Abstract
Isoquercetin, a naturally occurring dietary flavonoid glycoside, has shown many promising biological activities, such as antioxidant, anti-inflammatory, and anticancer. It also exhibits broad-spectrum antiviral activity, significantly reducing cell infection by influenza, Zika, Ebola, and dengue viruses, among others. Its beneficial effect for mitigating the disease of COVID-19 can be explained due to its inhibitory effects on several stages of the viral life cycle, from entry to replication and virus infectivity, and thus isoquercetin can be a promising anticoronaviral flavonoid to manage severe acute respiratory syndrome-coronavirus-2 (SARS-CoV-2) infection and warrants further investigation into its potential use in clinical settings.
Introduction
Isoquercetin (IQue) (Synonym: isoquercitrin, isotrifoliin, Figure 1) is a flavonoid glycoside having glucose linked to OH-3 of quercetin (3,3′,4′,5,7-pentahydroxyflavone), and thus it is also known as quercetin-3-
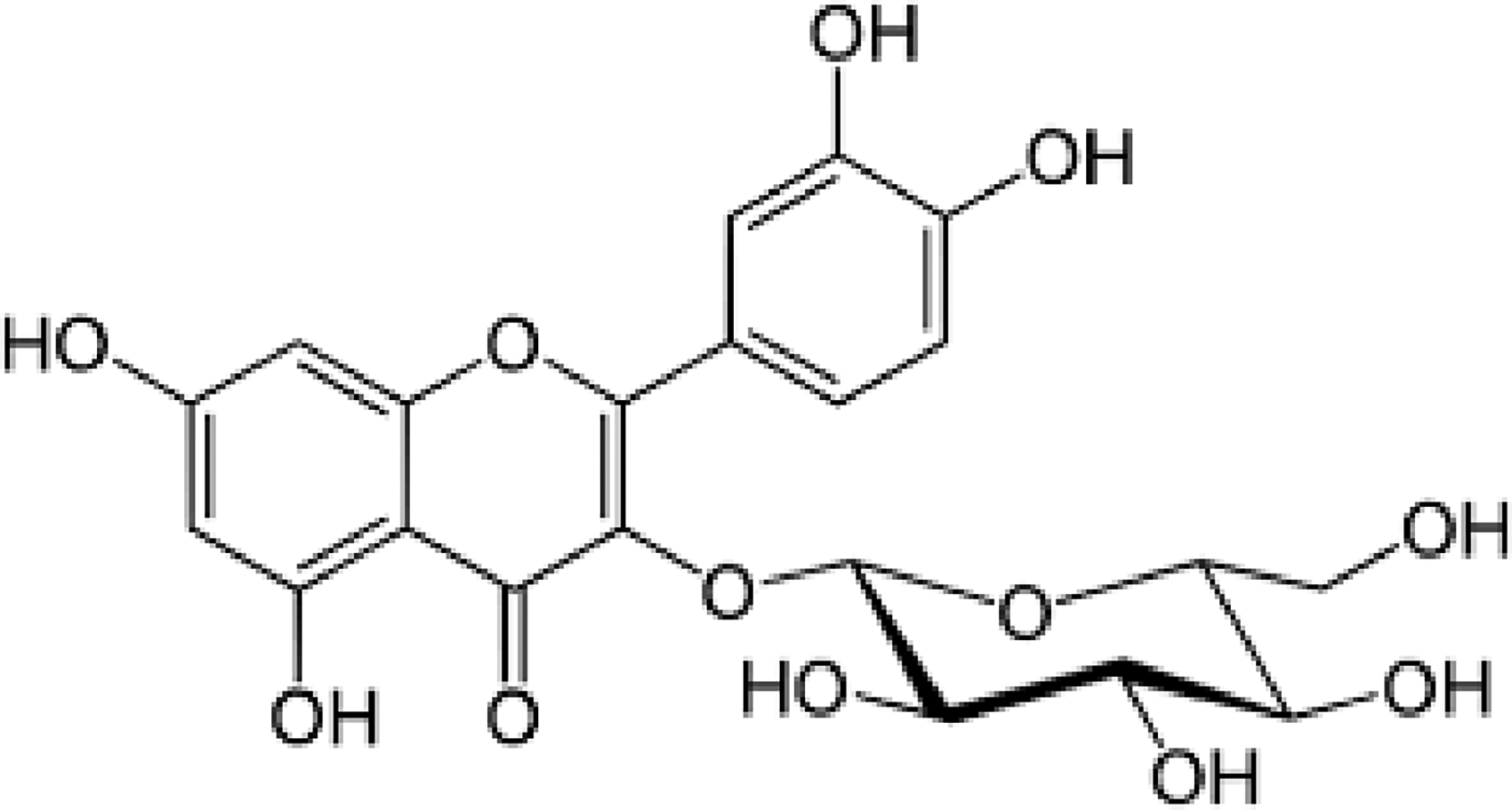
Chemical structure of isoquercetin (IQue).
IQue exerts many bioactivities including antioxidant, anti-inflammatory, anticancer, antidiabetic, antihypertensive, antibacterial, antimicrobial, anticoagulant, neuroprotective, and cytoprotective, as well as reducing the risk of cardiovascular disease.17-35 It has been reported to reduce the adverse effects of sunitinib, one of the most commonly prescribed drugs for advanced renal cell carcinoma. 36 According to a recent study, IQue targets extracellular protein disulfide isomerase (PDI), a thiol isomerase secreted by vascular cells, which is required for thrombus formation, and improves markers of coagulation in advanced cancer patients. 37
Several authors have reviewed the antiviral properties of quercetin-type flavonoids as these can work through several different mechanisms, such as blocking the entry of the virus into host cells, interfering with various stages of virus replication processes or translation, and/or preventing the release of the viruses to infect other cells.7,10-14,35,38-67 Several reports67-84 have demonstrated that IQue has antiviral significance against the influenza, Zika, Herpes, Ebola, dengue, varicella-zoster (VZV), human cytomegalovirus (HCMV), and Mayaro viruses.
Antiviral Significance of Isoquercetin
In a study related to antioxidant and antiviral evaluation of apple pomace extracts, Suárez et al found that methanol and acetone extracts (containing IQue) inhibit both herpes simplex virus type 1 (HSV-1) and 2 (HSV-2) replication in Vero cells by more than 50%, at noncytotoxic concentrations.
4
In another study related to identification of chemical constituents from
IQue has been shown to inhibit influenza A and B (IAB) virus replication in infected Madin-Darby canine kidney (MDCK) cells. The combination of IQue with amantadine showed a synergistic effect on IAB viral replication.
70
The IQue present in
IQue has been reported to protect mice from the Ebola virus (EBOV) when given prophylactically as little as 30 min prior to infection (IC50 5.3 µM). Even though the mechanism of action is unknown, it targets the early steps of viral entry. 76 These studies also suggested that IQue could be effective in the inhibition of a second common ebolavirus isolated in Sudan (SUDV). 76 IQue showed strong inhibition against strains of African historical and Asian epidemic strains of Zika virus (ZIKV) by interfering with its entrance process and blocking its internalization in host cells.77,78
The flavonoids, quercetin and its glycosides, including IQue from the Brazilian shrub
Virtual screening of 91 compounds found in Ginseng and Notoginseng against the Dengue viral protein NS5 methyl transferase domain suggested that IQue formed a relatively stable complex with its active sites. In vitro studies showed that IQue can inhibit Dengue virus by reducing viral RNA and viral protein synthesis with low toxicity to cells (CC50 > 20 mM), and thus its significance as an anti-DENV agent has been suggested. 82
Isoquercetin and COVID-19
The severe acute respiratory syndrome-coronavirus-2 (SARS-CoV-2), responsible for COVID-19, uses the spike glycoprotein present on its surface to interact with angiotensin-converting enzyme-2 (ACE2), which results in its entry and subsequent release of genomic RNA into the cytoplasm of the host cell.83,84 Two cysteine proteases, a papain-like protease (PLpro) and a 3C-like protease (3CLpro) or main protease (Mpro), are encoded by open reading frame 1a (ORF1a) and are involved in the cleavage of the first 3 and later 11 cleaving sites, respectively, of its polyprotein to release 16 nonstructural proteins (nsp1-nsp16), which are essential for viral replication and transcription of the genome.85,86 This is followed by virus assembly, and subsequently new virions are released from the infected cell. Thus, antiviral substances may act in various stages of coronavirus infection: (i) preventing the binding between virus and host cells and thus inhibiting internalization, (ii) blocking viral replication and/or multiplication, (iii) inhibiting the spread of the virus in the body, and (iv) modulating the inflammation.
Since enzymes such as Mpro and PLpro play crucial roles in the virus life cycle, therefore these are attractive targets. In this context, Mpro has attained an important position as it is not homologous with any human enzyme and therefore it has become an important molecular target in the development of anti-SARS-CoV-2 drugs. One of the promising strategies against COVID-19 virus infection is to identify potential natural compounds using in silico molecular docking studies in which these ligand compounds interact virtually through various intermolecular interactions such as hydrogen bonds, hydrophobic interactions and van der Waals’ forces with the active binding sites of the target receptor protein. Among various classes of natural products, flavonoids have been evaluated by many researchers to identify potential candidates for further experimental anticoronaviral evaluation. Moreover, flavonoids, being present abundantly in a variety of medicinal plants, and in several edible fruits and vegetables, are considered pleiotropic compounds, that is, the functional (hydroxyl, methoxy, and/or glycosyl) groups present in these polyphenolic compounds might interact with various sites of receptors and thus blocking and/or inhibiting several pathways related to virus inoculation and replication.40-82,87 Furthermore, flavonoids also exhibit antioxidant, anti-inflammatory and immunomodulatory properties that could reduce the severity of infection symptoms and enhance the immune response.61,70,88
ACE2 is a host receptor for SARS-CoV-2. Liu et al 59 have shown that flavonoids having 3′,4′-dihydroxylation inhibit recombinant human angiotensin-converting enzyme-2 [(rh)ACE2] activity. At 10 μM, Que was most active and IQue inhibited rhACE2 activity by 42% to 48%. Thus, it is plausible that dietary polyphenols (such as Que and IQue) intake could inhibit ACE2, particularly in the digestive tract. 59
A study by Jo et al, using a tryptophan-based fluorescence method, identified IQue as the blocker for the enzymatic activity of Middle East respiratory syndrome-coronavirus (MERS-CoV) 3CLpro, showing binding affinity (−9.751 kcal/mol) and an IC50 of 37.03 μM. 89 The docking study reflected that the hydroxymethyl group of the glucosyl moiety of IQue participated in hydrogen bond interactions between the hydroxyl and Glu169 of the S1 catalytic site of 3CLpro. 89
Inhibition of the 3 variants of 3CLpro of SARS-CoV and SARS-CoV-2 by 72 flavonoids was studied by Peterson using in silico molecular dynamics docking studies; 14 flavonoids were identified as hits, one of which was IQue. The mean binding energy across the 3 CLpro variants for IQue was −7.9 kcal/mol. 90
Another study, related to the screening of phytocompounds from
Virtual screening of 1064 phytochemicals for their inhibitory potential against RNA-dependent RNA-polymerase (RdRp) and Mpro using molecular docking and molecular dynamics simulation approaches, suggested IQue as one of the prominent constituents involved in high binding with Mpro. IQue forms a stable Mpro-IQue complex due to H-bond interactions with ASN142, Glu166 and Pro168 and through hydrophobic interactions with HIS41 (molecular docking score: −10.432), thus reflecting IQue as a possible lead molecule against Mpro. 94
Johnson et al employed computational techniques for the 168 chemical constituents of
Kushwaha et al, 96 based on results of molecular docking, showed that IQue was involved effectively with the spike, RdRp, and Mpro targets. IQue showed hydrogen bonding with Phe347, Val350, Asp442, and Phe497; and hydrophobic interactions with Arg509, Arg346, Ala348, Val401, Ser349, Gln493, Tyr495, Tyr351, Ser494, Gln498, Pro499, and Ser443 amino acid residues of the S1 binding site (binding efficacy −8.22 kcal/mol) of spike protein. IQue interacted with Mpro via hydrogen bonds with several amino acid residues, particularly with Leu27, His41, Met49, Asp142, Gly143, Leu167, Asp187, and Glu189, and with Phe140 by hydrophobic interaction, with a docking score of −9.73 kcal/mol. IQue formed hydrogen bonds with Trp617, Asp618, Tyr619, Asp760, and Lys798 amino acid residues of the active site of the RdRp protein, and hydrophobic interactions with residues Pro620, Lys621, Cys622, Cys799, Trp800, Gly616, Ala762, Asp761, Glu811, Ser814, and Ser759, having a binding efficacy of −6.86 kcal/ mol. Thus, IQue possesses significant efficacy in SARS-CoV-2 virulence mitigation. 96
In another molecular docking and simulation study about inhibition of the Mpro activity of SARS-CoV-2 and the replication of human coronavirus 229E for five flavonols and three dihydroflavonols, Zhu et al identified that IQue can bind to at least two subsites (S1, S1′, S2, and S4) in the binding pocket of SARS-CoV-2 Mpro. 97 Their studies predicted that the A-ring, B-ring, C-ring, and glucosyl group of IQue occupied S2, S1′, the space between S1 and S2, and S1, respectively. The affinity scores of IQue binding to Mpro and 229E Mpro were −7.5 and −8.7 kcal/mol, respectively. Further in vitro assays substantiated the prediction of docking simulation. In the tested concentrations, IQue, starting from 2.5 μM, showed a significant inhibition of HCoV-229E replication in Huh-7 cells. IQue inhibited the activity of SARS-CoV-2 Mpro with an IC50 value of 4.03 μM. These observations indicate IQue's usefulness as a promising antiviral agent. 97
Furthermore, Hiremath et al
98
studied, in silico, the ability of the constituents of
To investigate the use of Toujie Quwen Granules, a traditional Chinese medicine, composed of 16 medicinal materials, Ye et al 99 used network pharmacology, molecular docking and surface plasmon resonance technology (SPR) to explore the potential compounds and their interaction mechanisms for the treatment of COVID-19. Among the 4 prominent identified compounds was IQue. It exhibited affinity scores of −8.3, −8.1, −9.8, −6.6, and −8.8 kJ/mol with 3CLpro, RdRp, ACE2, interleukin 6 (IL-6), and S-protein, respectively. Hydrogen bond interactions and hydrophobic interactions between ligand and various amino acid residues were responsible for these bindings. The SPR titration suggested a Kd value of 4.54 × 10−6 M for IQue and S-protein, which led to speculation that IQue can bind to the receptor binding domain (RBD) region of S-protein and inactivate it to prevent its binding to ACE2 of the host surface. 100
Intriguingly, the extract of European black elderberry (
Clinical Trials
An open-labelled, randomized and multi-center clinical trial on the effect of IQue in subjects with RT-PCR confirmed SARS-CoV-2 infection with mild-to-moderate symptoms was investigated to determine its clinical efficacy in patients with COVID-19. This trial has been registered in ClinicalTrials.gov with the ID code (NCT04733651). This study, which is being conducted in Nepal, aims to determine whether the daily use of IQue, 1000 mg as 4 × 250 mg IQue capsules for 28 days, can either prevent or treat COVID-19. The results of this trial have not been published as of this writing. 101
In Canada, a trial to evaluate the effect of IQue (IQC-950AN) for the treatment of disease progression in patients with confirmed COVID-19, as well as on the reduction of SARS-CoV-2 viral titers, in addition to standard of care, has been registered (ID code NCT04536090). Patients will be randomized to receive treatment for 28 days and then will return 30 days following the discontinuation of treatment for a final safety visit. However, results of this trial are not yet available. 102
A randomized, double-blind, placebo-controlled phase 2 clinical trial to evaluate the safety and efficacy of Masitinib combined with IQue, and best supportive care in hospitalized patients with moderate and severe COVID-19 is also under way and results are awaited. 103
Results from a systematic review and meta-analysis of 30 randomized controlled trials about the use of flavonoids for treating viral acute respiratory tract infections (ARTIs) also suggested that flavonoids are efficacious and safe in treating viral ARTIs and COVID-19. 104 In a phase I trial study related to the use of IQue as an adjunct therapy in patients with kidney cancer receiving first-line Sunitinib (QUASAR), Buonerba et al found that 900 mg/day IQue is safe to use for a median 81 days. 36 IQue, being water-soluble, was found to improve endothelial function in high-risk adult participants with cardiovascular disease. 27
Formulation
IQue is about 4-fold more soluble in water (∼206 μM) than quercetin (∼50 μM), 105 and is highly bioavailable in the lumen of the intestine. 23 The biological activities of IQue are generally rendered from its conversion to Que. When IQue is taken orally, it is better absorbed in the intestines and rapidly converted into Que and its metabolites. 35
IQue has been formulated as an inclusion complex in
IQue formed a large network of interactions with Mpro; the ring-B-dihydroxy phenyl moiety occupies the S1 subpocket, ring-A meta-hydroxyl groups form hydrogen bond interactions with the side chain of His163; while the aromatic ring interacts with Leu141, Asn142, Ser144, Cys145, and Glu166. However, the glucoside moiety accommodates the S2 cavity, perhaps forming a hydrogen bond to Gly143 and also interacts with the catalytic His41; the S3 cavity binds the flavone nucleus, also via hydrogen bond interactions with Gln192. According to Wurtele et al, IQue-cyclodextrin inclusion complex formulations could be interesting for the development of future COVID-19 drugs, as well as for other emerging viral diseases, due to the binding with Mpro, which is essential for virus replication. 106
Synergistic Effect of Remdesivir with Isoquercetin
In an investigation of the efficacy of the antiviral effects of Remdesivir, the first-approved drug for the treatment of severe COVID-19, in combination with IQue, Rabezanahary et al noted strong antiviral synergistic effects in vitro. 107 The 50% effective concentration (EC50) to inhibit SARS-CoV-2 replication for 3QG in monotherapy was 23.99 ± 1.07 mM. These authors also investigated the EC50 for the individual reference compounds by mixing different concentrations of each compound, Remdesivir and 3QG, with a constant concentration of the second one. The combination of remdesivir and 3QG exhibited highly potent synergism as compared with their individual EC50 values. Considering Remdesivir as the reference compound, the addition of 15.65 mM of IQue reduced the Remdesivir EC50 values by at least half (8.18 ± 4.88 to 2.29 ± 0.02 mM). The EC50 was reduced 5 times with 31.5 mM EGCG. IQue showed a synergy score of 11.07. These results indicate that low concentrations of IQue can potentiate the activity of Remdesivir, thus reflecting an interesting avenue for combinational therapies for the treatment of COVID-19. 107 Another study showed that use of 1000 mg of quercetin daily in combination with the antiviral drugs, remdesivir and favipiravir in severe forms of COVID-19 disease was able to reduce the hospitalization period, reflecting its therapeutic significance. 16
Bioavailability and Safety
IQue and its alpha-glycosyl derived glycosides (AGIQ) showed a favorable safety profile 31 and the aglycone, quercetin, has been established by the US Food and Drug Administration (FDA) as a generally recognized as safe (GRAS) compound.51,108 Alpha-glycosyl isoquercitrin with enhanced solubility and bioavailability is used in Japan as a food additive and has GRAS status. 31 IQue, being glycosylated, is more soluble, and highly bioavailable80,109 in the lumen of the intestine, and is well tolerated in humans. 20
Concluding Remarks
Taking all the literature studies into account, IQue can be considered as a prophylactic option for preventing and managing COVID-19. Its multifactorial beneficial action can also be corroborated due to its broad-spectrum antiviral, antioxidant, anti-inflammatory, immunomodulatory and anticoagulant significance. IQue could act as a multi-targeted ligand due to its binding to (i) spike glycoprotein, disrupting the viral–host recognition interface and hence preventing SARS-CoV-2 entry, and (ii) viral replication by interfering with the activity of main protease (Mpro), a PLpro, and RNA-dependent RNA polymerase (RdRp). Thus, the beneficial effect of IQue for mitigating COVID-19 disease can be explained due to its inhibitory effects on several stages of the viral life cycle, from entry to replication, its function on targeting various mechanisms of oxidative stress, excessive inflammation, and virus infectivity, and warrants further investigation into its potential use in clinical settings.
Footnotes
Acknowledgement
PKA thanks Alexander von Humboldt Foundation for providing financial assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
