Abstract
Background/Objective
Vitis vinifera leaves harvested from the developing canes are used in Balkan cuisines. The objective was to determine the chemical composition of the essential oil and supercritical carbon dioxide (SC CO2) extract of V. vinifera leaves harvested after the flowering of the new vine shoots.
Methods
The V. vinifera leaf oils were obtained by hydrodistillation and SC CO2 extraction. The application of different SC CO2 extraction parameters, determined by the Box–Behnken design, influenced the extraction yield and the chemical composition of the SC CO2 extracts. The optimal extraction conditions were set for the extraction yield and eucalyptol amount. The essential oil and SC CO2 extracts obtained were analyzed by gas chromatography–mass spectrometry.
Results
The most abundant compounds present in the essential oil obtained by hydrodistillation were: phytol (11.3%), α-cadinol (8.3%), trans-calamenene (8.2%), hexahydrofarnesyl acetone (7.0%), and τ-cadinol (6.6%). The extracts obtained by SC CO2 extraction consisted of twenty five compounds among which the most abundant were: phytol, squalene, eucalyptol, camphene and hexadecan-1-ol. Over the ranges of SC CO2 process parameters studied, those of pressure and extraction time had significant effect on the extraction yield and eucalyptol amount.
Conclusion
The results obtained determined the compounds present in V. vinifera leaves essential oil and SC CO2 extracts which contribute to the flavor and aroma of V. vinifera leaves.
Keywords
Introduction
Vitis vinifera L. leaves stuffed with meat, rice, and vegetables are a dish known as dolmathes. 1 V. vinifera leaves are a rich source of flavonols: myrcetin, quercentin, and kaempferol.2,3 V. vinifera leaves from ten varieties, four red and six white, were investigated in order to select the variety that can be used for consumption. 4 It was suggested that the white variety Malvasia Fina is the one of choice for culinary use. 4 Volatile compounds present in white variety Malvasia Fina and red variety Touringa Franca, after boiling under pressure, were extracted by headspace solid-phase microextraction. 5 The obtained extracts were analyzed by gas chromatography–mass spectrometry (GC–MS). The chromatograms obtained revealed the presence of eight aldehydes, four alcohols, one ketone, three norisoprenoid derivatives, eight esters, six sesquiterpenes, and two terpenes. 5 V. vinifera leaves grown in a greenhouse and in the field were extracted with acetone. 6 The acetone extracts of the leaves grown in the greenhouse contained sterols, terpenes, and free fatty acids, while the acetone extracts of leaves grown in the field contained terpenes, free and esterified fatty acids, and ketones. 6 Extraction of V. vinifera leaves with ethanol revealed the presence of phenolic compounds, mainly hydroxycinnamic acid derivatives and quercetin glycosides. 7 The extraction of V. vinifera leaves with chloroform revealed the presence of β-amyrin, lupeol, taraxerol, α-tocopherol, and β-sitosterol. 8 The compounds present in V. vinifera leaves can be used as cosmetic ingredients and their safety assessment is well defined. 9
With the introduction of supercritical solvents, extractions with organic solvents have been replaced by SC CO2 extraction. Due to the CO2 characteristics in its supercritical state, the SC CO2 extracts consist of nonpolar or slightly polar compounds. SC CO2 extraction is a technique of choice for obtaining nonpolar compounds. 10 V. vinifera leaves were extracted with SC CO2 at 30 MPa, in the temperature range from 40 to 80 °C and with CO2 modified with 5% and 10% of either ethanol or ethyl acetate. 10 The SC CO2 extract yields ranged from 1.86% to 7.52% and consisted of β-amyrin, lupeol, hexacosan-1-ol, octacosan-1-ol, triacontan-1-ol, α-tocopherol, and β-sitosterol. 10 The longer extraction time increased the abundance of lupeol and β-amyrin in the SC CO2 extracts. 10 SC CO2 extraction using the modifier showed that the major components in V. vinifera leaves SC CO2 extracts were two terpenoids, three long chain aliphatic alcohols, α-tocopherol, and one phytosterol. 10
A long list of process parameters affect SC CO2 extraction and investigated can be pressure, temperature, extraction time, particle size, CO2 flow rate, the percentage of co-solvent added, and supercritical fluid composition. 11 The most important process parameters can be obtained either from screening or from previous experiments done. The process parameters can be reduced to the important independent parameters after screening. 11 Then, a second-order model can be used to obtain a curvature near to that of the optimum. 11 Box–Behnken design (BBD) is a second-order model that can be used for optimization of SC CO2 extraction. 11
Starting from the assumption that V. vinifera var. White Riesling ssp. sativa L. leaves can be used for consumption, the aim was to determine the nonpolar or slightly polar compounds present in the leaves. The assumption was that nonpolar compounds obtained by hydrodistillation represent the flavor released during cooking dolmathes. The nonpolar and slightly polar compounds obtained by supercritical CO2 extraction represent the substances influencing the flavor and aroma that can be released during cooking V. vinifera leaves in a form of dolmathes.
The aim of the study was to analyze the chemical compounds of V. vinifera var. White Riesling leaves, obtained by hydrodistillation and SC CO2 extraction, by GC–MS, to investigate the influence of pressure, temperature and time on the total extraction yield and eucalyptol amount, and determine the optimal SC CO2 extraction parameters by BBD.
Results and Discussion
V. vinifera leaves harvested from the developing canes in early June were hydrodistilled to give a pale green essential oil with a yield of 0.9%. GC–MS analysis of the essential oil led to identification of 43 compounds (Table 1). In the essential oil determined were: C13-norisoprenoid (0.3%), monoterpenoid p-menthane (0.3%), sesquiterpenes (28.4%), oxygenated sesquiterpenes (28.4%), alkanes (7.9%), alcohol (0.9%), polycyclic aromatic hydrocarbon (0.9%), terpene ketone (2.2%), fatty acid esters (11.3%), norterpene (1.1%), diterpene (0.8%), and acyclic diterpene alcohols (11.6%). The most abundant compounds in the essential oil obtained by hydrodistillation were acyclic diterpene alcohol phytol (11.3%), oxygenated sesquiterpene α-cadinol (8.3%), sesquiterpene trans-calamenene (8.2%), long-chain fatty acid ester ethyl hexadecanoate (7.3%), oxygenated sesquiterpene hexahydrofarnesyl acetone (7.0%), and oxygenated sesquiterpene τ-cadinol (6.6%). The only norterpene present was vitispirane (0.3%), indicating that there are stronger potential contributors to flavor notes of V. vinifera leaves. The sesquiterpenes which are potential contributors to the spicy flavor of V. vinifera leaves were α-muurolene (4.6%), γ-cadinene (4.0%), γ-himachalene (3.4%), α-cadinene (2.2%), α-selinene (1.5%), δ-selinene (1.3%), and β-selinene (1.0%). The sesquiterpenoids are associated with balsamic flavor notes. The potential oxygenated sesquiterpenes contributing to the flavor of V. vinifera leaves are α-cadinol (8.3%), τ-cadinol (6.6%), and α-muurolol (2.8%). Phytol enriches the complex V. vinifera flavor with a mild grassy type scent. Isophytol exhibits similar flavor nuances of grassy type scent, but its presence in V. vinifera leaves essential oil was only 0.3%. The long-chain fatty acid esters flavor can be characterized as bitter, acidic, sour bitter or sharp contributing to the characteristic flavor attributes of V. vinifera leaves essential oil flavor.
Chemical Composition of V. vinifera Leaves Essential Oil Obtained by Hydrodistillation.

RI exp—Retention indices obtained experimentally.
The SC CO2 extraction was done according to the BBD. The extraction pressure (X1), temperature (X2) and extraction time (X3) were independent variables investigated to optimize the extraction process in terms of obtaining the highest extraction yield and eucalyptol amount. The investigated process parameters and levels tested are depicted in Table 2.
Coded and Real Levels of Independent Process Parameters for the BBD.
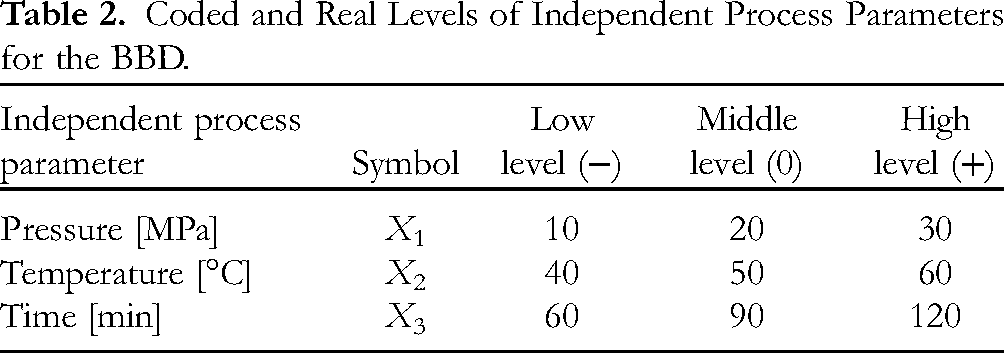
Fifteen experiments were carried out to investigate the influence of experimental process parameters on the extraction yield. The extraction yield was the lowest at a pressure of 20 MPa, temperature of 40 °C and an extraction time of 60 min. The highest extraction yield was at the pressure of 30 MPa, temperature of 60 °C and an extraction time of 90 min. The results depicted in Table 3 indicate that the extraction yield varied significantly on the applied process parameters. The influence of pressure (X1), temperature (X2) and extraction time (X3) on the extraction yield, Y is depicted in Table 3.
The BBD for V. vinifera Extraction Yield and Results Obtained.

Analysis of variance (ANOVA) was used to determine which process parameters and which interactions have a significant influence on the process. The influence of linear, quadratic, and interaction coefficients on the investigated responses are depicted in Table 4.
The ANOVA of the Responses for the Extraction Yield.
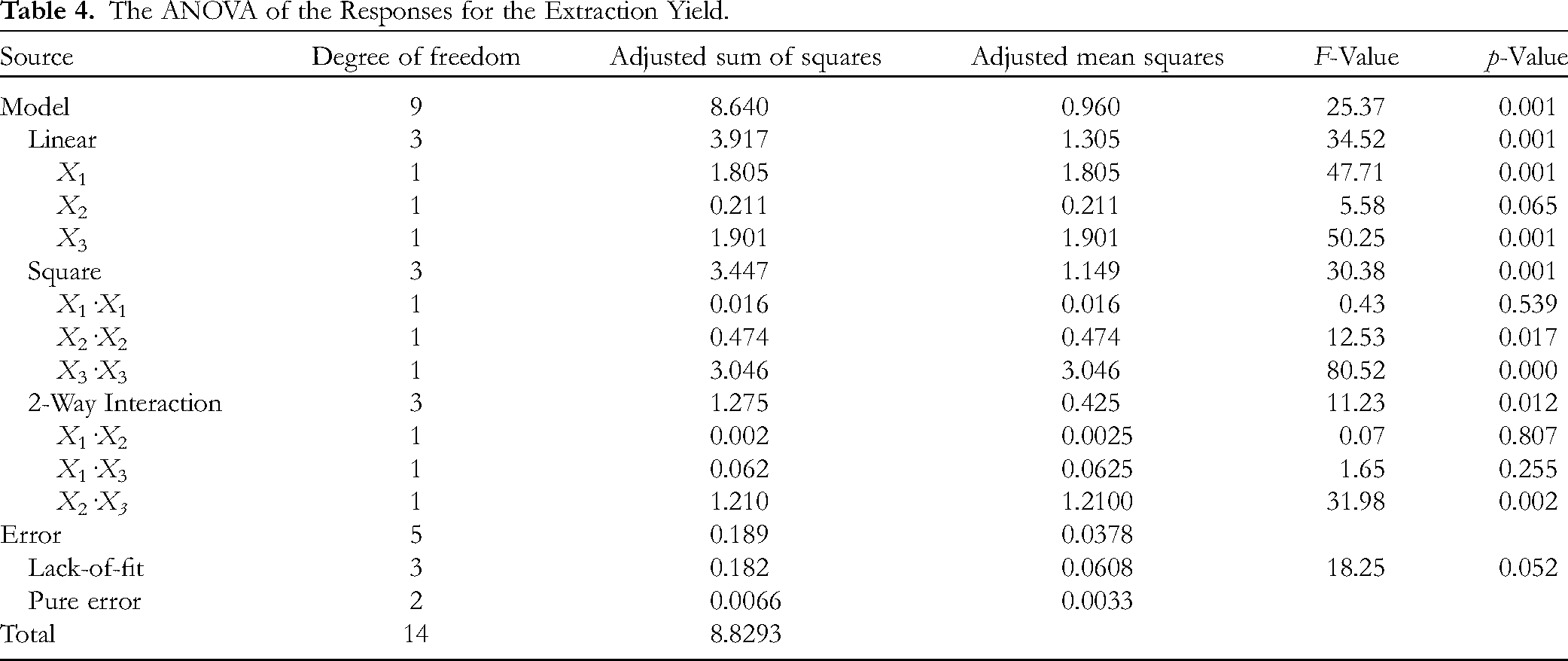
In Table 4 the degree of significance of each process parameter is presented with the p-value. The linear terms of pressure and time exhibited the most statistically significant influence on the extraction yield (p < .05). The quadratic terms of temperature and time had a statistically significant influence on the extraction yield. Interaction between temperature and time exhibited statistically significant influence on the investigated response. The coefficient of determination (R2) was 0.97 and the adjusted R2 was 0.94. The obtained results were used to create three-dimensional (3D) graphs of the response surface (Figure 1).

3D plots for the extraction yield as a function of extraction pressure, temperature, and time.
The response surface plot for the obtained extraction yield as a function of pressure and temperature indicated that the extraction yield increases with an increase in extraction pressure. The extraction yield as a function of extraction pressure and extraction time increased with the increase in extraction time and then, after reaching the maximum, decreased. The extraction yield as a function of temperature and extraction time increased with time and after reaching the maximum at 90 min started to decrease. The optimization was made by applying the optimal process parameters to obtain the highest extraction yield. The process parameters applied for obtaining the highest extraction yield were: 30 MPa, temperature 51.5 °C and extraction time 94.5 min. Under the optimal process parameters the extraction yield was 3.3 g of SC CO2 extract per 100 g of plant material.
The chemical compounds present in the obtained SC CO2 extracts after the range of applied process parameters were determined by GC–MS (Table 5). The chromatograms obtained revealed the presence of three monoterpenes (camphene, sabinene, and limonene), two oxygenated monoterpenes (eucalyptol and linalool), two phenylpropanoids (eugenol and methyleugenol), an aldehyde (dodecanal), a diterpene (neophytadiene), an alcohol (octadecan-1-ol), a long chain fatty acid (linoleic acid), an alkane (tricosane), and a triterpene (squalene). Compounds that were present in the essential oil and SC CO2 extracts were sesquiterpenes (α-selinene, β-selinene, γ-selinene, and α-cadinene), oxygenated sesquiterpenes (hexahydrofarnesyl acetone and farnesyl acetone), an alcohol (hexadecan-1-ol), acyclic diterpene alcohols (phytol and isophytol), and long chain fatty acids (ethyl hexadecanoate, ethyl linoleate, and ethyl oleate) (Table 5). The results obtained indicated several differences among the compounds present in the essential oil and SC CO2 extracts. The monoterpenes, oxygenated monoterpenes and phenylpropanoids were present in SC CO2 extracts probably due to the temperature interval from 40 to 60 °C applied for the extractions, which favored their solubility and extraction. Octadecan-1-ol, linoleic acid, tricosane and squalene were present in the SC CO2 extract probably due to the higher pressures applied in the SC CO2 extraction.
Phytochemical Profile of V. vinifera Leaves SC CO2 Extracts.

Different applied extraction process parameters influenced the amount of compounds in the obtained SC CO2 extracts. The amount of compounds obtained in SC CO2 extracts is depicted in Table 6 in eucalyptol equivalents per 100 g of plant material.
Amount of Compounds in V. vinifera SC CO2 Extracts in mg Eucalyptol Equivalents per 100 g of Plant Material.

Investigated was the influence of pressure, temperature and time on eucalyptol amount in SC CO2 extracts. The effect of linear, square and two-way interactions is depicted in Table 7.
The ANOVA of the Responses for the Eucalyptol Amount.
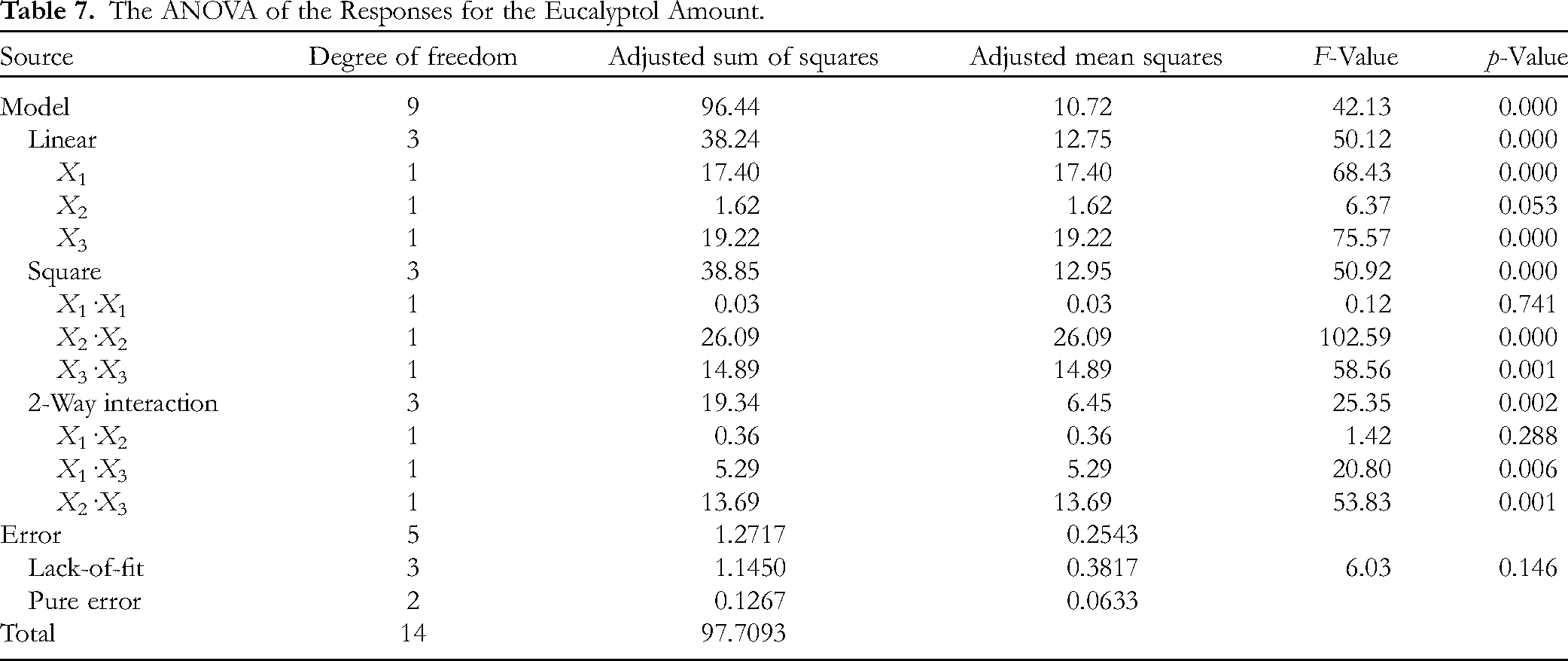
In the investigated responses the linear term of pressure and time showed statistically significant influence on the eucalyptol yield. Square interactions of temperature and time showed statistically significant influence on the eucalyptol amount. In the case of two-way interactions, the interactions between pressure and time and between temperature and time exhibited statistically significant influence on the investigated response. The R2 was 0.9870 and the adjusted R2 was 0.9636 indicating that the predicted model for investigating eucalyptol amount was appropriate. The obtained data were used for plotting 3D graphs of the response surface (Figure 2).

3D plots for the eucalyptol amount as a function of extraction pressure, temperature, and time.
The 3D plots showed statistically significant interactions between the applied extraction process parameters on eucalyptol amount (Figure 2). The eucalyptol amount increased with temperature till 49 °C and then decreased. The eucalyptol amount increased with longer SC CO2 extraction time, while the highest eucalyptol amount was achieved at a pressure of 30 MPa.
The optimal conditions for obtaining the highest eucalyptol amount were 30 MPa, 48.9 °C and an extraction time of 111.5 min. Under these extraction process parameters the obtained eucalyptol amount was 11.70 mg per 100 g of plant material and the theoretically predicted amount was experimentally confirmed with a deviation of ±3%.
The studies on compounds present in white variety Malvasia Fina and red variety Touringa Franca after boiling under pressure and extraction by headspace solid-phase microextraction revealed the presence of five aldehydes: pentanal, heptanal, octanal, nonanal, and decanal. 5 Pentanal was the most abundant aldehyde in both varieties. 5 The amount of all five aldehydes decreased with longer boiling under pressure. 5 In the SC CO2 extract of V. vinifera var. White Riesling leaves, the aldehyde dodecanal was present. Its amount was the highest with the applied process parameters of 30 MPa, at a temperature of 50 °C and an extraction time of 120 min (Table 6). The amount of dodecanal obtained in SC CO2 extracts was lower when the pressure was lower than 30 MPa, in cases when the SC CO2 extraction lasted 120 min (Table 6). The alcohol octan-1-ol was present in Malvasia Fina and Touringa Franca extracts obtained after the leaves were bleached and submerged for 5 min in boiling water. 5 Octan-1-ol was present only in Touringa Franca leaves when the boiling under pressure was 75 min. 5 The essential oil of White Riesling leaves contained the alcohol hexadecan-1-ol (Table 1), and the SC CO2 extracts hexadecan-1-ol and octadecan-1-ol (Table 5). The amount of alcohols in White Riesling SC CO2 extracts was the highest in the extract obtained at the pressure of 20 MPa, temperature of 60 °C and extraction time of 120 min (Table 6). In Malvasia Fina and Touringa Franca methyl, hexenyl and hexyl esters were present. 5 In White Riesling essential oil and SC CO2 extracts ethyl esters were present. The terpenes limonene and linalool were present in Malvasia Fina and Touringa Franca leaves after the culinary treatment, but only in Touringa Franca fresh leaves limonene was detected. 5 White Riesling SC CO2 extracts contained limonene and linalool (Table 5). The highest amount of limonene was obtained under the pressure of 30 MPa, at a temperature of 50 °C and an extraction time of 120 min (Table 6). The highest amount of linalool was obtained under the pressure of 10 MPa, at a temperature of 40 °C and an extraction time of 120 min (Table 6). The amount of linalool in White Riesling SC CO2 extracts was in the range from 0.1 to 1.5 mg, in mg eucalyptol equivalents, per 100 g of plant material (Table 6). Solvent extraction, using acetone, of the red variety Storgozia leaves revealed the presence of sterols, terpenes, fatty acids, esters, ketones, one furan, and a pyrimidine nucleobase thymine. 6 The solvent extraction, using chloroform, of eight V. vinifera cultivars: Chassels, Gewurztraminer, Muscat, Pinot auxerrois, Pinot gris, Pinot noir, Riesling and Sylvaner leaves, where the investigations were determined on triterpenoids, revealed the presence of β-amyrin, lupeol, taraxerol, α-tocopherol, and β-sitosterol. 8 The solvent extraction of one cultivar using acetone and eight cultivars using chloroform revealed that the common constituents of V. vinifera leaf cultivars were β-amyrin, lupeol, α-tocopherol, and β-sitosterol.6,8 The SC CO2 extracts of V. vinifera leaves, collected in October, contained β-amyrin, lupeol, α-tocopherol, and β-sitosterol. 10 The applied process parameters were 30 MPa, the temperature was in the range from 40 °C to 80 °C and with CO2 modified with 5% and 10% of either ethanol or ethyl acetate. 10 In White Riesling leaves, collected in early June, these compounds were not detected. The chemical compounds present in V. vinifera leaves are, most probably, influenced by the harvest time and variety.
Conclusions
The chemical compounds present in the essential oil and SC CO2 extracts of V. vinifera leaves contain flavor and aroma characteristic components. It can be concluded that the essential oil obtained by hydrodistillation has a characteristic flavor which is mostly dominanted by sesquiterpenoids. The SC CO2 extracts were rich in grassy type scent compounds. The main compounds influencing the flavor and aroma of dolmates cannot be attributed to a few compounds. It can be concluded that the flavor and aroma of dolmates is a complex mixture where sesquiterpenoids and grassy type compounds dominate.
Material and Methods
Plant Material
V. vinifera var. White Riesling ssp. sativa L. leaves were collected in early June 2022 in Karlovacki vinogradi, Serbia. Leaves were air dried for several days. Dried V. vinifera leaves were pulverized. After drying, the water content was determined according to AOAC Official Method 925.40 and was 7.8 ± 0.3%. The pulverized plant material was sieved using a vertical vibratory sieve shaker (Labortechnik GmbH, Ilmenau, Germany). The average particle size distribution was determined using a nest of five sieves. The mass of fragments remaining on each sieve was used to calculate the distribution of fragments by Rosin–Rammler distribution function. 14 The distribution revealed that the average particle size was 0.43 ± 0.09 mm.
Chemicals
The CO2 used for extraction was 99.97% (Messer Tehnogas AD, Serbia). The standard compound eucalyptol (1,8-cineol) was from Fluka (Buchs, Switzerland). All other chemicals were of analytical reagent grade.
Hydrodistillation
Air-dried pulverized plant material was hydrodistilled for 3 h using a Clevenger-type apparatus. The essential oil obtained was dried over anhydrous sodium sulfate.
Supercritical CO2 Extraction Procedure
The SC CO2 extractions were done using a lab scale extraction system. V. vinifera leaves (50 g), in pulverized form, were placed in the extraction vessel. The extractions were performed according to the conditions determined by the BBD. The CO2 flow rate was 2 kg·h−1. The obtained SC CO2 extract, after each extraction, was collected in a separator in a glass tube with a previously determined weight. The obtained extracts were analyzed by GC–MS.
GC–MS Analysis
The extract sample analyses were carried out on an Agilent 7890A GC equipped with 5975 MSD. The capillary column was HP-5MS (5% phenyl-methyl polysiloxane, 30 m × 250 μm × 0.25 μm). The injection port temperature was 250 °C; split-less injection. The transfer line temperature was 280 °C. The initial oven temperature was set at 200 °C for 3 min, and programed at 8 °C·min−1 to 280 °C. The carrier gas was He. MS conditions were: scan (45–450 amu) and the MS source temperature was 250 °C. Injected sample volume was 1 μL. The chemical compounds present in the essential oil and SC CO2 extracts were identified on the basis of their mass spectra, retention times and retention indices (RIs). Their identification was carried out based on computer matching with the NIST 2008 MS library. Quantification of compounds was done using calibration curves. The standard compound was dissolved in n-hexane to prepare six different concentrations of eucalyptol. The R2 for the calibration curve was 0.997. All analyses were performed in triplicate.
Experimental Design
The analyses were performed using MINITAB 21 software. Analysis of variance (ANOVA) was used to evaluate the quality of the model. The confidence level was 0.95. Three factors investigated were: pressure (X1), temperature (X2) and processing time (X3).
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable. This article does not contain any studies with human or animal subjects.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
Not applicable. This article does not contain any guidelines followed for performing experimental procedures.
Statement of Informed Consent
Not applicable. This article does not contain any studies with human or animal subjects.
