Abstract
Background
Previously, we showed that Yikun Yitong Ping (YKYTP) can effectively and safely relieve endometriosis-related dysmenorrhea; however, the underlying mechanism remained unclear. This study aimed to assess the effects of YKYTP-containing serum on rat basophilic leukemia cell line, as mast cells (MCs) analog, and investigate the mechanisms by which YKYTP alleviates endometriosis-related dysmenorrhea.
Method
In this study, YKYTP drug-containing serum was used to treat rat basophilic leukemia cell line (RBL2H3). The effect of YKYTP-containing serum on the expression of ER-α, ER-β, NGF, and NGFRp75 in RBL2H3 cells was evaluated using enzyme-linked immunosorbent assay, quantitative real-time polymerase reaction, and Western blotting.
Results
Different concentrations (5%-40%) of YKYTP-containing serum reduced ER expression in RBL2H3 cells, and the optimum concentration was 40%. Compared to the blank group, the expression of ER and NGF significantly increased in the E2 group (P < .01). After co-administration with YKYTP-containing serum, the expression of ER-α, ER-β, NGF, and P75 significantly decreased (P < .01).
Conclusion
YKYTP-containing serum can efficiently inhibit the expression of ER-α, ER-β, NGF, and P75 in RBL2H3 cells. YKYTP may alleviate endometriosis-related dysmenorrhea by downregulating ER expression in MCs.
Introduction
Endometriosis is defined as the presence of functioning endometrium outside of the uterine cavity with an unclear pathogenesis.1,2 It presents with dysmenorrhea, dyspareunia, pelvic discomfort, and infertility1,2 and mainly affects women of reproductive age, with a prevalence of about 10%. 3 Recurrence and side effects restrict the efficacy of current treatments of endometriosis, such as surgical resection and hormone therapy.4–6 In China, people with endometriosis benefit from traditional Chinese medicine (TCM) for treating dysmenorrhea brought on by endometriosis, enhancing fertility, and reducing recurrence.7–9 Prof. Xurun San, a TCM expert, developed an herbal drug, namely Yikun Yitong Ping (YKYTP), which effectively and safely treated endometriosis-related dysmenorrhea in our previous study. 10 However, the mechanism by which YKYTP ameliorates endometriosis-associated dysmenorrhea remained unclear.
Although the exact causes of endometriosis-related dysmenorrhea have not been uncovered, estrogen-dependent neuroinflammation has been implicated in dysmenorrhea caused by endometriosis.4,11 Based on some reports, endometriosis is a systemic illness caused by immunological and endocrine perturbation.12,13 Mast cells (MCs) are important components of the immune system and play a key role in allergic response. 12 Some researchers hypothesized that the number and degranulation rate of MCs increase in endometriotic lesions.2,14,15 Interestingly, recent studies reported that estrogen activates MC in endometriosis and showed that treatment with estrogen (E2) promotes the recruitment of degranulation of MCs.16–20 Lin et al 21 reported that estrogen receptors (ERs) are expressed on MCs. Further studies have demonstrated that activated MCs can release nerve growth factor (NGF), which can promote nerve growth and induce peripheral sensitization in endometriosis-related dysmenorrhea.2,12,22 These findings suggested that high levels of local estrogen may be a key factor for recruiting and activating MCs. In our previous animal studies, we found that NGF expression is higher in rats with adenomyosis compared to control rats. We also observed that treatment with YKYTP decreased the expression levels of NGF. 23 Therefore, we hypothesized that YKYTP can reduce the expression levels of NGF in MCs by blocking ER in these cells.
In the current study, we measured the expression levels of estrogen receptor-α (ER-α), estrogen receptor-β (ER-β), NGF, and NGFRp75 in a rat basophilic leukemia cell line (RBL2H3) after treatment with E2, fulvestrant (an estrogen inhibitor), and YKYT to explore the mechanism by which YKYTP mitigates endometriosis-associated dysmenorrhea.
Methods
Reagents and Antibodies
β-Estradiol 17-acetate (HY) and fulvestrant (HY-13636) were purchased from Haoyuan Chemexpress (Shanghai, China). Rat estradiol receptor (MM-0273R2) was obtained from Mianmian Biology (Jiangsu, China). TRIzol Reagent (CW0580S), HiFiScript cDNA (CW2569 M), UltraSYBR Mixture (CW0957 M), Ultrapure RNA (CW0581 M), and BCA Protein Assay Kit (CW0014S) were all purchased from CoWin Biosciences (Jiangsu, China).
Cell Culture and Treatment
Rat basophilic leukemia cell line (RBL2H3), as the counterpart of mucosal MC, was purchased from the Chinese Academy of Sciences (Beijing, China). At 37 °C with 5% CO2, cells were grown in Dulbecco's modified Eagle's medium provided by KeyGEN Biotech in China. In addition, 10% fetal bovine serum and 1% antibiotic solution (100 U/mL penicillin and 0.1 mg/mL streptomycin) were added to the culture media. For each experiment, cells in the logarithmic phase of growth were employed.
Preparation of YKYTP
The Pharmacy Division of the China-Japan Friendship Hospital (Beijing, China) provided all medicinal herbs for YKYTP decoction, and Professor Runsan Xu of the hospital provided identification. Table 1 comprises their Chinese names, English names, Latin names, family, proportions in the drug, countries of origin, and daily adult dosages (g). The combination of the components was cooked in 8 volumes of water (v/w) for 60 min, followed by 2 extraction steps to prepare YKYTP. The clinical preparation process was the same. YKYTP was extracted and kept in a −20 °C freezer until usage.
The Composition of Yikun Yitong Ping (YKYTP). 23

Animals and Drugs
Twenty male Sprague-Dawley (SD) rats, aged 4 to 5 weeks and weighing between 240 and 300 g, were obtained from Beijing Vital River Laboratory Animal Technology Co., Ltd (China). The Animal Ethics Committee of China-Japan Friendship Hospital approved the protocol of this study (No: 190205).
Animals had free access to water and food and were kept in a pathogen-free (SPF) environment (22-24 °C, 50%-70% humidity, and a 12:12 h light:dark cycle). The health status of rats was checked once a day while they were housed, and no abnormalities were found. After 1 week of adapting to the new diet, 20 rats were randomly divided into 2 groups: a saline-treated control group and a YKYTP-containing serum-treated group. The drug (100 mL/g of body weight) was given twice daily for 3 consecutive days, equivalent to a human dosage based on body surface area.
At the end of the experiment, euthanasia was accomplished using pentobarbital sodium and cervical dislocation to lessen the pain and distress of animals. The rats were weighed and then given a lethal dose of pentobarbital sodium (45 mg/kg, Sigma, USA) intraperitoneally 1 h after the last dose of YKYTP. Subsequently, blood samples were collected from the aorta with strict sterility. After centrifuging blood samples for 5 min at 2000 r/min, serum samples were extracted. Serum samples from the same group were mixed and inactivated in 56 °C water for 30 min to prevent viral and other interferences. The samples were stored at −20 °C until assay.
Grouping and Intervention
RBL2H3 cells were divided into 5 different groups. Group 1 served as the control group; group 2 received estradiol (500 pmol/L); group 3 received fulvestrant (10-5 mol/L), an estradiol inhibitor; group 4 received both estradiol and fulvestrant (E2 + F group); and group 5 received estradiol and 40% drug-containing serum (E2 + YKYTP group). After 24 h of culture, cells from each group were analyzed.
Enzyme-Linked Immunosorbent Assay
The cells were seeded in plates before treatment. After 24 h, 1.5 mL of growth media was collected from each well, and rat ER level was analyzed using enzyme-linked immunosorbent assay (ELISA). After collecting the supernatant at each time point, we measured protein levels using an ELISA kit (MeiMian, JS, China) and according to manufacturer's instructions. The operator simultaneously analyzed samples and calibration standards to generate a standard curve of optical density based on ER concentration. Finally, we calculated ER concentration for each sample based on the standard curve.
Quantitative Real-Time Polymerase Chain Reaction
The mRNA expression of the ER was measured using real-time polymerase chain reaction (RT-PCR). TRIzol reagent was used to extract total RNA following manufacturer's instructions (CW0580S, CWBIO, JS, China). Then, an Ultrapure RNA kit (CW0581M, CWBIO, JS, China) was used for reverse transcription following manufacturer's instructions. According to the package recommendations, Step Plus real-time PCR was used to conduct quantitative measurements of various gene levels. Biotechnology Company designed the primers (Shanghai, China). Table 2 displays the primer sequences in detail.
Primer Sequences Used in Real-Time PCR.

Western Blotting
After extraction, cells were boiled in the RIPA lysis buffer (Puli Gene Technology Company, Beijing, China). Supernatants were obtained during centrifugation. The SDS-PAGE was used to extract, resolve, and transfer the total proteins to PDVF membranes. Membranes were first pre-incubated with primary antibodies at 4 °C overnight, washed with TBST buffer, and then re-incubated with the appropriate secondary antibody at room temperature for 1 h. Chemi DocTM XRS + was used to clean and analyze the membranes (Bole Life Medical Products Company, Shanghai, China).
Statistical Analysis
GraphPad Prism 7.0 was used for analysis, and the results are presented as the mean ± standard error. Differences between groups were analyzed using 1-way analysis of variance. The cut-off for significance was set at P < .05. Post-hoc Tukey's test was used for between-group comparisons. Each experiment was conducted at least 3 times.
Results
Decreased Expression of ER in RBL2H3 Cells After Exposure to YKYTP-Containing Serum
We cultured RBL2H3 cells and subjected them to various dosages of YKYTP to determine whether the concentration of drug-containing serum was correlated with ER expression. ELISA revealed that the drug-containing serum altered ER expression in RBL2H3 cells. Various concentrations of drug-containing serum decreased E2 expression. The minimum expression level of ER was observed after treatment with 40% drug-containing serum (Figure 1A). Although 40% drug-containing serum downregulated the expression level of ER within 10, 30, and 60 min, the minimum level of ER was observed after 10 min (Figure 1B).
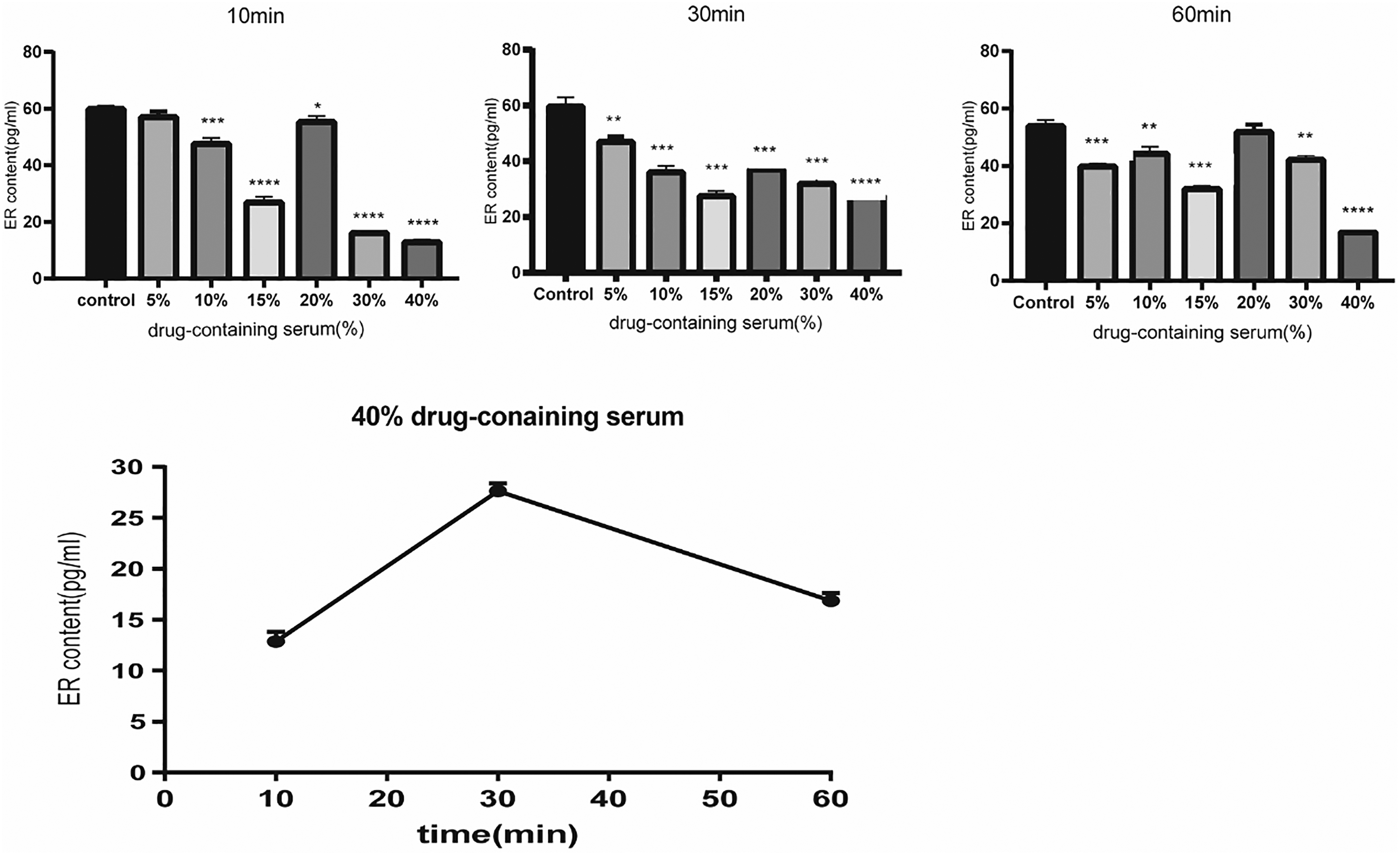
ER level in various concentrations of YKYTP-containing serum. ELISA showed that the expression levels of ER decreased in RBL2H3 cells after 10, 30, and 60 min of treatment with various concentrations of drug-containing serum. The most effective concentration of drug-containing serum was 40%, and the minimum level of ER expression was observed after 10 min.
ELISA Exhibited the Effects of YKYTP Drug-Containing Serum on ER Expression in RBL2H3 Cells
The protein expression levels of ER in RBL2H3 were detected by ELISA (Figure 2). The expression of ER in RBL2H3 cells was significantly increased after treatment with E2, suggesting the role of E2 in RBL2H3 cells. In contrast, ER expression significantly decreased in the anti-E2 (fulvestrant) group compared with the control group. ER expression levels considerably decreased in the E2 + YKYTP group compared to the E2 group, whereas there was no discernible difference in ER levels between the E2 + YKYTP and E2 + anti-E2 groups. The results of ELISA revealed that YKYTP suppressed ER levels in RBL2H3 cells.

ER expression in 5 different groups of RBL2H3 cells. (A) RBL2HS cell growth in different groups. (B) The protein expression levels of ER by RBL2H3 cells were detected by ELISA. The expression levels of ER increased by E2 (P < .01), while decreased by YKYTP (P < .01).
RT-PCR Showed That YKYTP-Containing Serum Reduced the mRNA Levels of ER in RBL2H3 Cells
We measured the mRNA level of ER by RT-PCR to verify the mechanisms by which YKYTP affects RBL2H3 cells. ELISA was used to measure ER expression level in RBL2H3 cells (Figure 3). Treatment with E2 dramatically enhanced ER expression in RBL2H3 cells, indicating that E2 affects these cells. However, ER expression levels were considerably lower in the fulvestrant group than in the control group. ER expression was considerably lower in the E2 + YKYTP group than in the E2 group. However, ER expression was not significantly different between the E2 + YKYTP group and the E2 + anti-E2 group (P > .05).

mRNA levels of ER and NGF in different groups. RT-PCR indicated that the mRNA levels of ER and NGF significantly increased after treatment with E2 (P < .01), and decreased after treatment with YKYTP (P < .01).
RT-PCR Indicated That YKYTP-Containing Serum Suppressed the Expression of NGF
NGF is released by MC degranulation; thus, we examined the mRNA levels of NGF to investigate the effect of YKYTP on RBL2H3 cell degranulation. RT-PCR exhibited that NGF mRNA levels were significantly higher in the E2 group than in the control group; however, the mRNA levels of NGF considerably reduced after adding YKYTP (P < .01). These findings suggest that YKYTP can inhibit E2-induced degranulation of RBL2H3 cells.
Western Blotting Exhibited That YKYTP-Containing Serum Downregulated ER-α, ER-β, NGF, and P75 Proteins
Using Western blotting, we assessed the protein levels of ER-α, ER-β, NGF, and P75 to delve into the mechanism by which YKYTP controls ER and RBL2H3 cell degranulation. Following E2 therapy, a dramatic increase was observed in the expression levels of ER-α, ER-β, NGF, and P75 (Figure 4). The protein levels of ER-α, ER-β, NGF, and P75 dramatically reduced when YKYTP was introduced. There was no statistically significant difference between the E2 + anti-E2 group and the E2 + YKYTP group concerning the expression levels of ER-α, ER-β, NGF, and P75 (P > .05). Based on these findings, YKYTP may regulate ER-α and ER-β expression to suppress the release of NGF and P75 via MC degranulation.
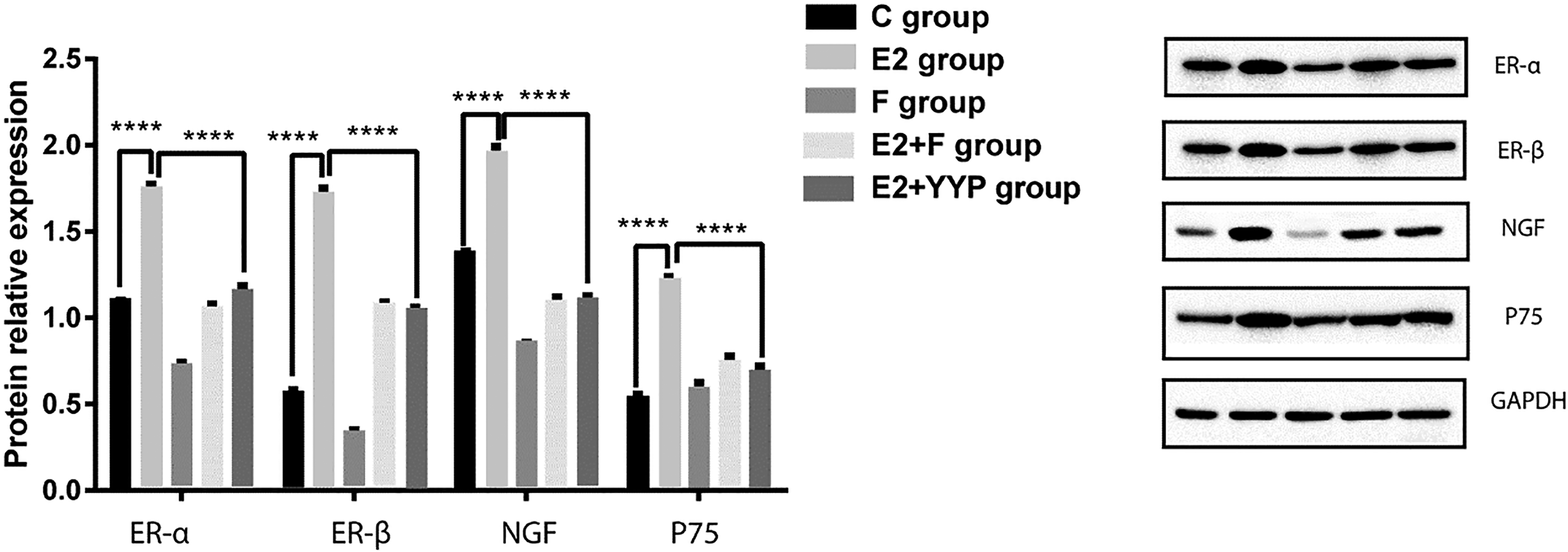
ER, NGF, and P75 protein expression in different groups. Western blotting revealed that the protein levels of ER-α, ER-β, NGF, and P75 significantly increased after treatment with E2 (P < .001) and decreased after treatment with YKYTP (P < .001).
Discussion
Here, we showed that YKYTP-containing serum can suppress ER expression in RBL2H3 cells, which means YKYTP can prevent MC degranulation by inhibiting ER. NGF and P75 both were upregulated in RBL2H3 cells following treatment with E2 while downregulated after adding YKYTP, suggesting that YKYTP may suppress ER-mediated NGF and P75 release by MCs.
Chronic pelvic discomfort, dysmenorrhea, and infertility are hallmarks of endometriosis, a prevalent gynecological disease that undermines patients’ quality of life, physical and emotional health, and productivity. 24 Our previous clinical investigation validated the efficacy and safety of YKYTP in the management of dysmenorrhea caused by endometriosis 10 ; however, the mechanism by which YKYTP mitigates endometriosis-related dysmenorrhea remained unclear.
Estrogen-dependent neuroinflammation has been implicated in the pathogenesis of endometriosis-related dysmenorrhea.3,25,26 Previous findings suggested that E2 may increase NGF and P75 levels in peritoneal endometriosis by increasing inflammation-nerve interactions. 11 As a key player in neuropathic pain, NGF is involved in various processes, including pain sensation, brain plasticity, immune cell aggregation, and inflammatory factor release.12,13,27,28 Previous studies indicated that increased E2 levels play a critical role in MC degranulation and recruitment and induce the release of NGF.20,29 Increased expression of NGF and platelet-derived growth factor in endometriotic lesions may also induce the ingrowth of nerve fibers into endometriotic tissue, which causes pain and local tenderness.18,29–32 Researchers have shown that NGF and P75 are overexpressed in patients with endometriosis compared to those without endometriosis, suggesting that these factors are involved in disease pathogenesis.33–35 We previously observed that YKYTP can suppress NGF and P75 expression in the rat model of adenomyosis. 23 In this study, we set out to investigate whether YKYTP regulates ER expression in MCs and neurons.
We employed a rat basophilic leukemia cell line, which is widely used for in-vitro studies of MCs, to assess our hypothesis (RBL2H3). The results of RT-PCR, Western blotting, and ELISA showed that treatment with estradiol (E2) increased the expression of ER and NGF, suggesting that E2 can induce RBL2H3 cells to release NGF by regulating the expression of ER. Our findings were consistent with those reported by Zhu et al. 8 Based on these findings, E2 was identified as a key mediator involved in inflammation-nerve crosstalk in endometriosis-associated dysmenorrhea.
Consistent with ELISA, Western blotting indicated markedly reduced protein levels of ER-α, ER-β, NGF, and P75 in the E2 + YKYTP group compared to the E2 group. In addition, compared to the E2 group, the E2 + YKYTP group had considerably decreased mRNA levels of ER and NGF. Our data showed that YKYTP can mitigate endometriosis-induced dysmenorrhea by reducing ER levels and preventing ER-mediated NGF release by MCs. There was no difference in the expression levels of ER and NGF between the E2 + YKYTP group and the E2 + F group. Compared to estrogen inhibitors, the efficacy and safety of YKYTP have been verified in clinical settings.
We conclude that E2 mediates inflammation-nerve crosstalk in endometriosis-associated dysmenorrhea by stimulating MC degranulation and NGF release. We also observed that YKYTP-containing serum decreased ER expression in RBL2H3 cells, thereby inhibiting their activation and reducing NGF release (Figure 5). YKYTP may alleviate endometriosis-related dysmenorrhea via downregulating ER expression in MCs. Our findings can advance the use of TCM in the treatment of endometriosis. However, RBL2H3 cells were used in this study, they do not behave the same as MCs in patients with endometriosis. Therefore, future studies should employ rat models of endometriosis and primary MCs from the peritoneal fluid of patients with endometriosis to unravel the mechanisms by which YKYTP improves endometriosis-related dysmenorrhea.

The mechanism by which YKYTP relieves endometriosis-associated dysmenorrhea. YKYTP reduced the expression levels of NGF and P75 in MCs by blocking estrogen receptors on Mast Cells.
According to these data, we showed that YKYTP-containing serum can suppress ER expression in RBL2H3 cells, suggesting that YKYTP can prevent MC degranulation by inhibiting ER. NGF and P75 both were upregulated in RBL2H3 cells following treatment with E2 while downregulated after adding YKYTP, suggesting that YKYTP may suppress ER-mediated NGF and P75 release by MCs. Therefore, we can assume that YKYTP inhibits ER on the surface of RBL2H3 cells to inhibit MC degranulation.
We conclude that E2 mediates inflammation-nerve crosstalk in endometriosis-associated dysmenorrhea by stimulating MC degranulation and NGF release. We also observed that YKYTP-containing serum decreased ER expression in RBL2H3 cells, thereby inhibiting their activation and reducing NGF release (Figure 5). YKYTP may alleviate endometriosis-related dysmenorrhea by downregulating ER expression in MCs. Our findings can advance the use of TCM in the treatment of endometriosis.
While providing insights into the effects of YKYTP on the RBL2H3 cell line, we acknowledge several limitations. We used RBL2H3 cells to explore the possible mechanisms by which YKYT affects MCs. However, RBL2H3 cells possess the characteristics of MCs rather than being MCs in the true sense. They do not behave the same as MCs in patients with endometriosis. In addition, we only indirectly proved the effect of YKYTP on MC degranulation through ER, NGF, and P75. Therefore, in future experiments, we will further verify the mechanism by which YKYTP affects MCs through animal experiments and measure changes in the number of MCs in the human peritoneal cavity. Besides, YKYT is a compound preparation for clinical application with various medicinal constituents. In the future, we will investigate which specific constituent of YKYTP plays a specific role in endometriosis. Our findings might not be completely generalizable to endometriosis-related dysmenorrhea in humans due to the complexity of human MC responses. Additionally, focusing solely on a specific cell line limits the comprehensive understanding of YKYTP's active components. Future studies are needed to address these limitations, aiming to deepen our understanding and promote the clinical application of YKYTP.
Conclusion
This study provides a deeper insight into the treatment of endometriosis-associated dysmenorrhea with YKYTP and helps find drug targets for pain inhibition. Endometriosis-related dysmenorrhea is treatable, and YKYTP can be prescribed as an alternative treatment in clinical settings. Still, more studies are needed to fully uncover the underlying mechanisms behind the clinical findings.
Footnotes
Data Availability
Data will be provided by the corresponding author upon a reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
The Animal Ethics Committee of China-Japan Friendship Hospital approved the protocol of this study (No: 190205).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Beijing Administration of Traditional Chinese Medicine (grant number 100108-1-02).
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the Institutional Animal Care guidelines of Nantong University, China, and approved by the Animal Ethics Committee of China-Japan Friendship Hospital, China.
