Abstract
Introduction
Plant-parasitic nematodes cause substantial economic losses to agriculture worldwide and threaten the sustainable development of modern agriculture, 1 with estimated annual economic losses of $157 billion worldwide.2,3
Root-knot nematodes (Meloidogyne sp.) are among the groups with the highest pathogenic capacity and the largest number of hosts. Specifically, Meloidogyne javanica is an important pathogen of vegetables worldwide, impacting both the quantity and the quality of marketable products. 4 On the other hand, the control of Meloidogyne has been mainly based on chemical nematicides. Despite their effectiveness, most of these compounds are withdrawn from the market because of their toxicity and severe impact on the environment and human health.
Therefore, developing new nematicidal agents with high efficiency and low toxicity is essential. 1 In this context, natural products (NPs) have gained interest in recent years due to their potential to control soil nematodes and because NPs provide diverse structural models for the bioinspired synthesis of new nematicidal compounds. 5
Phenolic compounds are elicitated by root-knot nematodes6,7 and have been associated with plant resistance to the pathogens in relation to the hypersensitivity reaction.8,9 Furthermore, phenolic compounds can attract Meloidogyne species second-stage juveniles 10 and also have direct nematicidal effect.5,9,11 Among natural and synthetic nematicidal phenolic compounds are aldehydes, 12 lactones, acetophenones, and chalcones,13–15 phenolic terpenes such as thymol and carvacrol, eugenol, 15 and phenolic acids such as salicylic. 16
The phenylketone group is a part of many natural molecules, such as flavonoids, chromones, some alkaloids, and acetophenones, and show a wide range of biological activities.17,18 Natural acetophenones are involved in plant defenses19,20 and usually have phenolic hydroxy groups and additional free or cyclized isoprenyl units. The nematicidal potential of acetophenones substituted in the 4′position with electrophilic groups 14 and haloacetophenones 21 has been explored, and the nematicidal effect of 2,4-dihydroxyacetophenone has been reported. 9
On the other hand, chalcones, (E)-1,3-diaryl-2-propen-1-ones, are characterized by two aromatic rings with an array of different substituents, and have biological activities including antimalarial, antiinflammatory, antibacterial, antiprotozoal, antifilarial, antifungal, and mosquito larvicidal. 22 Natural and synthetic chalcones have also been reported as being nematicidal. For example, E-chalcone exhibited activity against Globodera pallida, 23 while the ferrocenyl chalcones (2E)-1-(4-methylphenyl)-3-ferrocenyl-prop-2-en-1-one and (2E)-1-(4-methoxyphenyl)-3-ferrocenyl-prop-2-en-1-one decreased the incidence of M. incognita in tomato plants. 13 However, organic chalcones had better nematicidal activity than ferrocenyl chalcones against the model nematode Caenorhabditis elegans. 24 Other synthetic chalcones such as 2,4,5-trimethoxy-4´-nitrochalcone, 25 1-(4-fluoro-phenyl)-3-phenyl-propenone and 1,3-diphenyl-propenone, 26 (1E,4E)-1,5-di(4-nitrophenyl)-2-butylpenta-1,4-dien-3-one 27 were effective against M. exigua, M. graminicola, and M. incognita, and according to in silico studies, the target enzyme is cytochrome P450. 27
In a recent work, 26 phenolic molecules, including acetophenones and chalcones, have been synthesized to study the importance of the hydroxyl and isoprenyl substituents and the pyran-4-one systems in their trypanocidal activity. 18 However, their nematicidal activity has not been explored.
In this study, a series of hydroxylated and prenylated acetophenones and chalcones have been synthesized and evaluated on the nematode M. javanica. Additional phenolic compounds, including salicylic acid, isopropyl salicylate, thymol, carvacrol, cumic acid, eugenol, vanillin, and vanillic acid, were also tested to increase the chemical diversity and further understand the nematicidal structure-activity relationships of phenolic compounds.
Results and Discussion
Compounds

Synthetic strategy for preparing derivatives 5-21. Reagents and conditions: (I) Ac2O, BF3-Et2O, 50 °C, 12 h, Yield 16.5-56%; (II) 3-Methyl-2-buten-1-ol, BF3-Et2O, room temperature, 24 h, Yield 5.1-12.3%; (III) K2CO3, MOMCl, ACN, 60 °C, MW, 1 h, Yield 48.0-77.5%; (IV) KOH/EtOH, 40 °C, 12 h, Yield 46.3-77.5%.
Compounds
Additional compounds (

Chemical structures of additional phenolic compounds tested.
Considering the importance of phenolic compounds in the nematode plant interactions, we have studied the nematicidal activity of
Effects of Compounds

aCorrected according to Scheider-Orelli's formula. Values are means of four replicates.
bLethal doses to give 50% and 90% mortality (Probit analysis).
c95% Confidence Limits.
dCompound
The phenolic compounds resorcinol (
Prenylation of the acetophenones removed the activity in almost all cases. Thus, compounds of the type 2′-hydroxy-prenyl acetophenones (
Moreover, additional compounds (
The nematicidal action of the most effective compounds (
Effects of the Nematicidal Compounds
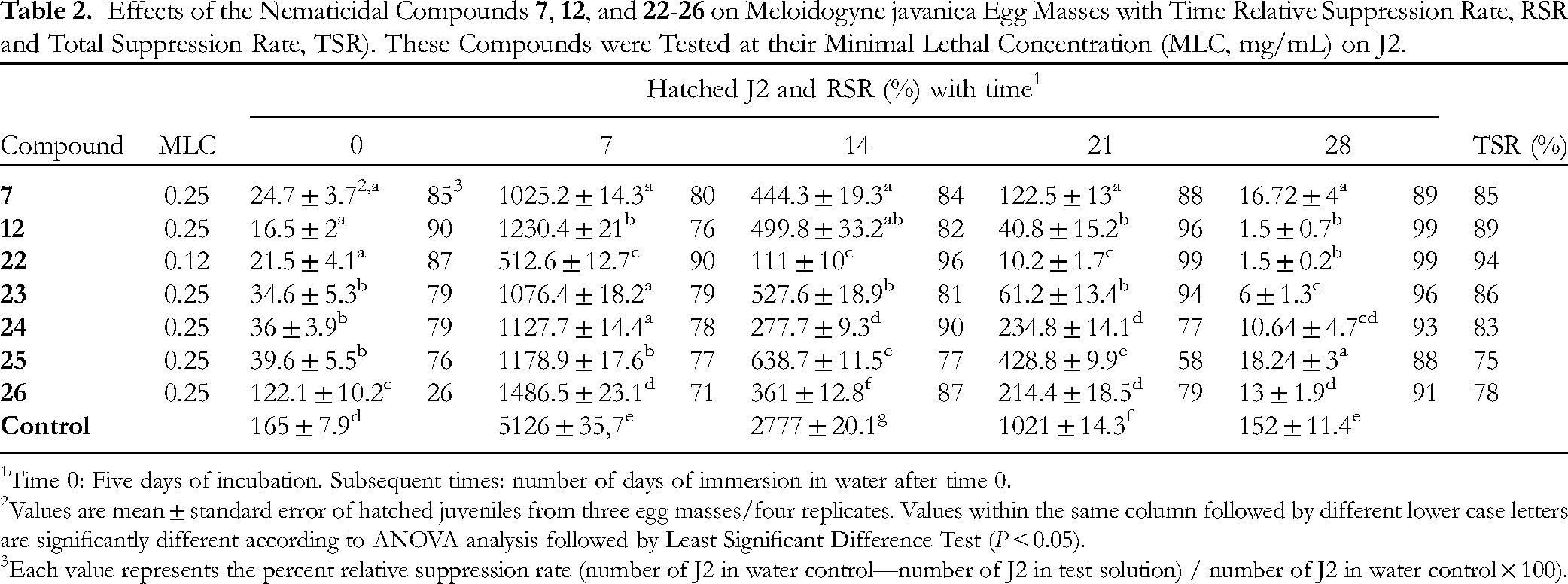
1Time 0: Five days of incubation. Subsequent times: number of days of immersion in water after time 0.
2Values are mean ± standard error of hatched juveniles from three egg masses/four replicates. Values within the same column followed by different lower case letters are significantly different according to ANOVA analysis followed by Least Significant Difference Test (P < 0.05).
3Each value represents the percent relative suppression rate (number of J2 in water control—number of J2 in test solution) / number of J2 in water control × 100).
This is the first report on the nematicidal and egg hatching inhibition effects of compounds
Several compounds studied here such as
Carvacrol (
Eugenol (
Conclusions
Developing new nematicidal agents with high efficiency and low toxicity is essential. In this context, NPs have gained interest in recent years due to their potential to control soil nematodes and their diverse structural models for the bioinspired synthesis of new nematicidal compounds. In the search for substances with nematicidal activity against M. javanica, 22 synthesized compounds were tested to determine the role of hydroxy groups and an isoprene residue (5 carbon atoms) in an acetophenone system. Among the hydroxylated acetophenones (
Our results confirm previous reports about the effect of hydrophilic groups in the decrease of the nematicidal action of acetophenones. It also highlights the importance of a benzoic acid core group and the need for a 2′-OH type group to highlight the activity of phenols.
48
Moreover, this study contributes to the generation of new structure leads for the development of new nematicidal compounds, such as hydroxylated acetophenones (
Materials and Methods
General Procedure
All commercially available reagents and solvents were obtained from commercial suppliers and used without further purification. Phenols
Chemical Synthesis
Synthesis and prenylation of 2-hydroxyacetophenone derivatives: The synthesis of the acetophenones
Preparation of prenylated chalcones: The synthesis of the prenylated chalcones
NMR-spectroscopy and Mass Spectrometry
Compounds
Compound (
3-(4-(dimethylamino)phenyl)-1-(2-hydroxy-4-(methoxymethoxy)-5-(3-methylbut-2-en-1-yl)phenyl)prop-2-en-1-one: Yield 77.5%, m.p.: 112 °C-113 °C; IR (KBr) ʋ/cm:1. 1H NMR (300 MHz, CDCl3) δ 13.54 (s, 1H), 7.86 (d, J = 15.3 Hz, 1H), 7.63 (s, 1H), 7.56 (d, J = 8.8 Hz, 1H), 7.37 (d, J = 15.2 Hz, 1H), 6.74 (d, J = 8.6 Hz, 1H), 6.63 (s, 1H), 5.33-5.17 (m, 3H), 3.47 (s, 2H), 3.29 (d, J = 7.1 Hz, 2H), 3.06 (s, 6H), 2.17 (s, 6H), 1.76 (s, 6H).13C-NMR (75 MHz, CDCl3) δ 191.97, 164.65, 161.01, 145.06, 132.74, 130.60, 129.96, 122.54, 121.67, 114.45, 112.39, 112.14, 102.14, 93.96, 56.35, 40.56, 30.99, 28.42, 25.85, 17.92. HRMS (ESI) m/z, calculated for C24H30NO4 [M + H]+ 396.2169; found 396.2176
Compound (
4-(3-(2-hydroxy-4-(methoxymethoxy)-5-(3-methylbut-2-en-1-yl)phenyl)-3-oxoprop-1-en-1 yl)benzonitrile: Yield 72.3%, m.p.: 121-122 °C; IR (KBr) ʋ/cm:1. 1H NMR (300 MHz, CDCl3) δ 13.09 (s, 1H), 7.88 (d, J = 15.6 Hz, 1H), 7.84 (s, 1H), 7.78 (s, 4H), 7.70 (d, J = 15.9 Hz, 1H), 7.64 (s, 1H), 6.71 (s, 1H), 5.30 (sbr, 3H), 3.53 (s, 3H), 3.29 (d, J = 6.9 Hz, 2H), 1.80 (s, 3H), 1.79 (s, 3H).13C-NMR (75 MHz, CDCl3) δ 191.09, 165.18, 162.03, 141.53, 139.21, 133.02, 132.76, 130.06, 128.78, 123.82, 122.24, 118.45, 114.06, 113.52, 102.26, 94,02, 56.46, 28.47, 25.84, 17.95. HRMS (ESI) m/z, calculated for C23H24NO4 [M + H]+ 379.1771; found 379.1778
Compound (
7-(methoxymethoxy)-6-(3-methylbut-2-en-1-yl)-2-(4-nitrophenyl)-4H-chromen-4-one: Yield 75.5%, m.p.: 157-158 °C; IR (KBr) ʋ/cm:1. 1H NMR (300 MHz, CDCl3) δ 8.23 (d, J = 8.7 Hz, 1H), 7.97 (d, J = 8.7 Hz, 1H), 7.54 (s, 1H), 7.01 (s, 1H), 6.73 (s, 1H), 5.35 (s, 1H), 5.26 (t, J = 7.2 Hz, 1H), 3.52 (s, 1H), 3.29 (d, J = 7.1 Hz, 1H), 1.75 (s, 3H), 1.69 (s, 3H). 13C-NMR (75 MHz, CDCl3) δ 183.05, 167.38, 163.14, 149.49, 147.38, 139.01, 134.05, 131.57, 128.09, 124.79, 123.94, 121.01, 114.15, 108.17, 97.56, 94.42, 56.58, 28.31, 25.89, 17.79. HRMS (ESI) m/z, calculated for C22H22NO6 [M + H]+ 396.1441; found 346.1443
Nematicidal Activity
A field-selected M. javanica population from Barcelona, Spain, was maintained on tomato plants (Solanum lycopersicum L. var. Marmande) cultivated in pot cultures in environmentally controlled growth chambers (at 25 ± 1 °C, >70% relative humidity). Egg masses of M. javanica were handpicked from infected tomato roots 2 months after inoculation of the seedlings. Second-stage juveniles (J2) were obtained by incubating egg masses in a water suspension at 25 °C for 24 h.
In vitro Effects on Juveniles J2
Second-stage juveniles (J2) were obtained from hatched eggs by incubating egg masses in a water suspension at 25 °C for 24 h. evaluated as described.
7
Briefly, 100 J2 were placed per well of a 96-well plate in 95 ml of water. Treatments and control consisted of 5 mL of a DMSO-Tween solution (0.2% Tween 20 in DMSO) added to each well. The stock test solutions were prepared at 10 mg/ml resulting in an initial concentration of 0.5 mg/ml (except for compound
In vitro Effects on Egg Hatching
Three egg masses of uniform size were washed with sterilized distilled water and transferred to a 12-well plate containing 400 μL of distilled water. Treatments and controls consisted of 22 mL of a DMSO-Tween solution (0.2% Tween 20 in DMSO, with or without the test compound) added to each well. Each experiment was replicated four times. The plates were covered to prevent evaporation and incubated in the darkness at 25 °C. After the incubation period (5 days), the hatched J2 s were counted, and the test solutions were replaced with sterilized distilled water. The egg masses were monitored weekly (immersion time). 9 The number of hatched J2 was calculated after four immersion times. Values were transformed by Log10 (x + 1), analyzed by ANOVA, and means separated by Least Significant Difference Test at P < .05. The relative suppression rate was calculated for each immersion time as follows:
Relative suppression rate (%) = (number of J2 in control—number of J2 in test solutions) / number of J2 in control × 100.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241227689 - Supplemental material for Potential of 2-Hydroxyacetophenone Derivatives and Simple Phenol's for the control of Meloidogyne javanica
Supplemental material, sj-docx-1-npx-10.1177_1934578X241227689 for Potential of 2-Hydroxyacetophenone Derivatives and Simple Phenol's for the control of Meloidogyne javanica by Luis Alberto González López, Maria Fe Andres, Wiston Quiñones, Fernando Echeverri and Azucena Gonzalez-Coloma in Natural Product Communications
Footnotes
Acknowledgements
We acknowledge Felipe de la Peña (ICA-CSIC) for technical support with nematicidal tests.
Author Contributions
LAG and AGC wrote the first draft of the manuscript and formatted the last version. AGC edited the manuscript. MFA, FE, and WQ revised the manuscript. LAG, FE, and WQ performed the chemical synthesis; MFA and AGC performed the biological experiments. All authors have read and agreed to the published version of the manuscript.
Data Availability Statement
Data available upon request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported by grants ICOOP-CSIC: COOPA20324L, Ministerio de Ciencia, Tecnología e Innovación Minciencias and Universidad de Antioquia, grant number 111571249860-061-2016, contract 061-2016A and grant PID2019-106222RB-C31/SRA (Agencia Española de Investigación AEI, 10.13039/501100011033). Minciencias and Universidad de Antioquia scholarship in Doctorados Nacionales Program, 647-2014 was granted to Luis Alberto González López.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
