Abstract
Background
Black raspberry (Rubus occidentalis L.) has various bioactive properties, including antioxidant, anti-inflammatory, and anticancer activities. Recently, it has been proposed that the pharmacological efficacy of black raspberry varies with its maturation stage. In the current study, we explored the relationship between maturation stage and anticancer activity in gastric cancer cell lines.
Materials and Methods
Black raspberry extracts were prepared from both ripe samples 41 days after the fruit set and unripe samples 22 days after the fruit set. Their anticancer activities were compared by assessing the effects on proliferation, apoptosis, invasiveness, and clonogenicity in gastric cancer cell lines.
Results
Both the ripe and unripe black raspberry samples were effective in inhibiting cell proliferation, invasion, and clonogenic activity. However, the unripe black raspberry extract showed a stronger effect on these activities compared to ripe black raspberry extract. Additionally, the unripe fruit extract increased apoptotic body formation, caspase-3 activity, Bak expression and decreased Bcl2 expression. Furthermore, the unripe black raspberry sample was found to contain higher contents of ellagic acid and gallic acid, and had a stronger suppressive effect on p-ERK and p-AKT protein levels compared to the ripe black raspberry sample.
Conclusion
Unripe black raspberry has greater anti-cancer effects than fully ripe black raspberry in gastric cancer cells.
Introduction
Gastric cancer (GC) is a formidable global health challenge and a leading cause of cancer-related mortality, responsible for over 1 million deaths every year across the world, with varying incidence rates geographically. 1 Despite advances in the diagnosis and treatment of GC, it often remains undetected until advanced stages and even after complete resection, it frequently recurs. Thus, effective chemotherapy is indispensable for managing unresectable cases and preventing disease recurrence.
Natural products have recently gained attention as a potential source of novel cancer treatments due to their selective induction of cell death in cancer cells with minimal damage to normal cells. 2 Among natural products, black raspberries, Rubus occidentalis, a member of the Rosaceae family, are particularly noteworthy as they are rich in flavonoids and polyphenols that have demonstrated diverse bioactive properties including antioxidant, anti-inflammatory, and potential cancer inhibitory activities.3,4 Among these properties, its potential role in the prevention and treatment of cancer, as well as its potential as an adjuvant to enhance chemotherapeutic activity, is of considerable interest.
Previous studies have shown that black raspberry powder inhibits tumorigenesis induced by various carcinogens in animal models of esophageal, colonic, and oral cancers.5–7 Similarly, black raspberry preparations and extracts have been found to decrease proliferation and induce apoptosis in cervical cancer cells and highly tumorigenic esophageal cells.8,9 Furthermore, dietary intake of black raspberry has been shown to suppress mammary carcinogenesis in animal models10,11 and inhibit multiple stages of gastrointestinal cancer in preclinical and clinical studies.12,13
The anticancer activity of black raspberry is primarily due to its high concentration of active compounds, including ellagic acid.8,14 Multiple studies have reported that black raspberry contains ellagic acid levels approximately 35 to 300 times higher than blueberries, 4 to 8 times higher than strawberries, and 2 to 4 times higher than raspberries. Ellagic acid, which is a key active component of black raspberry, has demonstrated anticancer effects in various cancer cell types, including GC cells, and lung cancer cells, oral cancer, and pancreatic cancer in both in vitro and vivo studies.15–19 Additionally, black raspberry contains a higher concentration of total phenolics than other berry fruits such as blueberries, strawberries, blackberries, and raspberries.20–23
However, it is important to note that the composition and content of polyphenols in black raspberry may exhibit variability based on the level of maturity of the fruit. Hence, the pharmacological activities may differ depending on the ripeness of the black raspberry fruit. 24 Furthermore, previous investigations have revealed that unripe black raspberries have an elevated concentration of bioactive compounds such as flavonoids and phenolic acids, which possess superior antioxidant capabilities. Nevertheless, there has been a limited exploration of whether the degree of fruit maturity in black raspberry is linked to the anticancer properties exhibited in GC. Thus, we conducted a comparative analysis of the anticancer effects of ripe black raspberry fruits (gathered on the 41st day following fruit set) and unripe black raspberry fruits (collected on the 22nd day following fruit set) on cellular proliferation, apoptosis, clonogenicity, and invasion in GC cells.
Results
Effects of Ripe and Unripe Black Raspberry Fruit Extracts on Cancer Cell Growth and Viability
To evaluate the potential growth inhibitory effect of ripe and unripe black raspberry fruits on cancer cells, corresponding extracts were prepared. The fully ripe black raspberry fruits harvested on the 41st day exhibiting a deep red color and reaching the peak of sweetness. The unripe fruits, on the other hand, were collected on the 22nd day, corresponding to a stage where the fruits had maximized in size, and maintained a high quantity of active ingredients, but had not yet changed color to red, as reported in a previous study. 24
To assess the growth inhibitory effect of the extracts, AGS and SNU601 GC cells were exposed to either ripe or unripe black raspberry extracts at concentrations ranging from 50 to 200 μg/mL for 1 to 3 days. Cell growth rate was assessed by counting cell numbers, as absorbance measurements could have been affected by fruit color. In AGS cells, treatment with ripe black raspberry concentrations of 50, 100, and 200 μg/mL for 3 days resulted in a reduction of proliferation by 7.8%, 20.6%, and 33.3%, respectively. In contrast, treatment with unripe black raspberry at concentrations of 50, 100, and 200 μg/mL for 3 days resulted in a greater reduction of cell growth of 25.5%, 44.1%, and 58.8%, respectively. Similarly, in SNU601 cells, treatment with ripe and unripe black raspberry extracts showed growth inhibitory effects of 2.3%, 11.6%, and 25.6%; and 13.9%, 20.5%, and 59.4%, respectively after 3 days of exposure (Figure 1).

Effect of ripe and unripe BR extract on GC cell growth. AGS (A–C) and SNU601 (D–F) cells were treated with 50, 100, and 200 μg/mL of either ripe or unripe BR extracts for the indicated durations, then cell numbers were counted to assess cell proliferation rate. Abbreviations: GC, gastric cancer; BR, black raspberry.
These results indicated that both ripe and unripe black raspberry extracts had a growth inhibitory effect on AGS and SNU601 cells, with unripe black raspberry extracts demonstrating a greater inhibitory effect in both cell lines.
Effects of Ripe and Unripe Extracts on Induction of Apoptosis
To uncover the molecular mechanisms underlying the inhibitory role of unripe black raspberry in cancer cell growth, the induction of apoptosis was evaluated by assessing the proportion of nuclear condensation or nuclear cleavage after Hoechst 33342 staining. The results, depicted in Figure 2, indicated that treatment with unripe extracts led to the formation of apoptotic bodies in both cancer cell lines at concentrations of 100 and 200 μg/mL. In contrast, ripe extracts did not induce the formation of apoptotic bodies.
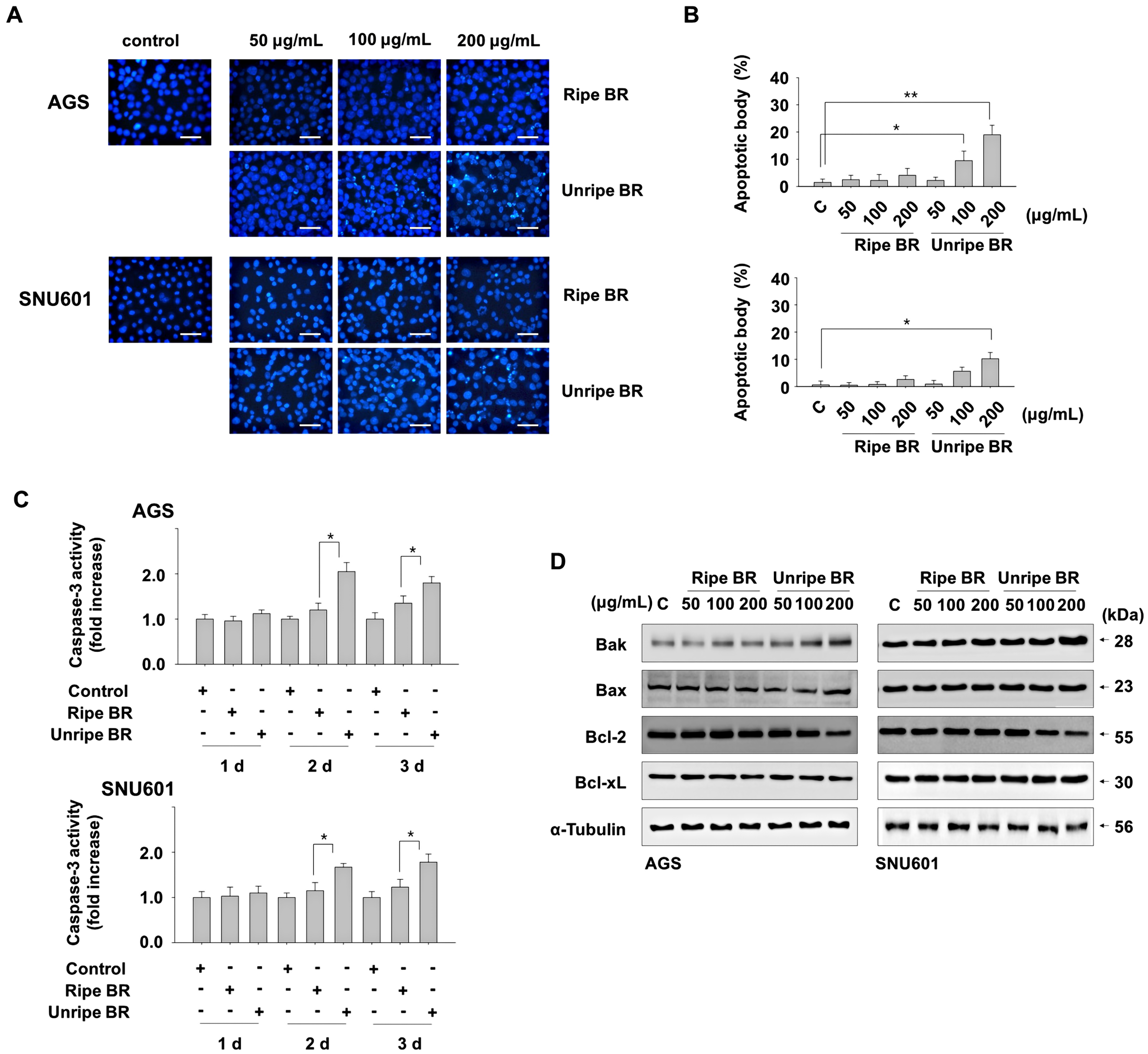
Unripe BR induces apoptosis in GC cells. (A, B) Cells were exposed to indicated concentrations of either ripe or unripe BR extracts for 2 days, and apoptotic bodies were detected after staining with Hoechst 33342. The images were taken under a fluorescence microscope (A) and apoptotic bodies expressed as a percentage of the total counted cell numbers (B). (C) Cells exposed to 200 μg/mL of ripe or unripe BR extracts for the indicated durations were harvested and total protein lysates were subjected to a caspase-3 activity assay. (D) Cells exposed to the indicated concentrations of either ripe or unripe BR extracts for 2 days were analyzed by immunoblotting. *P < .05, **P < .01. Scale bar = 100 μm. Abbreviations: GC, gastric cancer; BR, black raspberry.
Consistent with this result, higher caspase-3 activity was observed in cells treated with unripe extract compared to ripe extract. When cells were treated with 200 μg/mL of ripe and unripe extract for 1 to 3 days, ripe extract treatment did not affect caspase-3 activity, but unripe extract significantly increased caspase-3 activity after 2 and 3 days of treatment in both cell lines (Figure 2C).
We also analyzed the expression levels of the pro-apoptotic Bcl-2 family members, Bax and Bak, as well as the anti-apoptotic Bcl-2 and Bcl-xL proteins. Exposure to unripe black raspberry extract led to a reduction in the expression of the antiapoptotic Bcl-2 protein and an increase in the proapoptotic Bak protein. In contrast, the ripe extract had little effect on these protein levels. Expression of other pro-apoptotic Bcl-2 family members Bax and antiapoptotic Bcl-xL were not altered in response to either ripe or unripe extracts (Figure 2D). Based on these results, treatment with unripe black raspberry extract at a concentration of 200 μg/mL may induce apoptosis through the upregulation of Bak and downregulation of Bcl-2 protein levels in these cells.
Effects of Ripe and Unripe Extracts on Metastatic Behaviors
Next, we examined the effects of ripe and unripe black raspberry extracts on the metastatic behavior of cancer cells, specifically with regard to cell invasion and colony formation. Invasion of cancer cells is a crucial step in cancer metastasis, and, therefore, we utilized a Matrigel infiltration assay system to assess cancer cell invasiveness. Our results demonstrated that both ripe and unripe extracts reduced the infiltration of AGS and SNU601 cells when compared to the untreated control group. However, the unripe extract-treated group exhibited a more pronounced inhibitory effect on cell invasion than the ripe extract-treated group. (Figure 3A and B).
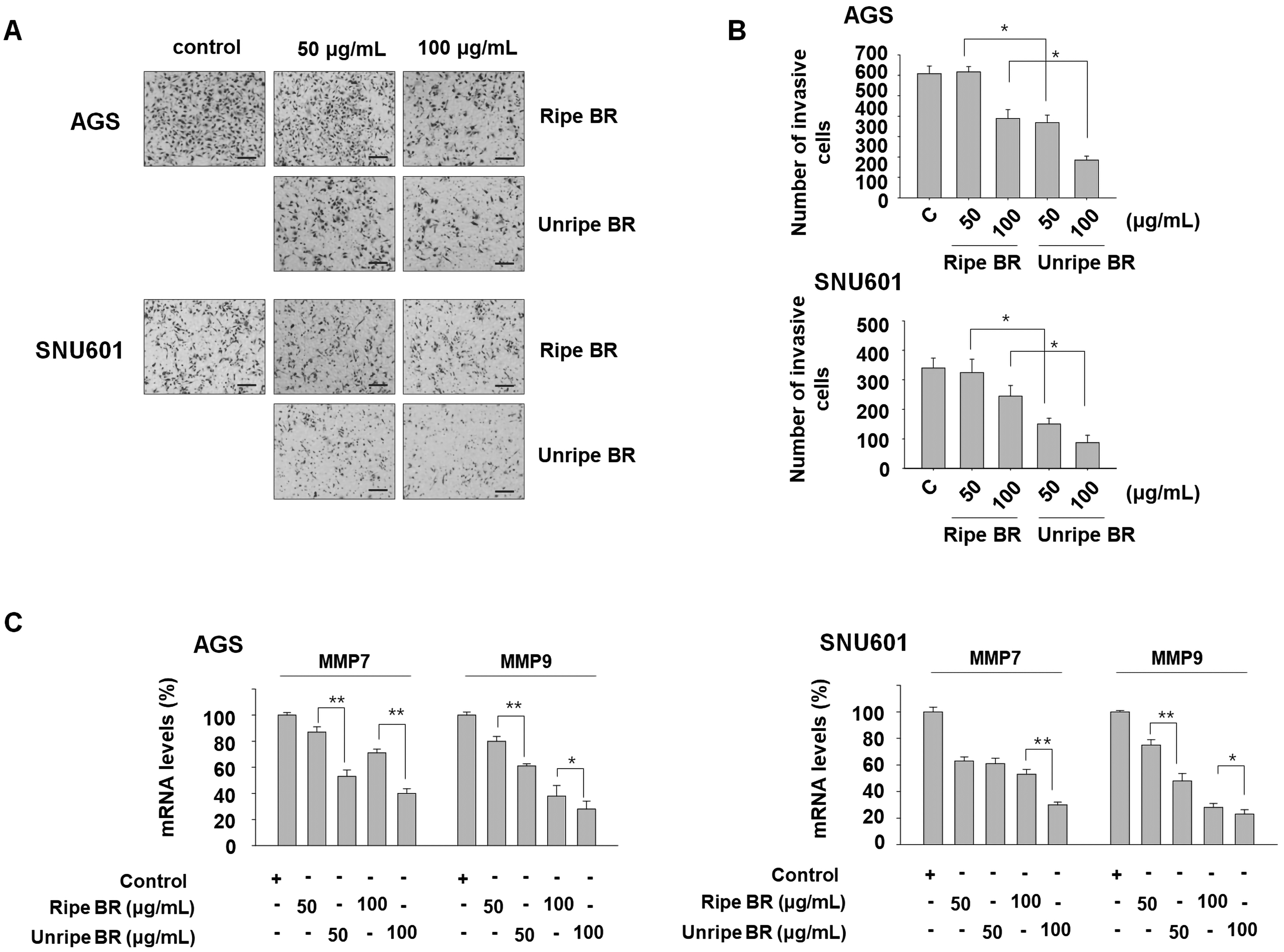
Anti-invasive activity of ripe and unripe BR. (A, B) Cells were exposed to indicated concentrations of ripe or unripe BR extracts for 1 day and equal numbers of viable cells were used for invasion assay. The cells which invaded through the transwell membrane were observed under a microscope (A) and counted (B). (C) After exposure of cells to either ripe or unripe BR extracts for 2 days, the cells were analyzed by real-time PCR to detect the mRNA expression of the genes encoding MMP7 and MMP9. *P < .05, **P < .01. Scale bar = 200 μm. Abbreviations: MMP, matrix metalloproteinase; PCR, polymerase chain reaction; BR, black raspberry.
We also examined the expression of the matrix metalloproteinase (MMP) family members that are associated with the progression and metastasis of cancer. The findings revealed that the mRNA expression of MMP7 and MMP9 decreased significantly in the presence of both extracts, with the unripe extract demonstrating a greater inhibitory effect than the ripe extract (Figure 3C). In addition, the clonogenicity of these cancer cells was found to decrease in a concentration-dependent manner with the administration of black raspberry extract. Notably, treatment with the unripe extract resulted in a significant reduction in clonogenicity, surpassing the effect of the ripe extract (Figure 4). This result indicates that unripe black raspberry extracts may have greater potential in preventing metastatic behaviors compared to ripe extracts.

Effect of ripe and unripe BR on clonogenicity. Cells exposed to the ripe or unripe extracts for 1 day were replated on 60 mm dishes and cultured for 2 weeks. After staining the colonies with crystal violet, the images were taken (A) or colonies were counted (B). *P < .05, **P < .01.
Previously, it has been postulated that the enhanced anticancer activity of unripe berries, relative to ripe ones, may be attributable to their higher abundance of bioactive ingredients. 24 Indeed, the analysis of the content of constituent compounds using high-performance liquid chromatography (HPLC) revealed that the ripe and unripe black raspberry extracts contained 1600 and 8502 μg/g of ellagic acid, and 68 and 3624 μg/g of gallic acid, respectively (Figure 5A).

Possible involvement of ellagic acid and gallic acid in anticancer effect of RB. (A) Contents of ellagic acid and gallic acid in ripe and unripe BR were measured by HPLC. (B) Cells were exposed to the indicated concentrations of either ellagic acid or gallic acid for 1 day. Cell viability was assessed by the EZ-cytox assay. (C) Cells were exposed to the indicated concentrations of either ellagic acid or gallic acid for 1 day. Then equal numbers of viable cells were taken and used for the invasion assay. Subsequently, images of the cells that invaded through the transwell membrane were captured under a microscope. (D) Cells were treated with 10 μM EA, 10 μM GA, and ripe or unripe BR extracts for 1 day, after which total protein lysates were collected and subjected to immunoblotting analysis. **P < .01 versus polyphenol quantity in ripe BR. Scale bar = 200 μm. Abbreviation: HPLC; high performance liquid chromatography; BR, black raspberry; EA, ellagic acid; GA, gallic acid.
To ascertain the anticancer activity of these compounds on these cells, we assessed their inhibitory effect on cell invasion after incubating with 5 and 10 μM ellagic acid and 5 and 10 μM gallic acid for 1 day. As shown in Figure 5B and C, a notable suppressive effect was observed, although they did not significantly inhibit cell viability.
Additionally, we evaluated the activity levels of ERK and AKT, 2 crucial signaling modulators in cancer progression and malignancy. The results indicated that ellagic acid and gallic acid down-regulated p-ERK and p-AKT levels. Interestingly, the unripe black raspberry extract exhibited a more significant reduction in p-ERK and p-AKT levels than ripe black raspberry, as shown in Figure 5D. These results indicate that unripe black raspberries contain elevated concentrations of ellagic acid and gallic acid, and demonstrated pronounced anticancer effects in GC cells, evidenced by potentiated anti-proliferative and anti-invasive activities and induction of pro-apoptotic activity.
Discussion
Black raspberries possess chemo-preventive and anticancer activities in several types of cancer, including breast, colon, and cervical.9,10,25 Thus, black raspberry fruits have been widely processed and consumed as food products and health supplements for their potential health benefits. Black raspberries are fragrant and sweet as they ripen and are usually consumed as ripe fruits. However, previous studies have reported that immature berries are more effective in preventing inflammation and hyperlipidemia.26,27
In this study, we investigated the anticancer properties of extracts derived from ripe and unripe black raspberry fruits on AGS and SNU601 GC cell lines. We found that the inhibitory effect of the unripe raspberry extract on cell proliferation was stronger than that of the ripe extract, although both ripe and unripe raspberry extracts reduced the proliferation of these cancer cells. The unripe black raspberry extract induced apoptotic bodies, altered Bcl-2 and Bak expression, and triggered caspase-3 activation, but the same concentration of ripe black raspberries extract had no effect. Moreover, the inhibitory effect of unripe extracts on clonogenicity and invasiveness was stronger than that of the ripe extract. Therefore, it is demonstrated that unripe black raspberries have more effective anticancer activity in GC cells than ripe black raspberries.
In support of our results, extracts from unripe black raspberry showed greater antiproliferative activity than their ripe or overripe counterparts in colon cancer cells. 25 Similarly, studies using ripe black raspberry showed no anticancer effects in prostate cancer cells and rat prostate carcinogenesis models 28 ; however, studies using unripe fruits of black raspberry have shown significant inhibitory activity on the migration and invasion of prostate cancer cells by decreasing MMP expression. 29 These results are consistent with our observation that unripe black raspberry fruits have better anticancer effects.
The precise mechanism underlying the enhanced anticancer activity of unripe black raspberry, relative to ripe black raspberry, remains unclear, although it is hypothesized that this may be attributable to the greater abundance of active phenolic compounds, such as ellagic acid and gallic acid, present in unripe black raspberry. Indeed, a uniquely high content of phenolic compounds was detected in unripe berry fruits harvested 15-25 days after fruit setting. 24 The contents of almost all phenolic components investigated, except for anthocyanins, were significantly high during this period and then decreased with maturation. About 5-10 times higher levels of ellagic acid, gallic acid, kaempferol, p-coumaric acid, caffeic acid, and myricetin were detected in unripe fruit extract. 24 Similarly, our study also showed that the contents of ellagic acid and gallic acid were 5.3 and 53.2 times higher in unripe black raspberry extract than in ripe extract, respectively. Ellagic acid and gallic acid have been well-established as potent anticancer agents against various types of cancers, including GC.30,31 Ellagic acid induces apoptosis and inhibits tumor growth, angiogenesis, and metastasis in in vitro and in vivo models by affecting various genes and regulators related to cancer progression, including PI3 K/AKT, ERK, JAK2/STAT3, and Bcl-2 family members..32,33 Gallic acid also induces chemotherapeutic efficacy by modulating various signaling pathways, including EGFR/ERK, PI3 K/AKT, NF-kB, and JAK/STAT3.34,35 Consistently, our results showed that ellagic acid and gallic acid have strong inhibitory effects on the invasiveness of AGS and SNU601 GC cells. Moreover, the study revealed that unripe black raspberry extract had a more significant impact on reducing p-ERK and p-AKT levels compared to ripe black raspberry extract, indicating a stronger inhibitory effect on the activity of ERK and AKT. Additionally, treatment with either ellagic acid or gallic acid alone also reduced p-ERK and p-AKT levels. Therefore, these findings suggest that the potent anticancer effects of unripe black raspberry may be attributed to the effects of ellagic acid and gallic acid.
Besides, kaempferol, p-coumaric acid, caffeic acid, myricetin, rutin, and ferulic acid, which have been suggested as either promising anticancer candidates or effective therapeutic sensitizers, both alone and in combination with other anticancer agents, have been reported to be present in high amounts in unripe black raspberry. These compounds have been also reported to be involved in the prevention of several signal regulatory kinases including Ras/Raf/ERK, PI3 K/AKT, and TGF-β2/Smad2/3.36–38 Thus, the strong anticancer effect of unripe black raspberry may be due to the synergistic action of these high-content multiphytochemical combinations.
Although the effects of unripe black raspberry on normal cells were not observed in this study, previous reports have demonstrated that black raspberry can induce apoptosis in certain malignant cancer cells without affecting normal cell viability. 39 Specifically, the presence of ellagic acid in substantial quantities in unripe raspberries has been identified as selectively inducing apoptosis in cancer cells while leaving normal cells unaffected. 40 In relation to the inhibitory effects of unripe black raspberry and its key component, ellagic acid, on the abnormally hyper-activated AKT and ERK pathways in cancer cells, as observed in our study and other studies,32,33 the anticancer effect of unripe black raspberry appears to be selective to cancer cells. Taking these results into consideration, it is anticipated that unripe black raspberry could yield a potent anticancer effect in GC. However, further investigation through clinical studies is required to validate these findings.
Conclusion
This study demonstrates that unripe black raspberry extract exhibits significant anticancer effects in GC cells. These effects are evidenced by enhanced anti-proliferative and anti-invasive activities, as well as the induction of pro-apoptotic activity. It is noteworthy that unripe black raspberry extract contains elevated concentrations of ellagic acid and gallic acid, which may contribute to pronounced anticancer effects, partially through the downregulation of ERK and AKT activity.
Materials and Methods
Cell Culture
SNU-601 and AGS cell lines derived from human GC were purchased from the Korean Cell Line Bank and the American Type Culture Collection, respectively. Both cell lines were maintained in RPMI 1640 medium (Invitrogen) supplemented with 10% (v/v) fetal bovine serum (FBS) and 1% antibiotics at 37 °C in a 5% CO2 atmosphere.
Measurement of Cell Growth
SNU-601 and AGS cells were plated in 6-well plates at a density of 1 × 105 cells per well and incubated overnight. The cells were then treated with either ripe or unripe black raspberry extracts at concentrations of 50, 100, or 200 µg/mL in triplicate. The cells were harvested at daily intervals over a period of 3 days by rinsing, trypsinizing, and collecting by centrifugation. The harvested cells were stained with trypan blue, and the number of unstained live cells was counted. The experiment was replicated 3 times, and the outcomes were shown as the mean value accompanied by standard deviation.
Measurement of Cell Viability
The EZ-cytox viability assay was performed following the manufacturer's protocol. Briefly, cells were plated at a density of 5–8 × 104 cells/well in a 24-well plate. On the following day, they were incubated in the growth medium with or without ellagic acid and gallic acid for 1 day. The EZ-cytox solution (Daeillab) was added to the wells and incubated at 37 °C in a CO2 incubator for the last 2 h of incubation, and the plates were read using an enzyme-linked immunosorbent assay plate reader at 450 nm. The absorbance of the untreated cells was set as 100% and cell survival was expressed as a percentage of this value.
Preparation and Treatment of Ripe and Unripe Black Raspberry Extracts
The ripe and unripe black raspberry extracts used in this study were prepared and provided by the Berry & Biofood Research Institute. The black raspberry fruits used for the extracts were collected from 5 farms in Gochang province. The fruits were extracted using a reflux condenser by adding a 10-fold volume of water and heating at 100 °C for 2 h. This process was repeated twice to ensure thorough extraction. The resulting extracts were filtered to remove any particulate matter and then concentrated using a vacuum concentrator. Finally, the concentrated extracts were lyophilized using a freeze dryer (PVTFD10R; Ilshinbiobase) to obtain a powdered extract that could be stored and used for further experimentation. For treatment, lyophilized black raspberry extract was solubilized in distilled water at a final concentration of 100 mg/mL, filtered, and then treated by adding 50 to 200 μg/mL of black raspberry extract to the cells and incubating for 1 to 3 days.
Hoechst 33342 Staining
Cells were incubated with black raspberry extract obtained from either ripe or unripe berries. In the final 10 min of the incubation period, the cells were stained with Hoechst 33342 at a concentration of 1 µg/mL. Both floating and attached cells were collected through centrifugation, and the resulting pellets were washed with ice-cold phosphate-buffered saline (PBS). The cells were then fixed on ice using a 3.7% formaldehyde solution for 15 min and washed again with PBS. To prepare slides, a fraction of the suspension of cells was centrifuged using a Shandon centrifuge (Thermo Fisher Scientific), and the resulting slides were air-dried and mounted in an anti-fade solution. Subsequently, the slides were examined using a fluorescence microscope (DM5000, Leica) to identify apoptotic nuclei. A total of 500 cells, randomly distributed throughout different fields, was tallied, and the percentage of apoptotic cells was calculated by dividing the number of apoptotic cells by the total number of cells.
Caspase-3 Activity Assay
For the measurement of caspase-3 activity, a colorimetric assay kit (BioVision) was employed. To perform the assay, either treated or untreated cells were collected and lysed. Subsequently, a protein lysate of 200 μg was combined with the reaction buffer and DEVD-pNA substrate to generate a reaction mixture of 50 μL. The mixture was incubated for 90 min, and the absorbance of the reaction mixture was measured at 405 nm. The fold increase in caspase-3 activity was determined by comparing the absorbance values of the treated samples with those of the untreated control.
Immunoblotting
The treated cells were harvested, washed, and subsequently incubated with a lysis buffer composed of 50 mM HEPES, 150 mM NaCl, 1% Triton X-100, 5 mM EGTA, and a protease inhibitor cocktail to induce cell lysis. Following that, protein extracts at a concentration of 30 μM were mixed with 2X sampling buffer and subjected to boiling. The resulting samples were then separated by electrophoresis using 12% SDS-PAGE. The separated proteins were then transferred onto a nitrocellulose membrane using standard techniques. Subsequently, the membrane was blocked in TBS buffer containing 5% skim milk and probed with antibodies against Bak (06-536, EMD Millipore), Bax (BD610983, BD Biosciences), Bcl-2 (2876, Cell Signaling Technology), Bcl-xL (2762, Cell Signaling Technology), and α-tubulin (sc-5286, Santa Cruz Biotechnology), p-ERK (sc-7383, Santa Cruz Biotechnology), total ERK (sc-1647, Santa Cruz Biotechnology), p-AKT (9271, Cell Signaling Technology), and total AKT (9272, Cell Signaling Technology). The antibodies were diluted in 2% skim milk in TBS buffer and incubated overnight at 4 °C. The signals were visualized using the FUSION Solo X imaging system (Vilber).
Invasion Assay
Cell invasion was assessed using Matrigel-coated Transwell chambers (Corning Costar). After exposing the cells to black raspberry extract from ripe and unripe fruits for the indicated time period, an equal number of cells was taken and suspended in 200 μL of RPMI medium containing 1% FBS. After preparing the cell suspension, it was placed on the top of the insert, while RPMI medium containing 5% FBS was added to the lower chamber. The cells were then incubated at 37 °C in 5% CO2 for either 6 h (for AGS) or 15 h (for SNU601). Noninvasive cells were wiped off from the upper surface of the transwell membrane. Then the membrane was rinsed, fixed with 3.7% formaldehyde, and stained with a crystal violet solution. The invading cells were observed and counted under a microscope (×200, Olympus).
Clonogenic Assay
The clonogenic activity was assessed following established procedures with certain modifications. 41 Specifically, for the clonogenic assay, 2.5 × 105 cells were seeded in 35 mm dishes and treated with various concentrations of either unripe or ripe black raspberry extracts for 1 day. The cells were then trypsinized, counted, replated (2000 cells/60-mm dishes), and maintained under a humidified atmosphere at 37 °C in 5% CO2 for 14 days. Afterwards, the cells were fixed and stained with 0.5% crystal violet, and images were taken and colonies with a diameter greater than 0.7 mm were scored to determine the extent of cell proliferation.
Real-Time PCR
Real-time PCR was conducted using the Fast Start DNA Master SYBR Green I Kit (Roche) with Light Cycler 2.0 (Roche). To confirm the amplification product, PCR products were examined on a 2% agarose gel stained with ethidium bromide. The primer sequences used for β-actin were as follows: 5′-GACTATGACTTAGTTGCGTTA-3′ and 5′-GCCTTCATACATCTCAAGTTG-3′. Primers for MMP7 (P310408) and MM9 (P323207) were purchased from Bioneer (Daejeon). PCR was performed at 95 °C for 10 min, followed by 45 cycles at 95 °C for 15 s, 60 °C for 5 s, and 72 °C for 7 s. The presence of a single product was confirmed via a melt curve analysis. For each run, negative controls containing no templates were included, and the data were analyzed using the Light Cycler software (ver. 4.0; Roche). The relative gene expression was analyzed using the 2ΔΔCt method.
HPLC Analysis
The pretreated sample was analyzed for phenolic compounds using HPLC (Agilent) equipped with an autosampler/injector and ultraviolet–visible detector. An OP C18 column (5 μm, 4.6 × 250 mm) was used for separation. Both mobile phase A (0.2 M ortho-phosphoric acid, pH1.57) and B (20% 50 mM ammonium dihydrogen phosphate, pH2.6 in 80% acetonitrile) were used for elution. A gradient was employed as follows: 0–10 min, 80% A; 10–15 min, 70% A; 15–20 min, 60% A; 20–25 min, 10% A; 25–30 min, 10% A; 30–32 min, 95% A, 32–40 min, 95% A. All phenolic compounds were identified and quantified using external standards. Data scanned at 260 nm (ellagic acid) and 280 nm (gallic acid) were collected.
Statistical Analysis
The data are presented as mean ± standard deviation from 3 independent experiments. Simple comparisons were evaluated using Student's t-test, while multiple comparisons were performed using 1-way ANOVA with Tukey's test. A P-value less than .05 was considered statistically significant.
Footnotes
Acknowledgments
The authors thank Ms. Jeong Eun Choi and Ms. Sang Mi Kang for their excellent technical assistance.
Authors’ Contributions
Conceptualization was handled by S.H., T.L., and K.K.. Methodology was designed by K.K., T.L., and J.K.. Writing-review and editing were done by S.H., T.L., and K.K.. Funding acquisition was handled by S.H., T.L., and K.K. All authors have read and agreed to the final version of manuscript.
Data Availability
The data presented in this study are available upon reasonable request from the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a research fund from the Clinical Medicine Research Institute of the Chosun University Hospital (2020).
