Abstract
Objective
This study aims to convert cativic acid, which was isolated from Ageratina jocotepecana, into cistendiol (
Methods
Cativic acid was isolated from Ageratina jocotepecana flowers, reduction methodology was realized with THF and LiAlH4, epoxidation reaction was carried out with mCPBA in CH2Cl2 and K2CO3, oxidation of alcohol with PCC in CH2Cl2 and MgSO4, photooxidation reaction was realized with blue and white light using singlet oxygen (1O2) and photosensitizers such as eosin, methylene blue and Ru(bpy)3(PF6)2 in acetonitrile. Geometry optimizations for transition states were performed using MMFF94 with Monte Carlo protocol, and GDFT was used B3LYP/DGDZVP level of theory with Gaussian 09.
Results
Cistendiol was synthesized by photooxidation reaction of
Conclusion
Herein, we described the conversion of cativic acid isolated from Ageratina jocotepecana employing a new photooxidation methodology and classic epoxidation techniques, to produce an analog of cistenolic acid isolated from Cistus symphytifolius. The results, which include Supplemental material, enable comparison of stereochemistry, specifically the 13S for C-13 of Ageratina jocotepecana with other compounds such as cistenolic or salvic acid, which had been the subject of study by researchers.
Introduction
Labdane-type diterpenes are widely distributed compounds in nature that display numerous pharmacological activities.1,2 Several of these compounds exhibit antifungal, antibacterial, cytotoxic, anti-inflammatory, or analgesic properties. These bicyclic-based skeletons occur naturally in two series of antipodal labdane, referred to as “normal” and the “ent”-labdanes. 3
On the other hand, cativic acid [
Furthermore, the 13S configuration of C-13 has been determined by vibrational circular dichroism.
6
Previously, a study of Cistus symphytifolius species resulted in the identification of two labdane-type diterpenes, cativic acid (

Structures of cativic acid (
The oxygen-dependent photosensitization reaction is extensively employed in natural products synthesis, and is found ubiquitously within nature. 16
The transformation of cativic acid (
This serves as a supplementary confirmation of the stereochemistry of C-13 in both enantiomer
Results and Discussion
Cistenolic acid (
Diterpene
The reduction of the carboxylic functional group of

Reagents and conditions: (i) mCPBA, K2CO3, CH2Cl2, 3 h, R.T.; 85%, (7R,8S)-
The ratio of 7R and 7S isomers remained constant 9:1 after these procedures. The main isomer was confirmed to have (7R)-
In an effort to improve both, the yield and diastereoselectivity of achieving compound

Reagents and conditions: (i) LiAlH4, THF, 4 h, 94%; (ii) PCC, MgSO4, CH2Cl2, 5 h, 92%; (iii) mCPBA, K2CO3, CH2Cl2; 3 h, R.T, 93% for
The epoxidation reaction of
Molecular modeling of transition states structures of alpha and beta epoxides
The global minimum energy conformers for
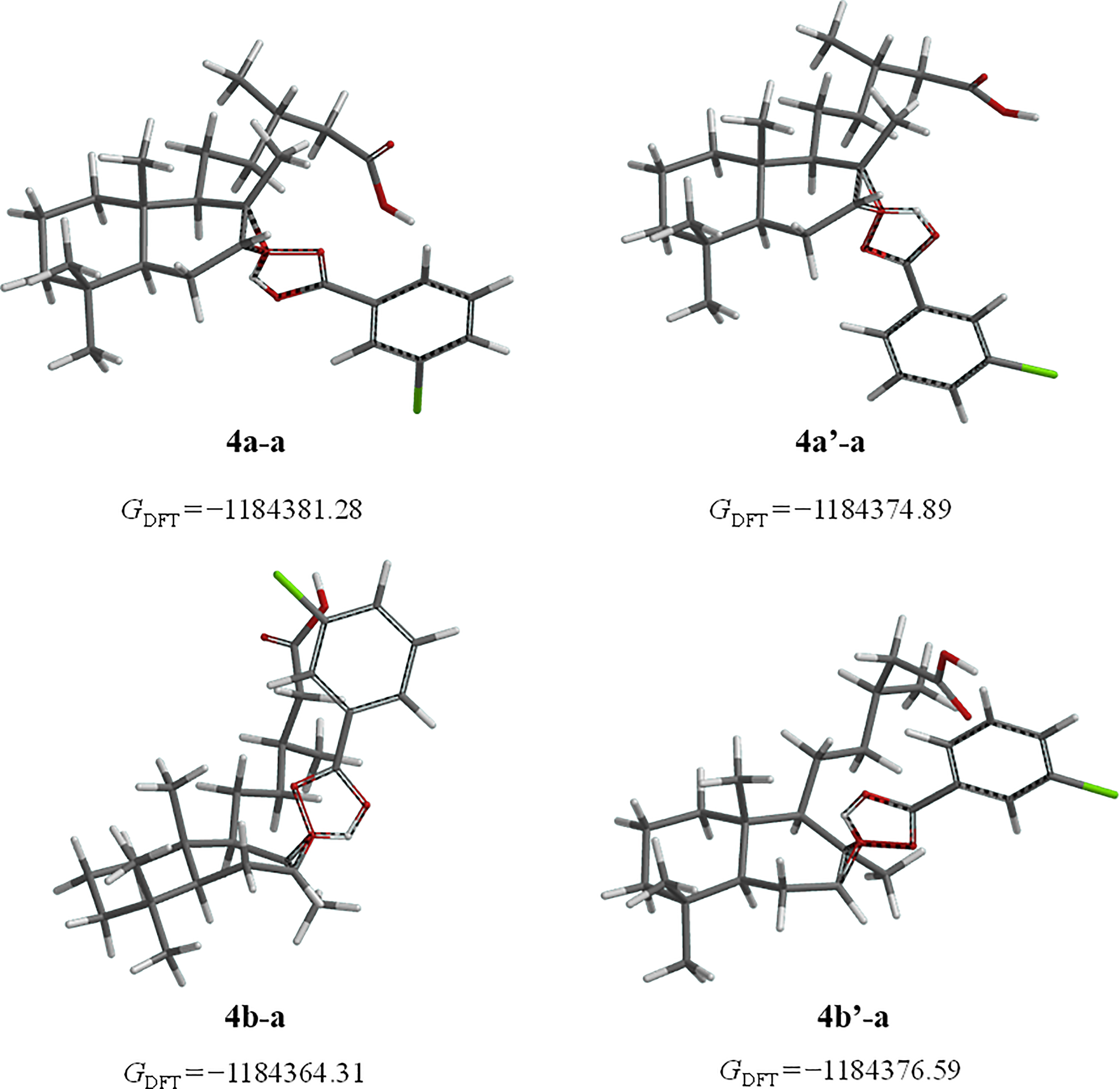
Transition states of epoxides of cativic acid (
Epoxides
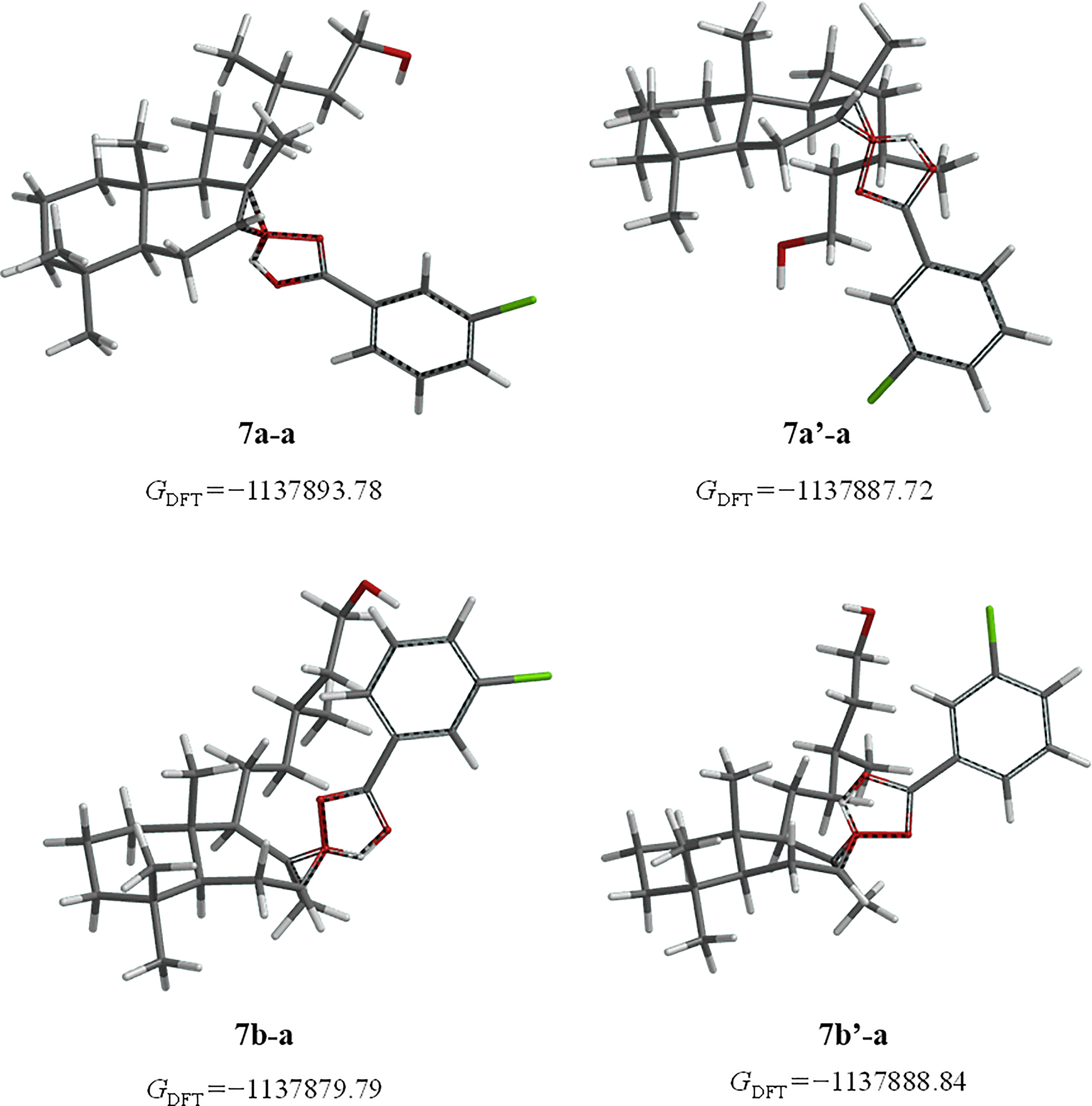
Transition states of epoxides of alcohol
A comparable pattern was noted with the epoxides alpha

Transition states of epoxides of aldehyde
All calculations realized demonstrate that the alpha epoxide is the favored, being in agreement with the NMR results. The preference is attributed to steric hindrance of peracid approach with the allylic chain located in C-9 in a beta orientation.
As the separation of the diastereomers a/b of compounds

Reagents and conditions: (i) Ru(bpy)3(PF6)2, air, white light, in CH3CN, 50%,
The active form of molecular oxygen is formed in situ through a photosensitizer that absorbs energy to excite oxygen, resulting in an excited singlet state 1O2, 21 which is involved in the oxidation reactions in certain natural products synthesis. 22 The reactions between 1O2 and organic compounds involve: [4 + 2], ene, and [2 + 2] additions to alkenes, 23 the unbiased alkenes react with atmospheric oxygen under visible light irradiation with a photosensitizer to give allyl hydroperoxides. 24
Encouraged by the target position of hydroxyl group at C-7 in compound
Reactions of Photooxidation of
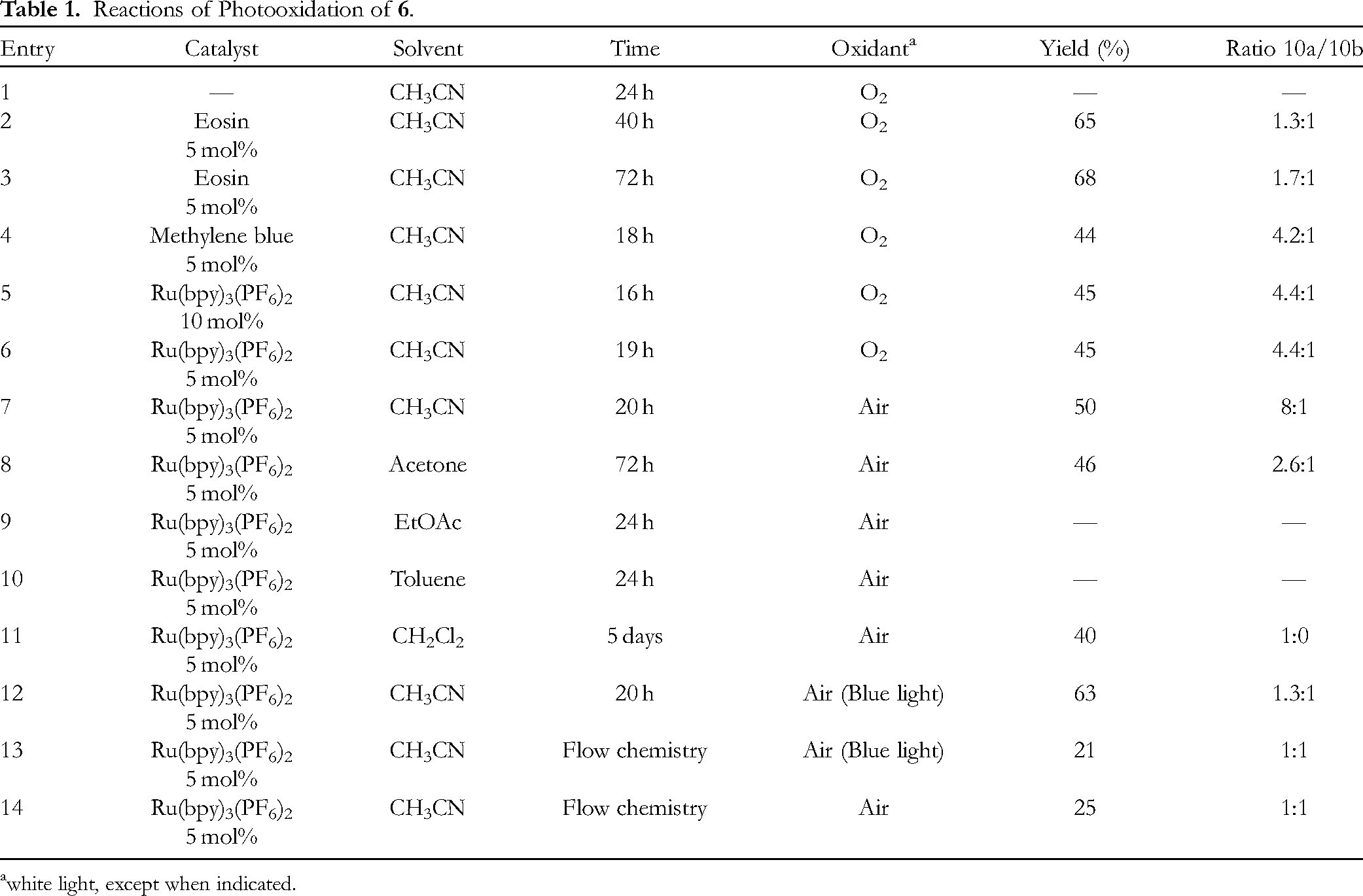
white light, except when indicated.
In particular, the mass spectrometry analysis of the mixture exhibited the molecular ion [M − OH]+ at m/z 307.2634 corresponding with the expected formula C20H35O2. The 1H NMR spectrum of
In a first time, the influence of the catalysts has been tested (Table 1, entries 2-6) for the photooxidation reaction of compound
It is interesting to note, a variation in the source of oxidant from O2 (bottle) to air induced a slight increase in yield to 50% and a notable stereoselectivity ratio up to 8:1 after 20 h of reaction (entry 7). Different solvents were evaluated (entries 8-11), and found that acetone led the product over 72 h of reaction with no notable change in the ratio compared with acetonitrile, while at 24 h, ethyl acetate and toluene solvents proved to be ineffective.
The reaction in dichloromethane was particularly interesting, despite extended reaction time: only the epimer
To further demonstrate the effect of wavelength, according to the maximum absorption of the catalyst Ru(bpy)3(PF6)2, the reaction was conducted under conditions similar to entry 7, but with blue light (400-490 nm). The results obtained showed a higher yield of 63%, however with a reduced ratio to 1.3:1 (entry 12). Finally, the reaction of
In addition, this study employs molecular modeling to examine the transition state of the photooxidation reaction for the alcohol

Transition states of photooxidation reaction of alcohol
According to results, the alpha transition state
Conclusions
In this study, we present the first interconversion reaction of cativic acid, which was isolated from Ageratina jocotepecana, using two pathways: epoxidation and photooxidation reaction to afford the cistendiol the reduced product of cistenolic acid, which was previously only isolated from Cistus symphytifolius. Furthermore, has already determinate its configuration as 13S to cativic acid, providing a supplementary proof of the stereochemistry as 13S for cistendiol compared with their enantiomer salvic acid. The relationship of analytical values of cistendiol and their enantiomer is in agreement. Additionally, is a contribution to understanding a possible biochemical pathway for the synthesizing cistenolic acid from cativic acid in the Cistus symphytifolius species.
Experimental
General Experimental Procedures
Commercially grade chemicals were used. The 1D and 2D NMR spectra were acquired on Bruker AVANCE1 300MHz spectrometer. Chemical shifts (δ) were reported in parts per million (ppm), as internal reference was used TMS at 0.00 ppm. Coupling constants (J) are reported in Hertz (Hz). Silica gel Merck 60 (230−400 mesh) was used for column chromatography and reactions were monitored by TLC. HRESIMS were acquired on Xevo Q-Tof WATERS Quadrupole Hybrid Time-of-Flight Mass Spectrometer, Versailles France. In the Supplemental material are fully described the compounds
Plant Materials
The procedure and isolation were realized as reported by del Rio et al 6 Ageratina jocotepecana (B.L. Turner) specimens were collected during the flowering stage, on March-February, 2018, near km 51 of the Morelia-Carapan federal road no. 15. A voucher specimen (No.188459) is deposited at the Herbarium of Instituto de Ecología, A. C., Centro Regional del Bajío, Pátzcuaro, Michoacán, Mexico, where Prof. Jerzy Rzedowski identified the plant material.
Extraction and Isolation
Flowers from A. jocotepecana were macerated employing hexanes at room temperature 3 times (7 d each). After evaporating the solvents under reduced pressure, the dried extract was acquired. Aliquots of extracts were column chromatographed using mixtures of hexanes-EtOAc in ascending polarity, to afford cativic acid [
Reduction Methodology with LiAlH4
Acid
Epoxidation Methodology with mCPBA
Cativic acid (
Oxidation Methodology with PCC
Compound
Photooxidation Methodology
To alcohol
Preparation of Compound 5 from 4 or by Photooxidation Reaction of 6
I. Epoxide
II. Alcohol
Molecular Modeling
Geometry optimizations for transition states from epoxides
Spectral Data
7α,8α-epoxy-cativic acid (4)
Colorless oil. HRESIMS m/z 323.2586 (calcd for C20H34O3 + H+, 323.2581). 1H NMR (300 MHz, CDCl3) δ 2.96 (1H, br s, H-7), 2.37 (1H, dd, J = 15.1, 5.8 Hz, H-14a), 2.13 (1H, dd, J = 15.1, 8.1 Hz, H-14b), 2.06 (1H, m, H-6a), 1.92 (1H, m, H-13), 1.76 (1H, m, H-1a), 1.69 (1H, m, H-6b), 1.57 (1H, m, H-11a), 1.55 (1H, m, H-12a), 1.49 (1H, m, H-2a), 1.40 (1H, m, H-2b), 1.37 (1H, m, H-3a), 1.30 (3H, s, H-17), 1.21 (1H, m, H-11b), 1.18 (1H, m, H-12b), 1.17 (1H, m, H-9), 1.08 (1H, m, H-3b), 0.99 (1H, m, H-5), 0.99 (3H, d, J = 6.6 Hz, H-16), 0.84 (3H, s, H-19), 0.83 (1H, m, H-1b), 0.82 (3H, s, H-18), 0.72 (3H, s, H-20). 13C NMR (75 MHz, CDCl3) δ 179.2 (C-15), 61.1 (C-7), 59.1 (C-8), 55.8 (C-9), 46.0 (C-5), 42.2 (C-3), 41.5 (C-14), 39.0 (C-12), 38.9 (C-1), 36.1 (C-10), 33.2 (C-18), 32.8 (C-4), 30.9 (C-13), 23.4 (C-11), 23.0 (C-6), 22.8 (C-17), 22.1 (C-19), 19.9 (C-16), 18.8 (C-2), 14.3 (C-20).
Labd-8,17-ene-7α,15-diol (5)
The HRESIMS m/z 309.2790 (calcd for C20H36O2 + H+, 309.2788) and spectral NMR data of
(−)-(5S,9S,10S,13S)-labd-7-en-15-ol (6)
The HRESIMS m/z 293.2847 (calcd for C20H36O + H+, 293.2839) and spectral NMR data of
7α,8α-epoxylabdan-15-ol (7)
Colorless oil. HRESIMS m/z 309.2788 (calcd for C20H36O2 + H+, 309.2788). 1H NMR (300 MHz, CDCl3) δ 3.67 (2H, m, H-15), 2.95 (1H, br s, H-7), 2.26 (1H, br s, OH), 2.08 (1H, dd, J = 15.3, 4.7 Hz, H-6a), 1.78 (1H, m, H-1a), 1.70 (2H, m, H-14a and H-14b), 1.63 (1H, m, H-6b), 1.52 (1H, m, H-1b), 1.51 (1H, m, H-13), 1.49 (1H, m, H-12a), 1.46 (1H, m, H-11a), 1.37 (2H, m, H-2a and H-2b), 1.35 (1H, m, H-3a), 1.30 (3H, s, H-17), 1.20 (1H, m, H-11b), 1.16 (1H, m, H-9), 1.09 (1H, m, H-12b), 1.03 (1H, m, H-3b), 1.00 (1H, dd, J = 12.5, 4.7 Hz, H-5), 0.91 (3H, d, J = 6.1 Hz, H-16), 0.84 (3H, s, H-19), 0.82 (3H, s, H-18), 0.71 (3H, s, H-20). 13C NMR (75 MHz, CDCl3) δ 61.2 (C-15), 61.0 (C-7), 59.1 (C-8), 55.9 (C-9), 46.0 (C-5), 42.2 (C-3), 38.9 (C-14), 39.7 (C-12), 39.6 (C-1), 36.1 (C-10), 33.2 (C-18), 32.8 (C-4), 30.3 (C-13), 23.2 (C-11), 23.0 (C-6), 22.9 (C-17), 22.1 (C-19), 19.8 (C-16), 18.8 (C-2), 14.3 (C-20).
(−)-(5S,9S,10S,13S)-labd-7-en-15-al (8)
The spectral NMR data of
7α,8α-epoxylabdan-15-al (9)
Colorless oil. HRESIMS m/z 307.2628 (calcd for C20H34O2 + H+, 307.2632). 1H NMR (300 MHz, CDCl3) δ 9.77 (1H, br s, H-15), 2.95 (1H, br s, H-7), 2.45 (1H, dd, J = 16.2, 5.4 Hz, H-14a), 2.25 (1H, dd, J = 16.2, 8.0 Hz, H-14b), 2.08 (1H, m, H-6a), 2.04 (1H, m, H-13), 1.74 (1H, m, H-1a), 1.63 (1H, m, H-6b), 1.57 (1H, m, H-12a), 1.51 (1H, m, H-11a), 1.40 (2H, m, H-2a and 2b), 1.34 (1H, m, H-3a), 1.31 (3H, s, H-17), 1.21 (1H, m, H-12b), 1.16 (1H, m, H-11b), 1.45 (1H, m, H-9), 1.07 (1H, m, H-3b), 1.00 (1H, m, H-5), 0.99 (3H, d, J = 6.6 Hz, H-16), 0.86 (3H, s, H-19), 0.84 (3H, s, H-18), 0.82 (1H, m, H-1b), 0.73 (3H, s, H-20). 13C NMR (75 MHz, CDCl3) δ 203.0 (C-15), 58.8 (C-8), 61.0 (C-7), 55.8 (C-9), 46.0 (C-5), 42.2 (C-3), 51.0 (C-14), 39.3 (C-12), 39.0 (C-1), 36.1 (C-10), 33.2 (C-18), 32.8 (C-4), 28.8 (C-13), 23.4 (C-11), 23.0 (C-6), 22.9 (C-17), 22.1 (C-19), 20.2 (C-16), 18.8 (C-2), 14.3 (C-20).
7α-hydroperoxylab-8(17)-en-15-ol (10)
Colorless oil. HRESIMS m/z 307.2634 (calcd for C20H35O2 − OH−, 307.2632). 1H NMR (300 MHz, CDCl3) δ 7.89 (1H, br s, OOH), 5.16 (1H, s, H-17a), 4.85 (1H, s, H-17b), 4.50 (1H, s, H-7), 3.67 (2H, m, H-15), 2.02 (1H, m, H-6a), 1.94 (1H, m, H-9), 1.76 (1H, m, H-1a), 1.66 (1H, m, H-14a), 1.56 (1H, m, H-2a), 1.55 (1H, m, H-6b), 1.53 (1H, m, H-13), 1.53 (1H, m, H-11a), 1.44 (1H, m, H-12a), 1.42 (1H, m, H-5), 1.37 (1H, m, H-3a), 1.36 (1H, m, H-2b), 1.34 (1H, m, H-14b), 1.15 (1H, m, H-11b), 1.14 (1H, m, H-3b), 1.04 (1H, m, H-1b), 1.00 (1H, m, H-12b), 0.90 (3H, d, J = 4.8 Hz, H-16), 0.85 (3H, s, H-18), 0.78 (3H, s, H-19), 0.66 (3H, s, H-20). 13C NMR (75 MHz, CDCl3) δ 146.0 (C-8), 113.3 (C-17), 87.2 (C-7), 61.3 (C-15), 52.1 (C-9), 48.5 (C-5), 42.1 (C-3), 39.8 (C-10), 39.6 (C-14), 38.8 (C-1), 36.1 (C-12), 33.3 (C-18), 33.3 (C-4), 30.1 (C-13), 27.6 (C-6), 21.3 (C-19), 20.5 (C-11), 20.0 (C-16), 19.4 (C-2), 13.6 (C-20).
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231222764 - Supplemental material for Chemical Correlation of Cistendiol Obtained from Cativic Acid in Two Different Pathways †
Supplemental material, sj-docx-1-npx-10.1177_1934578X231222764 for Chemical Correlation of Cistendiol Obtained from Cativic Acid in Two Different Pathways † by David Calderón-Rangel, Hugo A García-Gutiérrez, Rosa E del Río and Christine Thomassigny in Natural Product Communications
Footnotes
Acknowledgements
Thanks CIC-UMSNH and CONAHCYT-Mexico (Grant No. A1-S-47352) for partial financial support. D.C.R. is grateful to CONAHCYT-Mexico for scholarships 576044 and 291276 (international exchange scholarship). Molecular modeling at the High-Performance Computing Laboratory at IIQB-UMSNH.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable to this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the CIC-UMSNH, Consejo Nacional de Humanidades, Ciencias y Tecnologías (grant number A1-S-47352).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Human and Animal Rights
There are no human subjects in this article and informed consent is not applicable.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
