Abstract
Keywords
Ginkgo biloba L. is the only extant species within the division Ginkgophyta.1-4 G. biloba extracts are herbal remedies used for many centuries. 5 G. biloba herbal remedies have maintained historical and cultural alternatives for pharmaceutically made medicaments.6,7 G. biloba is a medicinal plant and most special extracts are made from its leaves.8,9 The leaves are green in spring and summer and turn to grey-yellow, yellow, brown, or blackish in autumn. Ginkgo leaves major constituents are alkanes, lipids, sterols, terpenoids, carotenoids, carboxylic acids, benzenoids, phenylpropanoids, flavonoids, biflavonoids, catechins, proanthocyanidins, and carbohydrates.10-13 Characteristic constituents are terpenetrilactones with the trivial name ginkgolides, while sesquiterpene lactones are bilobalides.12,14
Thin-layer chromatographic analysis is used for ginkgolides and bilobalide identification. 14 High-performance liquid chromatographic analyses for determination of flavonoids, ginkgolides, and bilobalide have been developed.10,15 Chemical assays have been developed for qualitative and quantitative flavonoid glycoside determination and are carried out after hydrolysis of the aglycones. 16 The qualitative biflavones identification can be determined by high-performance liquid chromatography. 17 The qualitative and quantitative diterpene ginkgolides and sesquiterpene bilobalide determination can be done by high-performance liquid chromatography. 12
The changes in G. biloba leaf lipids through the early stages of degradation were done in order to identify biomarkers of the Ginkgoale. 18 The fresh, senescent, and litter leaves were extracted with CH2Cl2:MeOH (2:1, v/v) and obtained were internal lipids. 18 It was found that α-tocopherol is present in G. biloba leaves and can be a useful biomarker for the Ginkgoale. 18
The G. biloba leaves, collected in September, where the essential oil was obtained by hydrodistillation, contained hydrocarbons (31.14%), alcohols (5.61%), ketones (46.17%), aldehydes (1.78%), acids (5.41%), and esters (1.1%). 19 The essential oil obtained by hydrodistillation, when the leaves were collected in June, consisted of sesquiterpenes (42.11%), diterpenes (14.08%), hydrocarbons (5.89%), esters (6.12%), aldehydes (2.59%), alcohols (2.36%), monoterpenes (1.94%), acids (0.57%), and a ketone (0.11%). 20 Essential oils, obtained after hydrodistillation of leaves, collected from May to late November from three different trees, two young and one old tree, composition varied from month to month. The constituents were hydrocarbons, esters, aldehydes, ketones, and acids. 21 The ionones were detected only in one young tree and limonene only in the old one. 21
G. biloba green leaves have been used for the extraction of biologically active compounds using various extraction techniques.22-25 The carbon dioxide (CO2) in its supercritical state is a solvent enabling the extraction of lipophilic compounds. To increase the polarity of CO2 fluid, in the process, cosolvents were added to improve extraction efficiency. 25 For the extraction of flavonoids and terpenoids from G. biloba green leaves, after the primary extraction with 70% ethanol, followed the extraction with the supercritical CO2 (SC CO2) using the CO2 containing 5% ethanol as a cosolvent. 25 The most favorable experimental conditions were 30 MPa, 60 °C, and CO2 containing 5% ethanol as a cosolvent. 25 The influence of pressure, temperature, and time of the SC CO2 extraction on the yield, using the cosolvents, was investigated. 23
It is well known that the leaf's development stage influences the phytochemical profile. Leaf senescence is characterized by chloroplast degeneration and catabolism of phytochemicals. Phytochemicals disassembled from senescent leaves, which were not relocated to phloem tissues, were extracted by hydrodistillation and SC CO2 extraction in order to gain insight into G. biloba yellow leaves essential oils. Data on the SC CO2 extraction of yellow G. biloba leaves are not available. The SC CO2 was the method of choice to study the lipophilic phytochemicals in G. biloba yellow leaves under the most favorable experimental condition.
The aim of the study was to determine the phytochemical profile of G. biloba yellow leaves essential oils obtained by hydrodistillation and SC CO2 extraction.
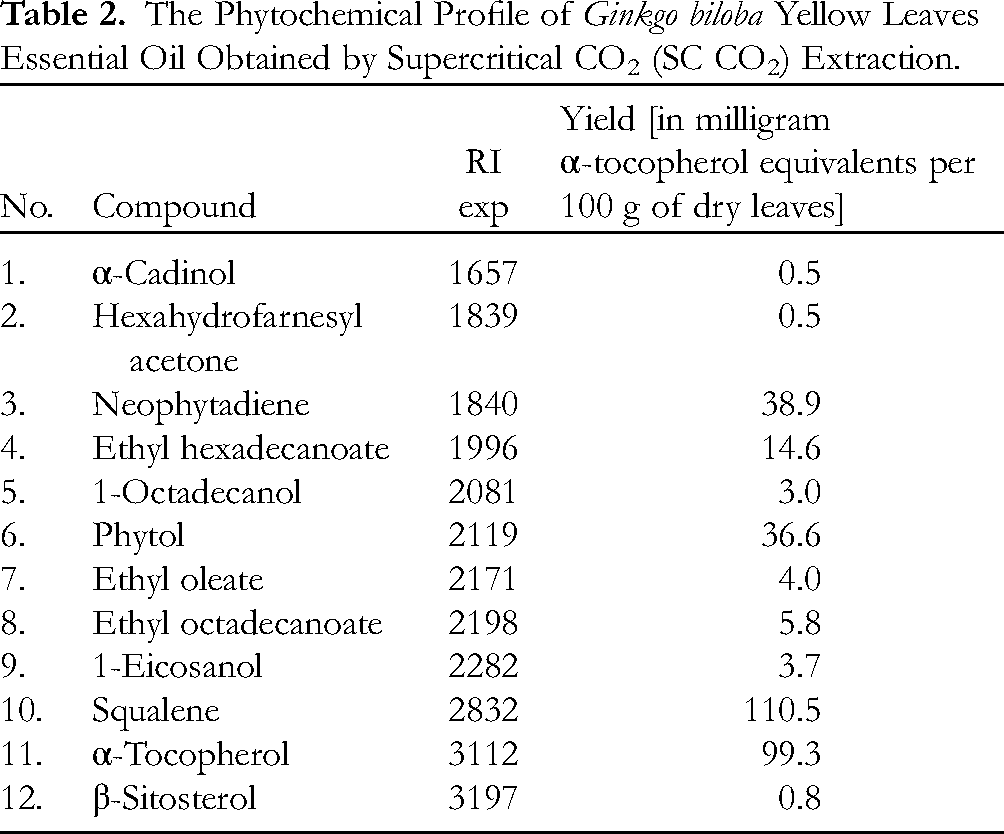
The essential oil obtained by hydrodistillation yielded 0.2% and after the SC CO2 extraction, the yield was 1.4%. In the essential oil, obtained by hydrodistillation, 50 compounds were identified (Table 1). In the essential oil, obtained by SC CO2 extraction, 12 compounds were identified. The yield of the phytochemicals in the G. biloba yellow leaves essential oil, obtained by SC CO2 extraction, is expressed in milligrams of α-tocopherol equivalents per 100 g of dry leaves (Table 2).
Phytochemical Profile of Ginkgo biloba Yellow Leaves Essential Oil Obtained by Hydrodistillation.

aRI exp: retention indices on HP-5MS column relative to C8-C40 n-alkanes.
bRI lit: retention indices obtained from the literature and NIST Webbook.
The Phytochemical Profile of Ginkgo biloba Yellow Leaves Essential Oil Obtained by Supercritical CO2 (SC CO2) Extraction.
The essential oil obtained by hydrodistillation consisted of monoterpenes (3.1%), oxygenated monoterpenes (15.3%), sesquiterpenes (6.2%), oxygenated sesquiterpenes (51.4%), esters (1.3%), the oxolane vitispirane (1.5%), two benzopyrans (7.1%), and a rose ketone trans-α-ionone (0.8%). The essential oil obtained by hydrodistillation of leaves collected in September, contained 1.1% of esters while the essential oil, from the leaves collected in November, had 1.32% of esters. 19 Ketones dominated in the essential oil from the leaves collected in September, while in the essential oil, from the leaves collected in November, the geranyl acetone presented 3.95% and hexahydrofarnesyl acetone was present in 1.09%. 19 The rose ketone was identified previously only in essential oil obtained from young tree leaves while in the essential oil obtained from leaves collected in November, it presented 0.85%. 21 The most abundant compounds in the essential oil obtained from the leaves collected in November were: caryophyllene-4 (12),8(13)-dien-5α-ol (7.1%), 14-hydroxy-(Z)-caryophyllene (5.8%), caryophyllene oxide (5.8%), hydroxydihydroedulan (5.6%), iso-italicene epoxide (4.8%), clovenol (4.6%), spathulenol (4.4%), geranyl acetone (4.0%), and 4-hydroxy-9-epi-(E)-caryophyllene (3.4%) (Table 1). Oxygenated sesquiterterpenes were the most abundant compounds present in the essential oil (Table 1).
The essential oil obtained by SC CO2 extraction was characterized by high amounts of squalene and α-tocopherol, followed by neophytadiene and phytol (Table 2). The α-cadinol, hexahydrofarnesyl acetone, and β-sitosterol were present in small amounts. The SC CO2 extraction of green G. biloba leaves revealed the presence of flavonoids and terpenoids. 25 The phytochemical analysis of the essential oil of G. biloba yellow leaves collected in November, revealed the presence of one sesquiterpenoid alcohol α-cadinol; a diterpene neophytadiene; a ketone hexahydrofarnesyl acetone; three esters: ethyl hexadecanoate, ethyl oleate, and ethyl octadecanoate; alcohols: 1-octadecanol, phytol, and 1-eicosanol; a triterpenoid squalene, α-tocopherol, and one phytosterol β-sitosterol (Table 2). The hexahydrofarnesyl acetone and three esters were present in both essential oils. The operating parameters applied for the SC CO2 extraction yielded the presence of α-cadinol, neophytadiene, phytol, 1-octadecanol, 1-eicosanol, squalene, α-tocopherol, and β-sitosterol. The presence of α-tocopherol can be attributed to the applied operating temperature of 40 °C during the SC CO2 extraction. The essential oil obtained by hydrodistillation did not contain α-tocopherol due to the high temperatures applied during the hydrodistillation. The essential oil obtained by SC CO2 extraction had low terpenoid content due to the low operating temperature during the SC CO2 extraction process. The differences in chemical composition of essential oils obtained by two different extraction techniques can be attributed to the temperature applied. The novelty of this study is the presence of α-tocopherol in the SC CO2 essential oil of G. biloba yellow leaves. The quantity of α-tocopherol is high and yellow leaves can be a reliable source for obtaining α-tocopherol from the SC CO2 extract.
This study reports the phytochemical profile of G. biloba yellow leaves essential oils obtained by hydrodistillation and SC CO2 extraction. It can be concluded that the essential oil obtained by hydrodistillation is a valuable source of oxygenated sesquiterpenes. The essential oil obtained by SC CO2 extraction is rich in squalene and α-tocopherol. The identification of α-tocopherol is a starting point for further investigations in order to find the optimal conditions for its isolation from the G. biloba yellow leaves.
Experimental
Plant Material
G biloba yellow leaves were collected in November 2021 in Zrenjanin, Serbia (45°22′51″ N, 20°23′20″ E). Yellow leaves were air-dried for several days. Dried G. biloba yellow leaves were grounded in a rotating blade coffee grinder Bosch MKM 6003. After drying, the water content was determined according to AOAC Official Method 925.40 and was 9.3 ± 0.1%. The grounded plant material was sieved for 20 min using the vertical vibratory sieve shaker (Labortechnik GmbH). The average particle size distribution was determined using a nest of 3 sieves of aperture sizes 0.5, 0.4, and 0.315 mm. The mass of fragments remaining on each sieve was used to calculate the distribution of fragments by the Rosin-Rammler distribution function and was normalized concerning the total mass. 26 The distribution revealed that the average particle size was 0.37 ± 0.08 mm.
Chemicals
The CO2 used for extraction was 99.97% (Messer Tehnogas AD). The standard compound α-tocopherol was from Dr. Ehrenstorfer (Augsburg, Germany). All other chemicals were of analytical reagent grade.
Hydrodistillation
Air-dried grounded plant material was hydrodistilled for 3 h using Clevenger-type apparatus. The essential oil obtained was dried over anhydrous sodium sulfate.
SC CO2 Extraction Procedure
Grinded yellow leaves (50 g), in a powder form, were poured into the extraction vessel made from stainless steel bar (AISI 304) 100 mm outside diameter and height 500 mm, with the center hole of Ø 40 mm. The volume of the extraction vessel was 500 mL. The cap of the extraction vessel held a plug connected to the extraction vessel through the thread. The plug had O-ring seals in two places. The plug had a filter element with the ability to filter particles of 2 to 10 µm (Norman Ultraporous 4202T-6T-2M) to prevent the withdrawal of the plant material. The extract was collected in a separator with the glass tube with previously determined glass tube mass. The temperature was controlled by a controller. The input CO2 line leading towards the extraction vessel was preheated by a heat exchanger supported by the water heating system. Liquid CO2 was precooled through the stainless steel coil by an ethylene glycol/ethanol cooling bath. Before the extraction vessel, CO2 was preheated through the stainless steel coil at the extraction temperature. The CO2 flow rate was controlled by a flow meter. The mass of the plant material in the extraction vessel, grounded plant material particle size, and the extraction time (90 min) were kept constant during the experiment. The extraction lasted for 90 min since a longer extraction time did not increase the extraction yield significantly.
Gas Chromatography–Mass Spectrometry Analysis
The extract sample analyses were carried out on Agilent 7890A GC equipped with 5975 MSD. The capillary column was HP-5MS (5% phenyl-methyl polysiloxane, 30 m × 250 μm × 0.25 μm). The injection port temperature was 250 °C, split-less injection. The transfer line temperature was 280 °C. The initial oven temperature was set at 200 °C for 3 min, and programmed with 8 °C·min−1 to 280 °C. The carrier gas was He. Mass spectrometry (MS) conditions were: scan (45 to 450 amu) and the MS source temperature was 250 °C. Injected sample volume was 1 μL. The phytochemicals present in the essential oils were identified on the basis of their mass spectra, retention times, and retention indices (RIs). The identification of phytochemicals was carried out based on computer matching with NIST 2008 MS library. Quantification of compounds was provided using calibration curves. The standard compound was dissolved in n-hexane to prepare six different concentrations of α-tocopherol. The R2 for the calibration curve was 0.999. All analyses were performed in triplicate.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
