Abstract
As part of the search for new steroid substances with potential application in oncology, a series of endoperoxide steroid derivatives (
This is a visual representation of the abstract.
Introduction
Cancer is caused by malignant cell proliferation and is recognized worldwide as a disease with a high mortality rate.1,2 Chemotherapy remains one of the main effective cancer treatments available today, but serious side effects greatly limit the clinical use of many chemotherapy drugs.3,4 Therefore, the development of new chemotherapy drugs with low toxicity and high efficiency is a meaningful and important task in the field of pharmaceutical chemistry. Natural active products have a very long history of medicinal use, and structural modification based on a natural active product skeleton is one of the main sources and methods of production of novel anticancer drugs. 5
Natural endoperoxides are a class of compounds with peroxy-bridged structures (-O-O-) that are abundant and biologically diverse in nature, and many endoperoxide natural products have been shown to have significant antitumor activities and potential medicinal values. 6 Artemisinin, 7 gracilioether A, 8 schinalactone A, 9 and other natural peroxides have been verified to have significant anticancer activities. It is desirable that the novel structure of endoperoxide is utilized to provide a research direction for the research and development of new pharmacophores or new drugs.
Ergosterol peroxide (EP) is a natural endoperoxide steroid that was extracted from the traditional Chinese medicine Ganoderma lucidum,10,11 which has broad-spectrum antitumor activity (Figure 1).12‐18 As a lead compound with potential research value, appropriate structural modification of EP is the key to increase its antitumor activity.
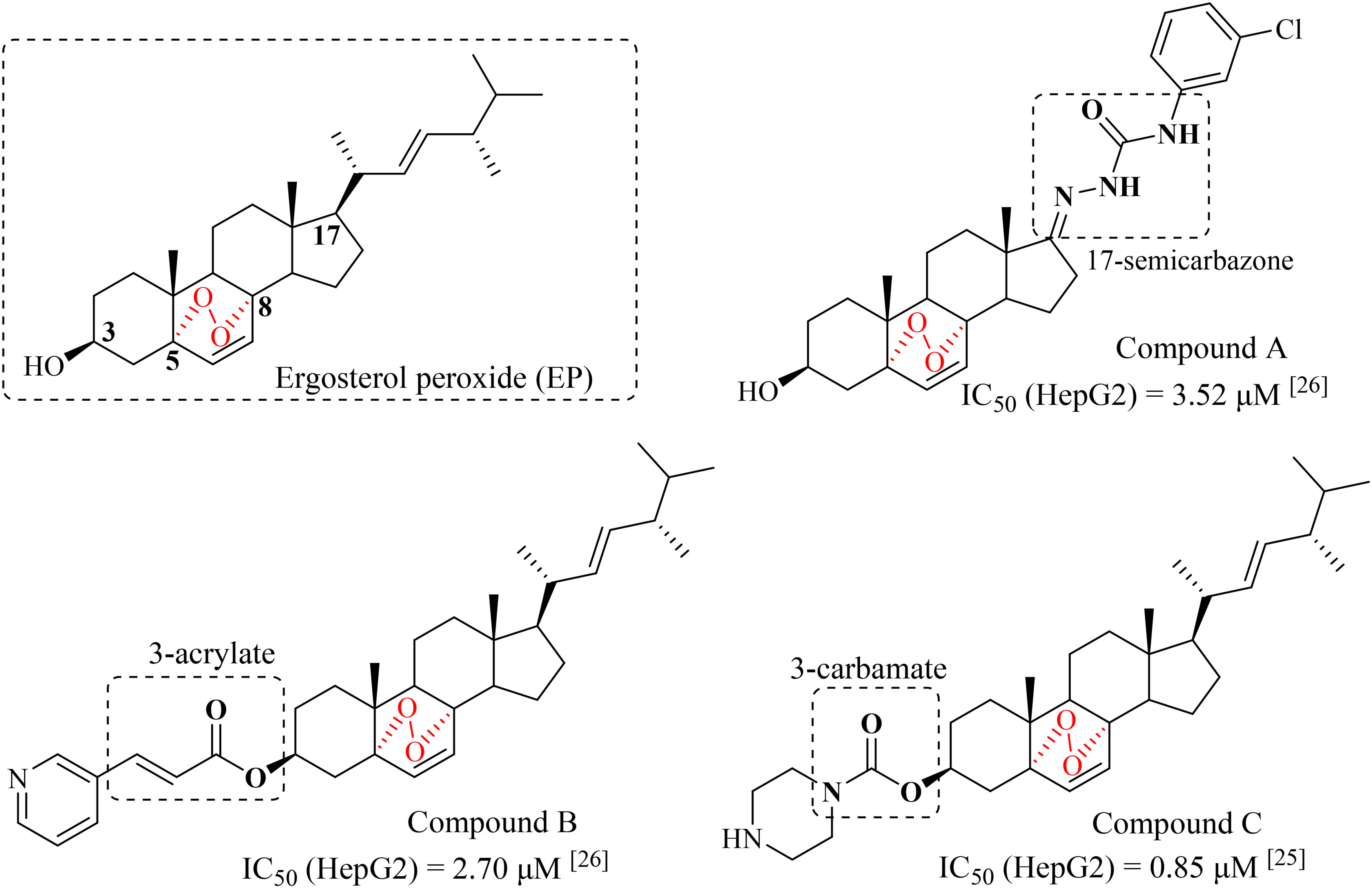
Structures of ergosterol peroxide (EP) and its derivatives.
Previously, we have prepared a series of C-17 modified derivatives of EP, and many of them exhibited strong inhibitory efficacy against different kinds of tested tumor cell lines, such as compound
Thiazole is a five-membered heterocyclic molecule containing nitrogen and sulfur atoms and is the central reactive group in many natural products. 26 The biological activities of thiazoles and compounds containing thiazole structures cover a wide range of aspects, including antitumor, antioxidant, anti-inflammatory, antiviral, antibacterial, and other activities, with antitumor activity being particularly prominent.27‐31 Compounds containing thiazole can exhibit anti-cancer activities through different pathways of action. Clinically applied antitumor agents containing thiazole nuclei are partially exhibited in Figure 2. Therefore, the synthesis and pharmacological studies of compounds containing thiazole and other multitarget groups are of great significance.

Representative antitumor drugs with thiazole structure.
Based on the above-mentioned encouraging results, our research group investigated the further modification of EP by introducing richer types of heterocyclic substituents. In the present work, fourteen endoperoxide steroid derivatives (
Results and Discussion
Chemistry
The general synthesis route of new endoperoxide steroid derivatives
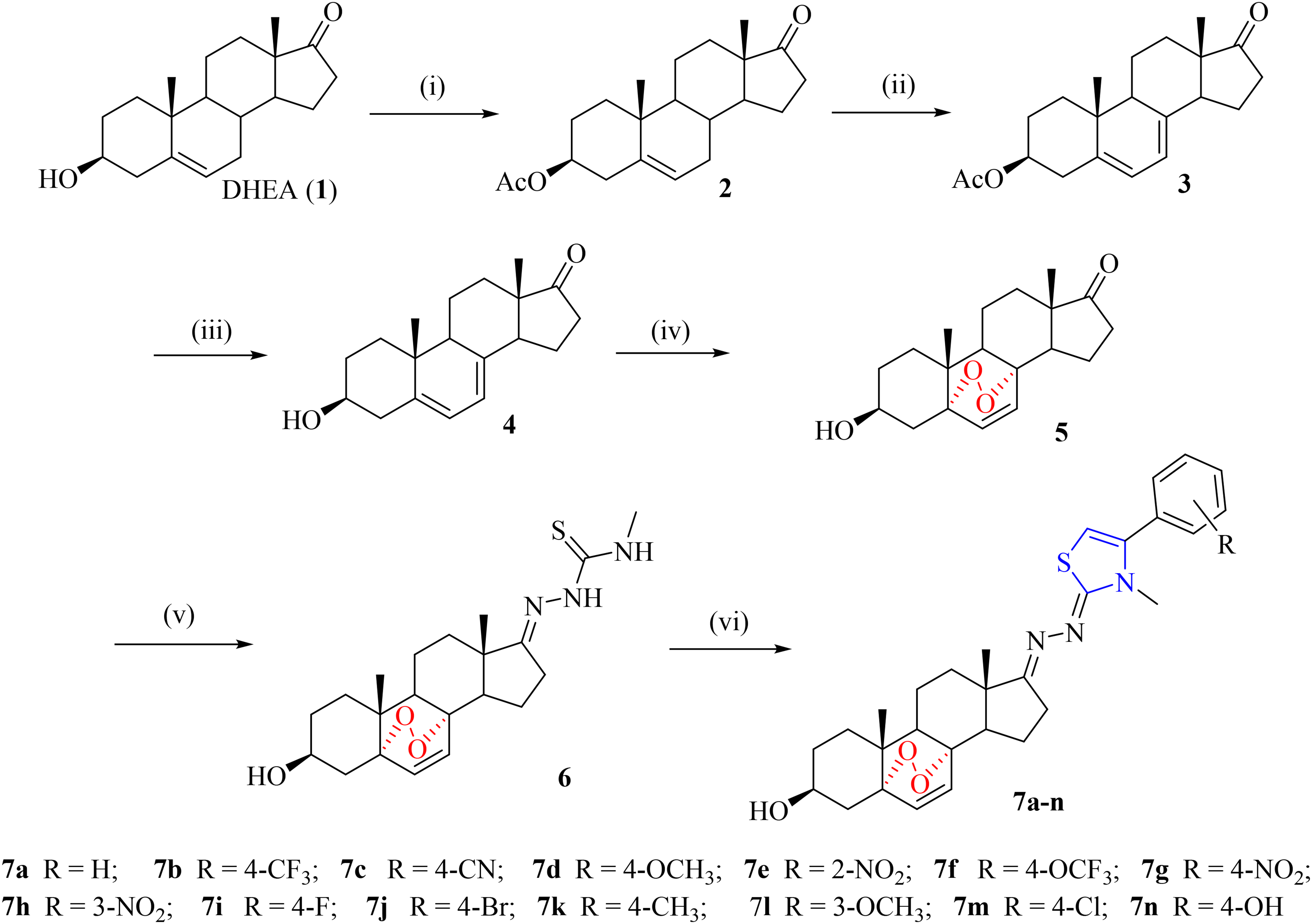
Synthesis of endoperoxide steroid derivatives
Biology
In vitro cytotoxic effects of all new compounds
In Vitro Cytotoxic Activities of Compounds
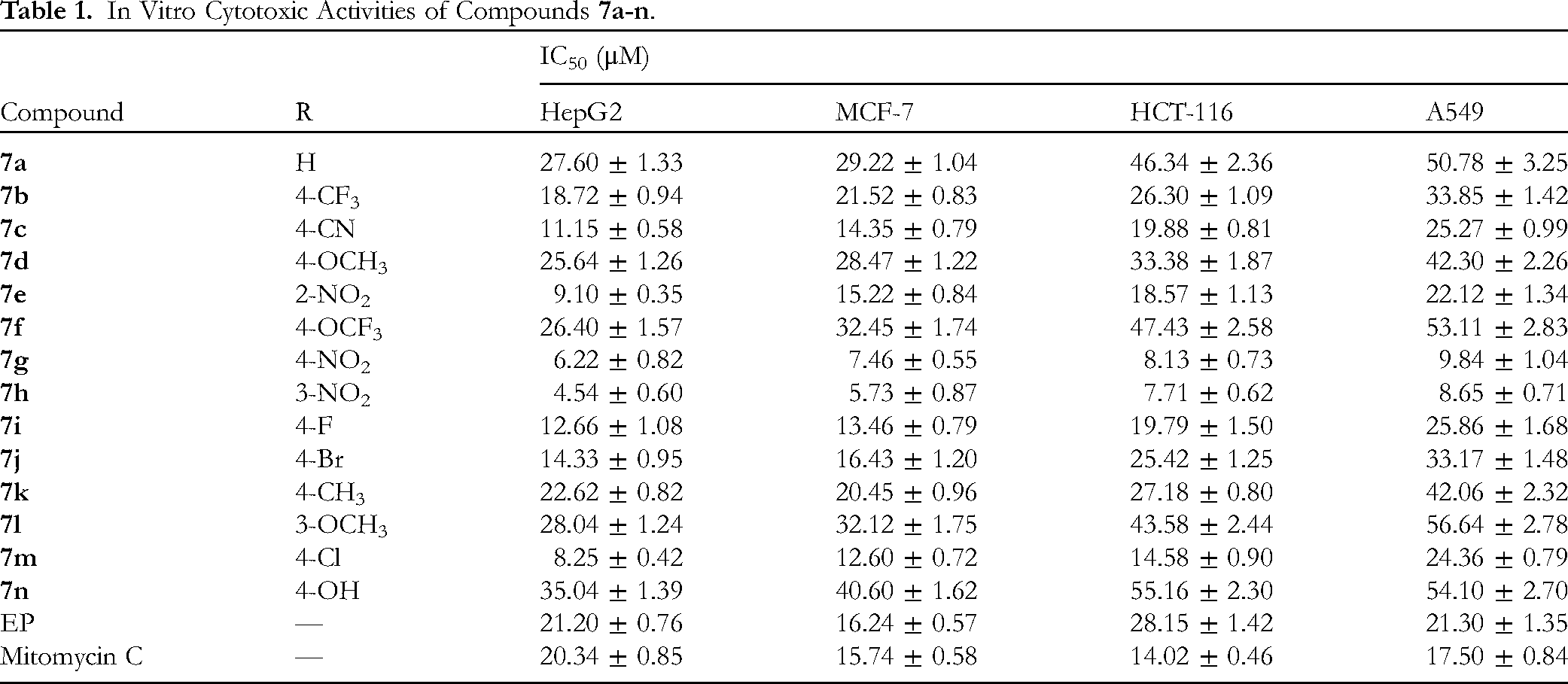
For the HepG2 cell line, most of the derivatives showed conspicuous cytotoxic activities to EP (IC50 = 21.20 μM) against HepG2 cells. Among them, compounds
Taken together, the above results suggested that both the thiazole moiety and aromatic ring conjugated substituents are significant for the cytotoxicity of the novel endoperoxide steroid derivatives. In addition, compounds
To assess whether
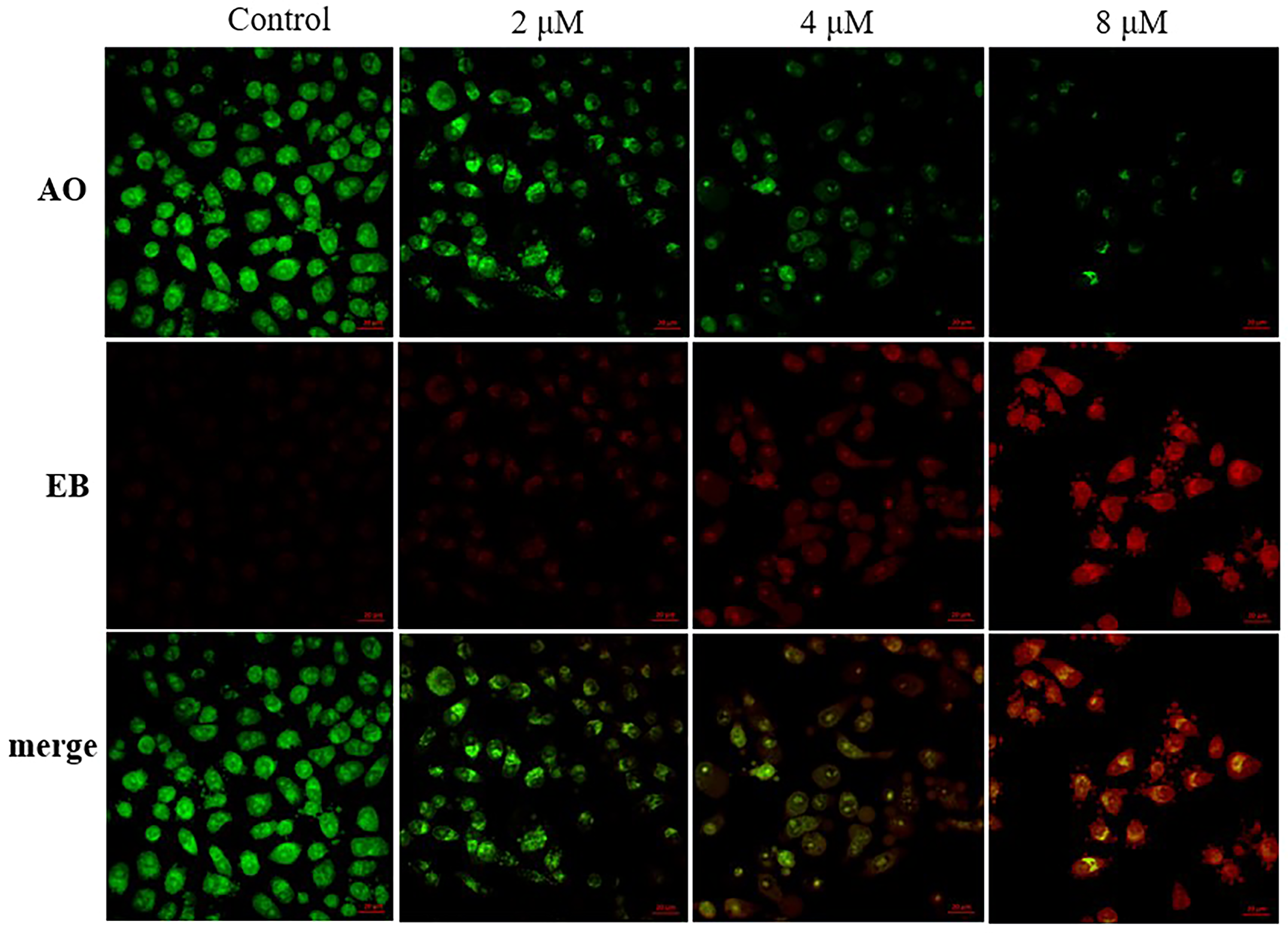
The effect of
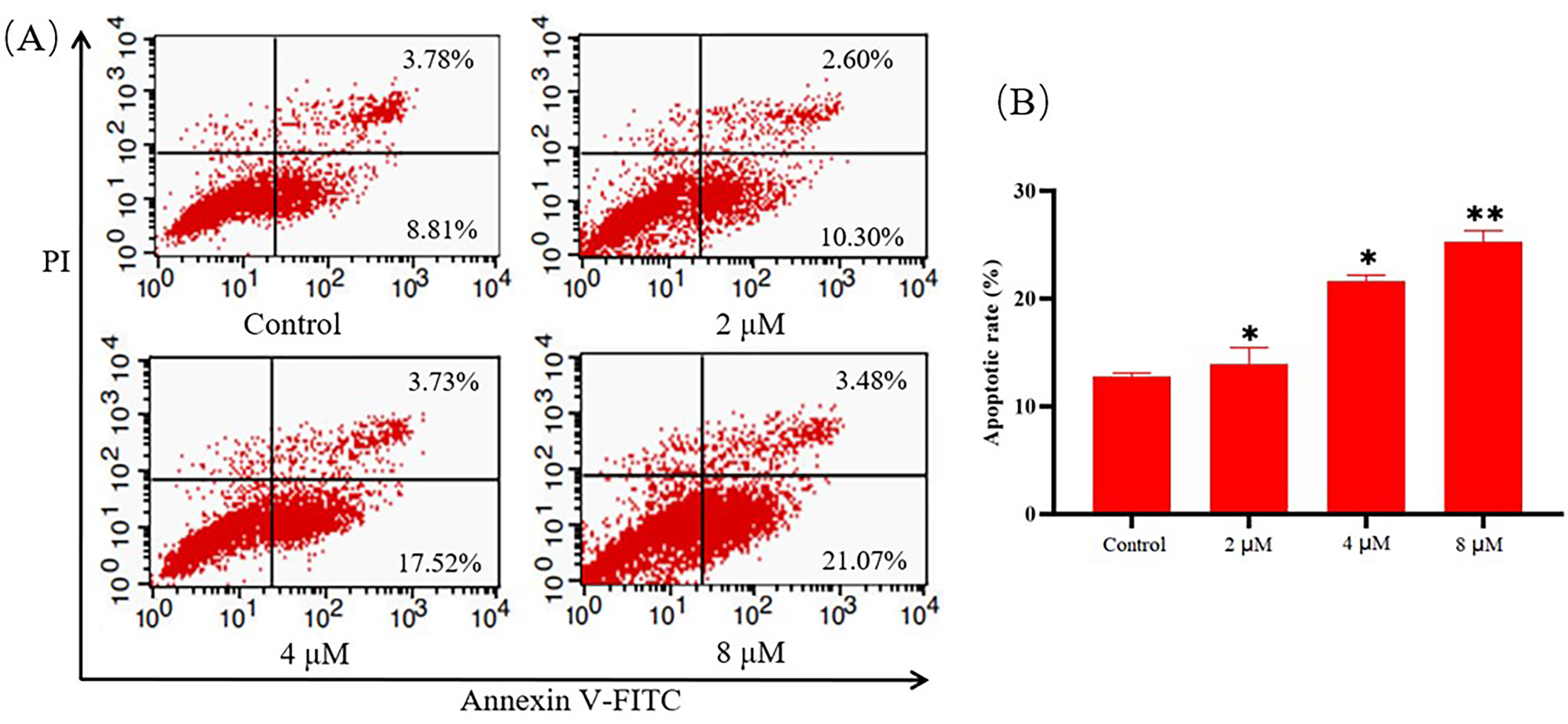
Compound
The loss of mitochondrial membrane potential (MMP) is an important sign of early events in the tumor cells apoptosis cascade. Hence, we decided to evaluate the effect of

Compound
It is well known that changes in MMP trigger cytochrome c release, and subsequently apoptosis via caspase-9 activation. Here, we measured the expression levels of mitochondrial apoptosis related proteins using Western blotting. As shown in Figure 6, the content of cytochrome c in the cytoplasm increased, while cytochrome c triggered the activation of cleaved-caspase-9. Accordingly, as the end point of the signaling pathway, after treatment with 7h, the expression of apoptosis-related protein Bax began to increase, while the Bcl-2 expression was significantly suppressed. These effects were all achieved in a concentration-dependent manner. These results illustrate that 7h was able to induce HepG2 cell death through the mitochondrial apoptosis pathway.
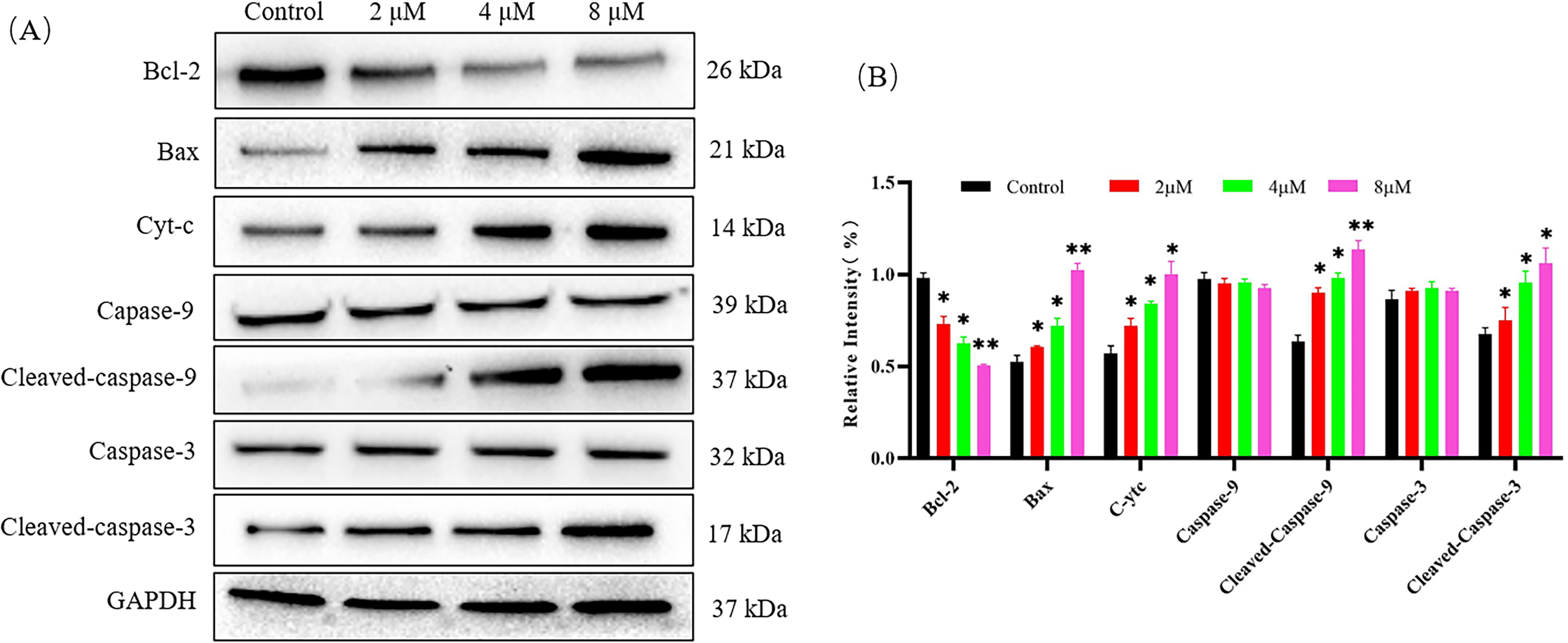
Effects of
Experimental
Chemistry
All chemical reagents and materials were commercially available from Energy Chemical (Zesheng Technology Co., LTD). The reaction process was monitored by TLC (Rongman Biotechnology Co., Ltd). The crude product was purified by flash column chromatography (200-300 mesh silica gel, Yantai Chemical Industry Research Institute). Melting points (mp) of new derivatives were obtained with an MP120 melting point apparatus (Haineng Electrical Co., Ltd). NMR spectra were recorded using an AVANCE NEO 600 Bruker spectrometer (Bruker Optics). HRMS data were measured on a Waters G2-XS UPLC QTOF (Waters Corporation).
Syntheses
Synthesis of Intermediate 2
DHEA (
Synthesis of Intermediate 3
Intermediate
Synthesis of Intermediate 4
A solution of intermediate
Synthesis of Intermediate 5
A mixture of intermediate
Synthesis of 2-(3β-Hydroxy-17H-5α,8α-epidioxyandrost-6-en-17-ylidene)-N-phenylhydrazine-1-carbothioamide (6 )
Intermediate
General Procedure for Synthesis of 7a-n
Intermediate
17-((3-Methyl-4-phenylthiazol-2(3H)-ylidene)hydrazineylidene)-2H-5α,8α-epidioxyandrost-6-en-3β-ol (7a )
Yellow solid, yield 82%, mp 147.8–149.5 °C; 1H NMR (600 MHz, CDCl3) δ 7.53 (s, 1H, Ar-H), 7.35 (s, 2H, Ar-H), 7.27 (s, 2H, Ar-H), 6.54 (d, J = 8.6 Hz, 1H, C6-H), 6.30 (d, J = 8.5 Hz, 1H, C7-H), 5.89 (s, 1H, C = CH), 4.31 (s, 1H, C3-H), 3.97 (s, 1H, OH), 3.29 (s, 3H, N-CH3), 2.72 (s, 1H), 2.64 (s, 1H), 2.13 (s, 1H), 2.06 (s, 1H), 1.96 (d, J = 12.7 Hz, 2H), 1.90 (s, 1H), 1.73 (s, 2H), 1.66 (s, 1H), 1.58 (s, 1H), 1.54 (s, 1H), 1.45 (s, 1H), 1.32 (s, 1H), 1.25 (s, 2H), 1.08 (s, 3H), 0.92 (s, 3H); 13C NMR (150 MHz, CDCl3) δ 172.7, 168.5, 140.8, 135.8, 130.4, 129.1, 128.8, 99.0, 82.4, 79.3, 66.4, 65.6, 51.6, 49.5, 45.8, 37.2, 36.9, 34.7, 34.4, 33.5, 30.6, 30.1, 29.7, 27.6, 23.1, 20.5, 18.5, 18.3, 13.8; HRMS (ESI) Calcd for C29H35N3O3S [M + H]+ 506.2477, found: 506.2442.
17-((4-(4-(Trifluoromethyl)phenyl)-3-methylthiazol-2(3H)-ylidene)hydraziney-lidene)-2H-5α,8α-epidioxyandrost-6-en-3β-ol (7b )
Yellow solid, yield 79%, mp 173.4–175.6 °C; 1H NMR (600 MHz, CDCl3) δ 7.70 (d, J = 8.1 Hz, 2H, Ar-H), 7.48 (d, J = 8.0 Hz, 2H, Ar-H), 6.54 (d, J = 8.5 Hz, 1H, C6-H), 6.30 (d, J = 8.5 Hz, 1H, C7-H), 5.96 (s, 1H, C = CH), 4.31 (s, 1H, C3-H), 3.97 (s, 1H, OH), 3.29 (s, 3H, N-CH3), 2.71 (s, 1H), 2.63 (s, 1H), 2.13 (d, J = 13.8 Hz, 1H), 2.06 (d, J = 13.2 Hz, 1H), 1.96 (d, J = 11.8 Hz, 2H), 1.90 (s, 1H), 1.73 (s, 2H), 1.67 (s, 1H), 1.58 (s, 1H), 1.50 (s, 1H), 1.44 (d, J = 7.5 Hz, 1H), 1.33 (s, 1H), 1.26 (s, 2H), 1.07 (s, 3H), 0.94 (s, 3H); 13C NMR (150 MHz, CDCl3) δ 173.3, 168.1, 139.3, 135.9, 131.0, 130.4, 128.9, 125.8, 100.6, 82.4, 79.3, 66.4, 65.6, 51.6, 49.5, 45.8, 37.1, 36.9, 34.6, 34.4, 33.5, 30.6, 30.1, 27.6, 23.1, 20.4, 19.2, 18.5, 18.2, 13.7; HRMS (ESI) Calcd for C30H34F3N3O3S [M + H]+ 574.2351, found: 574.2302.
17-((4-(4-(Cyano)phenyl)-3-methylthiazol-2(3H)-ylidene)hydrazineylidene)-2H-5α,8α-epidioxyandrost-6-en-3β-ol (7c )
Yellow solid, yield 86%, mp 156.1–168.2 °C; 1H NMR (600 MHz, CDCl3) δ 7.73 (d, J = 8.2 Hz, 2H, Ar-H), 7.48 (d, J = 8.2 Hz, 2H, Ar-H), 6.54 (d, J = 8.5 Hz, 1H, C6-H), 6.30 (d, J = 8.5 Hz, 1H, C7-H), 6.01 (s, 1H, C = CH), 4.31 (s, 1H, C3-H), 3.97 (s, 1H, OH), 3.31 (s, 3H, N-CH3), 2.72 (s, 1H), 2.63 (s, 1H), 2.12 (s, 1H), 2.06 (d, J = 13.1 Hz, 1H), 1.96 (d, J = 13.6 Hz, 2H), 1.90 (s, 1H), 1.73 (s, 2H), 1.64 (s, 1H), 1.57 (s, 1H), 1.50 (s, 1H), 1.44 (d, J = 7.6 Hz, 1H), 1.31 (d, J = 13.5 Hz, 1H), 1.26 (s, 2H), 1.07 (s, 3H), 0.93 (s, 3H); 13C NMR (150 MHz, CDCl3) δ 173.6, 167.9, 135.9, 132.6, 131.0, 130.3, 128.9, 112.6, 101.7, 82.4, 79.2, 66.3, 65.6, 51.6, 49.4, 45.8, 37.1, 36.9, 34.6, 34.3, 33.8, 30.6, 30.1, 27.6, 23.0, 20.4, 19.2, 18.5, 18.2, 13.7; HRMS (ESI) Calcd for C30H34N4O3S [M + H]+ 531.2430, found: 531.2397.
17-((4-(4-Methoxyphenyl)-3-methylthiazol-2(3H)-ylidene)hydrazineylidene)-2H-5α,8α-epidioxyandrost-6-en-3β-ol (7d )
Yellow solid, yield 84%, mp 165.5–167.4 °C; 1H NMR (600 MHz, CDCl3) δ 7.62 (d, J = 102.5 Hz, 1H, Ar-H), 7.25 (s, 1H, Ar-H), 6.95 (d, J = 6.9 Hz, 2H, Ar-H), 6.54 (d, J = 8.5 Hz, 1H, C6-H), 6.30 (d, J = 8.5 Hz, 1H, C7-H), 5.83 (s, 1H, C-CH), 4.31 (s, 1H, C3-H), 3.97 (s, 1H, -OH), 3.84 (s, 3H, OCH3), 3.28 (s, 3H, N-CH3), 2.73 (s, 1H), 2.64 (s, 1H), 2.12 (s, 1H), 2.05 (s, 1H), 1.96 (d, J = 11.6 Hz, 2H), 1.90 (s, 1H), 1.73 (s, 2H), 1.64 (s, 1H), 1.58 (s, 1H), 1.51 (s, 1H), 1.44 (d, J = 9.7 Hz, 1H), 1.32 (s, 1H), 1.25 (s, 2H), 1.07 (s, 3H), 0.92 (s, 3H); 13C NMR (150 MHz, CDCl3) δ 167.8, 160.2, 135.8, 132.3, 131.0, 130.4, 130.1, 128.8, 114.1, 82.3, 79.3, 66.4, 65.6, 55.4, 51.6, 49.4, 45.8, 37.1, 36.9, 34.6, 34.4, 30.6, 30.1, 27.6, 23.1, 20.4, 19.2, 18.5, 18.2, 13.7; HRMS (ESI) Calcd for C30H37N3O4S [M + H]+ 536.2583, found: 536.2539.
17-((4-(2-Nitrophenyl)-3-methylthiazol-2(3H)-ylidene)hydrazineylidene)-2H-5α, 8α-epidioxyandrost-6-en-3β-ol (7e )
Yellow solid, yield 77%, mp 172.8–174.1 °C; 1H NMR (600 MHz, CDCl3) δ 8.11 (s, 1H, Ar-H), 7.71 (d, J = 3.2 Hz, 2H, Ar-H), 7.65 (s, 1H, Ar-H), 7.53 (s, 1H, C6-H), 7.43 (s, 1H, C7-H), 5.86 (s, 1H, C = CH), 3.94 (s, 1H, C3-H), 3.35 (s, 1H, OH), 3.14 (s, 3H, N-CH3), 2.98 (s, 1H), 2.71 (s, 1H), 2.44 (s, 1H), 2.38 (s, 1H), 2.20 (s, 2H), 2.06 (s, 1H), 1.83 (s, 1H), 1.71 (d, J = 7.8 Hz, 2H), 1.64–1.62 (m, 1H), 1.44 (d, J = 7.7 Hz, 2H), 1.25 (s, 2H), 1.20 (s, 1H), 0.96 (s, 3H, C18), 0.84 (s, 3H); 13C NMR (150 MHz, CDCl3) δ 167.8, 149.0, 135.8, 135.3, 133.0, 132.3, 131.0, 130.8, 128.9, 126.2, 124.8, 68.5, 67.1, 65.8, 65.6, 62.6, 46.9, 44.2, 39.1, 38.2, 30.8, 30.6, 29.7, 23.6, 23.0, 22.7, 19.2, 16.3, 13.7; HRMS (ESI) Calcd for C29H34N4O5S [M + H]+ 551.2328, found: 551.2294.
17-((4-(4-(Trifluoromethoxy)phenyl)-3-methylthiazol-2(3H)-ylidene)hydrazine-ylidene)-2H-5α,8α-epidioxyandrost-6-en-3β-ol (7f )
Yellow solid, yield 81%, mp 174.4–176.6 °C; 1H NMR (600 MHz, CDCl3) δ 7.62 (d, J = 105.6 Hz, 1H, Ar-H), 7.39 (d, J = 8.7 Hz, 2H, Ar-H), 7.29 (s, 1H, Ar-H), 6.54 (d, J = 8.4 Hz, 1H, C6-H), 6.30 (d, J = 8.4 Hz, 1H, C7-H), 5.91 (s, 1H, C = CH), 4.31 (s, 1H, C3-H), 3.97 (s, 1H, OH), 3.29 (s, 3H, N-CH3), 2.72 (s, 1H), 2.63 (s, 1H), 2.12 (s, 1H), 2.05 (s, 1H), 1.96 (d, J = 13.8 Hz, 2H), 1.90 (s, 1H), 1.73 (s, 2H), 1.65 (s, 1H), 1.57 (s, 1H), 1.50 (s, 1H), 1.44 (d, J = 7.7 Hz, 1H), 1.32 (d, J = 9.7 Hz, 1H), 1.25 (s, 2H), 1.07 (s, 3H), 0.93 (s, 3H); 13C NMR (150 MHz, CDCl3) δ 173.1, 168.2, 135.9, 131.0, 130.4, 130.2, 128.9, 121.3, 99.8, 82.4, 79.3, 66.3, 65.6, 51.6, 49.5, 45.8, 37.1, 36.9, 34.6, 34.4, 33.4, 30.6, 30.1, 27.6, 23.1, 20.4, 19.2, 18.5, 18.2, 13.7; HRMS (ESI) Calcd for C30H34F3N3O4S [M + H]+ 590.2300, found: 590.2245.
17-((4-(4-Nitrophenyl)-3-methylthiazol-2(3H)-ylidene)hydrazineylidene)-2H-5α, 8α-epidioxyandrost-6-en-3β-ol (7g )
Yellow solid, yield 86%, mp 175.0–176.5 °C; 1H NMR (600 MHz, CDCl3) δ 8.30 (s, 1H, Ar-H), 7.71 (s, 1H, Ar-H), 7.54 (d, J = 6.7 Hz, 2H, Ar-H), 6.54 (d, J = 8.5 Hz, 1H,C6-H), 6.30 (d, J = 8.4 Hz, 1H, C7-H), 6.09 (s, 1H, C = CH), 4.31 (s, 1H, C3-H), 3.98 (s, 1H, OH) 3.35 (s, 3H, N-CH3), 2.74 (s, 1H), 1.96 (d, J = 11.4 Hz, 2H), 1.73 (s, 2H), 1.45 (s, 1H), 1.44 (s, 1H), 1.34–1.32 (m, 1H), 1.25 (s, 2H), 1.08 (s, 3H), 0.93 (s, 3H); 13C NMR (150 MHz, CDCl3) δ 167.8, 147.9, 135.9, 132.3, 130.9, 130.3, 129.1, 128.9, 124.2, 82.4, 79.2, 66.4, 65.6, 51.6, 49.4, 45.9, 37.2, 36.9, 34.7, 34.3, 30.6, 30.1, 29.7, 23.1, 20.4, 19.2, 18.5, 18.2, 13.7; HRMS (ESI) Calcd for C29H34N4O5S [M + H]+ 551.2328, found: 551.2286.
17-((4-(3-Nitrophenyl)-3-methylthiazol-2(3H)-ylidene)hydrazineylidene)-2H-5α, 8α-epidioxyandrost-6-en-3β-ol (7h )
Yellow solid, yield 85%, mp 174.6–166.1 °C; 1H NMR (600 MHz, CDCl3) δ 7.70 (d, J = 13.7 Hz, 2H, Ar-H), 7.64 (s, 1H, Ar-H), 7.53 (s, 1H, Ar-H), 6.54 (d, J = 8.5 Hz, 1H, C6-H), 6.30 (d, J = 8.5 Hz, 1H, C7-H), 6.03 (s, 1H, C = CH), 4.31 (s, 1H, C3-H), 3.98 (s, 1H, OH), 3.32 (s, 3H, N-CH3), 2.72 (s, 1H), 2.63 (s, 1H), 2.14 (d, J = 8.8 Hz, 1H), 2.06 (d, J = 13.2 Hz, 1H), 1.96 (d, J = 13.8 Hz, 2H), 1.90 (s, 1H), 1.73 (d, J = 7.3 Hz, 2H), 1.65 (s, 1H), 1.58 (s, 1H), 1.44 (d, J = 7.5 Hz, 2H), 1.33 (s, 1H), 1.25 (s, 2H), 1.08 (s, 3H), 0.93 (s, 3H); 13C NMR (150 MHz, CDCl3) δ 173.6, 167.8, 148.5, 135.9, 134.2, 131.0, 130.3, 129.9, 128.9, 123.4, 101.4, 82.4, 79.2, 66.4, 65.6, 51.6, 49.4, 45.8, 37.1, 36.9, 34.3, 33.5, 30.6, 29.7, 27.6, 23.1, 20.4, 19.2, 18.2, 13.7; HRMS (ESI) Calcd for C29H34N4O5S [M + H]+ 551.2328, found: 551.2294.
17-((4-(4-Fluorophenyl)-3-methylthiazol-2(3H)-ylidene)hydrazineylidene)-2H-5α,8α-epidioxyandrost-6-en-3β-ol (7i )
Yellow solid, yield 80%, mp 167.3–168.7 °C; 1H NMR (600 MHz, CDCl3) δ 7.25 (s, 2H, Ar-H), 7.06 (s, 2H, Ar-H), 6.47 (d, J = 8.5 Hz, 1H, C6-H), 6.23 (d, J = 8.5 Hz, 1H, C7-H), 5.81 (s, 1H, C = CH), 4.23 (s, 1H, C3-H), 3.90 (s, 1H, OH), 3.21 (s, 3H, N-CH3), 2.66 (s, 1H), 2.56 (s, 1H), 2.06 (s, 1H), 1.98 (s, 1H), 1.88 (d, J = 12.6 Hz, 2H), 1.83 (s, 1H), 1.65 (s, 2H), 1.57 (s, 1H), 1.51 (s, 1H), 1.38 (s, 1H), 1.36 (s, 1H), 1.25 (s, 1H), 1.18 (s, 2H), 1.00 (s, 3H), 0.85 (s, 3H); 13C NMR (150 MHz, CDCl3) δ 167.8, 135.9, 132.3, 131.0, 130.6, 130.4, 128.9, 116.0, 115.9, 82.4, 79.3, 66.4, 65.6, 51.6, 49.5, 45.8, 37.2, 36.9, 34.7, 34.4, 30.6, 30.1, 29.8, 23.1, 20.5, 19.2, 18.5, 18.3, 13.7; HRMS (ESI) Calcd for C29H34FN3O3S [M + H]+ 524.2383, found: 524.2344.
17-((4-(4-Bromophenyl)-3-methylthiazol-2(3H)-ylidene)hydrazineylidene)-2H-5α,8α-epidioxyandrost-6-en-3β-ol (7j )
Yellow solid, yield 75%, mp 171.2–172.5 °C; 1H NMR (600 MHz, CDCl3) δ 7.57 (s, 1H, Ar-H), 7.53 (s, 1H, Ar-H), 7.22 (d, J = 8.1 Hz, 2H, Ar-H), 6.54 (d, J = 8.5 Hz, 1H, C6-H), 6.30 (d, J = 8.5 Hz, 1H, C7-H), 5.90 (s, 1H, C = CH), 4.31 (s, 1H, C3-H), 3.97 (s, 1H, OH), 3.28 (s, 3H, N-CH3), 2.72 (s, 1H), 2.63 (s, 1H), 2.12 (s, 1H), 2.05 (s, 1H), 1.95 (d, J = 13.9 Hz, 2H), 1.90 (s, 1H), 1.72 (s, 2H), 1.64 (s, 1H), 1.57 (s, 1H), 1.44 (d, J = 7.6 Hz, 2H), 1.32 (s, 1H), 1.25 (s, 2H), 1.07 (s, 3H), 0.92 (s, 3H). 13C NMR (150 MHz, CDCl3) δ 173.1, 167.8, 135.8, 132.0, 130.9, 130.2, 128.8, 123.3, 82.3, 79.2, 66.4, 65.6, 51.6, 49.4, 45.8, 37.1, 36.9, 34.6, 34.4, 30.6, 30.1, 29.7, 27.6, 23.1, 20.4, 19.2, 18.5, 18.2, 13.7; HRMS (ESI) Calcd for C29H34BrN3O3S [M + H]+ 584.5744, found: 584.5762.
17-((4-(p-Tolyl)-3-methylthiazol-2(3H)-ylidene)hydrazineylidene)-2H-5α,8α-epidioxyandrost-6-en-3β-ol (7k )
Yellow solid, yield 79%, mp 165.7–166.4 °C; 1H NMR (600 MHz, CDCl3) δ 7.72 (s, 1H, Ar-H), 7.53 (s, 1H, Ar-H), 7.27 (s, 2H, Ar-H), 6.54 (d, J = 8.5 Hz, 1H, C6-H), 6.30 (d, J = 8.4 Hz, 1H, C7-H), 5.84 (s, 1H, C = CH), 4.31 (s, 1H, C3-H), 3.97 (s, 1H, OH), 3.28 (s, 3H, N-CH3), 2.72 (s, 1H), 2.63 (s, 1H), 2.40 (s, 3H), 2.13 (s, 1H), 2.06 (s, 1H), 1.96 (d, J = 11.4 Hz, 2H), 1.90 (s, 1H), 1.73 (s, 2H), 1.63 (s, 1H), 1.58 (s, 1H), 1.45 (d, J = 7.5 Hz, 1H), 1.44 (s, 1H), 1.32 (s, 1H), 1.25 (s, 2H), 1.07 (s, 3H), 0.92 (s, 3H); 13C NMR (150 MHz, CDCl3) δ 172.6, 167.8, 135.8, 132.3, 131.0, 130.4, 129.4, 128.9, 128.6, 82.4, 79.3, 66.4, 65.6, 51.6, 49.5, 45.7, 37.2, 36.9, 34.7, 34.4, 30.6, 30.1, 27.6, 23.1, 21.3, 20.5, 19.2, 18.5, 18.3, 13.8; HRMS (ESI) Calcd for C30H37N3O3S [M + H]+ 520.2634, found: 520.2594.
17-((4-(4-Methoxyphenyl)-3-methylthiazol-2(3H)-ylidene)hydrazineylidene)-2H-5α,8α-epidioxyandrost-6-en-3β-ol (7l )
Yellow solid, yield 82%, mp 167.6–168.2 °C; 1H NMR (600 MHz, CDCl3) δ 7.34 (s, 1H, Ar-H), 6.94 (d, J = 10.8 Hz, 2H, Ar-H), 6.86 (s, 1H, Ar-H), 6.54 (d, J = 8.5 Hz, 1H, C6-H), 6.30 (d, J = 8.4 Hz, 1H, C7-H), 5.90 (s, 1H, C = CH), 4.31 (s, 1H, C3-H), 3.98 (s, 1H, OH), 3.83 (s, 3H, N-CH3), 3.30 (s, 3H), 2.73 (s, 1H), 2.64 (s, 1H), 2.13 (s, 1H), 2.05 (s, 1H), 1.96 (d, J = 11.4 Hz, 2H), 1.90 (s, 1H), 1.73 (s, 2H), 1.64 (s, 1H), 1.57 (s, 1H), 1.44 (d, J = 7.7 Hz, 2H), 1.32 (s, 1H), 1.26 (s, 2H), 1.07 (s, 3H), 0.92 (s, 3H); 13C NMR (150 MHz, CDCl3) δ 167.8, 159.7, 135.8, 132.3, 131.0, 130.4, 129.8, 128.9, 121.0, 114.4, 82.3, 79.3, 66.4, 65.6, 55.4, 51.6, 45.8, 37.1, 36.9, 34.6, 34.4, 30.6, 30.1, 29.7, 23.1, 20.4, 19.2, 18.5, 18.2, 13.7; HRMS (ESI) Calcd for C30H37N3O4S [M + H]+ 536.2583, found: 536.2554.
17-((4-(4-Chlorophenyl)-3-methylthiazol-2(3H)-ylidene)hydrazineylidene)-2H-5α,8α-epidioxyandrost-6-en-3β-ol (7m )
Yellow solid, yield 86%, mp 174.1–175.5 °C; 1H NMR (600 MHz, CDCl3) δ 7.62 (d, J = 105.5 Hz, 1H, Ar-H), 7.41 (d, J = 8.5 Hz, 2H, Ar-H), 7.29 (s, 1H, Ar-H), 6.54 (d, J = 8.5 Hz, 1H, C6-H), 6.30 (d, J = 8.5 Hz, 1H, C7-H), 5.90 (s, 1H, C = CH), 4.31 (s, 1H, C3-H), 3.97 (s, 1H, OH), 3.28 (s, 3H, N-CH3), 2.72 (s, 1H), 2.63 (s, 1H), 2.15 (s, 1H), 2.05 (s, 1H), 1.95 (d, J = 13.8 Hz, 2H), 1.90 (s, 1H), 1.73 (s, 2H), 1.64 (s, 1H), 1.57 (s, 1H), 1.50 (s, 1H), 1.44 (d, J = 7.6 Hz, 1H), 1.32 (s, 1H), 1.25 (s, 2H), 1.07 (s, 3H), 0.92 (s, 3H); 13C NMR (150 MHz, CDCl3) δ 173.0, 167.8, 139.6, 135.8, 132.3, 130.9, 130.4, 129.9, 129.1, 128.8, 82.3, 79.2, 66.3, 65.6, 51.6, 49.4, 45.8, 37.1, 36.8, 34.6, 34.4, 30.5, 30.1, 27.6, 23.1, 20.4, 19.2, 18.2, 13.7; HRMS (ESI) Calcd for C29H34ClN3O3S [M + H]+ 540.2088, found: 540.2062.
17-((4-(4-Hydroxyphenyl)-3-methylthiazol-2(3H)-ylidene)hydrazineylidene)-2H-5α,8α-epidioxyandrost-6-en-3β-ol (7n )
Yellow solid, yield 85%, mp 165.5–167.3 °C; 1H NMR (600 MHz, CDCl3) δ 7.11 (d, J = 8.5 Hz, 2H, Ar-H), 6.79 (d, J = 8.4 Hz, 2H, Ar-H), 6.46 (d, J = 8.4 Hz, 1H, C6-H), 6.22 (d, J = 8.4 Hz, 1H, C7-H), 5.74 (s, 1H, C = CH), 4.24 (s, 1H, C3-H), 4.05 (s, 1H, OH), 3.92 (s, 1H, OH), 3.19 (s, 3H, N-CH3), 2.64 (s, 1H), 2.55 (s, 1H), 2.06 (s, 1H), 2.01 (s, 1H), 1.89 (d, J = 11.6 Hz, 2H), 1.80 (s, 1H), 1.65 (s, 2H), 1.57 (s, 1H), 1.50 (s, 1H), 1.38 (s, 1H), 1.36 (s, 1H), 1.23 (s, 1H), 1.18 (s, 2H), 0.99 (s, 3H), 0.85 (s, 3H); 13C NMR (150 MHz, CDCl3) δ 166.8, 134.8, 131.3, 130.0, 129.4, 129.2, 127.8, 114.7, 81.3, 78.3, 65.5, 64.6, 59.5, 50.5, 48.4, 44.8, 36.1, 33.6, 29.5, 29.1, 28.7, 26.5, 22.1, 19.4, 18.2, 17.5, 17.2, 13.1, 12.7; HRMS (ESI) Calcd for C34H37N3O4S [M + H]+ 522.2427, found: 522.2403.
in Vitro Cytotoxic Activity
Human MCF-7, HepG2, HCT-116, and A549 cancer cells in exponential growth period were cultured in moderate RPMI 1640 medium (Corning, USA) containing 100 kU/L of penicillin, 10% fetal bovine serum (FBS) and 100 mg/L streptomycin. The culture vessels were placed in a humidified atmosphere of 5% CO2. The cytotoxic activity was evaluated by MTT assay (Beyotime). Cells were cultured into 96-well plates (100 μL per well) at a concentration of 1 × 104/mL for 12 h. Then the cancer cells were treated with different concentrations of synthesized compounds for 48 h before MTT solution (10 µL) was added to each well for another 2 h. The MTT solution was poured off and then dimethyl sulfoxide (DMSO) 100 μL was added. The optical density (OD) value of each well was recorded using a microplate reader. The IC50 value of the compound was calculated by SPSS analysis. 32
Determination of Morphological Changes of Cells
HepG2 cells were seeded into 6-well plates at a concentration of 1 × 106 cells/well and cultured for 12 h. Following
Apoptosis Analysis by Flow Cytometry
HepG2 cells were seeded on six-well plates, and then incubated with
Analysis of Mitochondrial Membrane Potential
HepG2 cells were treated with
Western Blot Analysis
HepG2 cells were treated with
Conclusions
In summary, a series of endoperoxide steroid derivatives (
Supplemental Material
sj-doc-1-npx-10.1177_1934578X231175588 - Supplemental material for Novel Endoperoxide Steroid Derivatives Containing Thiazole Moiety as Potential Antitumor Agents: Design, Synthesis, and Cytotoxic Evaluation
Supplemental material, sj-doc-1-npx-10.1177_1934578X231175588 for Novel Endoperoxide Steroid Derivatives Containing Thiazole Moiety as Potential Antitumor Agents: Design, Synthesis, and Cytotoxic Evaluation by Gang Li, Xiaoshan Guo, Wenkang Ren, Jiafeng Wang, Zhen Lv, Hongling Li, Mingrui Sun, Xiaoming Li, Gang Chen, Zhiguo Zhang, Wenting Zhang and Ming Bu in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Fundamental Research Funds for Education Department of Heilongjiang Province (No. 2019-KYYWF-1217).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
