Abstract
Introduction
Alzheimer's disease (AD), an age-related neurodegenerative disease, is characterized by memory loss, decline in language, and a cognitive and behavioral disorders. 1 AD is one of the most dangerous diseases, ranking fifth in terms of mortality rate among adults aged 65 and older, as well as seventh in mortality rate among all adults in 2020 and 2021. 2 Studies demonstrated that many factors are involved in the pathophysiology of AD. For example, there is a deficient cholinergic neurotransmission function, 3 accumulation of aggregates of amyloid-β (Aβ) 4 and hyperphosphorylated tau proteins, 5 increased production of reactive oxygen species (ROS), 6 imbalance of biometals (zinc, copper, calcium, and iron) in the brain, 7 and deficits in acetylcholine (ACh). 8 Acetylcholinesterase (AChE), a form of cholinesterases, is known to immediately hydrolyze ACh in cholinergic synapses and stimulate the aggregation of amyloid-β peptide fragments and thus contributing to amyloid formation. 9 Therefore, common therapeutic treatments for AD have been mainly based on enhancing cholinergic function by utilizing AchE inhibitors (AChEIs) to improve the levels of acetylcholine (ACh) in cholinergic synapses. 10 Up to now, the FDA has approved four AChEIs (donepezil, tacrine, galantamine, and rivastigmine), an N-methyl-D-aspartic acid (NMDA) antagonist (memantine), and a humanized IgG1 monoclonal antibody (lecanemab) for the treatment of AD.11,12 However, the increasing use of AChEIs has led to reduced AChEIs efficacy, increased adverse drug reactions (ADRs), and serious side effects.13–15 Accordingly, the pursuit of new AChE inhibitors with fewer side effects has attracted great interest among scientists.16–19
In this regard, pyrimidine, a N-based heterocyclic ring, is a privileged substructural motif in a diversity of drugs/lead, including antimicrobial,
20
antibacterial,
21
antitumor,
22
anticancer,
23
cytotoxicity,
24
HMG-CoA reductase inhibitors,
25
and more. Notably, a novel series of pyrimidine-based molecules have been developed with roles as potential treatments for AD, functioning as γ-secretase modulators, cholinesterase inhibitors, Aβ aggregation inhibitors, and BACE1 (β-secretase) inhibitors.26–29 For example, Rao et al have reported pyrido[3,2-d]pyrimidine bioisostere

A number of reports have appeared in the literature regarding the multicomponent synthesis of pyrano[2,3-d]pyrimidines, which are unsaturated six-membered heterocycles formed by fusion of pyran and pyrimidine rings.48–52 Nevertheless, less works have employed the 2-amino-4H-pyran-3-carbonitrile derivatives to construct pyrano[2,3-d]pyrimidines skeleton directly. Furthermore, numerous effective synthetic methods have been recorded for generating 2-amino-4H-pyran-3-carbonitrile intermediates.53–62 Therefore, as part of our ongoing research focused on the discovery of novel bioactive compounds,63–67 in this study, we focused on the synthesis and biological evaluation of novel pyrano[2,3-d]pyrimidine compounds
Results and Discussion
Nowadays, organocatalysts have been used extensively in green chemistry and small molecule drug discovery because of their efficiency, stability, purity, and selectivity.
68
Diazabicyclo[2.2.2]octane (DABCO), an organocatalyst, has received considerable attention as an inexpensive, eco-friendly, highly reactive, and nontoxic base catalyst in the synthesis of pyran derivatives through multicomponent reactions.69,70 In our previous study, utilizing DABCO as a base catalyst and acetonitrile as a solvent, we successfully synthesized 2-amino-5,10-dioxo-4-aryl-5,10-dihydro-4H-benzo[g]chromene-3-carbonitrile compounds in good yields via the microwave-assisted three-component reaction of 1,4-naphthoquinone, malononitrile, and various arylaldehydes.
49
Therefore, in this study, 0.2 mmol of DABCO as a base catalyst was added to the mixture of cyclohexane-1,3-dione (1 mmol), malononitrile (1 mmol), and 4-methoxyaldehyde (1 mmol) in acetonitrile. The reaction mixture was stirred at 82 °C in 15 min under microwave irradiation to furnish 2-amino-4-(4-methoxyphenyl)-5-oxo-5,6,7,8-tetrahydro-4H-chromene-3-carbonitrile (

Synthesis of pyrano[2,3-d]pyrimidine compounds
The plausible mechanism for the formation of products

The plausible reaction mechanism.
The ability of pyrano[2,3-d]pyrimidines to inhibit the AChE enzymes was then evaluated by the modified Ellman's method.
29
AChE inhibitory activity results were expressed as inhibitory concentration 50% (IC50, µg/mL) and donepezil was used as a reference. As shown in Table 1, while products
Inhibitory Activity of Pyrano[2,3-d]Pyrimidine Derivatives (
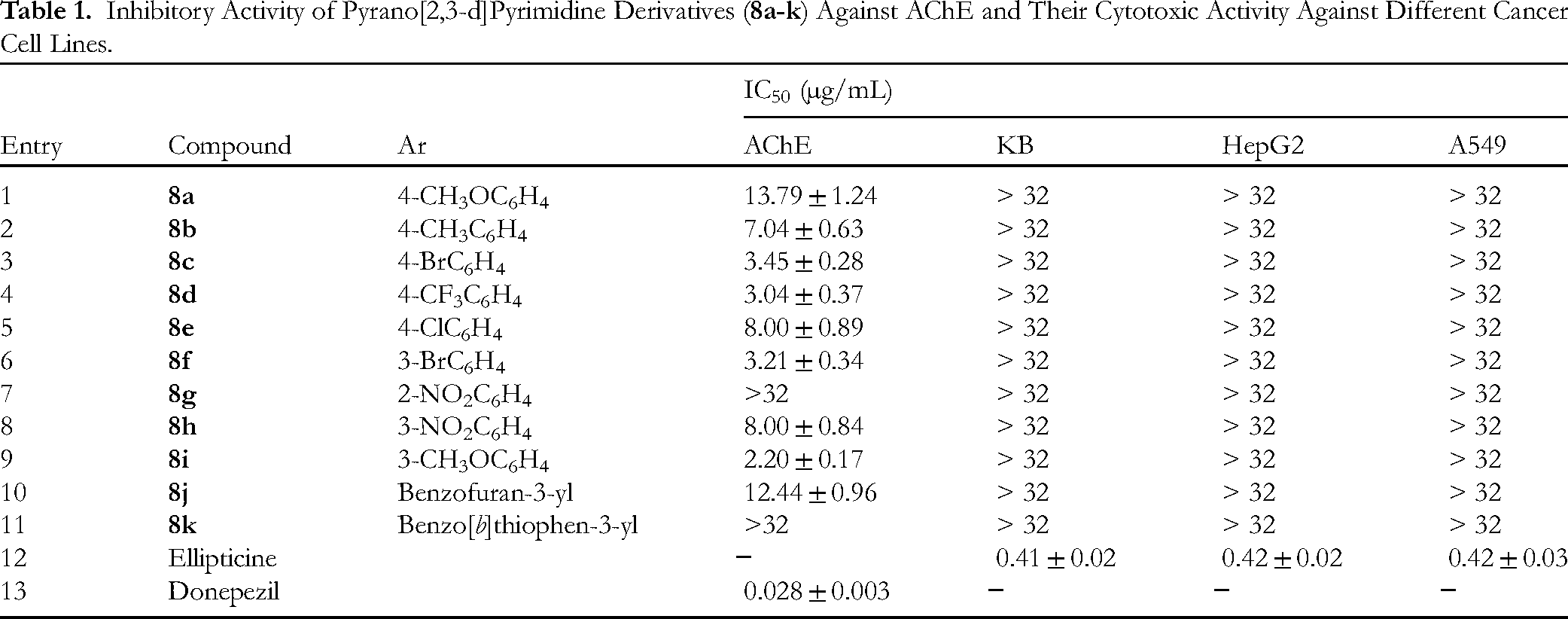
Besides that, all synthesized compounds were also evaluated for their cytotoxic activities against human cancer cell lines, namely human epidermoid carcinoma cancer cell line (KB), human lung cancer cell line (A549) and human hepatoma carcinoma cancer cell line (HepG2) by MTT (3-[4,5-di-methylthiazol-2-yl]-2,5-diphenyltetrazolium bromide) assay,71,72 with ellipticine as a reference. The results indicated that all synthesized products have shown no cytotoxic activity against selected cancer cell lines with IC50 > 32 µg/mL (Table 1).
Materials and Methods
Experimental procedures for the synthesis of products
5-(4-Methoxyphenyl)-2-Methyl-5,7,8,9-Tetrahydro-4H-chromeno[2,3-d]Pyrimidine-4,6(3H)-Dione (8a )
Yield 264 mg (78%), white solid, mp. 268-269 °C. Rf = 0.48 (EtOAc/Acetone 1:20). IR (KBr) 3496, 3265, 3102, 3010, 2943, 2785, 1662, 1602, 1510, 1456, 1366, 1302, 1258, 1243, 1202, 1179, 1150, 1125, 1029, 978, 809, 578 cm−1. 1H NMR (CDCl3, 600 MHz): δ 13.19 (1H, s, NH), 7.22 (2H, dd, J = 7.2, 2.4 Hz), 6.74 (2H, dd, J = 7.2, 2.4 Hz), 4.90 (1H, s), 3.72 (3H, s, OCH3), 2.78-2.73 (2H, m), 2.42-2.32 (2H, m), 2.35 (3H, s, CH3), 2.09-2.01 (2H, m). 13C NMR (CDCl3, 125 MHz): δ 196.5, 165.0, 164.7, 160.8, 158.4, 158.2, 135.7, 129.4 (2C), 116.1, 113.5 (2C), 103.0, 55.2, 36.9, 32.1, 27.3, 21.2, 20.3. HRMS (ESI+) m/z [M + H]+ calc. for: C19H19N2O4: 339.1339, found: 339.1340, m/z [M + Na]+ calc. for: C19H18N2NaO4: 361.1159, found: 361.1154.
Yield 258 mg (80%), white solid. Rf = 0.50 (EtOAc/Acetone 1:20). IR (KBr) 3440, 3101, 3038, 2950, 2781, 1656, 1605, 1573, 1479, 1429, 1362, 1304, 1239, 1205, 1179, 1149, 1125, 1064, 1027, 980, 864, 802, 709, 579 cm−1. 1H NMR (DMSO-d6, 600 MHz): δ 12.49 (1H, s, NH), 7.09 (2H, d, J = 7.8 Hz), 7.01 (2H, d, J = 7.8 Hz), 4.67 (1H, s), 2.74-2.61 (2H, m), 2.35-2.23 (2H, m), 2.24 (3H, s, CH3), 2.20 (3H, s, CH3), 2.00-1.96 (1H, m), 1.91-1.86 (1H, m). 13C NMR (DMSO-d6, 150 MHz): δ 196.2, 165.6, 162.2, 159.9, 158.5, 141.1, 135.5, 128.6 (2C), 127.9 (2C), 115.0, 101.9, 36.5, 31.9, 26.7, 20.9, 20.6, 20.0. HRMS (ESI+) m/z [M + H]+ calc. for: C19H19N2O3: 323.1390, found: 323.1385, m/z [M + Na]+ calc. for: C19H18N2NaO3: 345.1210, found: 345.1212.
5-(4-Bromophenyl)-2-Methyl-5,7,8,9-Tetrahydro-4H-chromeno[2,3-d]Pyrimidine-4,6(3H)-Dione (8c )
Yield 314 mg (81%), white solid, mp. 299-301 °C. Rf = 0.51 (EtOAc/Acetone 1:20). IR (KBr) 2920, 2867, 2785, 2650, 1912, 1795, 1679, 1650, 1598, 1501, 1424, 1366, 1327, 1240, 1205, 1186, 1152, 1106, 1065, 1020, 982, 934, 895 cm−1. 1H NMR (DMSO-d6, 600 MHz): δ 12.55 (1H, s, NH), 7.41 (2H, dd, J = 7.8, 1.8 Hz), 7.18 (2H, dd, J = 7.8, 1.8 Hz), 4.68 (1H, s), 2.75-2.62 (2H, m), 2.37-2.25 (2H, m), 2.26 (3H, s, CH3), 2.01-1.96 (1H, m), 1.94-1.87 (1H, m). 13C NMR (DMSO-d6, 150 MHz): δ 196.0, 165.8, 162.0, 159.9, 158.8, 143.2, 130.7 (2C), 130.3 (2C), 119.4, 114.2, 101.1, 36.3, 32.1, 26.6, 20.9, 19.8. HRMS (ESI+) m/z [M + H]+ calc. for: C18H16BrN2O3: 387.0339 and 389.0319, found: 387.0343 and 389.0326, m/z [M + Na]+ calc. for: C18H15BrN2NaO3: 409.0159 and 411.0138, found: 409.0162 and 411.0136.
2-Methyl-5-(4-(Trifluoromethyl)Phenyl)-5,7,8,9-Tetrahydro-4H-chromeno[2,3-d]Pyrimidine-4,6(3H)-Dione (8d )
Yield 294 mg (78%), white solid. Rf = 0.49 (EtOAc/Acetone 1:20). IR (KBr) 2920, 2867, 2785, 2650, 1912, 1795, 1679, 1650, 1598, 1501, 1424, 1366, 1327, 1240, 1205, 1186, 1152, 1106, 1065, 1020, 982, 934, 895 cm−1. 1H NMR (DMSO-d6, 600 MHz): δ 12.57 (1H, s, NH), 7.59 (2H, d, J = 7.8 Hz), 7.46 (2H, d, J = 7.8 Hz), 4.78 (1H, s), 2.77-2.65 (2H, m), 2.38-2.27 (2H, m), 2.26 (3H, s, CH3), 2.02-1.98 (1H, m), 1.95-1.89 (1H, m). 13C NMR (DMSO-d6, 150 MHz): δ 196.1, 166.0, 162.0, 160.0, 159.0, 148.4, 128.9, 127.0 (1C, q, J = 31.5 Hz), 124.8 (2C, d, J = 4.5 Hz), 124.2 (1C, q, J = 270 Hz, CF3), 114.0, 100.8, 36.3, 32.7, 30.6, 26.7, 20.9, 19.8.
5-(4-Chlorophenyl)-2-Methyl-5,7,8,9-Tetrahydro-4H-chromeno[2,3-d]Pyrimidine-4,6(3H)-Dione (8e )
Yield 271 mg (79%), white solid, mp. 293-295 °C. Rf = 0.53 (EtOAc/Acetone 1:20). IR (KBr) 3444, 2935, 2842, 2778, 1668, 1638, 1487, 1405, 1369, 1324, 1249, 1237, 1195, 1176, 1144, 1125, 1083, 1009, 950, 857, 802, 743, 593, 578, 518 cm−1. 1H NMR (DMSO-d6, 600 MHz): δ 12.54 (1H, s, NH), 7.27 (2H, dd, J = 6.6, 2.4 Hz), 7.24 (2H, dd, J = 6.6, 1.8 Hz), 4.70 (1H, s), 2.75-2.63 (2H, m), 2.37-2.25 (2H, m), 2.26 (3H, s, CH3), 2.02-1.96 (1H, m), 1.94-1.87 (1H, m). 13C NMR (DMSO-d6, 150 MHz): δ 196.1, 165.8, 162.1, 160.0, 158.8, 142.9, 131.0, 129.9 (2C), 127.9 (2C), 114.4, 101.2, 36.4, 32.1, 26.7, 20.9, 19.9. HRMS (ESI+) m/z [M + H]+ calc. for: C18H16ClN2O3: 343.0844, found: 343.0842, m/z [M + Na]+ calc. for: C18H15ClN2NaO3: 365.0664, found: 365.0651.
5-(3-Bromophenyl)-2-Methyl-5,7,8,9-Tetrahydro-4H-chromeno[2,3-d]Pyrimidine-4,6(3H)-Dione (8f )
Yield 299 mg (77%), white solid, mp. 300-302 °C. Rf = 0.62 (EtOAc/Acetone 1:20). IR (KBr) 3437, 2937, 2837, 2758, 1667, 1638, 1604, 1564, 1493, 1469, 1417, 1370, 1329, 1240, 1196, 1147, 1127, 1069, 972, 825, 785, 696, 672, 608, 586 cm−1. 1H NMR (DMSO-d6, 600 MHz): δ 12.57 (1H, s, NH), 7.40 (1H, s), 7.34-7.31 (1H, m), 7.19 (2H, d, J = 4.8 Hz), 4.68 (1H, s), 2.76-2.71 (1H, m), 2.68-2.62 (1H, m), 2.34-2.25 (2H, m), 2.25 (3H, s, CH3), 2.01-1.96 (1H, m), 1.93-1.87 (1H, m). 13C NMR (DMSO-d6, 125 MHz): δ 196.1, 166.0, 162.0, 160.0, 158.9, 146.5, 130.9, 130.2, 129.3, 127.0, 121.1, 114.1, 100.9, 36.3, 32.4, 26.7, 20.9, 19.8. HRMS (ESI+) m/z [M + H]+ calc. for: C18H16BrN2O3: 387.0339 and 389.0319, found: 387.0333 and 389.0320, m/z [M + Na]+ calc. for: C18H15BrN2NaO3: 409.0159 and 411.0138, found: 409.0146 and 411.0127.
2-Methyl-5-(2-Nitrophenyl)-5,7,8,9-Tetrahydro-4H-chromeno[2,3-d]Pyrimidine-4,6(3H)-Dione (8g )
Yield 258 mg (73%), white solid, mp. 273-275 °C. Rf = 0.60 (EtOAc/Acetone 1:20). IR (KBr) 3444, 2937, 2872, 2781, 1674, 1646, 1607, 1532, 1424, 1364, 1327, 1243, 1190, 1154, 1067, 982, 861, 829, 786, 733, 702, 581 cm−1. 1H NMR (DMSO-d6, 600 MHz): δ 12.47 (1H, s, NH), 7.81 (1H, d, J = 7.8 Hz), 7.55 (1H, t, J = 7.8 Hz), 7.37 (1H, d, J = 7.8 Hz), 7.36 (1H, t, J = 7.8 Hz), 5.64 (1H, s), 2.70-2.65 (2H, m), 2.34-2.29 (1H, m), 2.25 (3H, s, CH3), 2.25-2.20 (1H, m), 2.00-1.96 (1H, m), 1.92-1.86 (1H, m). 13C NMR (DMSO-d6, 125 MHz): δ 196.0, 166.1, 161.8, 159.8, 159.1, 149.1, 138.0, 132.9, 130.7, 127.3 123.9, 113.6, 100.6, 36.3, 28.4, 26.7, 20.9, 19.8. HRMS (ESI+) m/z [M + H]+ calc. for: C18H16N3O5: 354.1085, found: 354.1083, m/z [M + Na]+ calc. for: C18H15N3NaO5: 376.0904, found: 376.0893.
2-Methyl-5-(3-Nitrophenyl)-5,7,8,9-Tetrahydro-4H-chromeno[2,3-d]Pyrimidine-4,6(3H)-Dione (8h )
Yield 265 mg (73%), white solid, mp. 291-293 °C. Rf = 0.58 (EtOAc/Acetone 1:20). IR (KBr) 3478, 3373, 3094, 3015, 2938, 2877, 2794, 1677, 1663, 1600, 1525, 1429, 1350, 1324, 1241, 1203, 1186, 1154, 1123, 985, 917, 825, 772, 731, 683, 578 cm−1. 1H NMR (DMSO-d6, 600 MHz): δ 12.57 (1H, s, NH), 8.07 (1H, t, J = 1.8 Hz), 8.03-8.00 (1H, m), 7.69 (1H, d, J = 7.8 Hz), 7.54 (1H, t, J = 7.8 Hz), 4.83 (1H, s), 2.79-2.73 (1H, m), 2.72-2.66 (1H, m), 2.38-2.32 (1H, m), 2.31-2.26 (1H, m), 2.27 (3H, s, CH3), 2.04-1.97 (1H, m), 1.96-1.89 (1H, m). 13C NMR (DMSO-d6, 150 MHz): δ 196.1, 166.3, 162.0, 160.0, 159.2, 147.4, 145.9, 134.8, 129.5, 122.7, 121.5, 113.7, 100.5, 36.2, 32.8, 26.7, 20.9, 19.8. HRMS (ESI+) m/z [M + H]+ calc. for: C18H16N3O5: 354.1085, found: 354.1073, m/z [M + Na]+ calc. for: C18H15N3NaO5: 376.0904, found: 376.0887.
5-(3-Methoxyphenyl)-2-Methyl-5,7,8,9-Tetrahydro-4H-chromeno[2,3-d]Pyrimidine-4,6(3H)-Dione (8i )
Yield 257 mg (76%), white solid, mp. 282-283 °C. Rf = 0.65 (EtOAc/Acetone 1:20). IR (KBr) 3429, 3000, 2936, 2869, 1770, 1664, 1635, 1596, 1487, 1460, 1363, 1325, 1314, 1243, 1188, 1160, 1036, 980, 823, 780, 693, 617, 572, 457 cm−1. 1H NMR (DMSO-d6, 600 MHz): δ 12.52 (1H, s, NH), 7.14 (1H, t, J = 7.8 Hz), 6.77 (1H, d, J = 1.6 Hz), 6.76 (1H, td, J = 7.8 Hz), 6.72 (1H, dd, J = 7.8, 2.4 Hz), 4.71 (1H, s), 3.70 (3H, s), 2.76-2.71 (1H, m), 2.68-2.63 (1H, m), 2.36-2.27 (2H, m), 2.25 (3H, s, CH3), 2.02-1.97 (1H, m), 1.93-1.89 (1H, m). 13C NMR (DMSO-d6, 150 MHz): δ 196.1, 165.7, 162.1, 160.0, 158.9, 158.5, 145.3, 129.0, 120.0, 114.6, 114.4, 11.2, 101.5, 54.8, 36.4, 32.1, 26.6, 20.9, 19.9. HRMS (ESI+) m/z [M + H]+ calc. for: C19H19N2O4: 339.1339, found: 339.1333, m/z [M + Na]+ calc. for: C19H18N2NaO4: 361.1159, found: 361.1152.
5-(Benzofuran-3-yl)-2-Methyl-5,7,8,9-Tetrahydro-4H-chromeno[2,3-d]Pyrimidine-4,6(3H)-Dione (8j )
Yield 233 mg (67%), white solid, mp. 325-326 °C. Rf = 0.42 (EtOAc/Acetone 1:20). IR (KBr) 3428, 2859, 2781, 1673, 1638, 1607, 1449, 1364, 1328, 1239, 1190, 1153, 1127, 1099, 1080, 1028, 979, 950, 854, 802 cm−1. 1H NMR (DMSO-d6, 600 MHz): δ 12.58 (1H, s, NH), 7.77 (1H, s), 7.59 (1H, dd, J = 7.2, 0.6 Hz), 7.49 (1H, dd, J = 7.2, 0.6 Hz), 7.25 (1H, td, J = 7.2, 1.2 Hz), 7.21 (1H, td, J = 7.2, 1.2 Hz), 4.98 (1H, s), 2.79-2.73 (1H, m), 2.71-2.65 (1H, m), 2.35-2.25 (2H, m), 2.25 (3H, s, CH3), 2.02-1.96 (1H, m), 1.92-1.86 (1H, m). 13C NMR (DMSO-d6, 125 MHz): δ 196.1, 166.0, 162.1, 160.3, 158.7, 154.7, 143.6, 126.3, 123.9, 122.5, 122.1, 120.0, 113.2, 111.3, 100.0, 36.3, 26.6, 22.5, 20.9, 19.9.
5-(Benzo[b]Thiophen-3-yl)-2-Methyl-5,7,8,9-Tetrahydro-4H-chromeno[2,3-d]Pyrimidine-4,6(3H)-Dione (8k )
Yield 230 mg (63%), white solid, mp. 323-324 °C. Rf = 0.48 (EtOAc/Acetone 1:20). IR (KBr) 3422, 3050, 3005, 2939, 2866, 2775, 1671, 1634, 1601, 1490, 1457, 1424, 1363, 1325, 1312, 1238, 1189, 1150, 1126, 1055, 1026, 979, 907, 890, 797, 767, 742, 575 cm−1. 1H NMR (DMSO-d6, 600 MHz): δ 12.48 (1H, s, NH), 8.14 (1H, d, J = 7.8 Hz), 7.88 (1H, d, J = 7.8 Hz), 7.40 (1H, td, J = 7.8, 1.2 Hz), 7.36 (1H, s), 7.32 (1H, t, J = 7.8 Hz), 5.18 (1H, s), 2.78-2.72 (1H, m), 2.71-2.65 (1H, m), 2.35-2.28 (1H, m), 2.27-2.22 (1H, m), 2.25 (3H, s, CH3), 2.02-1.95 (1H, m), 1.92-1.84 (1H, m). 13C NMR (DMSO-d6, 150 MHz): δ 196.5, 1656.0, 162.6, 160.5, 158.9, 140.1, 139.9, 138.3, 125.7, 124.4, 124.2, 123.4, 123.0, 115.2, 101.9, 36.9, 27.2, 26.6, 21.4, 20.4. HRMS (ESI+) m/z [M + Na]+ calc. for: C20H16N2NaO3S: 387.0774, found: 387.0755.
2-Amino-4-(4-Methoxyphenyl)-5-oxo-5,6,7,8-Tetrahydro-4H-Chromene-3-Carbonitrile (6a )
Yield 263 mg (89%), white solid, mp. 224-225 °C. IR (KBr) 3459, 3322, 3211, 3175, 2924, 2186, 1682, 1663, 1601, 1509, 1450, 1409, 1365, 1259, 1243, 1210, 1171, 1132, 1067, 1030, 1001, 895, 843, 779, 696, 626, 566, 535 cm−1. 1H NMR (CDCl3, 600 MHz): δ 7.16 (2H, dd, J = 6.6, 2.4 Hz), 6.82 (2H, dd, J = 6.6, 2.4 Hz), 4.52 (2H, s, NH2), 4.39 (1H, s), 3.77 (3H, s, OCH3), 2.63-2.51 (2H, m), 2.41-2.30 (2H, m), 2.08-1.95 (2H, m). 13C NMR (CDCl3, 125 MHz): δ 196.0, 162.9, 158.7, 157.4, 135.5, 128.7 (2C), 118.7, 115.5, 114.0 (2C), 63.8, 55.2, 36.8, 34.7, 27.0, 20.1.
Conclusions
In summary, new pyrano[2,3-d]pyrimidine compounds have been synthesized in moderate to good yields using a “one-pot” two-step multicomponent reaction under microwave irradiation. The result of cytotoxic activity against cancer cells (KB, HepG2, and A549 cells) revealed that active products exhibited no cytotoxicity, suggesting a safety profile of these products. With respect to the AChE inhibitory activity, it can be concluded that almost all of the products displayed moderate to good AChE inhibitory activity. Products
Supplemental Material
sj-doc-1-npx-10.1177_1934578X231201037 - Supplemental material for Synthesis and Evaluation of Acetylcholinesterase Inhibitory and Cytotoxic Activities of Pyrano[2,3-d]pyrimidines
Supplemental material, sj-doc-1-npx-10.1177_1934578X231201037 for Synthesis and Evaluation of Acetylcholinesterase Inhibitory and Cytotoxic Activities of Pyrano[2,3-d]pyrimidines by Nguyen Ha Thanh, Nguyen Thi Quynh Giang, Nguyen Van Ha, Hoang Thi Phuong, Le Nhat Thuy Giang, Nguyen Tuan Anh, Ba Thi Cham, Le Duc Huy, Dang Thi Tuyet Anh, Phan Van Kiem and Nguyen Van Tuyen in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Institute of Chemistry, Vietnam Academy of Science and Technology (grant number VHH.2023.01).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
