Abstract
Curcumin, a natural polyphenol compound found in turmeric, exhibits significant anti-cancer activity in preclinical studies. However, its clinical application is limited by poor bioavailability and low solubility in aqueous media. To overcome these challenges, various curcumin delivery systems (CDSs) have been developed to enhance the pharmacokinetics and pharmacodynamics of curcumin, aiming to improve its solubility, stability, and targeted delivery to tumor cells. Preclinical studies have shown that CDSs can improve pharmacokinetics, enhance anti-tumor efficacy, and reduce toxicity in various cancers. Several CDSs have also advanced into clinical trials, demonstrating safety and efficacy in cancer patients. Despite the promising results, challenges remain, such as optimizing formulation and dosage, investigating curcumin's influence on the carriers in drug delivery and release, evaluating long-term safety and toxicity, and conducting large-scale clinical trials. Understanding the effect of gut microbiota on CDSs metabolism and bioavailability is critical for enhancing their clinical effectiveness in tumor therapy. Further research and development are necessary to fully exploit the potential of CDSs in cancer treatment. This review summarizes recent progress in CDSs research and their potential application in tumor therapy, encompassing formulation strategies, delivery routes, as well as preclinical and clinical evaluations. It not only provides valuable insights into the future rational design and development of curcumin dosage forms and their cancer therapeutic potential but also facilitates the clinical translation of curcumin and CDSs.
Introduction
Curcumin is a prominent polyphenol compound derived from Curcuma longa L. rhizomes. 1 It has garnered significant attention in the scientific community due to its wide range of pharmacological activities, including antioxidant, anti-inflammatory, anti-carcinogenic, anxiolytic, antiatherosclerotic, hepatoprotective, antiposttraumatic stress disorder, and antidiabetic.1–6 The historical use of curcumin dates back to 1937 when it was first tested in a clinical trial for biliary disease, later reported in Lancet. 7 Subsequently, Nature published the first study on curcumin's biological activity as an antibacterial agent. 8 Its multi-faceted approach to targeting various signaling pathways, such as inhibiting tumor cell proliferation, 9 inducing cell death, 10 inhibiting inflammation, 3 preventing cell migration, 11 and displaying chemopreventive activity, 12 makes it particularly promising for anti-tumor therapy.9–13 Notably, researches have demonstrated its potential in suppressing tumors associated with head and neck cancer, 14 esophageal cancer, 15 breast cancer,10,11,16 lung cancer, 9 hepatocellular carcinoma,17,18 gastric cancer, 19 colorectal cancer,4,18 cervical cancer, 12 ovarian cancer, 18 and prostate cancer, 20 bone cancer, 21 osteosarcoma,13,22 melanoma, 23 squamous cell carcinoma, 23 papilloma. 24
However, despite its promising properties, the clinical applications of curcumin face several challenges. Issues like poor aqueous solubility, p-glycoprotein (P-gp) efflux by multidrug pump, extensive in-vivo metabolism, rapid elimination, low permeation, suboptimal targeting efficacy, as well as limited pharmacokinetics and bioavailability, restrict its effective use in cancer therapy.2–4,12,13,21,22,25 Consequently, these limitations hinder the realization of curcumin's full clinical potential.
To overcome these obstacles, innovative curcumin delivery systems (CDSs) have been developed, aiming to enhance the solubility, stability, and targeted delivery of curcumin. This review provides a comprehensive overview of recent advancements in CDSs research and its potential application in tumor therapy. The focus will be on formulation strategies, delivery routes, and preclinical and clinical evaluations. By summarizing the current state of research in this field, we highlight the potential of CDSs as promising strategies to augment the therapeutic efficacy of curcumin in cancer treatment. Nevertheless, it is essential to acknowledge that further research and development are warranted to overcome the existing limitations and fully exploit the clinical potential of CDSs.
Curcumin Delivery Systems: Types and Applications
Various forms of CDSs have been developed to address its challenges and enhance therapeutic efficacy. These systems offer innovative solutions to the limitations faced by curcumin, such as poor solubility, rapid metabolism, and limited bioavailability. Among the notable types of CDSs are liposomes (LPs), which are lipid-based vesicles that encapsulate curcumin, enhancing its stability and targeted delivery.16,17 Micelles, another form of delivery system, involve self-assembling structures that encapsulate curcumin, leading to improved solubility, increased the blood concentration of curcumin and increased the half-life of the drug, enhanced targeted delivery.15,21 Drug nanocarriers carrying curcumin can improve the shortcomings of curcumin, such as enhanced encapsulation or solubilization and accurate and selective distribution of therapeutic drugs to the desired target sites.14,26 In addition, to achieve thorough stabilization and internalization of drugs, they have a high surface-to-volume capacity, change surface functional groups, impact pharmacokinetics, and decreased body clearance.14,26 Cyclodextrin inclusion complexes are molecular complexes that not only enable curcumin to be encapsulated within their cavity, making it more water-soluble and more easily absorbed but also enhance the anti-inflammatory activity of curcumin and reduce interleukin-6 (IL-6) levels. 3 Additionally, dendrimers utilize dendritic structures to encapsulate curcumin, providing a protective environment and efficient delivery to the target site. 11 Hydrogel, a cross-linked hydrophilic macromolecule network with unique properties such as high flexibility, hydrophilicity, biocompatibility, and high water retention capacity, can carry curcumin to enhance its stability and antibacterial ability. It can also be used as an anti-infection biomaterial for cancer wound healing to reduce healing time and prevent damage to cells near the wound due to dehydration and angiogenesis.27,28 These diverse delivery systems have shown promising results in preclinical studies, demonstrating increased stability, sustained release, and enhanced therapeutic effects of curcumin.
Moreover, these CDSs can be administrated through various routes to suit different clinical needs. Conventional administration methods, such as oral intake, have been widely explored.4,20 However, the bioavailability issues of curcumin have prompted researchers to investigate alternative routes. Injectable administration allows for controlled release and direct delivery to the site of action. 17 Further, inhalation administration is being explored as a potential approach for respiratory disorders and targeted lung cancer therapy. 29 By delivering CDSs directly to the lungs, this approach maximizes local drug concentration while reducing systemic side effects. 9
The potential application of CDSs extends beyond traditional drug delivery. Studies have shown that CDSs have promising potential in gene and drug delivery, and the development of novel platforms for photothermal agent contrast agents and radiosensitizers.11,30,31 One of the most significant applications of CDSs is in cancer therapy. CDSs have demonstrated promising anti-cancer effects in various malignancies, including head and neck cancer, 14 esophageal cancer, 15 breast cancer, 16 lung cancer,9,25 hepatocellular carcinoma,17,18 gastric cancer, 19 colorectal cancer, 18 cervical cancer, 12 ovarian cancer, 18 prostate cancer, 20 bone cancer, 21 osteosarcoma, 22 melanoma,23,26 squamous cell carcinoma, 23 papilloma, 24 and glioblastoma. 32 Table 1 provides an overview of the specific types of CDSs, common cancers treated, results, and the major outcomes of optimizing curcumin's physicochemical properties and overcoming its limitations.
Summary of Application of Curcumin Delivery Systems in Various Cancers.
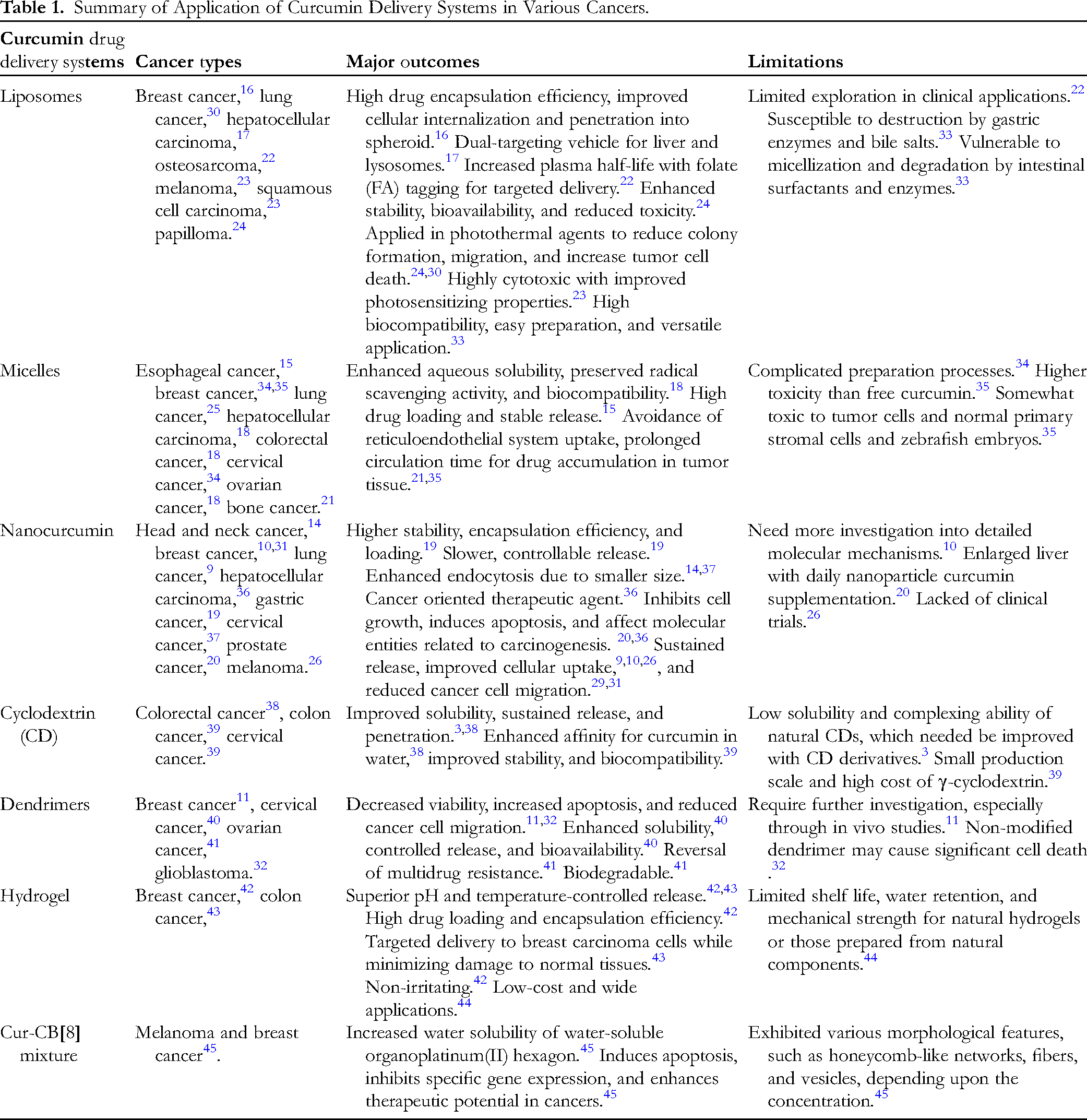
Although CDSs are available in a variety of dosage forms to improve the bioavailability of curcumin, different dosing regimens may also affect the bioavailability of curcumin to a certain extent. Recognizing that developing more effective dosing regimens is also a challenging task in the process of “improving curcumin's shortcomings and bioavailability,” it would be of great clinical value to combine the above two points. This involves studying the relationship between CDSs in different dosage forms and the oral first-pass effect, as well as the relationship between CDSs in different dosage forms and intestinal flora, to fine-tune the formulations, dosages, and delivery systems of curcumin to maximize their potential for cancer treatment. These studies will provide a better understanding of the drug's mechanism of action and enable optimal use in different cancer treatments.
Different dosage forms of CDSs have shown great potential not only in preclinical studies for the treatment of different cancers but also in phototherapy, radiotherapy, and immunotherapy. For example, LPs, nanoparticles (NPs), and other dosage forms of CDSs can enhance the sensitivity of tumor cells to radiotherapy, effectively destroying cancer cells. Moreover, CDSs in the form of NPs and other agents can modulate the immune response, stimulating the activity of immune cells and enhancing the host immune system's ability to fight cancer. These features make CDSs multifunctional anti-cancer drugs that can work synergistically with various therapeutic modalities, providing more possibilities for cancer treatment.
To enable the clinical translation of curcumin, it's crucial to select appropriate drug formulations and dosage forms to different tumor types and patients. The key to this selection process is how various CDSs affect its physicochemical properties. Table 1 outlines the primary cancer types studied in preclinical CDSs formulations and their impact on curcumin's properties. Despite these advancements, the unknown anti-cancer mechanisms of CDSs remain an obstacle to clinical translation. To address this, we summarized the anti-cancer mechanisms associated with CDSs, as shown in Figures 1–3.

Mechanisms of curcumin and curcumin delivery systems in anti-cancer cell proliferation. TGF-β, transforming growth factor-beta; Smad3/7, Drosophila mothers against decapentaplegic protein 3/7; E2F, E2F transcription; hTERT, human telomerase reverse transcriptase; Cyclin D1, involving in the G1 transition; Cyclin B1, involving in the G2/M transition; Ki67, a protein expressed in cell nuclei during cell division; PCNA, proliferating cell nuclear antigen; IL-6, interleukin-6; JAK, Janus Kinase; STAT3, signal transducer and activator of transcription 3; NF-κB, nuclear factor kappa-B; β-catenin, a key mediator of Wnt signaling; EGF, epidermal growth factor; RAS (H-ras, K-ras, and N-ras), the second largest mutated gene driver in various human cancers; MEK, Mitogen-activated protein kinase; ERK, extracellular signal-regulated kinase; TGF-β, transforming growth factor-beta.

Mechanisms of curcumin and curcumin delivery systems in anti-cancer cell invasion and metastasis. TGF-β, transforming growth factor-beta; Smad3, Drosophila mothers against decapentaplegic protein 3; NF-κB, nuclear factor kappa-B; BMI1, polycomb group protein; PTEN, phosphatase and tensin homolog deleted on chromosome ten; EMT, epithelial-to-mesenchymal transition; CXCR4, CXC chemokine receptor 4 axis; PTCH, Patched; SMO, frizzled class receptor; β-catenin, a key mediator of Wnt signaling; IL-6, interleukin-6; JAK: Janus Kinase; STAT3, signal transducer and activator of transcription 3; ZEB1, transcription factor; TGF-β, transforming growth factor-beta; ZEB1, zinc finger e-box-binding homeobox 1; BMI1, b lymphoma mo-mlv insertion region 1.
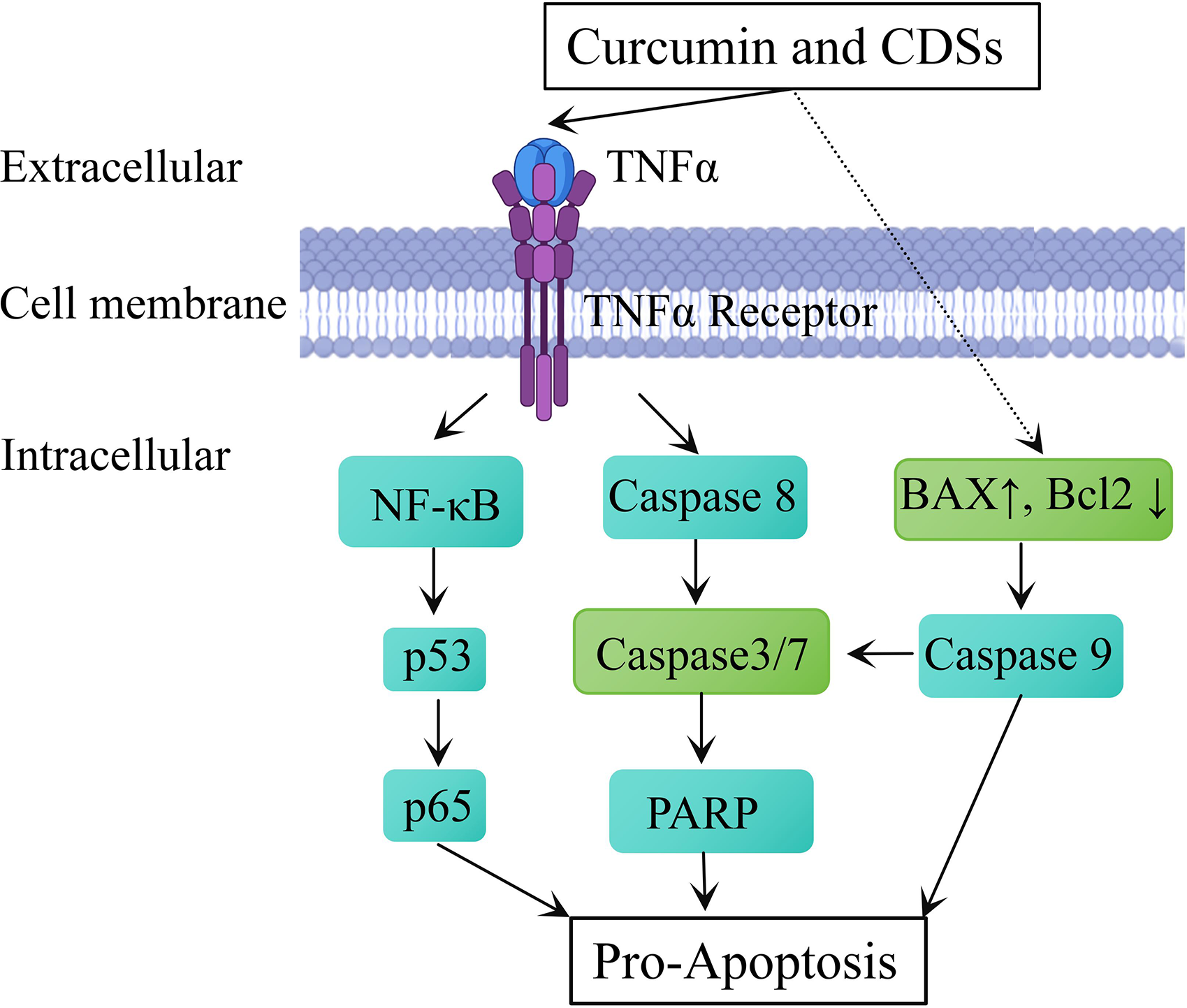
Mechanisms of curcumin and curcumin delivery systems (CDSs) in promoting cancer cell apoptosis. TNFα, tumor necrosis-like factors α; NF-κB, nuclear factor kappa-B; p53, a tumor suppressor protein; P65, an indicator of NF-κB activation; PARP, poly-ADP ribose polymerase; Bcl-2, b-cell lymphoma protein 2; Bax, Bcl-2-associated X protein.

Applications of curcumin delivery systems in various cancer treatments.
The Mechanisms of Action of CDS in Cancer
Effects on Cell Cycle and Cell Proliferation
CDSs have been found to influence key molecular mechanisms involved in cell cycle regulation and cell proliferation (Figure 1). An important target is the transforming growth factor-beta (TGF-β) signaling pathway. TGF-β is known to inhibit the expression of human telomerase reverse transcriptase (hTERT) by regulating smad3/7 and E2F. 46 HTERT is essential for maintaining chromosome termini and cell immortality in cancer cells. 47 CDSs, such as nanocurcumin, have been shown to up-regulate TGF- β1 by up-regulating smad3 and E2F, and down-regulating smad7. This results in down-regulation of hTERT and inhibition of cancer cell proliferation. 36 Furthermore, these CDSs can inhibit proliferation of lung cancer and cervical cancer cells by down-regulating Cyclin D1 and up-regulating Cyclin B1 in TGF-β pathway, further down-regulates Ki67 and inhibiting proliferating cell nuclear antigen overexpression and resulting in cell cycle arrest.9,22,36,37,48
IL-6 is a multifunctional cytokine that regulates the activity of STAT3 by acting on its downstream kinase JAK. 49 When IL-6 binds to its receptor IL-6R, JAK is activated, thereby phosphorylating STAT3 protein. Phosphorylated STAT3 forms a dimer and enters the nucleus, regulating the transcription of target genes and inhibiting nuclear translocation of nuclear factor kappa-B (NF-κB), thereby reducing its mediated gene transcription. It has been shown that CDSs can inhibit STAT3 phosphorylation and NF-κB activity by inhibiting IL-6 regulation of JAK. This playing an important role in inflammatory response, immune regulation, and inhibition of cervical cancer cell, papillary thyroid cancer and cancer stem-like cells proliferation.37,48,50–52
The Wnt signaling pathway is a complex signaling pathway involved in the regulation of cell growth, differentiation, and apoptosis. 53 It has been reported that CDSs can inhibit the nuclear accumulation of β-catenin, a key mediator of the Wnt signaling pathway, leading to the down-regulation of the expression level of Cyclin D1, and inhibit the cell cycle and cancer cell proliferation.22,37,48
Growth factors, such as EGF, binds to its receptor (EGFR) can activate Ras-MAPK signal transduction pathway, including Ras-MEK- extracellular signal-regulated kinase (ERK) triggers downstream signaling pathways involved in cell growth and division. 54 Curcumin has been shown to interfere with the EGF signaling pathway, and impact downstream components of the pathway, limiting the signaling cascade's progression, may induce cell cycle arrest, preventing cells from progressing through the cell cycle and dividing.9,55
Effects on Cell Invasion and Metastasis
CDSs can inhibit multi-faceted inhibitory effects on cancer invasion and metastasis by modulating key signaling pathways (Figure 2). Specifically, they suppress the TGF-β/Smad3 signaling pathway, leading to the down-regulate NF-κB signaling pathway, impacting inflammatory and immune responses. NF-κB, in turn, regulates pivotal genes such as b lymphoma mo-mlv insertion region 1 (BMI1), phosphatase and tensin homolog deleted on chromosome ten (PTEN), and components involved in epithelial mesenchymal transition (EMT), influencing tumor genesis and progression.9,11,37,48,56 Moreover, CDSs down-regulate components of the CXCL12/CXCR4 axis and Hedgehog signaling pathway, thereby inhibiting EMT and suppressing breast and ovarian cancer development.9,11,57 Furthermore, CDSs reduce the nuclear accumulation of β-catenin, inhibiting invasion and metastasis of cervical cancer cells.37,57 In the IL-6 signaling pathway, CDSs down-regulate IL-6, inhibit the phosphorylation of STAT3, and down-regulate ZBE1 and EMT, thereby inhibiting invasion and metastasis of cervical cancer and ovarian cancer cells.37,48
Induction of Apoptosis
Curcumin exerts its anti-cancer effects through inducing apoptotic-related proteins like Bax and modulating TNFα (tumor necrosis factor α) (Figure 3).22,38,58 TNFα is an important inflammatory cytokine, and CDSs regulate the survival and death of lung cancer cells by modulating TNFα. This modulation leads to the down-regulation of the NF-κB /p53 axis, resulting in the down-regulation of nuclear transcription factor P65 and its translocate to the nucleus.9,11,59 Notably, b-cell lymphoma protein 2 (Bcl-2), an anti-apoptotic protein inhibiting pro-apoptotic factors like Bax, caspase 8, and caspase 3/7 or 9, is impacted by curcumin. It is found that CDSs can down-regulate Bcl-2 and up-regulate Bax, promoting the activation of caspases and poly-ADP ribose polymerase (PARP), ultimately facilitating caspase-dependent cell death, including osteosarcoma, breast cancer, and HeLa cancer cells.22,38,58
The Potential of CDSs in Cancer Treatment
CDSs offer promising benefits in cancer therapy, enhancing the efficacy and safety of curcumin by protecting it from degradation, extending its circulation time, and improving its cellular uptake. One of the key advantages of CDSs is their ability to modulate multiple cellular signaling pathways involved in cancer development and progression. Through interactions with various molecule targets, CDSs can regulate key factors implicated in cancer cell proliferation, invasion, metastasis, and cell death. Additionally, CDSs have been shown to modulate the expression of microRNAs and lncRNAs, which play crucial roles in cancer pathogenesis.37,41 In addition to their direct effects on cancer cells, CDSs can also influence the tumor microenvironment and immune response. Some CDSs have been shown to modulate the NF-κB pathway, leading to reduced inflammation and immune suppression in the tumor microenvironment.9,11 This can enhance the efficacy of immune-based cancer therapies and promote anti-tumor immune responses.9,11 Moreover, some CDSs have been found to reverse multidrug resistance by down-regulating P-gp and inhibiting ATP activity, leading to increased sensitivity of cancer cells to chemotherapy.60,61 Changes in blood rheology are closely related to hematogenous metastasis of tumors. 62 It is found that curcumin is potentially valuable in improving the deformability of red blood cells and regulating blood rheological properties. 63 Therefore, studying the value and mechanism of multi-target drug curcumin and CDSs in regulating blood rheological properties, inhibiting tumor thrombus formation in circulating tumor cells, and reducing tumor metastasis can not only provide new ideas for the treatment of tumors, but also offer new research methods to improve the bioavailability of curcumin and promote its clinical translation. Clarifying the optimization of the physicochemical properties of curcumin and the anti-cancer mechanism of CDSs through different CDSs formulation strategies can provide more precise data, laying the foundation for conducting large-scale clinical trials.
Clinical Trials of CDSs
Extensive clinical trials have demonstrated that the safety and efficacy of CDSs with notable inhibitory effects on tumor cell proliferation and metastasis. Additionally, these delivery systems show promise in reducing the side effects associated with conventional cancer treatment methods. A comprehensive search on the World Health Organization International Clinical Trials Registry Platform (ICTRP) revealed a total of 75 clinical trials related to “curcumin, cancer therapy,” with six of them specifically focusing on “curcumin drug delivery system, cancer therapy.” Detailed information on these clinical trials can be found in Table 2.
Clinical Trials with the Curcumin Delivery Systems in Tumor Therapy.

Among the selected clinical interventional studies (six in total), two were conducted on breast cancer, one on colon cancer, one on prostate cancer, and two on advanced cancer cases. This underscores the high expectations placed on CDSs for the treatment of breast, colon, prostate, and locally advanced or metastatic cancer. It is worth noting that not all clinical trials have reported the results concerning CDSs in anti-cancer treatments, possibly due to ongoing research or unpublished studies at the time of searching. Nevertheless, the safety and tolerability of CDSs have been demonstrated in patients.9,67 For instance, a phase 1 dose-escalation study was conducted in Salzburg to evaluate the safety, tolerability, and pharmacokinetic characteristics of curcumin LP in cancer patients. The study observed reductions in relevant tumor markers in patients with prostate carcinoma, bone, and lymph node metastases, as well as lymphangiosis of the lung, along with patients with colon carcinoma and liver and lung metastases. 67 Furthermore, the clinical trials manifest that no dose-limiting toxicity was observed in 26 patients administered doses between 100 and 300 mg/ m2 over 8 h. 67 These encouraging findings from clinical trials indicate that CDSs hold significant potential as safe and effective adjuncts to traditional cancer therapies.
In conclusion, CDSs have shown great potential in preclinical studies. Their diverse dosage forms, unique mechanisms, and excellent pharmacokinetic properties make them an innovative approach to cancer treatment. However, we also need to recognize that there are still some problems and challenges with CDSs in clinical trials. First of all, the safety and side effects of its long-term use still need further research and evaluation. Second, devising more effective dosing regimens is a challenging task. To solve these problems, it's crucial to conduct an extensive series of clinical trials that assess the effectiveness and safety of CDSs. These trials will provide better understand into the drug's mechanism of action, facilitating the identification of its optimal use in different cancer treatments.
Future Direction for Clinical Translation of CDSs
Despite the satisfactory results achieved by CDSs in optimizing the physicochemical properties of curcumin, preclinical studies and clinical trials, there are still some challenges to be addressed in clinical translation (Table 3). Optimization of formulation and dosage is a key focus point for improving the clinical efficacy of drugs to enhance the clinical efficacy of curcumin and reduce side effects. Evaluating the effects of curcumin on carriers during drug delivery and release, long-term safety and toxicity is the basis for optimizing CDSs formulation and dosage. The prospect of studying three-dimensional (3D) cell growth on the basis of two-dimensional (2D) cells, investigating the effect of gut microbiota on the metabolism and bioavailability of CDSs in vivo, and conducting large-scale clinical trials are conducive to preclinical and clinical studies of curcumin and CDSs. Table 3 provides an overview of the specific types of CDSs, common cancers treated Results, the major outcomes of optimizing the of curcumin's physicochemical properties and limitations.
Challenges and Solutions to Clinical Translation of CDSs.

CDSs, curcumin delivery systems.
Challenges and Solutions to Clinical Translation of CDSs
Optimizing Formulation and Dosage in CDSs
Despite the promising results, there are challenges that need to be addressed for clinical translation of CDS. To effectively utilize CDSs, optimizing their formulation and dosage is crucial. It is discovered that key factors that influence entrapment efficiency, curcumin solubility, stability, and anti-cancer competence of CDSs are closely linked to the carrier/drug molar ratio and the type of carrier used.18,70,71 For example, the hydrophilic shell of the micelle, generated using the monomer oligo (ethylene glycol) methyl ether acrylate, plays a crucial role in protecting the micelle with a water-soluble shield. 72 Similarly, spherical cationic LP-polyethylene glycol (PEG)-polyethylenimine complex (LPPC) with hair-like projections on the surface acts as a rapid shuttle carrier for curcumin, facilitating its efficient delivery into cells and enhancing its cytotoxic efficacy. 73 Notably, CDSs like chitosan nanocurcumin have shown enhanced lung localization compared to free curcumin. 9 Additionally, coating the surface of porous poly lactic-co-glycolic acid (PLGA) NPs with red blood cell membranes enhances uptake by cancer cells and accumulation in the tumor due to the protein CD47, which signals the host's immune system to avoid phagocytosis and prolongs systemic circulation. 74 Furthermore, modifying erythrocyte membranes to prepare low immunogenicity carriers has shown promise for personalized treatment programs based on ABO blood group patients.74,75
Prescription and dose optimization play a pivotal role in the targeted delivery and release of CDS. Researchers have proposed that the sizes of drug-loaded micelles should strike a balance, being large enough to avoid renal filtration (greater than the renal clearance threshold, ∼8 nm) but small enough to penetrate through leaky vasculatures in the tumor region (smaller than the average gap of leaking blood vessels in cancer tumors, ∼200 nm). 61 Recent study has shown that micelles with a hydrodynamic size ranging from 8 to 200 nm are beneficial for tumor-targeted delivery. 61 Moreover, maintaining a slight negative charge on the micelles can reduce reticuloendothelial system-mediated clearance. 61
However, the medical applications occasionally face limitations due to problems in optimizing formulations and doses. For instance, LP agglomeration after 3 month's storage results in larger diameters, wide size distribution, and zeta potential reduction. thereby limiting their medical application.76,77 Researchers have identified strategies to address these issues, such as surface-coating LP's with hydrophilic polymers like PEG or carboxymethyl dextran (CMD), supplementary use of LP's surfactant, and employing freeze-drying LPs with cryo- or lyoprotectants to prolong stability.33,76–78 Remarkably, CMD coating not only stabilizes LPs against curcumin leakage in circulation but also reduces the risk of clearance by the reticuloendothelial system, promoting LP accumulation at pathological sites via the enhanced permeability and retention effect. 77
Avoiding Curcumin Negatively Effect on Carriers
Curcumin's disadvantages, such as poor water solubility and membrane interactions, can negatively affect various carriers, influencing drug delivery and releasing. Curcumin tends to decrease membrane fluidity of nanoliposomes by weakening hydrophobic interactions among acyl chains of phospholipids and perturbing the packing characteristics of the phospholipid bilayer. This inhibition of diffusivity and partitioning tendency of the permeant can hinder drug delivery.79–81 However, researchers have found that nanoliposomes made of soya lecithin, with a higher proportion of polyunsaturated fatty acids with short chains, exhibit higher membrane fluidity, potentially compensating for curcumin's effects on nanoliposome membrane. 80 Furthermore, curcumin's poor water solubility makes it difficult to penetrate into the matrix of the LPs. To achieve sustained release, LPs prepared with corrosive phospholipids can assist in increasing drug release while minimizing leakage before reaching targeted tumors and cells. 79
More in Vitro and in Vivo Studies
Another important aspect is the evaluation of CDSs in physiologically relevant models. Conventional 2D cell cultures do not fully mimic the complexity and heterogeneity of tumors in vivo, and therefore, the use of 3D cell models(cell spheroids) and multicellular spheroids is gaining prominence in drug screening and treatment efficacy assessment.82–84 These models provide a more representative environment for studying cell–cell interactions and drug responses. Most of the researches are in vitro studies, more in vivo study should be conducted. Additionally, large-scale clinical trials are essential to validate the efficacy and safety of CDS in real-world settings. These trials should explore the therapeutic potential of CDSs in different cancer types and stages, assess their long-term safety, and compare their efficacy with standard cancer treatments.
Investigating the Effect of Gut Microbiota on the Metabolism and Bioavailability of CDS
While numerous studies have demonstrated the promising effects of curcumin on cancer treatment, there are still some researchers who question its anti-tumor capabilities due to discrepancies observed between its application and certain in vitro and preclinical literatures.85–89 It has labeled both a PAINS (pan assay interference compounds) 86 and an IMPS (invalid metabolic panaceas) compound86,90 with its limitations including rapid degradation and low bioavailability, poor pharmacokinetic/pharmacodynamic properties, lack of a singular chemical species in some instances, and autofluorescence. 88 However, instead of disregarding its remarkable pharmacological effects, it is crucial to address these challenges and explore strategies to enhance our understanding and utilization of curcumin. Gut microbiota play a significant role in the metabolism of dietary polyphenols, including curcumin, and can affect the bioavailability and therapeutic efficacy of curcumin.85,91 Understanding the influence of gut microbiota on the metabolism and bioavailability of CDS in vivo is critical for their effective application in tumor therapy. Stevens JF et al in 2016 provided compelling evidence supporting the innovative concept that gut microbiota contribute to the metabolism of dietary polyphenols, such as curcumin, thereby influencing the bioavailability of both the parent compounds and their metabolites. 91 This reinforces the need to thoroughly investigate the impact of gut microbiota on the metabolism and bioavailability of CDSs in vivo.
By gaining deeper insights into how gut microbiota affect curcumin's actions, researchers can fine-tune CDS formulations, dosage, and delivery systems to maximize their potential for cancer treatment. Bridging the gap between in vitro studies and real-world clinical outcomes will strengthen the scientific foundation for the application of CDSs and pave the way for more successful and reliable tumor therapies. The knowledge obtained from these investigations will not only address the concerns of doubters but also provide a solid basis for the future development and widespread adoption of CDSs in the field of oncology.
CDSs’ Potential for Photoacoustic Imaging-Guided Photothermal/Chemo Combined Therapeutic Applications
Interestingly, CDSs with the capability of photoacoustic imaging, greater EPR-induced accumulation in tumor site and hyperthermia ablation for photothermal chemotherapy shows potential for photoacoustic imaging-guided photothermal/chemo combined therapeutic applications, which can improve the bioavailability of hydrophobic agent for photothermal treatment.92,93 CDSs, including targeted nanocomposites selectively absorbed by tumor cells, potentially functioned as efficient multiprobes for optical, magnetic resonance imaging, and photoacoustic imaging contrast agents in the field of cancer diagnostics via aptamer conjugation after the successful encapsulation of curcumin and superparamagnetic iron oxide nanoparticles inside a PLGA nanocapsule.92,93
Conclusion
In conclusion, CDSs offer promising advancements in cancer therapy by addressing curcumin's limitations (Figure 4). These innovative systems show improved pharmacokinetics and anti-tumor effects in preclinical studies. Clinical trials demonstrate their safety and potential for inhibiting tumor growth. Further research is needed to optimize formulations and explore their interactions with gut microbiota. CDSs' ability to target multiple pathways and modulate the tumor microenvironment makes them a transformative approach in cancer treatment. Overall, CDSs hold the key to maximizing curcumin's therapeutic potential and revolutionizing cancer care.
Contributions
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No.32060220) and Natural Science and Technology Foundation of Guizhou (No. QKH [2020]1Y094).
