Abstract
A facile and concise synthesis of β-carboline alkaloids, such as (–)-isocyclocapitelline and (+)-cyclocapitelline, has been achieved from commercially available geraniol through a unified strategy. The key steps involved in this synthesis are Sharpless epoxidation, intramolecular ring opening of epoxide, Pictet-Spengler reaction, and dehydrogenative aromatization using 10% palladium/carbon in xylene under neutral conditions.
Keywords
Substituted tetrahydrofuran (THF) ring containing natural products possess a highly complex structure with a diverse range of biological properties. 1,2 The tetrahydrofuran ring is frequently found in natural products, as well as in many biologically and pharmaceutically active compounds. 3 -9 In particular, 2,5-disubstituted THFs is a key structure for a variety of biologically active natural products. 10,11 On the other hand, THFs are the synthons for the synthesis of complex natural products such as pheromones, pharmaceutical agents, polyether antibiotics, and marine toxins. 12 -14 These fascinating structural features and intrinsic biological activities attracted many scientists toward the total synthesis of these natural products. 15 -25
β-Carboline alkaloids (Figure 1), such as (–)-isocyclocapitelline (
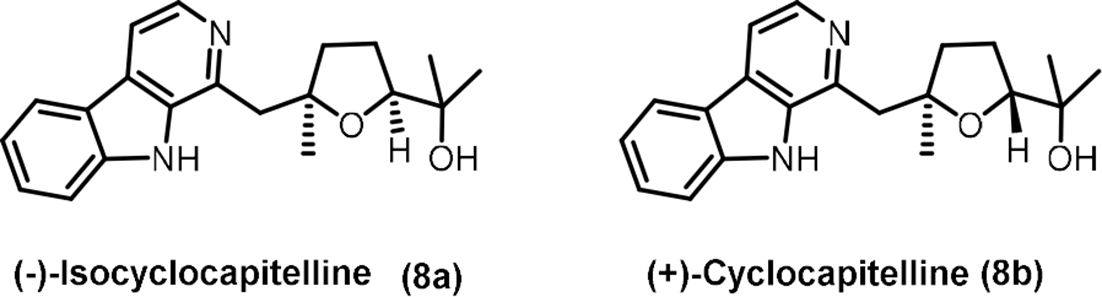
Representative examples of β-carboline alkaloids.
Results and Discussion
Our synthesis commenced with a familiar transformation in organic synthesis, which is Sharpless asymmetric epoxidation of the readily available monoterpene geraniol (

Reagents and conditions: (a) (–)-diethyltryptamine, Ti(O
i
Pr)4, tert-butyl hydroperoxide, 4 Å MS, CH2Cl2, –30 °C, 95%; (b) dry tetrahydrofuran, sodium bis(2-methoxyethoxy)aluminum hydride (3.5 M in toluene, 1.1 equiv.), 90%; (c) meta-chloroperoxybenzoic acid, dichloromethane (CH2Cl2), 0 to room temperature, 4 hours, 80% (
Coupling of Aldehyde 5a and 5b With Tryptamine (6)
Aldehyde

Reagents and conditions: (f) tryptamine (6), trifluoroacetic acid, dichloromethane, 78 °C, 3 hours; (g) palladium/carbon, xylene, reflux, 8 hours, 75%.
Similarly, synthesis of (+)-cyclocapitelline (
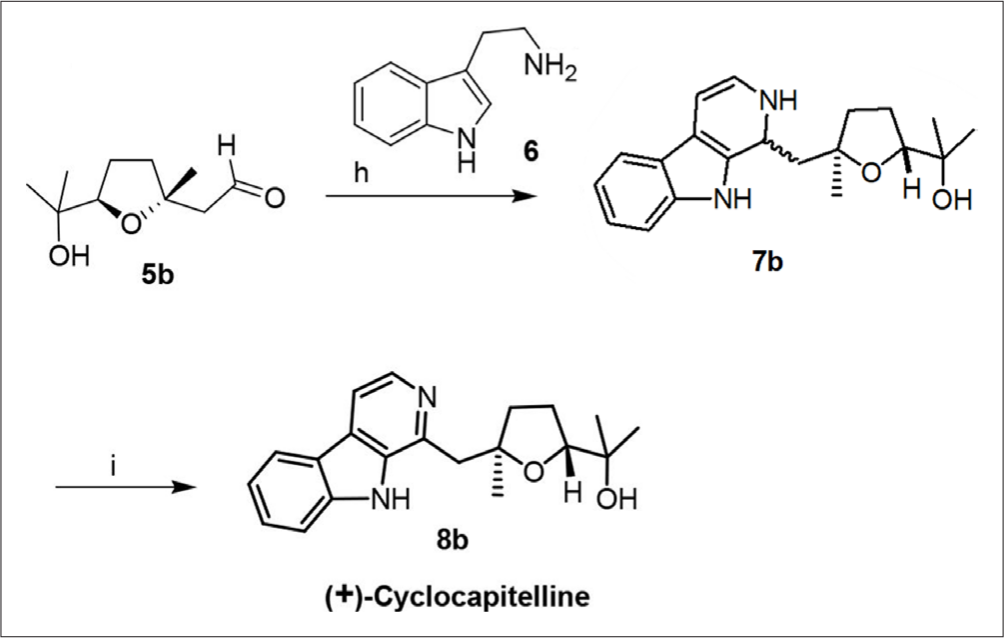
Reagents and conditions: (h) tryptamine (6), trifluoroacetic acid, dichloromethane, 78 °C, 3 hours; (i) palladium/carbon, xylene, reflux, 8 hours, 70% over 2 steps.
The 1H and 13C NMR spectra of the synthesized product
General experimental details and spectroscopic data have been included in Supplementary Material 1.
Conclusion
In summary, we have successfully established a unified strategy for the total synthesis of Hedyotis plant alkaloids (–)-isocyclocapitelline (
Supplemental Material
Supplementary Material 1 - Supplemental material for A Unified Total Synthesis of Isocyclocapitelline and Cyclocapitelline
Supplemental material, Supplementary Material 1, for A Unified Total Synthesis of Isocyclocapitelline and Cyclocapitelline by Marepally Srilatha, Vallabhareddy Satteyyanaidu, Chepyala Krishna Reddy and Venkata Subba Reddy Basireddy in Natural Product Communications
Footnotes
Acknowledgment
We thank the Vice-Chancellor of Osmania University for his support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
