Abstract
Objectives
Due to the urgent need to develop innovative, environmentally sustainable bactericides, we use the natural product isoquinoline as a lead compound to discover highly active bactericides from isoquinoline derivatives.
Methods
In this paper, the antibacterial activity of 49 isoquinoline derivatives was evaluated against three plant bacteria in vitro.
Results
Among all the derivatives, isoquinoline-3-carboxylic acid (IQ3CA) demonstrated significant antibacterial activity against Ralstonia solanacearum (Rs), Acidovorax citrulli (Ac), X. oryzae pv. oryzicola (Xoc), X. campestris pv. campestris (Xcc), P. carotovorum subsp. carotovorum (Pcc), and X. fragariae (Xf), with EC50 values ranging from 8.38 to 17.35 μg/mL. Furthermore, IQ3CA exhibited a potent protective effect against Ac, with an efficacy of 68.56% at 200 μg/mL, which was not significantly different from that of the positive control kasugamycin (72.48%) and was superior to that of the positive control thiosen copper (64.62%). The scanning electron microscopy observations revealed that treatment of Ac cells with IQ3CA at a concentration of 25 μg/mL resulted in a curved and sunken cell morphology, along with destroyed cell membrane integrity. Additionally, the motility and exopolysaccharides production of Ac were inhibited, and biofilm formation was prevented.
Conclusion
These results suggest that IQ3CA holds promise as a lead compound with antibacterial properties against plant diseases.
Introduction
Phytopathogenic bacteria-induced plant bacterial diseases have significant global economic importance as they severely impact crop yield and quality.1,2 The incidence of bacterial diseases continues to rise due to changing cropping patterns and global climate warming. Notably, bacterial diseases such as rice bacterial leaf blight (BLB) caused by Xanthomonas oryzae pv. oryzae (Xoo),3,4 tomato bacterial wilt caused by Ralstonia solanacearum (Rs), 5 and bacterial fruit blotch (BFB) of cucurbits caused by Acidovorax citrulli (Ac)6,7 result in substantial losses of agricultural crops worldwide. Currently, the management of bacterial infections in plants is primarily reliant on the application of copper derivatives and antibiotics. 8 Nevertheless, the irrational usage of existing antibacterial agents over an extended period has resulted in the emergence of bactericide-resistant strains, thereby causing a range of adverse effects such as environmental contamination and jeopardized food safety.9,10 Consequently, there is an urgent need to develop of innovative and environmentally sustainable bactericides. Isoquinolines are heterocyclic aromatic compounds11,12 that possess various pharmacological functions, including anti-HIV, 13 antitumor,14,15 antifungal, 16 antibacterial,17–19 antioxidant, 20 anti-inflammatory, 21 and insecticidal activities. 22 Isoquinoline derivatives are used as anesthetics, antihypertension agents, disinfectants, and vasodilators. Isoquinoline is also employed in manufacturing dyes, paints, insecticides, antifungal agents, and as extraction solvents for resins and terpenes, as well as an antiseptic preservative agent. 23 Certain isoquinoline alkaloids, such as berberine, have been used as commercial botanical fungicides and bactericides for plant diseases in China.24,25 Despite their potential therapeutic applications, the antibacterial activity and underlying mechanisms of isoquinoline compounds have yet to be systematically investigated. Consequently, it is crucial to investigate the antibacterial activity and associated mechanisms of action of these substances to establish their potential as alternative bactericides and promising lead compounds for the development of environmentally safe, efficacious, and fully biodegradable pesticides.
To identify potential valuable isoquinoline compounds for subsequent investigation, this study initially conducted a screening process involving 49 isoquinoline compounds to evaluate their antibacterial activity against Xoo, Rs, and Ac. The compounds exhibiting significant activity in this screening process were subjected to further testing to determine their antibacterial spectrum and EC50 values against sensitive plant pathogenic bacteria. Finally, we assessed the impact of these highly active compounds on various physiological and biochemical indicators of sensitive bacteria, including bacterial morphology, motility, and cell membrane integrity, and discussed their potential bactericidal mechanisms. The findings from this research could provide valuable insights for the development of novel antibacterial agents based on isoquinoline compounds.
Materials and Methods
Chemicals and Bacterium
Chemicals
Forty-nine isoquinoline compounds were purchased from Bide Pharmatech Ltd (Shanghai, China), and the structures are shown in Figure 1. Kasugamycin (65%) and kasugamycin 2% aqueous solution were obtained from Guangxi Tianyuan Biochemical Co. Ltd (Nanning, China)and Jiangsu Jianpai Agrochemical Co. Ltd (Yancheng, China), respectively. Thiosen copper 20% suspension concentrate was purchased from Zhejiang Dongfeng Chemicals Co., Ltd (Wenzhou, China).

The structure of 49 isoquinulines.
Bacterium
Fourteen phytopathogenic bacterial strains assayed for in vitro antibacterial screening were A. citrulli (Ac, BFB), X. oryzae pv.oryzae (Xoo, BLB of rice), X. oryzae pv. oryzicola (Xoc, bacterial leaf streak of rice), X. axonopodis pv. citri (Xac, citrus bacterial canker), X. campestris pv. mangiferaeindicae (Xcm, bacterial black spot of mango), Pseudomonas syringae pv. lachrymans (Psl, cucurbit angular leaf spot), Ralstonia solanacearum (Rs, bacterial wilt of tomato), P. syringae pv. actinidiae (Psa, bacterial canker of kiwifruit), Pectobacterium carotovorum subsp. brasiliense (Pcb, bacterial soft rot of potato), P. carotovorum subsp. carotovorum (Pcc, bacterial soft rot of Chinese cabbage), X. axonopodis pv. manihotis (Xam, bacterial blight of cassava), Dickeya zeae (Dz, bacterial soft rot of banana), X. fragariae (Xf, bacterial angular leaf spot of strawberry), and X. campestris pv. campestris (Xcc, black rot of cabbage). All the strains were kindly provided by different researchers and then long-term preserved in our lab in 20% glycerol at −80 °C for further use. Strains were cultured on Luria-Bertani agar plates (containing 10 g of tryptone, 5 g of yeast extract, 10 g of NaCl, 16 g of agar and 1 L of distilled water) or in LB broth (without agar) at 28 °C in the dark.
Methods
In Vitro Antibacterial Assay
In vitro antimicrobial activities of all target compounds against Xoo, Rs and Ac were evaluated using a turbidity assay
26
with kasugamycin as the positive control and dimethyl sulfoxide (DMSO) as the blank control. The compounds were dissolved in DMSO and then diluted with culture medium to achieve the concentrations of 50 and 100 µg/mL. Ten microliters bacterial suspensions and 190 microliters drug-containing medium were added to a 96-well plate. The bacterial suspension solutions were incubated for 24-48 h at 28 ± 1 °C with the shaking rate of 180 rpm in a constant temperature shaker until the control solution turned turbid. The OD600 nm was determined and the inhibition rates were calculated by the following formula. In the following formulation, CK represents the OD value of the blank control group and T denoted the OD value of treatment groups. Each treatment was repeated three times.
EC50 of IQ3CA Against Sensitive bacteria
The present study investigated the toxicities of IQ3CA on five plant pathogenic bacteria including Ac, Dz, Rs, Xam, and Xoo, at varying concentrations of 50, 12, 12.5, 6.25 and 3.125 µg/mL. The EC50 values were determined by employing a probit analysis with SPSS software (version 20.0, IBM Corp., New York, USA), wherein the optical density inhibition rate was regressed against the log of bactericide concentrations. The experiment was conducted thrice to ensure the reliability of the results.
In Vivo Antibacterial Assay
In vivo antibacterial activity of IQ3CA against Ac was evaluated by greenhouse pot experiments. 27 IQ3CA was dissolved in DMSO and serially diluted with distilled water containing 0.1% Tween-80 to obtain the concentrations of 200, 100, and 50 µg/mL. Distilled water containing 0.1% Tween-80 was used as a blank control and 2% kasugamycin water solution applied as a positive control. Each treatment was performed in triplicate, and six plants were measured for each replicate. For the experiment of preventive effect, melon seedlings with two true leaves were sprayed with different concentrations of agents. After 24 h, the cotyledons were punctured and inoculated with a sterile needle, creating four wounds per cotyledon. Bacterial suspensions (OD600 = 1.0) of Ac were then inoculated into cotyledons of melon. For the experiment of curative effect, the cotyledons of melons were inoculated with the same bacterial suspensions, 24 h before the application of the test agents at different concentrations. All the inoculated melon seedlings were placed in the greenhouse (16 h light, 25 ± 2 °C, 60 ± 5% RH and 8 h dark, 20 ± 2 °C, 75 ± 5% RH) for 7 days, and then the disease index and control efficacy were calculated.
The disease index was graded as follows: Grade 0: Indicated no disease; Grade 1: Indicated the disease area account for less than 5% of the leaf area; Grade 3: Indicated the disease area accounts for 25-30% of the leaf area; Grade 5: Indicated the disease area accounts for 31-50% of the leaf area; Grade 7: Indicated the disease area accounts for 51-75% of leaf area; Grade 9: Indicated the disease area accounts for more than 76% of leaf area.
Growth Curve
Effect of IQ3CA on growth of Ac in vitro was determined using the method described by Silva-Angulo et al 28 with minor modification. The concentrations of IQ3CA were 50, 25, 12.5, 6.25, and 3.13 µg/mL, respectively. Three independent growth assays were conducted for each concentration.
Bacterial Morphology
Scanning electron microscopy (SEM) assayed was performed to evaluate the effect of IQ3CA on the ultrastructure of Ac. 29 The concentrations of IQ3CA were 50 and 25 µg/mL, respectively.
The morphologies and microstructures of compound IQ3CA against Ac were observed by SEM. 28 Bacterial culture of 10 mL was incubated to logarithmic phase (OD600 = 0.6) and centrifuged at 5000 r/min for 5 min at 4 °C and washed three times in 0.1 mol/L phosphate buffer (pH 7.2). Bacterial culture of 10 mL was incubated to logarithmic phase (OD600 = 0.6) and centrifuged at 5000 r/min for 5 min at 4 °C. IQ3CA was then supplemented to the bacterial suspension to reach final concentrations of 50 μg/mL and 25 μg/mL, respectively. The bacterial suspensions were cultivated at 28 °C for 24 h with shaking at 180 r/min, and then the cells were harvested by centrifugation at 5000 r.min for 5 min at 4 °C. After washing with a rinse solution (0.1 mol/L phosphate buffer, pH 7.2) three times, the cells were fixed in 2.5% glutaraldehyde for 4 h at 4 °C, and then washed with 0.1 mol/L phosphate buffer (pH = 7.2), three times for 5 min each again. Samples were dehydrated in a graded series of ethanol: 30%, 50%, 70%, 80%, 90%, and 100% for 15 min each and finally dehydrated by 100% isoamyl acetate for 20 min. After freeze-drying for 12 h and coating with gold, the morphology of the sample was observed.
Cell Membrane Permeability
Effect of IQ3CA on the cell membrane permeability of Ac was determined according to the method of Ernst et al 30 The concentrations of IQ3CA were 50, 25, 12.5, 6.25, and 3.13 µg/mL respectively. Assays were repeated three times, and each assay was conducted in triplicate and the same as following.
Swimming Assay
Swimming assay of Ac was implemented according to the method of Bonaventura et al 31 with slight modification. Cultures of Ac (OD600 = 0.6) were prepared. The LB with 0.3% agar powder was completely dissolved via a heating with a microwave oven. After the temperature decreased to 40 °C, IQ3CA were added to the culture medium at the final concentrations of 50, 25, 12.5, and 6.25 µg/mL. An equal volume of acetone was used as solvent control. The agent-contained medium was poured into sterile petri plates. After solidification, 5 µL of bacterial suspension was drop-inoculated at the center of semisolid medium plates and incubated at 28 °C for 48 h. Swimming motility was evaluated by measuring the diameter of the longest bacterial circles. The motility assays were performed three times.
EPS Content
EPS content was determined according to the method of Shi et al 32
Biofilm Formation Assay
Effect of IQ3CA on biofilm formation was assessed according to a previous description. 33
Statistical Analysis
Results were expressed as means ± standard deviation of three independent repeated experiments. Data were performed with a variance analysis using the SPSS software (version 20.0, IBM Corp., Armonk, NY, USA). Duncan's Multiple Range Test was conducted to compare a statistical significance at P <0.05 level. Graphs were generated with Sigma Plot (version 12.5, Systat Software Inc., San Jose, CA, USA).
Results and Discussion
Antibacterial Screening of Isoquinoline Compounds
The antimicrobial activities of 49 isoquinoline compounds against Xoo, Rs and Ac are summarized in Table 1. Notably, isoquinoline-3-carboxylic acid exhibited highest antibacterial activity against Xoo, Rs and Ac, with inhibition rates of 94.67%, 96.19%, and 97.27%, respectively, at a concentration of 50 μg/mL, comparable to the positive control kasugamycin. Additionally, the data revealed that 7-nitroisoquinoline had also broad-spectrum antibacterial activity at the same concentrations, albeit with lower inhibition rates than those of isoquinoline-3-carboxylic acid and positive control. Isoquinoline-1-carboxylic acid, 1-aminoisoquinoline, 3-isoquinolinecarbonitrile, 5-bromoisoquinoline, 5-iodoisoquinoline, and 4-methoxyisoquinoline exhibited notable selectivity against Xoo, with inhibition rates ranging from 84% to 97% at a concentration of 100 μg/mL. Additionally, isoquinoline-1-carboxylic acid demonstrated potent antibacterial activity against Ac, with an inhibition rate of 94.81% at the same concentration.
Antibacterial Activity of 49 Isoquinolines Against Xoo, Rs, and Ac.

The present study aimed to investigate the structure-activity relationships of isoquinoline derivatives against Xoo, Rs and Ac, as presented in Table 1. The results indicated that the substitution of electron-withdrawing groups at C-3 of the isoquinoline ring significantly influenced the antibacterial activities. Notably, the C-3 linked carboxyl group exhibited the highest antibacterial activity. Additionally, the electron-withdrawing cyanogroup at C-3 demonstrated promising antibacterial activity against Xoo. The introduction of an electron-withdrawing group at C-5 (5-iodoisoquinoline, 5-bromoisoquinoline) or an electron-rich group at C-4 (4-methoxyisoquinoline) was found to enhance antibacterial activity. Additionally, the incorporation of an electron-absorbing group, such as a nitro group at C-7, further increased activity. As a result, IQ3CA exhibited the highest level of activity and may serve as a promising lead compound. Notably, modifications at C-3, C-4, C-5 and C-7 significantly impacted activity, highlighting the importance of further optimization research.
Bactericidal spectrum of IQ3CA
Further evaluation of the bactericidal spectrum was conducted based on the antibacterial activity. As is shown in Figure 2, IQ3CA indicated remarkable bactericidal activity against Rs, Ac, Xoc, Xcc, Xoo, Xam, Psa, and Xf, with inhibition rates exceeding 90% at a concentration of 25 μg/mL. However, IQ3CA demonstrated limited antibacterial activity against Pcc, Pcb, Psl, Dz, and Xac, with inhibition rates ranging from 45.59% to 69.08% at a concentration of 25 μg/mL.

Bactericidal spectrum of IQ3CA.
Virulence Determination of IQ3CA Against Sensitive bacteria Virulence
Following the antibacterial screening, the virulence of IQ3CA was examined against Rs, Ac, Xoc, Xcc, Pcc, and Xf (Table 2). The findings indicated that Ac and Xoc exhibited the highest sensitivity to IQ3CA, with EC50 values of 8.38 and 9.19 μg/mL, respectively. The EC50 values of IQ3CA against remaining bacterial strains ranged from 11.12 to 17.35 μg/mL.
EC50 Values of IQ3CA Against Sensitive bacteria.

Bacterial Activity of IQ3CA Against Ac in Vivo
The protective and curative effects of IQ3CA against Ac are presented in Table 3 and Figure 3. The data indicated that IQ3CA exhibited a potent protective effect against Ac, with an efficacy of 68.56% at 200 μg/mL. This efficacy was not significantly different from that of the positive control kasugamycin (72.48%) and was superior to that of the positive control thiosen copper (64.62%). Furthermore, IQ3CA demonstrated a strong curative effect against Ac, with an efficacy of 62.11% at 100 μg/mL. This efficacy was not significantly different from that of the positive control kasugamycin (67.28%) and was superior to that of the positive control thiosen copper (56.84%).

Bacterial activity of
The Protective and Curative Effect of IQ3CA Against Ac.

Note: Values presented are means ± standard errors from three independent experiments. Means in a column followed by different letter are significantly different (P < 0.05) according to Duncan's multiple range test.
Effect of IQ3CA Concentration on the Growth of Ac
The effects of different concentrations of IQ3CA on growth curve of Ac are shown in Figure 4. IQ3CA delayed Ac growth in a concentration-dependent manner compared with the control group and at 50 μg/mL, IQ3CA completely inhibited the growth of Ac.
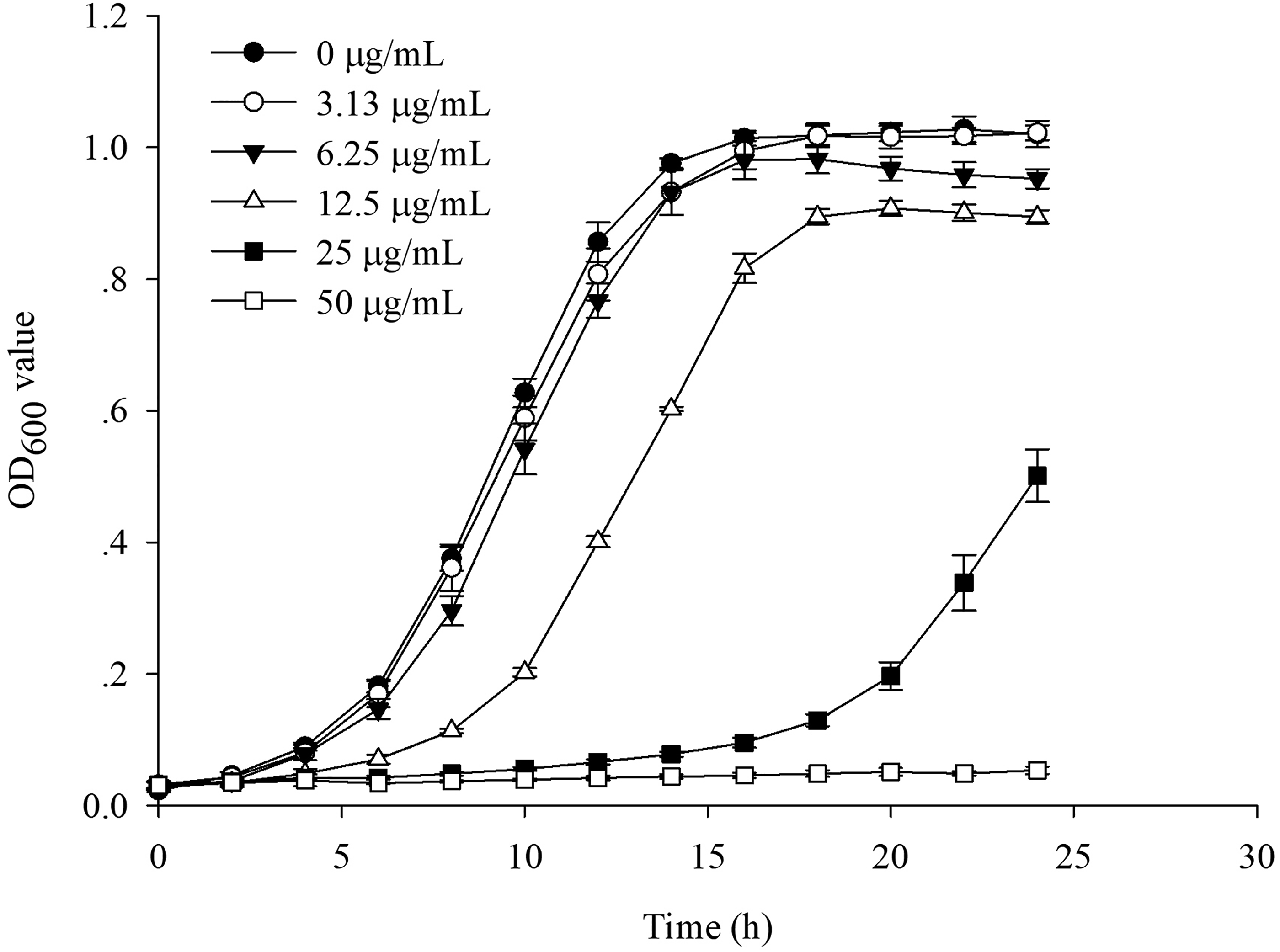
Effect of
Effect of IQ3CA on the Cell Morphology of Ac
The effect of IQ3CA on the ultrastructure of Ac was examined through the utilization of SEM, with the findings presented in Figure 5. The surface morphology of the control bacterial cells exhibited a smooth and rounded appearance, consistent with a typical rod-shaped morphology (5-A). Conversely, the morphology and structural integrity of the treated bacteria were significantly compromised, with the cell surface exhibiting a rugose and distorted appearance (5-B). The presence of depressions and sunken features became increasingly pronounced with rising concentration levels (5-C).
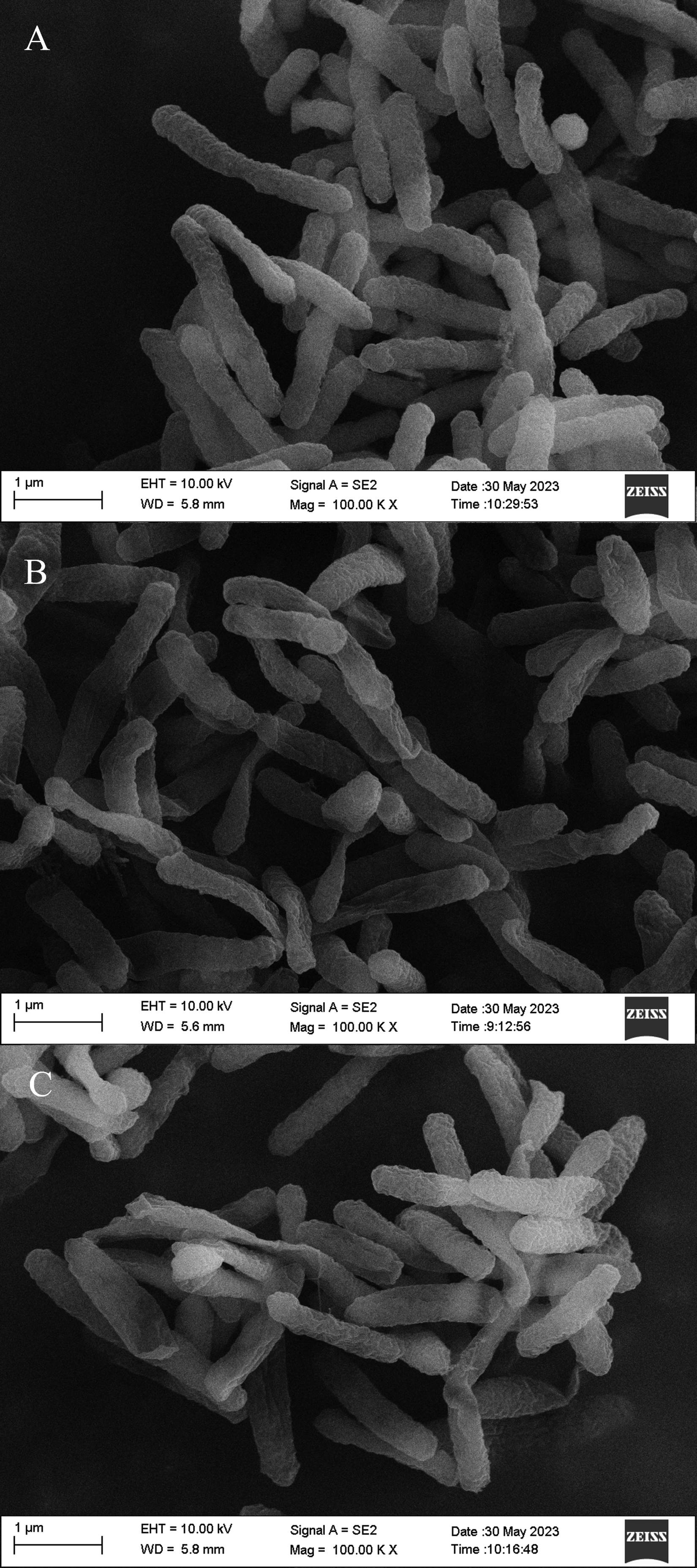
Effect of
Effect of IQ3CA on the Cell Membrane Permeability of Ac
The effect of IQ3CA on the permeability of cell membrane Ac is shown in Figure 6. The results indicated a positive correlation between the concentrations of IQ3CA and the conductivity. The relative electrical conductivity exhibited a gradual increase at 2 h post-treatment and a slower increase at 12 h post-treatment. Notably, the relative electrical conductivity reached 86.35% at 50 μg/mL after 12 h of treatment, which was significantly higher than the control and other treatments.

Effect of
Effect of IQ3CA on Bacterial Motility of Ac
As is shown in Figure 7, the inhibitory effects of IQ3CA on the motility of Ac were enhanced with an increase drug concentration. At concentrations of 12.5 μg/mL, 25 μg/mL and 50 μg/mL, the swimming diameters of Ac were reduced by 0.5-, 0.7-, and 1-fold, respectively, in comparison to the control group.

Effect of IQ3CA on bacterial motility of Ac.
Effect of IQ3CA on the EPS Content of Ac
The aforementioned information is depicted in Figure 8, indicating that IQ3CA exhibited a noteworthy inhibitory impact on the EPS production of Ac. Specifically, at a concentration of 50 μg/mL, the inhibition percentage was 82.86%.
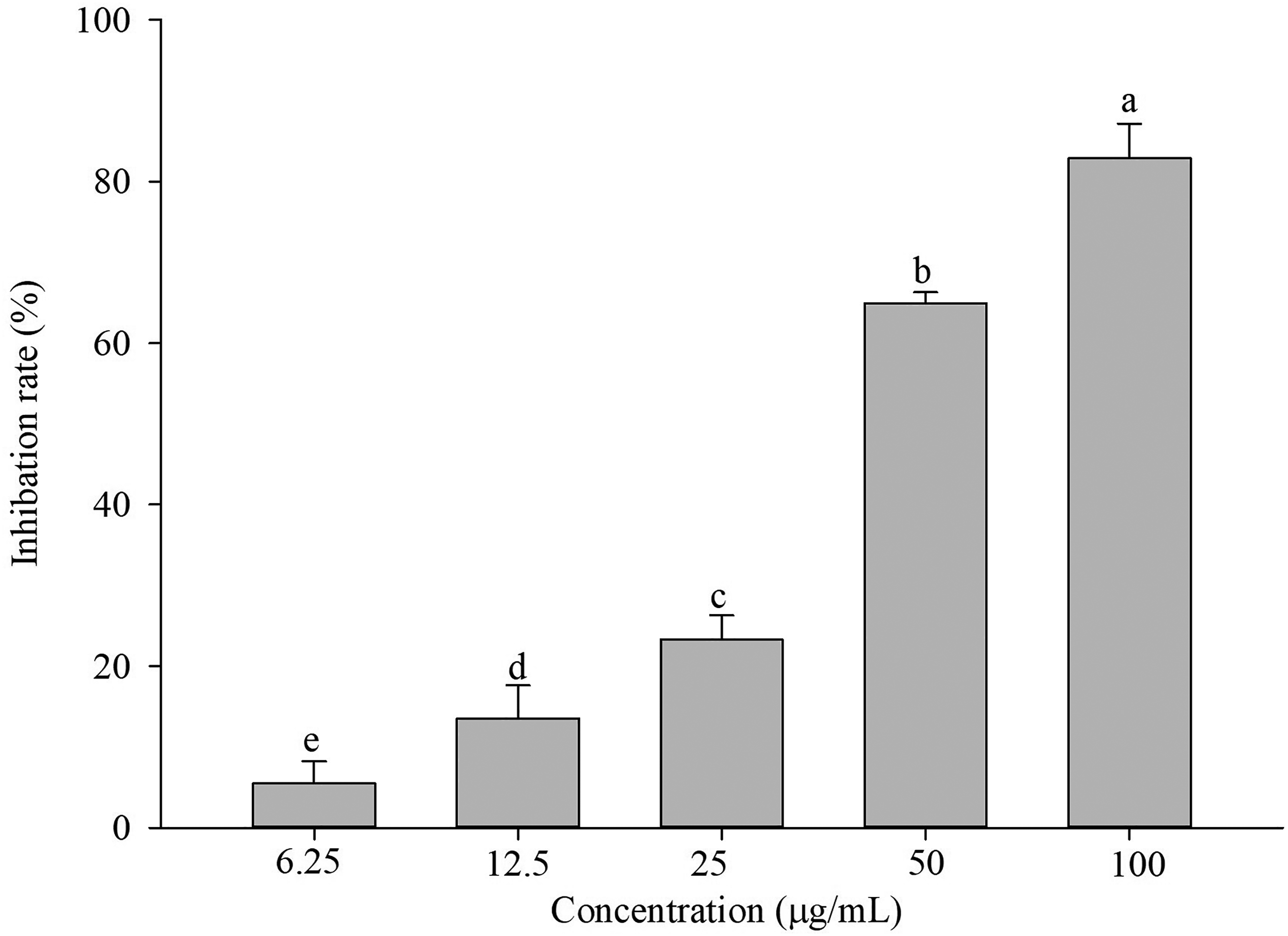
Effect of
Effect of IQ3CA on the Biofilm Formation of Ac
The results depicted in Figure 9 demonstrate that the application of IQ3CA at varying concentrations of 12.5 μg/mL, 25 μg/mL and 50 μg/mL led to a decrease in biofilm formation of Ac by 18.46%, 25.82%, and 45.67%, respectively, in comparison to the control group.

Effect of
Conclusions
In summary, this study evaluated the antibacterial activity of 49 isoquinolines against three pathogenic bacteria. The results revealed that certain compounds demonstrated enhanced antibacterial activity and broader antibacterial spectrum in comparison to the positive control kasugamycin. Notably, IQ3CA as derivative in natural products demonstrated the most potent antimicrobial activity against Ac. Moreover, it also conferred protection against Ac in vivo tests. IQ3CA inhibit bacterial growth of Ac by inducing alterations in cell morphology, damaging the bacterial cell membrane, diminishing the motility, reducing exopolysaccharides production, and preventing biofilm formation. However, the specific target of action of IQ3CA is not clear, and it can only be used as a new lead compound, which needs to be further embellished before becoming a commercial bactericide. Consequently, these results provide valuable insights into the potential antibactial properties of IQ3CA and its derivatives, thereby developing novel, highly efficient and environmentally friendly bactericides.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, Chinese Academy of Tropical Agricultural Sciences for Science and Technology Innovation Team of National Tropical Agricultural Science Center, (grant number NSFC No. 32372598, CATASCXTD202305).
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.



