Abstract
Background/Objective
Propolis, a resinous substance collected by honeybees from origin plants to protect their beehive, has been reported to have various biological activities and gained popularity as an alternative medicine in recent years. We have previously reported the plant origin, the chemical composition and some biological activities of native Thailand stingless bee propolis. In this study, we further elucidated its active components and their mode of action.
Methods/Results
Among nine prenylated xanthones isolated from this propolis, α-mangostin and γ-mangostin were shown to strongly inhibit tube formation of human umbilical vein endothelial cells (HUVECs), an indication of antiangiogenic activity in vitro. Further investigation of their effects at a molecular level revealed that both mangostins inactivated extracellular signal-regulated kinase 1/2 (ERK1/2) and activated p38 in tube-forming HUVECs in a concentration-dependent manner. Taken together, the two mangostins were shown to inhibit angiogenesis in vitro by inactivating angiogenic signal ERK1/2 and activating stress signal p38. Detailed comparison revealed that α-mangostin seemed to have a slightly stronger effect than γ-mangostin in these experiments. Combined with the fact that being the most abundant component among the nine prenylated xanthones tested, α-mangostin might be responsible for the antiangiogenic activity of this Thailand stingless bee propolis.
Conclusion
These results indicate that α-mangostin isolated from Thailand stingless bee propolis may be a good candidate for future applications in the prevention of angiogenesis-related diseases.
Introduction
Propolis, a resinous substance collected by honeybees from buds and exudates of specific types of plants to protect their hives, has been used as a folk medicine in many regions since ancient times. It has been reported to possess various biological properties, including antioxidant,1,2 antibacterial,2–4 anti-inflammatory,2,5 and anticancer activities.2,6,7 For this reason, propolis is extensively used in food and beverages these days to improve health and prevent some diseases, such as inflammation, diabetes, heart disease, and cancer. 8 Over the years, many types of propolis from around the world have been investigated. In general, propolis is known to have various chemical compositions depending on the vegetation near the beehive, because the origin plants from which bees collect resinous material differ from region to region.9,10 For example, propolis from Europe, whose plant origin is poplar, is the most commonly used and the most studied among all types of propolis in the world. It contains many kinds of flavonoids and phenolic acid esters. In contrast, propolis from Brazil, usually called Brazilian green propolis, has been shown to mainly contain derivatives of cinnamic acid and to be collected from its plant origin, Baccharis dracunculifolia.2,9,10 Propolis from subtropical regions of the Pacific area, such as Okinawa (Japan) and Taiwan, has been shown to mainly contain prenylated flavonoids with the plant origin being Macaranga tanarius.11,12
Stingless honeybees are widely found in tropical and some subtropical regions around the world, such as Thailand and Indonesia.13,14 They are very small, about 5 mm in length on average, do not sting, and play an important role in plant pollination in these regions. Apis mellifera, which is popularly known as the Western honey bee or European honey bee and by far the most commonly used bee to collect honey all over the world, is known to make its hives from beeswax and only use propolis to fill the gaps. In contrast, stingless bees make their hives in a totally different way, which are almost entirely made of propolis without the normal honeycomb structure. In Thailand and India, stingless bee propolis is often applied to treat various maladies, such as acne, diabetes, and inflammation.13–15 The antioxidant, anti-inflammatory, antiacne, and antitumor activities of several species of stingless bee propolis have been reported.16–19 Chemical composition and some health-promoting properties of stingless bee honey have also been investigated in recent years.20,21 However, to the best of our knowledge, studies on this relatively unknown native Thailand stingless bee propolis are still limited. Some of the prenylated xanthones in Thailand propolis have been reported to have anticancer activity,22–24 so we isolated these compounds and examined if these compounds also had any effect on angiogenesis in vitro.
Angiogenesis refers to the formation of new blood vessels from preexisting ones. Folkman first observed in the early 1970s that angiogenesis is required for tumor growth. 25 Tumor-induced neovessels carry oxygen and nutrients to tumor tissues and function as the primary path of metastasis. Cutting off the blood supply of oxygen and nutrients to solid tumors represents a useful antiangiogenic strategy for tumors. Thus, antiangiogenic treatment has been considered to be effective in the treatment and prevention of cancer progression. 26 In this context, food factors capable of inhibiting angiogenesis might be very useful to stop the progression of small cancers at an early stage.
Angiogenic factors such as bFGF and VEGF activate the differentiation signal in endothelial cells to induce angiogenesis. Mitogen-activated protein kinase (MAPK) signal plays important roles in the regulation of angiogenesis and, among the MAPK family members, extracellular signal-regulated kinase 1/2 (ERK1/2) have been reported to have essential roles in endothelial cell proliferation, survival, migration, differentiation, and tube formation.27–29 Inactivation of this signal is known to inhibit angiogenesis. In contrast, antiangiogenic stimuli activate the stress signal and inhibit angiogenesis. For instance, p38, another member of the MAPK family, has been shown to be strongly activated by various stress stimuli such as UV light and inflammatory cytokines and functions as a stress signal. 30
Recently, we have reported the possible plant origin, the chemical composition and some biological activities of Thailand stingless bee propolis.31,32 In the present study, to further investigate if the compounds isolated from this unique propolis had any useful biological activities, we examined antiangiogenic effects of its nine major components on tube formation of Human umbilical vein endothelial cells (HUVECs), one of the well-utilized angiogenic assays in vitro.29,33 Furthermore, we examined the mechanism of their antiangiogenic effects by investigating changes in intracellular signal transduction.
Results and Discussion
Antitube-Forming Activity of Mangostins In Vitro
We first examined the effects of nine xanthone analogs with one or two prenyl groups, isolated from Thailand stingless bee propolis, on angiogenesis in vitro. Figure 1 shows the chemical structures of these prenylated xanthones. First, the content of α-mangostin in ethanol extract of Thailand stingless bee propolis (EEP) was quantified and was shown to be 58.04 ± 0.50 mg/g of EEP. Compared with other prenylated xanthones, α-mangostin was contained in EEP over six-fold more than the next abundant γ-mangostin, which we previously reported to be 9.31 ± 0.07 mg/g of EEP. 32
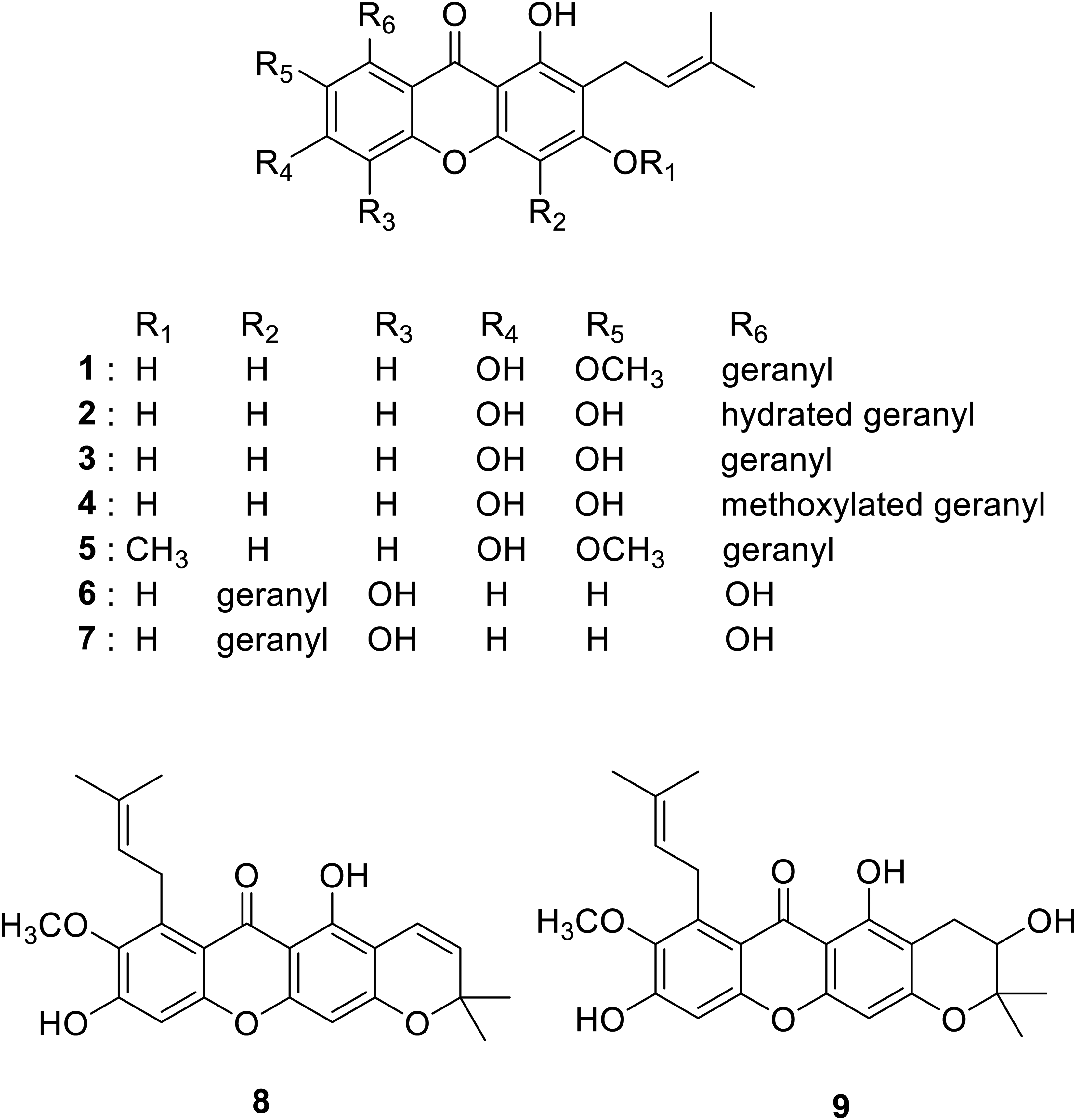
Chemical structures of nine compounds found in Thailand stingless bee propolis: α-mangostin (
In our angiogenic model, endothelial cells are induced to undergo tube formation (Figure 2). Under normal cell culture conditions, HUVECs will only undergo proliferation and will not form capillary-like tubes even if they are stimulated with angiogenic factors. However, when sandwiched between, or embedded inside, collagen gel and stimulated with proper angiogenic factors, they start to gather together and form a network of capillary-like tubes after the induction.
29
In the DMSO-treated control, properly stimulated cells migrated, gathered together, adhered to each other, and eventually elongated to form capillary-like tube network after the induction. Among the nine tested xanthones, α-mangostin (

Inhibitory effects of nine prenylated xanthones isolated from Thailand stingless bee propolis on tube formation of HUVECs. HUVECs were sandwiched between two layers of collagen gel and induced to form capillary-like tubes. The cells were treated with 3.13 or 12.5 μg/mL of these compounds and observed after 24 h. DMSO was used as a vehicle. Each experiment was repeated at least three times and representative data are shown. Arrowheads indicate cells or cell clusters that failed to form capillary-like tube network treated with α-mangostin (
The four compounds, α-mangostin (
Chemically, all of the four compounds that showed relatively strong antiangiogenic activity in this study have two prenyl groups compared to those with only one, suggesting that the number of prenyl groups may contribute to this activity. In general, compounds with prenyl groups are relatively more hydrophobic, which means that they are more likely to penetrate the cell membrane and exhibit some physiological activities inside the cells. In addition, the four compounds have also been previously reported to show antioxidant activity.36,37,38 Antiangiogenic activity of flavonoids, such as quercetin and kaempferol, has been found to correlate with their antioxidant activity.34,39 Our results suggest that antioxidant activity and angiogenesis inhibitory activity may also be correlated in xanthones.
Although gartanin (
Effects of Mangostins on Intracellular Signals
We further investigated how angiogenesis-related intracellular signal transduction was affected by these mangostins to examine their mechanism of inhibition at a molecular level. Treatment of tube-forming HUVECs with the two mangostins seemed to markedly decrease phosphorylation of ERK1/2, which is an indication of inactivation of this signal (Figure 3). On the other hand, p38 is known to function as a stress/cell death signal in HUVECs. The two mangostins seemed to increase the amount of phosphorylated p38, which is an indication of activation of this signal. Although the results indicated that changes in the level of phosphorylation of ERK1/2 and p38 were induced in a concentration-dependent manner by the two mangostins, the effects of γ-mangostin on these two signal proteins seemed to be weaker compared to those of α-mangostin.

Effects of α- and γ-mangostins on intracellular signal transduction in tube-forming HUVECs. HUVECs were embedded in collagen gel and induced to form capillary-like tubes. The cells were treated with 3.13, 6.25, or 12.5 μg/mL of these compounds as indicated. Cellular proteins were collected and changes in the phosphorylation state of ERK1/2 and p38 were analyzed by western blotting. Each experiment was repeated at least three times and representative data are shown.
In bovine retinal endothelial cells, α-mangostin was reported to inhibit angiogenesis by inducing inactivation of ERK1/2, but activation of p38 was not observed. 40 This suggests that α-mangostin may affect signaling pathways in HUVECs that are different from those in bovine retinal endothelial cells. Baccharis-derived propolis, which is mainly composed of cinnamic acid derivatives, and poplar-derived propolis, which is mainly composed of flavonoids and caffeic acid esters, have also been shown to inactivate ERK1/2 in tube-forming HUVECs.34,41 This suggests that α-mangostin may have a similar mechanism of action as these types propolis. We need to further investigate whether α-mangostin has any effects on signaling pathways in tube-forming HUVECs other than ERK1/2 and p38, such as those involved in apoptosis induction.41,42
Conclusions
In conclusion, α-mangostin was shown to be the primary active component of Thailand stingless bee propolis in terms of the amount and antiangiogenic properties. Our results suggest a possibility that angiogenesis-related diseases, such as tumor, could be prevented by daily intake of Thailand stingless bee propolis which contains α-mangostin.
Materials and Methods
Materials
Medium 199 was purchased from Sigma (St. Louis, MO, USA). Endothelial cell growth medium 2 was purchased from Promo Cell (Land Baden-Württemberg, Germany). Fetal bovine serum (FBS) was purchased from Moregate Biotech (Brisbane, Australia). Atelo Cell IPC-30 was obtained from Koken (Tokyo, Japan).
Propolis sample was obtained from an orchard in Chanthaburi, Thailand in May 2016, which we previously reported as being collected by a stingless bee species, Tetragonula pagdeni, from the fruit surface of Garcinia mangostana.
31
The prenylated xanthones used in this study were purified and identified to be α-mangostin (
Quantitative Analysis
Quantitative analysis of α-mangostin was performed using HPLC as previously reported. 32 An instrument equipped with a PU-2089 Plus quaternary gradient pump (Jasco Co., Inc., Tokyo, Japan), MD-4017 photo diode array detector (Jasco Co., Inc.), AS-4050 HPLC autosampler (Jasco Co., Inc.), and Capcell Pak C18 UG 120 column (5 μm, 4.6 × 250 mm, Osaka Soda, Osaka, Japan) was used. The mobile phases consisted of water with 0.1% trifluoroacetic acid (TFA) (A) and acetonitrile with 0.1% TFA (B). A linear gradient of 20%-100% B over 50 min followed by 100% B from 50 min to 60 min at a flow rate of 1 mL/min was applied. The injection volume was 10 μL. The calibration curve was produced for a standard compound. Analysis of EEP was conducted three times.
Cell Culture
HUVECs were grown in HUVEC growth medium (endothelial cell growth medium 2 with 0.02 mL/mL fetal calf serum, 5 mg/mL epidermal growth factor, 10 ng/mL basic fibroblast growth factor, 20 ng/mL insulin-like growth factor, 0.5 ng/mL vascular endothelial growth factor 165, 1 μg/mL ascorbic acid, 22.5 μg/mL heparin, and 0.2 μg/mL hydrocortisone; Promo Cell, Land Baden-Wüttemberg, Germany) and incubated at 37 °C under a humidified 95/5% (v/v) mixture of air and CO2. The cells were seeded on plates coated with 0.1% gelatin and allowed to grow to subconfluence before the experimental treatments.
Tube Formation Assay
Capillary tube-like structures formed by HUVECs in collagen gel were prepared as previously described with slight modifications. 43 Collagen gel was made by Atelo Cell IPC-30 (type Ι collagen). Two hundred μL of collagen solution (0.21% in Medium-199) was poured into the wells of a 24-well plate, which was then incubated at 37 °C for 30 min to solidify the gels. HUVECs in endothelial cell growth medium 2 with 10% FBS were seeded onto the collagen-coated wells and left at 37 °C in a 5% CO2 incubator for 1 h to attach to the collagen gel. After removal of the medium, 150 μL of the collagen solution was overlaid on the wells, and gelation was performed once more as described above. Next, 650 μL of endothelial cell growth medium 2 with 10% FBS supplemented with 8 nM phorbol 12-myristate 13-acetate (PMA), together with various concentrations of the samples, was added to the wells and incubated for up to 48 h. The resulting web-like capillary structures were viewed under a microscope, and images were captured using an Olympus E-620 digital camera (Olympus, Tokyo, Japan). For Western blot analysis, cells were suspended three-dimensionally in collagen gel (instead of being sandwiched between 2 layers of collagen gel).
Western Blots
After experimental treatment with various doses of samples for 3-6 h, cells embedded in collagen gel were treated with SDS sample buffer (50 mM Tris-HCl (pH 6.8), 2% SDS, 5.88% 2-mercaptoethanol, 10% glycerin, 1 mM β-glycerophosphate, 2.5 mM sodium pyrophosphate, 1 × phosphatase inhibitor cocktail 1, 1 × phosphatase inhibitor cocktail 2, and 1 × protease inhibitor cocktail 1) and boiled for 10 min. Each sample was electrophoresed in 6%-12% SDS-polyacrylamide gels and then transferred to a Hybond-ECL nitrocellulose membrane (GE Healthcare, Buckinghamshire, UK). Immunoreactive protein bands were detected by chemiluminescence (ECL Plus Western Blot Detection System, GE Healthcare).
Footnotes
Data Availability Statement
The data presented in this study are available upon reasonable request from the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable for this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Japan Society for the Promotion of Science (grant number KAKENHI 18KK0165).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
