Abstract
Fungi in the genus of Tolypocladium are often discovered as endophytes, soil inhabitants, and insect pathogens. Many biologically active secondary metabolites have been discovered from them in the past half-century, including medicinally important oligopeptide cyclosporine A, and other compounds like terpenes, polyketides, polyketide-non-ribosomal peptides. This review has summarized the chemistry and pharmacology of natural products discovered from Tolypocladium genus fungi, to provide a full record of all Tolypocladium secondary metabolites.
Introduction
The genus Tolypocladium was first established in 1970s, when 3 species of fungi were isolated from soil samples by Grams, 1 and was synonymized with Chaunopycnis and Elaphocordyceps by genetic phylogenetic analyses. 2 Distribution of this genus is wide, despite the original discovery from soil, most members in this genus are discovered as endophytic fungi, and they were possibly favored by plants for their abilities of producing insecticidal substances.
Fungi of this genus are medicinally and biologically important for their secondary metabolites (SMs), many of which are pharmacologically potent. Cyclosporin A, an oligopeptide discovered from T. inflatum, has been developed as a good immunosuppressant. Thus, metabolites in Tolypocladium sp. are worth exploring for their structural diversity and biological significance. Terpenes, polyketides (PKs) and PK-non-ribosomal peptides (PK-NRPs) and peptides including cyclosporines, have been discovered from fungus of Tolypocladium sp. Currently, there are review articles on taxonomy of Tolypocladium and research on cyclosporin A,3–5 but chemical and pharmacological properties of Tolypocladium SMs have never been summarized in full details. Thus, in this review, we have retrieved all papers on the chemistry and bioactivities of SMs isolated from fungi of Tolypocladium genus, and summarized structures and pharmacology of all the compounds, in order to show what we already discovered from this genus, and what to expect from it.
Secondary Metabolites
In the past half century, approximately 100 new Tolypocladium SMs have been found, they are mostly meroterpenoids, oligopeptides, PK-NRP and so forth. In this section, we summarized all the compounds isolated from Tolypocladium spp., and they are listed according to their structure types.
Terpenes and Meroterpenoids
Most commonly found terpenes in Tolypocladium sp. are diterpenes and terpenoid indole alkaloids. In 2011, Lin et al
6
first obtained 4 diterpenes from T inflatum and named them as aphidicolins (

Terpenes and meroterpenes isolated from fungi of the Tolypocladium genus.
Terpenes and Meroterpenoids and Their Origins.

Polyketides
A quite diversified collection of PKs can be produced by species of Tolypocladium, including pyrones, benzophenones, coumarins, etc, though mostly not active in bio-assays (Table 2 and Table S1). Structures and tested activities are illustrated in Figure 2 (Table 2 and Table S1).
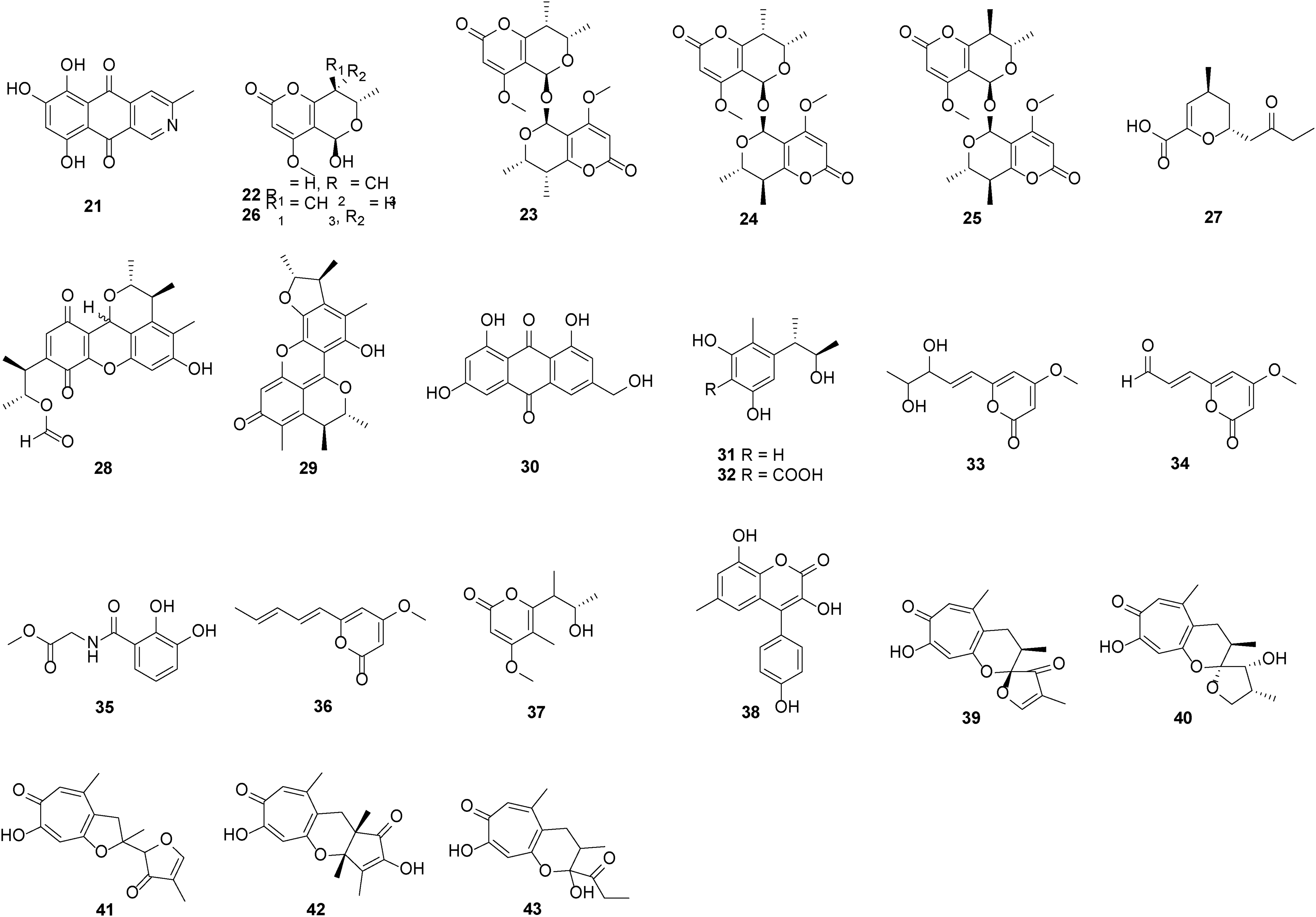
Polyketides isolated from fungi of the Tolypocladium genus.
Polyketides Discovered from Fungi in Tolypocladium Genus.

Aza-anthraquinones are not commonly discovered as natural products. Tolypocladin (
Polyketide-Non-Ribosomal Peptides
Tolypocladium-originated PK-NRPs are mostly tyrosine or alanine-derived pyridones and pyrrolidones, the latter is often referred to as tetramic acids. Tolypoalbin (
Li et al
14
discovered 6 PK-NRPs (
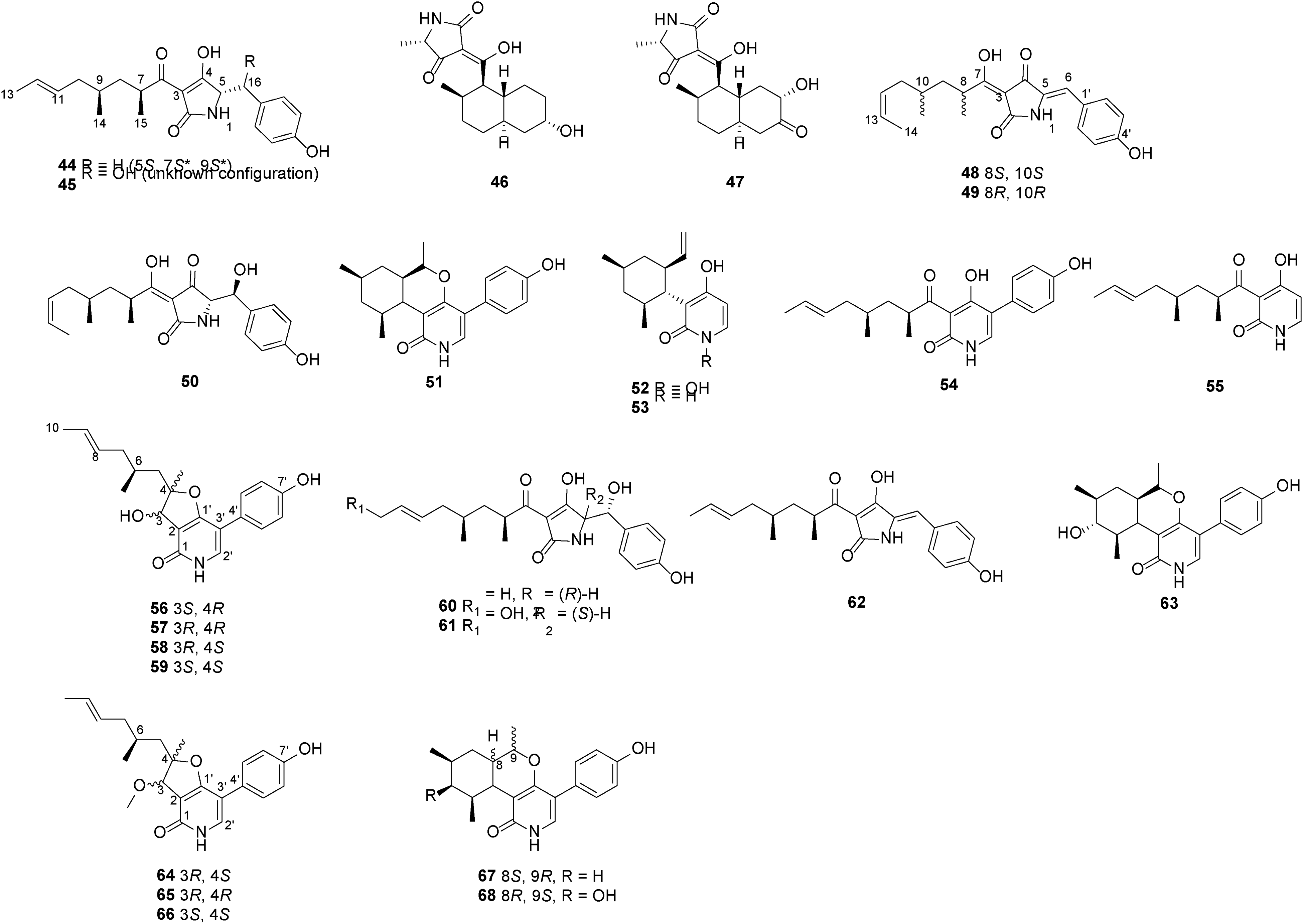
Polyketide-non-ribosomal peptides isolated from fungi of the Tolypocladium genus.
Polyketide-Non-Ribosomal Peptides Discovered from Fungi in Tolypocladium Genus.

*Obtained from heterologous expression of gene clusters of T. sp. 49Y in Aspergillus oryzae NSAR1.
Zhang et al
19
heterologously expressed a pyridone biosynthetic gene cluster from Tolypocladium sp. 49Y in Aspergillus oryzae NSAR1 and obtained 2 new compounds, tolypyridones C and D (
From a Chaunopycnis sp., Shang et al
20
discovered 4 new PK-NRPs (
Recently, 5 new pyridone derivatives (tolypyridones I-M,
NonRibosomal Peptides
Nonribosomal peptides (NRPs) are the most structurally diversified among all Tolypocladium SMs, over 50 cyclic or linear oligopeptides have been discovered till now. NRPs discovered in this genus are usually linear, cyclic, or branched oligopeptides consisted of at most 22 amino acid units which are sometimes acylated, glycosylated or N-methylated. This family of compounds is medicinally important. Cyclosporine A (amino acid sequence shown in Table 4, and structure in Figure 4), a cyclopeptide consists of 11 amino acid moieties, is now a first-line immunosuppressant (Sandimmun, Novartis) that is commonly used in transplantation and autoimmune diseases (eg psoriasis, rheumatoid arthritis, etc). 24

Structure of cyclosporin A.
Amino Acid Sequences of Cyclic Peptides in Tolypocladium sp. (Cyclosporines B-Z were from Traber 25 ).
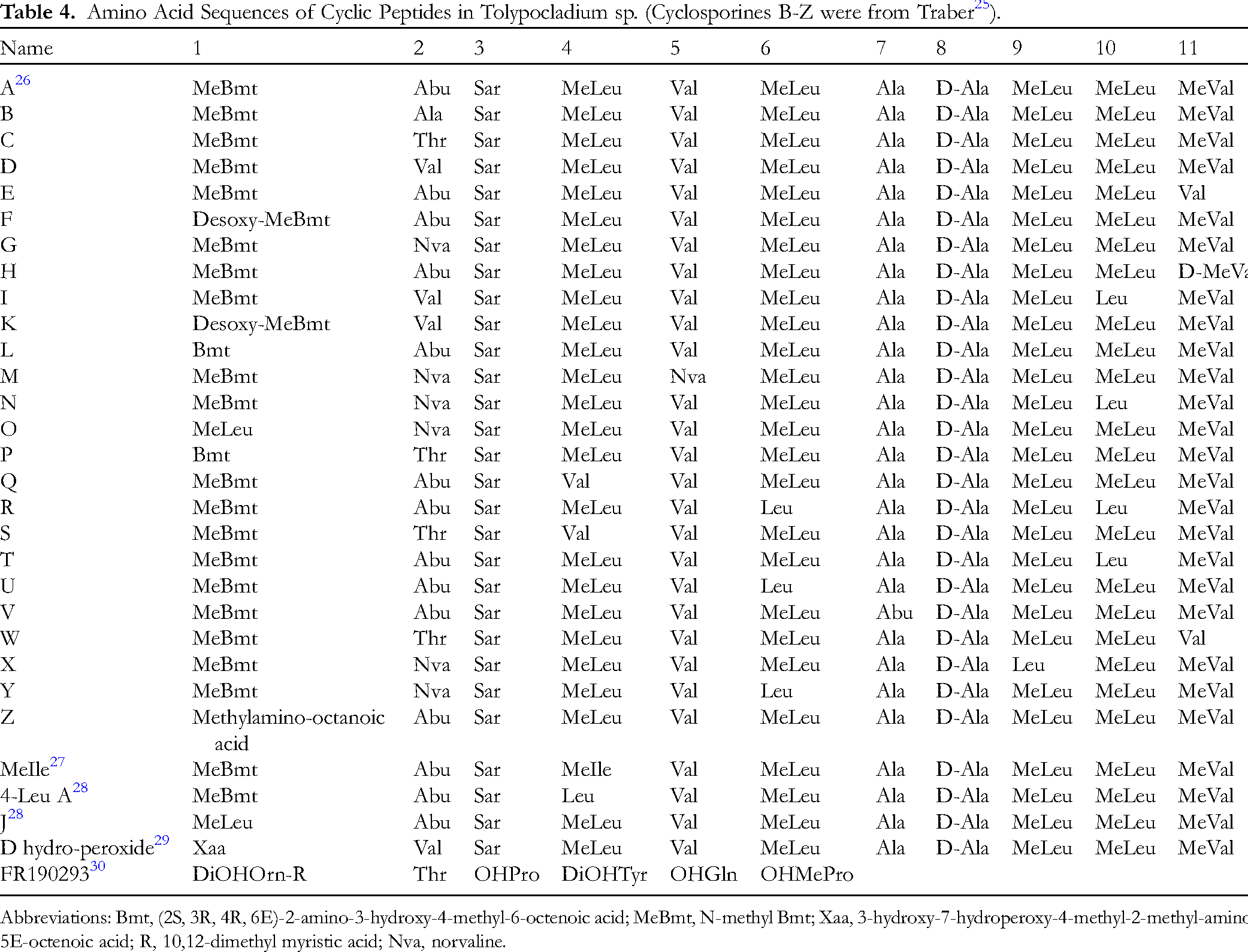
Abbreviations: Bmt, (2S, 3R, 4R, 6E)-2-amino-3-hydroxy-4-methyl-6-octenoic acid; MeBmt, N-methyl Bmt; Xaa, 3-hydroxy-7-hydroperoxy-4-methyl-2-methyl-amino-5E-octenoic acid; R, 10,12-dimethyl myristic acid; Nva, norvaline.
Cyclic Oligopeptides
Cyclosporin was first isolated from Trichoderma polysporum (Link ex Pers) (later redesignated as T inflatum Grams) 31 and identified in 1970s by Petcher et al. 26 It was a sarcosine-containing cyclo-undecapeptide (Figure 4). Then Traber et al25,32–34 discovered over 20 cyclosporins from T niveum, namely cyclosporins B-Z (Table 4). Structures ofcyclosporins were featured with unnatural amino acid moieties, (2S, 3R, 4R, 6E)-2-amino-3-hydroxy-4-methyl-6-octenoic acid (Bmt) and Bmt derivatives, and it was believed that this moiety was critical for immunosuppression, alternation of Bmt (deoxidation, replacement by other amino acids) caused loss of activity. They also found anti-HIV cyclosporine (MeIle) from T niveum fermentations supplemented with D-threonine, this compound can inhibit HIV-1 (strain IIIB)-induced cytopathic effect in MT4 cells. 27 With 60 cyclosporin derivatives, Loor et al 35 investigated the structure-activity relationship (SAR) of cyclosporin inhibiting human MDR-1 P-glycoprotein (P-gp) ABC transporter, the results indicated that hydrophobic residues favored P-gp inhibition, while N-desmethylation mostly abolished the activity.
Biosynthesis of Cyclosporin A
As cyclosporin A is chemically unique and clinically important, it is worth to give a brief summary on the biosynthesis of cyclosporin. Currently, it is commonly recognized that 3 major steps have been involved in the biosynthesis of cyclosporin A: isomerization of

Diketopiperazines isolated from fungi of the Tolypocladium genus.

Biosynthesis of cyclosporin A involves 3 steps: (1)
The unnatural amino acid
Assembly of the undecapeptide cycle was the most sophisticate step and it was catalyzed by a NRP synthase, 40 cyclosporin synthase (CySyn). CySyn, encoded by a 45.8 kb open reading frame (assigned as simA) in T niveum NRRL 8044, was a giant multimodule enzyme that consisted 11 sets of condensation-adenylation-thiolation domains (the C-A-T domains). Adenylation domain activated amino acids by adding adenyl group onto the amino group, thiolation domain with a free thiol group acted as a carrier of activated amino acids and peptidyls, and condensation domain catalyzed the formation of peptide bonds.41,42 Seven of the 11 modules contained a methyltransferase (MT) domain, and their function was catalyzation of N-methylation on the corresponding amino acid units (Bmt-1, Sar-3, Leu-4, Leu-6, Leu-9, Leu-10, and Ala-11). 40
Linear Oligopeptides
Efrapeptins are linear oligopeptides that are featured by their characteristic constituents α-amino-isobutyric acid (Aib) and pipecolic acid (Pip) (Table 5). Efrapeptins A-G were isolated from T niveum.43–45 Efrapeptins D and F were insecticidal (LC50 were 18.9 and 8.4 ppm) against Colorado potato beetle.
Sequences of Linear Peptides Discovered from Tolypocladium Species.

Abbreviations: AcAib, acetyl-2-aminoisobytyric acid; GlyOH, 2-hydroxyethylamine; Dec, decanoic acid; Non, nonanoic acid; Oc, octanoic acid; Lol, D-leucinol; Valol, valinol; mPro, 4-methyl proline, bAla, D-alanine.
Gichigamins are a family of 21 and 22-residue peptides, Du et al 15 comprehensively investigated the structures of gichigamins A-G by NMR, mass spectroscopy, electronic circular dichroism (ECD) or single-crystal x-ray diffraction. Gichigamin A was cytotoxic in vitro (GI50 0.55 ± 0.04 μM) and it showed antitumor activity in tumor xenograft mice as well. Gichigamin A can disrupt mitochondrial functions. In their semisynthetic structural modification research, by linking a coumarin to its C-terminus, antitumor potency increased to 60-fold of the prototype. Along with these peptides, the author also isolated 6 new 11-residue lipopeptaibols named dakwaabakains A-E and LP237-F7 along with 5 PKs.
In 2022, an array of new 11-residue peptaibols named tolypocladamides were discovered from T inflatum by 2 groups of researchers separately.50,51 Tolypocladamides A-G were tested for their Ras/Raf inhibitory activities, and tolypocladaminde C showed the lowest IC50 at 0.56 μM.
Recently in 2023, an endophytic Tolypocladium sp. from marine alga Spongomorpha arcta was investigated by Morehouse et al 52 Isolation led to the discovery of 2 antibacterial peptides named tolypocaibols A and B and an NRPS-PK-shikimate metabolite mixture maxinmiscin. Both peptides showed MIC values at 4 to 16 μM against methicillin-sensitive Staphylococcus aureus (MSSA), methicillin-resistant Staphylococcus aureus (MRSA), and other pathogenic bacteria.
Besides, diketopiperazines and other small-molecule peptides have been discovered alongside (
Diketopiperazines and Other Small-Molecule Peptides.

Discussion and Conclusions
Over the past half century since the genus Tolypocladium has been described and established, much has been done to understand its chemistry and discover pharmacologically active natural products from species in this genus. Gratifyingly, a potent immunosuppressant, cyclosporin A, is found and developed into a useful medicine.
Though species of this genus can be found from different sources including plants and insects, small molecules like PK-NPRs are rather similar (eg tolypyridones vs chaunolidones) among strains. This indicated that they are biosynthetically similar, and secondary metabolic gene clusters of small molecules are conserved between strains.
We believe that Tolypocladium genus is still a good resource of new natural products, especially oligopeptides. With the help of bio-informative methods and gene manipulation techniques, more cryptic biosynthetic gene clusters can hopefully be disclosed in the future. Noteworthily, pharmacology of many of the compounds, especially the oligopeptides, is somehow inadequately investigated, thus it is necessary to revisit these compounds for more useful bioactivities.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231225538 - Supplemental material for Diversity of Secondary Metabolites from Fungi of the Ascomycete Genus Tolypocladium
Supplemental material, sj-docx-1-npx-10.1177_1934578X231225538 for Diversity of Secondary Metabolites from Fungi of the Ascomycete Genus Tolypocladium by Ting-Ting Niu, Wei-Qiong Chen, Wen-Xing Li, Li Zhang, Ying-Qiao Shan and Li-Wei Ren in Natural Product Communications
Footnotes
Author Contributions
Investigation and Project administration were done by T.-T. N., L.-W. R.. Supervision was done by L. Z., L.-W. R. Data collection was done by T.-T. N., Y.-Q. S. Writing—original draft was done by T.-T. N, W.-Q. C. Writing—review & editing was done by W.-X. L, L.-W. R.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science, Technology and Innovation Commission of Shenzhen Municipality, Shenzhen Key Medical Discipline Construction Fund (grant numbers JCYJ20230807112402005, SZXK059).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
