Abstract
Objective
To introduce a nitrogen atom into the structure of the cacalol acetate and to evaluate the change in its ability to inhibit enzymes of therapeutic importance such as α-glucosidase and acetylcholinesterase (AChE), which are targeted in the development of drugs for the treatment of diabetes and Alzheimer’s disease (AD), respectively.
Methods
Cacalol acetate was obtained by subjecting the hexane and ethyl acetate extracts from the roots of Psacalium decompositum to acetylation conditions. Subsequently, the cacalol acetate was brought under nitration conditions using cerium (IV) ammonium nitrate and acetic anhydride to obtain 2-nitrocacalol acetate; this compound was characterized by nuclear magnetic resonance, mass spectrometry, and X-ray diffraction techniques. Additionally, the inhibitory activity of the ethyl acetate extract, the cacalol acetate, and the 2-nitrocacalol acetate was evaluated on the enzymes α-glucosidase and AChE. Finally, the possible mode of ligand–enzyme binding through molecular docking was established.
Results
The 2-nitrocacalol acetate was obtained with a 20% yield. Its crystallographic structure was obtained, to our knowledge, for the first time, corroborating the S configuration in C5, showing a mostly planar molecule, in which the C6, C7, and C14 atoms and the acetyl group leaving the plane. The ethyl acetate extract was that which significantly inhibited the α-glucosidase (95.8% at 100 μg/mL), while the 2-nitrocacalol acetate exhibited the highest inhibitory activity of the AChE (69.9% at 10 µg/mL). The 2-nitrocacalol acetate presented a favorable interaction energy with the AChE (−8.68 kcal/mol), highlighting the formation of two hydrogen bonds and hydrophobic interactions.
Conclusions
The 2-nitrocacalol acetate and its crystallographic structure were obtained, to our knowledge, for the first time providing a new modification to this sesquiterpene by introducing a nitro group, which was favored its ability to inhibit AChE. These structural modifications encourage future research in these sesquiterpenes as targets for the development of novel drugs that would be useful in clinical practice.
Introduction
Medicinal plants are considered an important source of natural products or secondary metabolites with unique structural and bioactive characteristics. Many enzyme inhibitors derive from secondary metabolites or structural modifications of them. 1
Psacalium decompositum (A. Gray) H. Rob & Brettel, belongs to Compositae plant family
2
and is a perennial plant found in the pine-oak forests of the Sierra Madre Occidental Mountain range in northeastern Mexico. The root, in the form of a tea or an herbal infusion, is employed against malaria, fever, diabetes, tumors, ulcers, rheumatism, kidney diseases, skin infections, and toothache.
3
The secondary metabolites that had been identified in the root of P. decompositum include cacalol, cacalone, maturin, maturinone, and maturone, which are the most abundant sesquiterpenes in the root.
4
Cacalol (
In the discovery of novel compounds with biological activity, natural products are a major source of structures, which through modifications in their functional groups or the introduction of new substituents, new molecules with different chemical and biological properties are generated. 16 The chemical and biological importance of some sesquiterpenes such as cacalol, could be enhanced by the exploration of new modifications around this structure, such as introducing a nitrogen atom through a nitro group. This functional group is unique because it could generate electron density deficient sites that allow the interaction with biological targets. In addition, the introduction of a nitro group provides the possibility of proposing other chemical modifications regarding this functional group. 17 In a previous work, the nitration of cacalol was reported with a mixture of nitrogen-oxide gases generated by means of the oxidation of tetrahydrofuran with nitric acid. The 2-nitrocacalol acetate was obtained with low yield. 18
The aim of this work was to introduce the nitro group to the cacalol acetate molecule and to evaluate the change in the inhibitory potential of enzymes that are therapeutic targets in the development of drugs for the treatment of diabetes and AD, such as α-glucosidase and acetylcholinesterase (AChE), respectively.
Results and Discussion
Chemistry
Cacalol (

Cacalol (
Cacalol acetate (
Once that the compound (
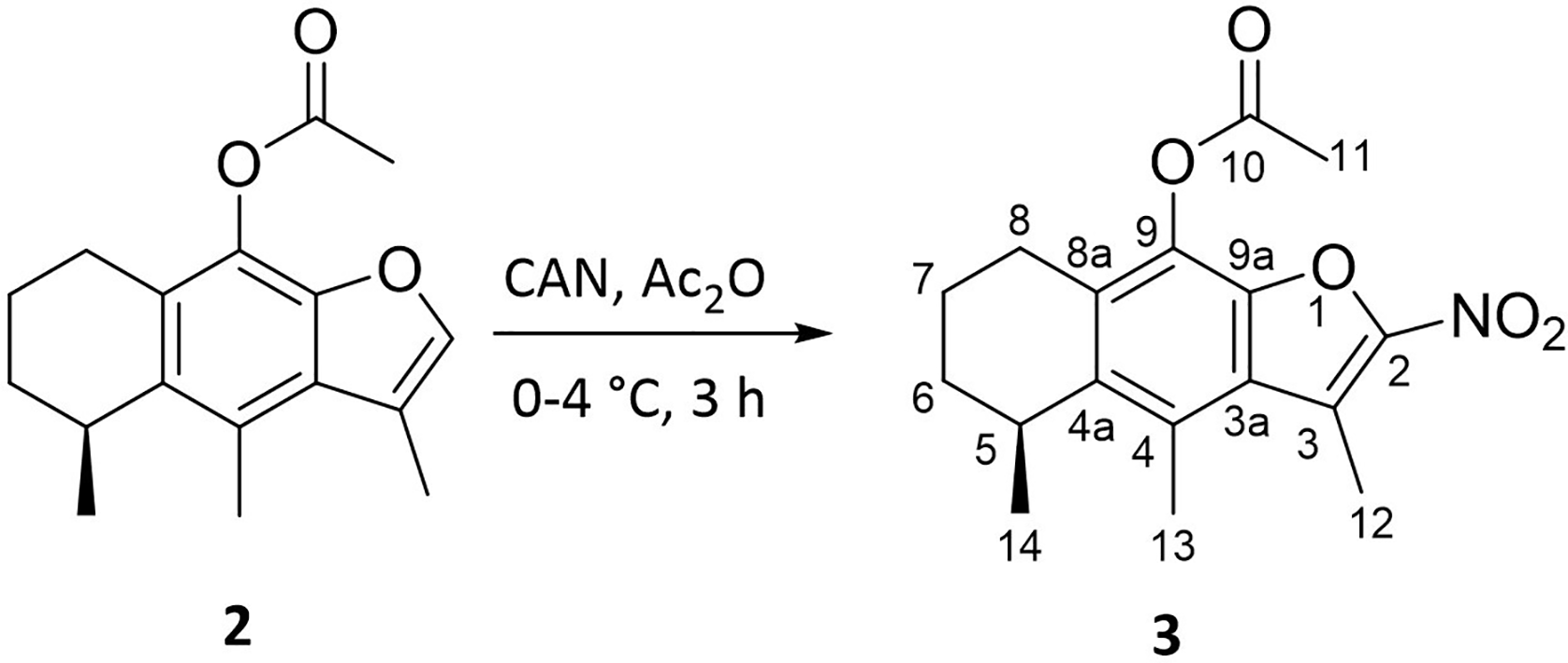
Reaction conditions to obtain 2-nitrocacalol acetate (
CAN has been used for the nitration of coumarins, 21 hydroxy aromatics, 22 and aromatics compounds 23 with success and under mild reaction conditions. In this case, nitration was achieved at position 2 of furan; the low yield can be attributed to the formation of other oxidation products and the degradation of the cacalol molecule.
The mechanism of the nitration reaction is by means of an electrophilic aromatic substitution where the benzofuran is activated by the ester group, which allows directing the electronic density in position 2 of the furan and adds it to a nitrate group of the CAN. Ganguly et al explain this mechanism in the nitration of hydroxycoumarins. 21
The 2-nitrocacalol acetate has been previously reported 18 ; however, we provide the spectroscopic details (Supplemental material). In the 1H NMR spectrum, a displaced doublet was observed at 1.19 ppm corresponding to the C14 methyl hydrogens; at 1.82 ppm there is a multiplet that integrates for four hydrogens attached to the C6 and C7 carbons. The C11, C13, and C12 methyl hydrogens displayed singlets shifted to 2.43, 2.63, and 2.90 ppm, respectively. At 3.29 ppm, a multiplet corresponding to C5 methine hydrogen was observed, and at 2.86 and 2.57 ppm, two multiplets corresponding with hydrogens attached to C8. Finally, the singlet corresponding to the furan hydrogen is absent, due to substitution by the nitro group.
The mass spectrum shows an m/z of 318 for the molecular ion [M+H]+, which corresponding to that expected for compound
Additionally, the structure of compound
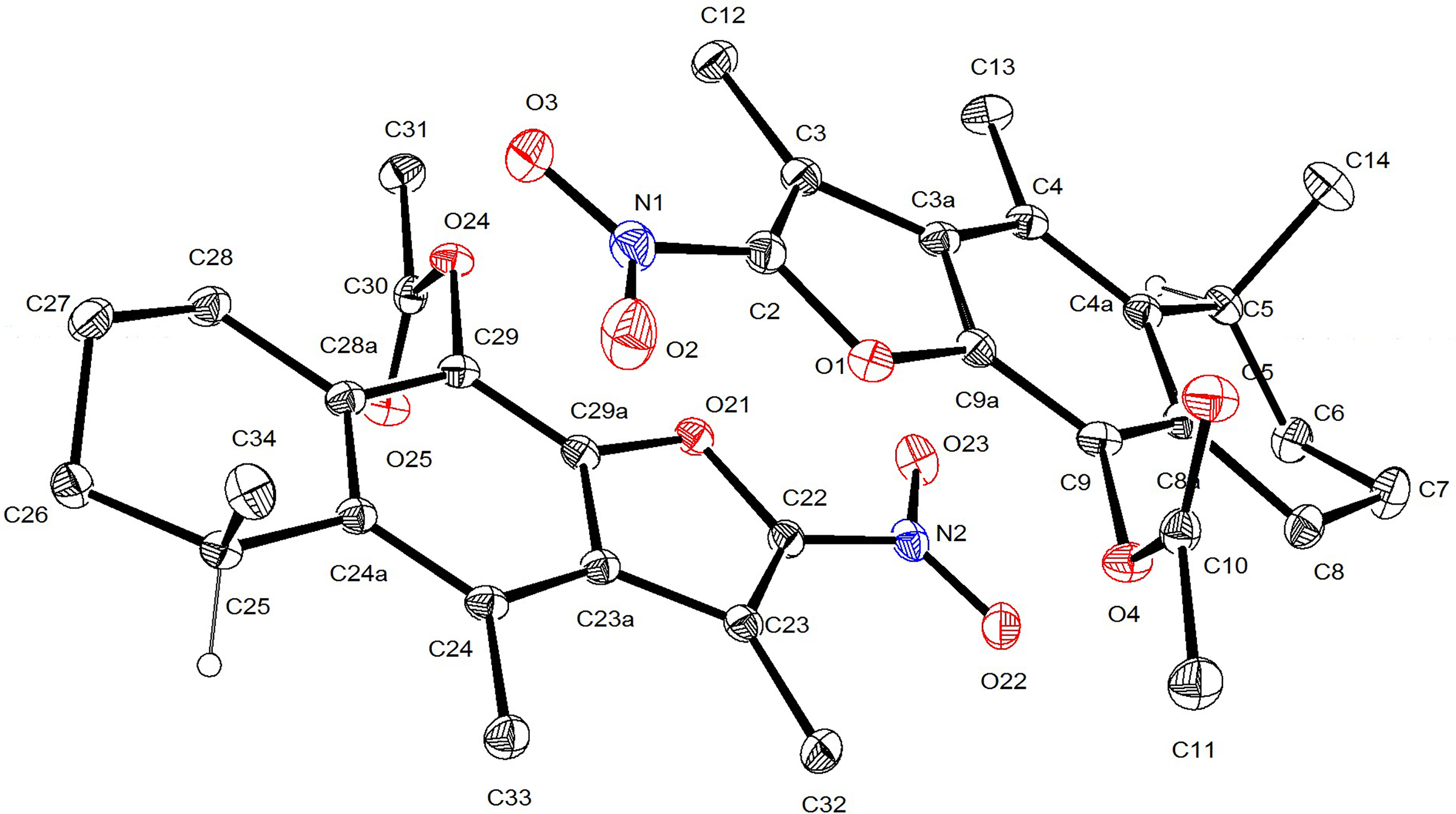
Crystallographic structure of 2-nitrocacalol acetate.
Biological Activity
Inhibition of α-Glucosidase
The α-glucosidase participates in the conversion of oligosaccharides and disaccharides into monosaccharides; therefore, its inhibition slows the digestion of carbohydrates and maintains the postprandial blood-glucose levels. 24 Preliminary tests of the inhibitory activity of the ethyl-acetate extract from the roots of P. decompositum and the derivatives cacalol acetate, and cacalol 2-nitroacetate were conducted in order to observe some change in their activity due to the structural modification.
Figure 4 shows that the ethyl-acetate extract of P. decompositum possesses outstanding inhibitory activity of the α-glucosidase at a concentration of 100 μg/mL, while the cacalol acetate and 2-nitrocacalol did not exhibit significant activity at the concentrations tested.
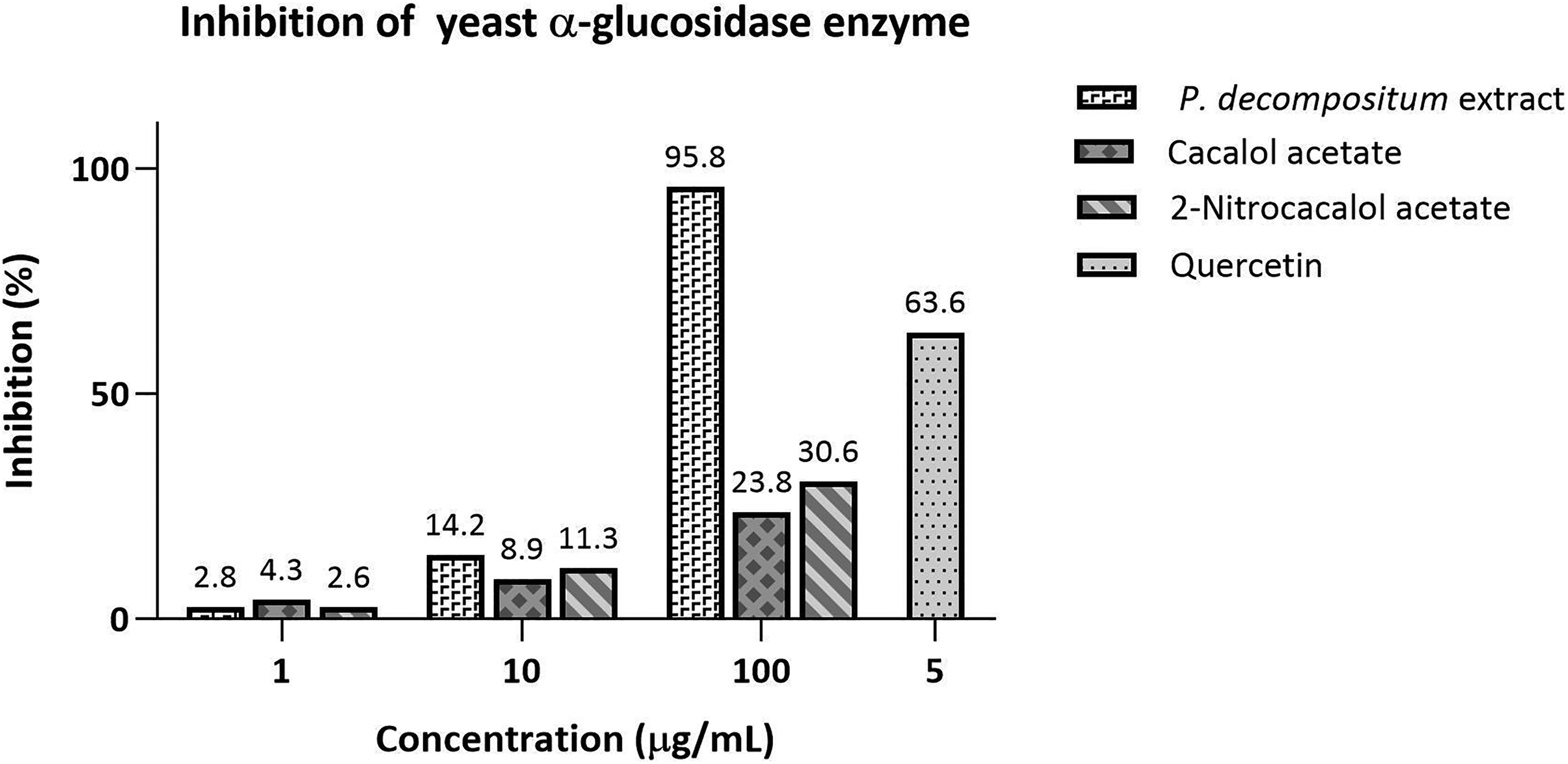
Inhibitory activity of the yeast α-glucosidase.
Previous investigations have reported the hypoglycemic effect on mice of the cacalol, the cacalol acetate, and the aqueous extract of the root, where the latter being that with the highest activity.4,25 These results corroborate that the presence of other components in the root extract is responsible for the inhibitory activity of the α-glucosidase and that the introduction of the nitro group in the C2 position of the cacalol acetate does not improve its ability to inhibit this enzyme.
Inhibition of AChE
Some natural products or plant extracts have been an important source in the search for AChE inhibitors,26,27 in that this is considered a target in the treatment of AD. By inhibiting this enzyme, the levels of the neurotransmitter acetylcholine are increased, causing an improvement in the cognitive abilities of patients with AD. 28
Figure 5 depicts the inhibition results of the AChE of the ethyl acetate extract of P. decompositum, the cacalol acetate, and the cacalol 2-nitroacetate at concentrations of 1, 10, and 100 μg/mL. Here, the ethyl acetate extract and the cacalol acetate exhibit a similar inhibitory activity of the enzyme in all concentrations; however, the 2-nitrocacalol acetate compound exerts greater inhibitory activity than the ethyl-acetate extract and the cacalol acetate at concentrations of 10 and 100 μg/mL. Therefore, the introduction of the nitro group into C2 improved its ability to inhibit this enzyme.

Inhibitory activity of the aChE.
Some natural products that are considered leaders in AChE inhibition are alkaloids, coumarins, flavonoids, and stilbenes. 29 Some terpenes, such as pentylcurcumen 30 and sesquiterpenes,31,32 have demonstrated the ability to inhibit the AChE, providing a guideline for structural modification in this type of natural products, in order to improve their inhibitory activity.
Molecular Docking
Concerning the potential of the 2-nitrocacalol acetate as an inhibitor of the AChE, a blind molecular docking was performed in Autodock 4.2.6, utilizing the crystallographic structure of the human AChE in a complex with dihydrotanshinone I, coded in the database of 4M0E proteins. Validation of the method was conducted with the co-crystallized ligand, and the energy obtained was −10.01 kcal/mol and an acceptable RMSD of 3.978 Å.
The energy affinity between the AChE and 2-nitrocacalol acetate was −8.68 kcal/mol and Figure 6a depicts this interaction, which occurred at the binding site of the co-crystallized ligand, dihydrotanshinone I, that is identified as a site peripheral to the catalytic site. 33 The interaction between the AChE and the acetate compound 2-nitro cacalol was established through two hydrogen bonds between the carbonyl of the acetate group and the amide group of the amino acid Arg296, as well as between the oxygen of the nitro group and the group amide of the amino acid Ser293. Other important interactions are hydrophic with the amino acids Tyr72, Asp74, Tyr124, Trp286, Phe295, Phe297, and Tyr341 (Figure 6b).

The (a) dihydrotanshinone I (green) and the 2-nitrocacalol acetate (blue) interaction in human AChE peripheral site; (b) hydrogen bonds and the hydrophobic contacts of 2-nitrocacalol acetate in the pocket.
Ali et al found β-Sitosterol-3-O-β-
Materials and Methods
General Experimental Procedures
Refer to Supplemental Materials.
Root Material
The roots of P. decompositum were acquired at the Pasaje Comercial de Plantas Medicinales in the Historic Center of Mexico City.
Extraction and Isolation
Refer to Supplemental Materials.
Cacalol acetate (
2-Nitrocacalol acetate (
The Determination of the Structure of Compound 2 by X-Ray Diffraction
Crystallographic data for compound
Crystal data: The crystal system is monoclinic, with space group C2, and accurate lattice constants of a = 37.2850 (12) Å, b = 7.8528 (2) Å, c = 10.8026 (3) Å, α = 90°, β = 97.437 (2)°, γ = 90°; crystal size (mm) 0.411 × 0.305 × 0.234 mm3; volume 3136.30(16) Å3; density (calcd) 1.344 Mg/m3; absorption coefficient 0.825 mm−1; F(000) = 1344. Theta range for data collection 2.390 to 68.279°. Reflections collected 19535; completeness to theta 67.679° 99.8%. Refinement method full-matrix least-squares on F2 full-matrix least squares; final R indices [I > 2sigma(I)] R1 = 0.0365, wR2 = 0.0822; largest diff. peak and hole 0.168 and −0.150 e.Å−3
The absolute configuration was determined on the basis of the Flack parameter 0.10(6).
Inhibition of the Yeast α-Glucosidase
Inhibition of the AChE
Molecular Docking
Refer to Supplemental Materials.
Conclusions
We established the conditions for the nitration reaction of the sesquiterpene cacalol acetate to obtain the product 2-nitrocacalol acetate. Its crystallographic structure, to our knowledge, for the first time, provides a new modification in this sesquiterpene by introducing a nitro group. The presence of the nitro group in the cacalol acetate produced an improvement in inhibiting the AChE and its interacting favorably with the site peripheral to the catalytic site. These results open the possibility of making this kind of modifications to other sesquiterpenes present in P. decompositum and evaluating in other biological activities, such as anti-inflammatory.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231188846 - Supplemental material for Preparation of 2-Nitrocacalol Acetate From a Sesquiterpene Isolated From Roots of Psacalium decompositum. Evaluation of α-Glucosidase and Acetylcholinesterase Inhibition
Supplemental material, sj-docx-1-npx-10.1177_1934578X231188846 for Preparation of 2-Nitrocacalol Acetate From a Sesquiterpene Isolated From Roots of Psacalium decompositum. Evaluation of α-Glucosidase and Acetylcholinesterase Inhibition by Manuel Jiménez-Estrada, Rosario Tavera-Hernández, J. Javier Alvarado-Sansininea and Maira Huerta-Reyes in Natural Product Communications
Footnotes
Acknowledgments
R. Tavera-Hernández received a fellowship from UNAM-PAPIIT, B221296. The authors are grateful to Instituto de Química, UNAM; Sac Nicté Flores Fabián, Antonio Nieto Camacho, Simón Hernández Ortega, María Teresa Ramírez, Beatriz Quiroz García, Elizabeth Huerta Salazar, María de los Ángeles Peña González, and Francisco Javier Pérez Flores for their technical assistance.
Authors’ Contributions
M. Jiménez-Estrada did research on the idea and writing. R. Tavera-Hernández performed reaction preparation, structure elucidation, molecular docking and writing. M. Huerta-Reyes carried out reviewing and editing. J. J. Alvarado-Sansininea did extract preparation and purification of cacalol acetate.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Dirección General de Asuntos del Personal Académico, Universidad Nacional Autónoma de México (grant number BG200321).
ORCID iDs
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
