next, the mice were intraperitoneally administered with 0.3 mL/mouse of physiological saline or 1 mg/kg of fluoxetine. After intraperitoneal administration, mice were left for 30 min. Finally, they were left again on the elevated platform for 10 min, and the postadministration pulse wave was measured in the “After” condition. The dosage of linalool was determined based on our past research results (Figure 5).8,9
Heart Rate Variability Analysis in Relation to Autonomic Nervous Activity
The obtained heart rate was analyzed for heart rate variability using LabChart 8. In heart rate variability, very low frequency (VLF), low frequency (LF), and high frequency (HF), which reflect sympathetic and parasympathetic nervous system activity, were set as follows: VLF, 0 to 0.15 Hz; LF, 0.15 to 1.5 Hz; and HF, 1.5 to 5 Hz. Sympathetic activity was expressed as LF/HF, and parasympathetic activity was expressed as HF. Although VLF is an indicator of dysregulation of the autonomic nervous system regulation mechanism due to mental stress, it was not specifically evaluated in this experiment.
Statistical Analysis
All statistical analyses were performed using Excel statistics (BellCurve for Excel, Social Survey Research Information Co., Ltd). Statistical significance was determined using a two-tailed paired samples t-test, with values of p < .01 considered significant.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the JSPS KAKENHI (Grant No. JP20K11562).
Ethical Approval
This article does not contain any studies involving human subjects. The animal experiments in this study were conducted with the approval of the International University of Health and Welfare Laboratory Animal Committee from the perspective of laboratory animal welfare (animal experiment plan approval number: 20015).
Informed Consent
This article does not involve any studies with human subjects.
Trial Registration
This article does not involve any clinical trials.
