Abstract
Sideritis caesarea H. Duman, Aytaç & Başer of the family Lamiaceae is an endemic plant with different ethnobotanical uses in Anatolia. In this present study, the antioxidant and biological activity potential of different S caesarea extracts and purified compounds were evaluated in vitro using DPPH•, CUPRAC, and ABTS• methods. In addition, their urease and chymotrypsin enzyme inhibitory activities, antimicrobial potential, and total phenol and flavonoid contents were determined. The experimental results showed that the two isolated flavonoids {4′-O-methylhypolaetin-7-O-[6″′-O-acetyl-β-D-allopyranosyl-(1→2)]-6″-O-acetyl-β-D-glucopyranoside, and isoscutellarein-7-O-[6″′-O-acetyl-β-D-allopyranosyl-(1→2)]-6″-O-acetyl-β-D-glucopyranoside}, as well as their fractions, except the n-hexane and water residue, showed remarkable antioxidant activity. Interestingly, the ethyl acetate and water residue fractions inhibited the urease enzyme activity by 49.6% and 49.1%, respectively; while the n-hexane and dichloromethane fractions attracted attention with their relative high chymotrypsin inhibitory activity of 85.8%, and 82.4%, respectively. When evaluated for their in vitro antimicrobial potential, including Helicobacter pylori, only Bacillus subtilis was susceptible to the n-butanol fraction at 1 mg/mL, while all of the studied samples showed activity against the human pathogenic yeast Candida albicans with the range of 0.25 to 1 mg/mL minimum inhibitory concentrations. The initial antioxidant activity, enzyme inhibitions, and antimicrobial results of S caesarea extracts and compounds suggest the further potential for in vivo evaluations.
Introduction
Herbal drugs and natural products reflect a wide range of diverse biological activities and also have an important potential for modern drug discovery and development. Ethnobotanical resources also contribute towards drug discovery. From this perspective, different in vitro and in vivo antioxidants, antimicrobial, and enzyme inhibitory assays are initial steps. Reactive oxygen species are unstable molecules such as superoxide anion, hydrogen peroxide, and hydroxyl radical, which are formed by partial reduction of oxygen in normal metabolic processes. In cases when their amount is higher than usual or when there is a decrease in cellular antioxidant capacity, this can cause lipid peroxidation and tissue damage. Studies have shown that this oxidative damage has an important role in the pathogenesis of gastric ulcers, and antioxidant compounds have a protective role in gastric ulcerogenesis. Therefore, it is preferable to support urease inhibitory activity with antioxidant activity.1,2
Helicobacter pylori, which is associated with gastritis, peptic ulcer, and gastric cancer, is one of the most common bacterial infections affecting about half of the world population. It was declared as a type-1 carcinogen by the World Health Organization in 1994. 3 H pylori has to reach the mucus layer to survive in the stomach lumen, which has a strong acid pH under normal conditions. The bacterium releases ammonia by breaking down urea secreted from stomach epithelium cells with the urease enzyme (EC 3.5.1.5) it produces. The released ammonia abnormally increases the pH and creates a suitable environment for the bacterium to live.4,5 Thus, the urease enzyme has a vital role in the survival and colonization of H pylori in the stomach tissue. In the current approach, urease inhibitors have a great impact on the discovery of new entities and compounds as new drug candidates for the treatment of peptic ulcers. Consequently, the discovery of effective and inexpensive urease enzyme inhibitors from natural products without side effects is extremely important in the search for new antiulcerogenic drugs.
Chymotrypsin (EC 3.4.21.1) is secreted from the pancreas as a proenzyme (chymotrypsinogen) and is converted to chymotrypsin by trypsin. It is important in many physiological events in mammals, such as the digestion of proteins, fibrin clots, and the prevention of cancer by removing proteins around cancer cells, among other roles. 6 On the other hand, excess amino acids are used as substrates in glucose or acetyl-CoA production, which is a precursor to the synthesis of triglycerides which are converted into fat for storage. Inhibition of chymotrypsin, which plays a role in protein digestion, may also be beneficial in the treatment of obesity.7,8 Therefore, it is important to find inhibitors that can be administered orally to regulate the activity of chymotrypsin.
The Sideritis genus is represented by more than 150 species, especially in the countries around the Mediterranean Sea. 9 The Anatolian Peninsula hosts up to 45 Sideritis species, according to “A Checklist of the Flora of Turkey (Vascular Plants).” 10 Sideritis species are recognized by different names such as “mountain grass, mountain tea, ironwort, té amarillo, and te de monte” in European countries, and are traditionally used in the treatment of diseases such as bronchitis, asthma, colds, hypertension, urinary system infections, peptic ulcer, rheumatism, and as an antimicrobial agent. 11 In Anatolia, it is mostly known as “adaçayı” and “dağ çayı,” and is used for ailments such as abdominal pain, digestive problems, stomach ache, hemorrhoids, colds, and as a stimulant, among many other uses.12–14 Since Sideritis caesarea H. Duman, Aytaç & Başer was introduced to the scientific world in 1998, the ethnobotanical data for this species is rather limited. However, decoction of this plant is used to treat stomach pain, and intestinal spasms, and as a sedative tea in the Pınarbaşı district of Kayseri. 13 Based on this ethnobotanical data, in an antiulcerogenic activity screening study by our team, S caesarea was determined to have a statistically significant activity for the prevention of ethanol-induced experimental ulcers in rats (95.8% ulcer inhibition, P < .001), 15 where mainly flavonoids were associated with the antiulcerogenic activity. 16
In this present study, to support the gastroprotective activity of S caesarea, in vitro antioxidant, urease inhibitory and antimicrobial activity on different microorganisms, including H pylori, were evaluated. In addition, total phenol, and total flavonoid content were also determined. Since the plant was recently introduced to the scientific world and considering the scarcity of scientific studies on it, its possible inhibition effect on chymotrypsin, which is considered one of the new drug discovery mechanisms against various cancer and inflammatory diseases, was also investigated, to the best of our knowledge for the first time.
Results and Discussion
S caesarea extraction and fractionation yielded 4.8 g n-hexane (SC-Hex), 4.4 g dichloromethane (SC-CH2Cl2), 23.2 g ethyl acetate (SC-EtOAc), 48.1 g n-butanol (SC-BuOH), and 40.4 g water residue fractions (SC-Rw), which were obtained by liquid–liquid fractionation of SC-EtOH. Two flavonoids {4′-O-methylhypolaetin-7-O-[6‴-O-acetyl-β-D-allopyranosyl-(1→2)]-6″-O-acetyl-β-D-glucopyranoside
In vitro urease and chymotrypsin inhibitory activity test results of the extract, fractions, and purified compounds obtained from S caesarea are listed in Table 1. After the evaluation, it was observed that the SC-EtOAc and SC-Rw fractions significantly inhibited urease enzyme by 49.61% and 49.14% (P < .0001), respectively, and the IC50 value of the SC-EtOAc fraction was determined as 177.5 µg/mL. The remaining studied extracts/fractions/compounds showed relatively low urease inhibitory activity. When the chymotrypsin inhibition test results were evaluated, the SC-Hex and SC-CH2Cl2 fractions showed relatively high inhibitory activity (85.8% and 82.4% inhibition, respectively, P < .0001) with IC50 values of 22.5 and 34.6 µg/mL, while the other extract/fractions/compounds showed relatively weak activity.
Urease and Chymotrypsin Inhibitory Activity Results of SC-EtOH Extract, Liquid–Liquid Fractions, and Flavonoids.

Standard error of the mean (n = 3).
% Inhibition at 200.0 µg/mL concentration.
% Inhibition at 76.9 µg/mL concentration.
Thiourea (reference for urease, inhibition at 25 μg/mL).
Phenylmethanesulfonyl fluoride (reference for chymotrypsin, inhibition at 4.8 μg/mL).
[*P < .05; **P < .01; ****P < .0001].
– No inhibitory activity.
The antimicrobial activity results of the studied extract and fractions are given in Table 2. Among the tested extract and fractions, only SC-BuOH showed an antibacterial effect against B subtilis at a concentration of 1 mg/mL; while all of them exhibited activity against C albicans (MIC values range from 0.25 to 1 mg/mL).
Antimicrobial Activity Results of S caesarea Ethanol Extract and Fractions (MIC, mg/mL).

–: no effect (>1 mg/mL); nt: not tested; *: µg/mL.
In vitro methods (CUPRAC, ABTS•, and DPPH•) were used to evaluate the antioxidant activity of the obtained extract/fractions/compounds. In the DPPH• method, SC-EtOAc, SC-BuOH,
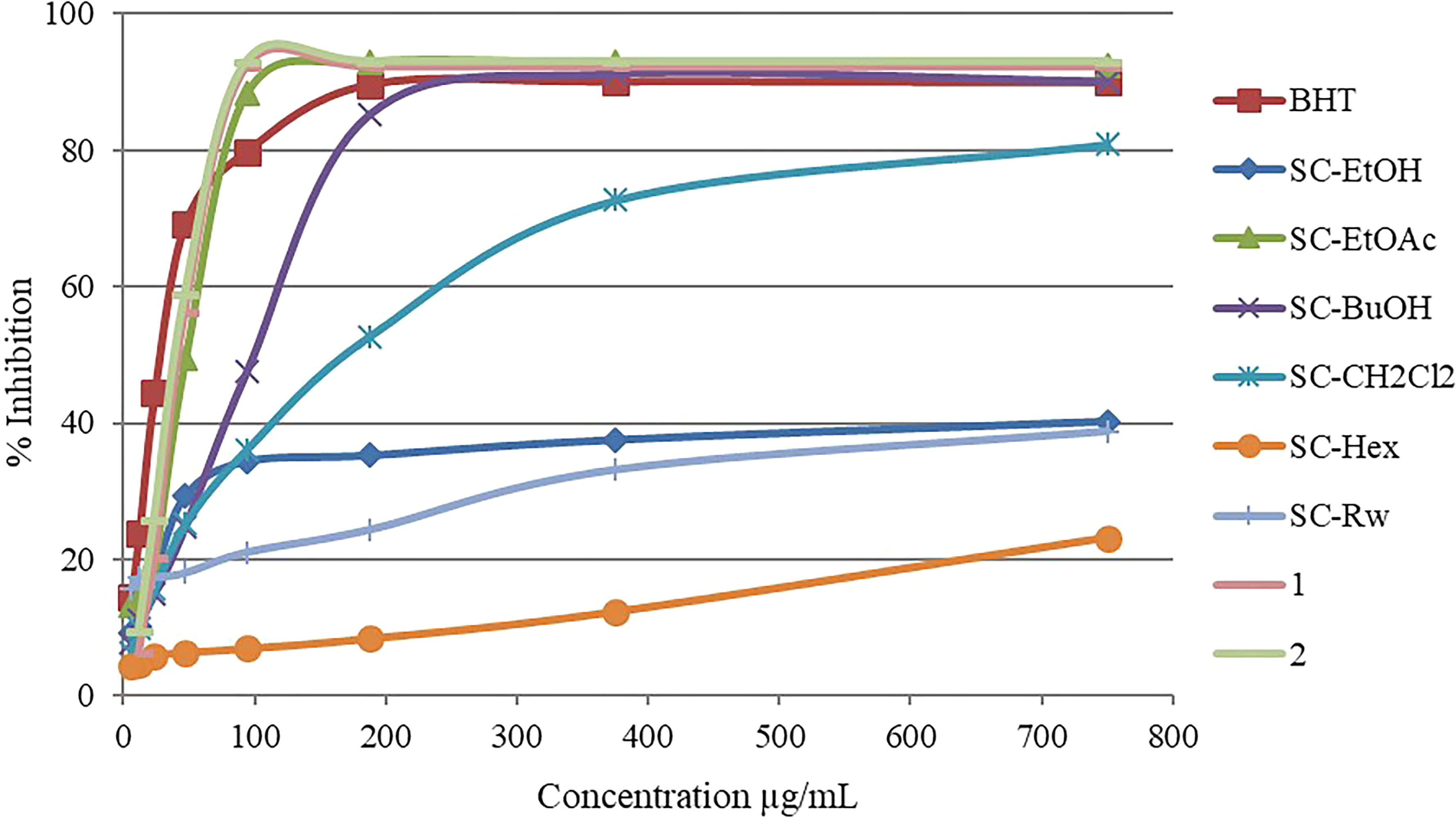
Free radical scavenging effect in DPPH• test at different concentrations of SC-EtOH, its liquid-liquid fractions, and isolated flavonoids.
Scavenging Effects and Cupric Ion Reducing Antioxidant Capacity of SC-EtOH, Liquid–Liquid Fractions, and Flavonoids.

Abbreviations: CUPRAC, cupric ion reducing antioxidant capacity assay; DPPH• (1,1-diphenyl-2-picrylhydrazyl) radical scavenging activity test; ABTS• [2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)] radical cation decolorization assay.
Standard error of the mean (n = 3), nt: not tested.
% Inhibition at 750.0 µg/mL concentration.
% Inhibition at 187.5 µg/mL concentration.
Gallic acid equivalent.
At 5.85 µg/mL concentration.
[**P < .01; ***P < .001; ****P < .0001].
In the ABTS• radical cation decolorization test, another method used to evaluate antioxidant activity, the highest activities were seen in the SC-EtOAc and SC-EtOH fraction (100% inhibition at 187.5 µg/mL). Although the increase in the concentrations of the studied test samples was observed to cause a serious increase in antioxidant activities, this effect was limited in the SC-Rw and SC-Hex fractions. The effects of the samples studied in this method are as follows: SC-EtOH = SC-EtOAc > SC-BuOH > SC-CH2Cl2 >

Free radical scavenging effect of the studied samples in ABTS• assay at different concentrations of SC-EtOH, its liquid–liquid fractions, and isolated flavonoids.
In the CUPRAC assay, the studied extract/fractions generally exhibited close activity to each other, but were lower than that of gallic acid (in the range of 121.55 and 279.51 mg/g gallic acid equivalent). However, flavonoids 1 and 2, purified from SC-EtOAc, exhibited concentration-dependent strong activity, although not as much as that of gallic acid, 665.41 and 650.96 mg/g gallic acid equivalents, respectively (Figure 3, Table 3).

Measured absorbances in the CUPRAC test at different concentrations of SC-EtOH, its liquid–liquid fractions, and isolated flavonoids. High absorbance indicates relatively higher antioxidant activity.
The total phenolic and flavonoid contents of the SC-EtOH extract, which was the main extract obtained from S caesarea, and its liquid–liquid fractions, are illustrated in Table 4, where the SC-EtOH extract showed 70.6 mg gallic acid equivalent/g extract, as well as total phenolic content and 121.0 mg rutin equivalent/g extract total flavonoid content. SC-EtOAc, which was obtained from the main extract, had relatively high total phenolic and flavonoid contents with 149.3 mg gallic acid equivalent/g extract and 269.2 mg rutin equivalent/g extract, respectively. Total phenolic contents of the liquid–liquid fractions obtained from the main extract were according to their amount: SC-EtOAc > SC-BuOH > SC-CH2Cl2 > SC-Hex > SC-Rw, while their total flavonoid contents were as follows: SC-EtOAc > SC-BuOH > SC-Rw > SC-CH2Cl2 > SC-Hex (Table 4).
Total Phenolic and Flavonoid Contents of S caesarea Ethanol Extract and Liquid–Liquid Fractions.

Gallic acid equivalent.
Standard error of the mean (n = 3).
Rutin equivalent.
As is well known, urease is a metalloenzyme with Ni+2 ions on its active site. Hence, aromatic hydroxyl groups of flavonoids or other phenolic molecules may interact with the nickel moiety of the enzyme and exhibit inhibitory activity. 17 Flavonoids, such as quercetin, apigenin, and luteolin, were previously reported to have urease inhibitory activity. 18 In the present research, the observation of urease inhibitory activity of the ethyl acetate extract (SC-EtOAc), which is rich in total phenol and flavonoid content, suggests confirmation of these findings; however, the ineffectiveness of the isolated flavonoids speculates that the activity of the ethyl acetate fraction may probably be due to other compounds.
To the best of our knowledge, the in vitro urease and chymotrypsin inhibitory activity of S caesarea was evaluated for the first time. However, the urease inhibitory activity of three different Sideritis species was previously reported.19,20 In the first of these studies, the n-hexane and methanol extract of S pisidica Boiss. & Held. (200 µg/mL) showed urease inhibitory activity close to that of the standard urease inhibitor thiourea with 78.9% and 66.6% inhibition, respectively, while the essential oil and acetone extract demonstrated low activity. 19 In another study, the hexane, acetone, and methanol extracts of S albiflora Hub.-Mor., and S leptoclada O. Schwarz & P.H. Davis extracts were determined to have strong urease inhibitory activity with IC50 values of 1.02 to 5.10 µg/mL. 20
In the present work, the ethanol extract, n-hexane, and dichloromethane fractions from S caesarea showed inhibitory activity of not more than 20% in the same concentration, as listed in Table 1. It is well known that members of the Sideritis genus offer a wide and diverse range of phytochemicals, such as terpenes, flavonoids, iridoids, coumarins, lignans, sterol derivatives, and essential oils. 11 Thus the reason for the partially lower activity of S caesarea, compared to the other three Sideritis species, may be associated with the differences in phytochemical content and composition, which could be influenced by the location of the collection area, climatic conditions, and genetic factors. The chymotrypsin inhibitory activity results of S caesarea are quite interesting. Nonpolar fractions (SC-Hex and SC-CH2Cl2) showed significant inhibition of chymotrypsin at very low concentrations, such as 22.5 and 34.6 µg/mL IC50 values, respectively (Figure 4).
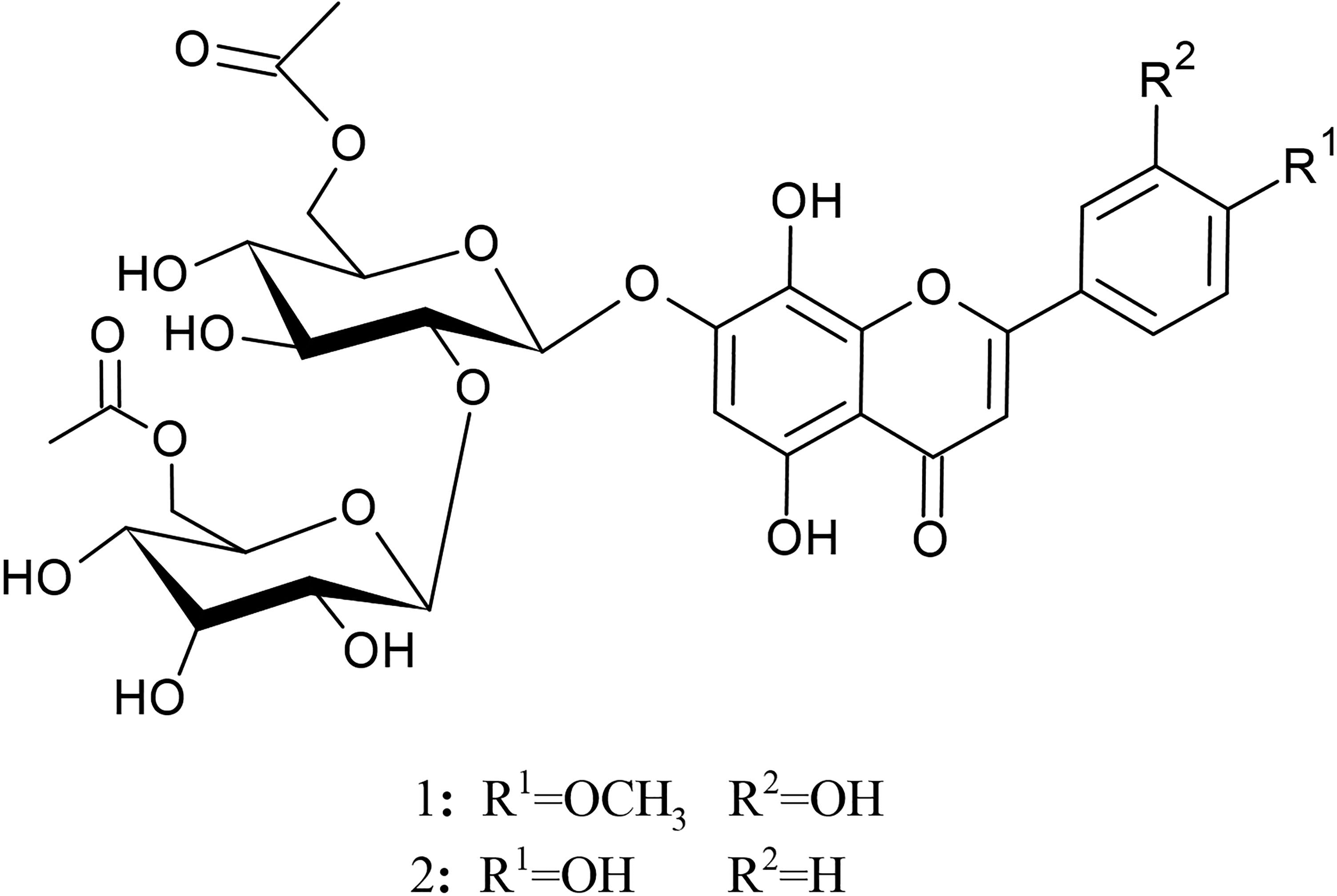
Structures of isolated flavonoids.
In the present research, only the n-butanol fraction inhibited the pathogen Bacillus subtilis at 1 mg/mL, while all of the studied samples showed activity against the human pathogenic yeast Candida albicans with 0.25 to 1 mg/mL minimum inhibitory concentrations. No inhibitory activity was observed against the other tested and evaluated human pathogenic microorganisms such as Staphylococcus aureus, Staphylococcus epidermidis, Pseudomonas aeruginosa, Klebsiella pneumonia, Listeria monocytogenes, and Helicobacter pylori. There are three previous studies on the antimicrobial evaluation of S caesarea. The first reported the antifungal activity of the methanol extract of the aerial parts of 8 plants from the Lamiaceae, including S caesarea. No fungicidal effect against Aspergillus flavus, A niger, A ochraceus, and Fusarium proliferatum was detected, with MIC values > 1.6 mg/mL. However, the tested extracts showed a fungistatic effect at concentrations of 0.1 mg/mL and above (P < .001). 21 In another study, antimicrobial activity of the methanol extract at varying degrees (inhibition zones between 8 and 16 mm at concentrations in the range of 1%-10%) was reported against Aeromonas hydrophila, Bacillus brevis, B cereus, B subtilis, B subtilis var. niger, Escherichia coli, Klebsiella pneumoniae, Morgenella morganii, Mycobacterium smegmatis, Proteus mirabilis, Pseudomonas aeruginosa, Staphylococcus aureus, Yersinia enterocolitica, Candida albicans, and Saccharomyces cerevisiae. 22 In a previous study of our group, antimicrobial effects of different S caesarea essential oils from different localities, against six pathogenic microorganisms (Escherichia coli, Staphylococcus aureus, Pseudomonas aeruginosa, Bacillus subtilis, Staphylococcus epidermidis, and Candida albicans) were evaluated. The tested essential oils showed varying degrees of growth inhibition (inhibition zones: 0.7-1.3 mm), where the highest inhibition zones (1.3 mm) were observed against Staphylococcus aureus; none of the essential oil samples showed inhibition against Candida albicans at the tested concentrations. 23
Free radicals are known to have a role in peptic ulcer formation. Therefore, the antioxidant activities of the studied extract/fractions/compounds were also investigated. Antioxidant activity determination methods may be evaluated under two headings based on mechanism, hydrogen transfer method, and electron transfer method. An example of a method based on hydrogen transfer is DPPH•; while CUPRAC is an example of an antioxidant activity method based on electron transfer. Moreover, ABTS• is an antioxidant activity determination assay that can be evaluated under both the hydrogen transfer and electron transfer methods.24,25 In the DPPH• method, purple-colored 1,1-diphenyl-2-picrylhydrazyl radical turns to yellow by hydrogen donation, and radical scavenging activity is determined by the decrease in absorbance of the purple color of the DPPH• radical at 517 nm wavelength. In the CUPRAC method, Cu2+ is reduced to Cu+, which forms a complex with neocuproine, thus increasing the characteristic absorbance of Cu+-neocuproin chelate at 450 nm, which is indicative of antioxidant activity.24,26 In the ABTS• method, the loss of green-blue color that of ABTS• radical produced by oxidation of 2,2 3-azinobis (3-ethylbenzothiazoline-6-sulfonic acid) with persulfate is determined by the reading absorbance at 734 nm.
27
In the current research, ethyl acetate and butanol fractions of S caesarea were shown to be quite rich in phenolics and flavonoids, which seemed to correlate with their antioxidant activity. The strong antioxidant activity of the flavonoids isolated from this fraction indicates that these flavonoids (
Although there are many studies evaluating the antioxidant activities of different Sideritis species, the literature survey revealed that there is only one study on the antioxidant activity and phenol content of S caesarea. In the aforementioned research, the total phenol content of the methanol extract (93.8 mg gallic acid equivalent/g extract) was comparable to the value observed for SC-EtOH in this present study (70.6 mg gallic acid equivalent/g extract), but in the DPPH• test, the methanol extract showed relatively higher radical scavenging activity (72% inhibition at a dose of 100 ppm) than SC-EtOH.
22
In a previous study, flavonoid
In another study, the antioxidant activity of flavonoid
When the phenol and flavonoid contents are examined, it can be justified that the results are generally compatible. Only a difference in the water residue fraction's phenol and flavonoid content was remarkable, with the lowest phenol content, but moderate flavonoid content. It was concluded that the flavonoids, with relatively high polar properties, were associated with their proportional concentration within the polar fractions such as in the ethyl acetate, butanol, and water residue fractions. On the contrary, phenolic compounds other than flavonoids in the extracts were relatively in the medium polar range with less solubility in nonpolar solvents such as dichloromethane and n-hexane.
As a result, the fractions and compounds obtained from S caesarea showed relatively strong antioxidant and antifungal activity, while strong inhibitory activity on chymotrypsin was observed in the n-hexane and dichloromethane fractions. According to the urease inhibition results, the ethyl acetate and the water residue fractions showed strong inhibition. According to our previous in vivo antiulcerogenic activity results 16 on S caesarea, the ethyl acetate fraction had the strongest antiulcerogenic activity. The water residue fraction also showed antiulcerogenic activity, but when compared to the other fractions, was relatively lower. The urease inhibitory activity results of the fractions, especially the ethyl acetate fraction, which showed also an vivo antiulcerogenic effect, suggested that these aforementioned fractions may help alleviate the unwanted effects of H pylori in the stomach. Therefore, it can be suggested that the most effective fraction against peptic ulcers is the ethyl acetate fraction along with the antiulcerogenic effective compounds, which were determined in the previous research.
Conclusion
The antiulcerogenic activity of S caesarea, used in folk medicines for symptoms resembling those of peptic ulcer, 13 is associated with multiple pathways such as antioxidant, cytoprotective activity, and inhibition of the urease enzyme produced by H pylori. Further development of new drugs for the treatment of peptic ulcer with multiple different mechanisms, including toxicity studies, are still needed with in vivo, and clinical evaluations.
Material and Methods
Materials
Jack bean urease (Type-III powder, 15.000-50.000 Unit/g solid, Sigma-Aldrich), ethanol (96% Tekkim), n-hexane (ACS Reagent, Reag. Ph. Eur., ≥ 99% Riedel-de Haën), dichloromethane (EMSURE® ACS, ISO, Reag. Ph Eur Merck), ethyl acetate (ACS Reagent, ≥ 99.5% Riedel-de Haën), n-butanol (EMSURE® ACS,ISO,Reag. Ph Eur Merck), DMSO (Emplura®, Merck), urea (ACS, Reag. Ph Eur., Merck), EDTA (anhydrous, crystalline BioReagent, suitable for cell culture, Sigma-Aldrich), lithium chloride (ACS reagent ≥ 99%, Sigma-Aldrich), phenol (ACS Reag. Ph Eur, GR for analysis, Merck), sodium nitroprusside dihydrate (purum p.a. ≥ 98.0%, Fluka), sodium hypochlorite (6%-14% active chlorine, Merck), thiourea (Reagent Plus® ≥ 99.0%, Sigma-Aldrich), sodium dihydrogen phosphate (Emprove® expert. Ph Eur, BP, USP, Merck), disodium hydrogen phosphate (Ph. Eur, BP, 98.5%-101%, Sigma-Aldrich), rutin (Merck), gallic acid (97.5%-102.5%, Sigma-Aldrich), butyl hydroxytoluene (European Pharmacopoeia Reference Standard, Sigma-Aldrich), copper (II) chloride (ACS reagent, ≥ 99.0%, Merck), copper (II) sulfate (ACS reagent, ≥ 98.0%, Sigma), sodium potassium tartrate (ACS, ISO, Reag. Ph Eur Merck), sodium hydroxide (for analysis ACS ISO, Carlo Erba), sodium carbonate (ACS reagent, anhydrous, ≥ 99.5% Sigma-Aldrich), sodium acetate (ACS reagent, ≥ 99.0%, Sigma-Aldrich), anhydrous sodium sulfate (ACS reagent, ≥ 99.0%, Sigma-Aldrich), aluminum chlorite (reagent grade, 98%, Sigma-Aldrich), Folin Ciocalteu reagent (Sigma-Aldrich), 1,1-diphenyl-2-picrylhydrazyl (97%, Sigma-Aldrich), potassium persulfate (ACS reagent, ≥ 99.0%, Merck), ammonium acetate (EMSURE® ACS, Reag. Ph Eur Merck), neocuproine (≥ 98% Sigma-Aldrich), 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (≥ 98% Sigma-Aldrich), HCl (Extrapure, DAB, PH Eur, BP, JP, NF, Merck), tris(hydroxymethyl)aminomethane hydrochloride (ACS reagent, ≥ 99.8% Sigma-Aldrich), α-chymotrypsin (from bovine pancreas Type II, lyophilized powder, ≥ 40 units/mg protein Sigma-Aldrich), N-succinyl phenylalanine-p-nitroanilide (Sigma-Aldrich) and phenylmethanesulfonyl fluoride [≥ 98.5% (GC) Sigma-Aldrich] were used.
Plant Material
Flowering aerial parts of Sideritis caesarea H. Duman, Aytaç & Başer were collected from the slopes on the east of the village (N: 38° 21′ 01″, E: 26° 30′ 57″) in Kayseri, Sarız district, Dayıoluk Village, and were dried at room temperature. Plant specimens were identified by Prof. Dr Ayşe Mine GENÇLER ÖZKAN, and herbarium specimens (GUE-3232) were stored in the Gazi University Faculty of Pharmacy Herbarium.
Extraction and Fractionation
The dried aerial parts of S caesarea (500 g) were coarsely ground and macerated at room temperature using 80% ethanol by mechanical stirring. The macerate was filtered, after several successive extractions. The filtrates were combined, and evaporated under reduced pressure, at temperatures below 45 °C. 137 g ethanol extract (SC-EtOH) was dissolved first in 90% methanol and further extracted with n-hexane. The remaining solvent was evaporated and dissolved using ∼ 800 mL of distilled water. The aqueous solution was successively extracted using dichloromethane, ethyl acetate, and n-butanol, respectively. Each fraction was evaporated under reduced pressure, where the remaining aqueous layer was lyophilized to yield a dry extract. The resulting ethyl acetate fraction was further fractionated by different chromatographic methods. Details of the S caesarea extraction and isolation were previously described. 16
Urease Inhibition Assay
Urease inhibitory activity was determined by measuring ammonia production using the indophenol method.
32
In this method, 25 µL of jack bean urease solution was incubated with 5 µL of the test sample at 30 °C for 15 min in a 96-well microplate. Then 55 µL of buffer containing 100 mM urea (0.01 M K2HPO4.3H2O, 1 mM EDTA, and 0.01 M LiCl) was added to this mixture and incubated again at 30 °C for 15 min. After incubation, 45 µL of phenol reagent (1% w/v phenol and .005% w/v sodium nitroprusside), and 70 µL alkaline reagent (0.5% w/v NaOH and NaOCl containing 0.1% active chloride) were added to each well. After 50 min, the increase in absorbance at 630 nm was measured using a microplate reader (VersaMax). The experiment was performed at pH 6.8, where thiourea was used as a standard urease inhibitor, in triplicate assays. IC50 values were calculated using GraphPad Prism 6 software. Percentage inhibition (I) was calculated by the formula below:
Chymotrypsin Inhibition Assay
α-Chymotrypsin inhibitory activity was determined by the method used by Cannell et al with slight modifications. Tris-HCl buffer (60 μL 50 mM, pH 7.6), 10 μL test solution, and 30 μL enzyme solution (36 U/mL) were pre-incubated at 37 °C for 15 min, and after this pre-incubation, the absorbance at 410 nm was measured using a microplate reader (Versa Max). After the addition of a 1.3 mM 30 μL substrate (N-succinyl phenylalanine-p-nitroanilide) solution, the reaction was initiated. After incubation for 75 min at 37 °C, the absorbances at 410 nm were measured again. Phenylmethanesulfonyl fluoride was used as a positive control, and the experiments were performed in triplicates. IC50 values were calculated using GraphPad Prism 6 software. Percentage inhibition (I) was calculated using the formula below
33
:
Antimicrobial Activity Assay
Staphylococcus aureus ATCC 25923, Staphylococcus epidermidis ATCC 14990, Pseudomonas aeruginosa ATCC 10145, Klebsiella pneumonia NCTC 9633, Listeria monocytogenes ATCC 19111, Bacillus subtilis NRRL B-4378, Helicobacter pylori ATCC 43504, and Candida albicans ATCC 90028 standard strains were obtained from the American Type Culture Collection (ATCC), Northern Regional Research Laboratory (NRRL), and National Collection of Type Cultures (NCTC). All microorganisms were stored at −85 °C or −20 °C.
Bacterial cultures were cultured on either Mueller Hilton Agar (MHA) or Mueller Hinton Broth (MHB) at 37 °C for 24 h for S aureus, S epidermidis, P aeruginosa, K pneumonia, L monocytogenes, and B subtilis. Columbia agar containing 5% (v/v) horse blood and Brucella broth containing 10% (v/v) fetal bovine serum were used for the H pylori strain, while the C albicans strain was incubated on Potato Dextrose Agar (PDA) and RPMI at 37 °C for 24 h. After incubation, microorganism culture densities were adjusted turbidometrically to McFarland No: 0.5 tube.34–36
A modified microdilution assay34,35,37,38 was used to determine the antimicrobial activity of the sample. A stock solution of the test sample was prepared in dimethyl sulfoxide (DMSO) and diluted with sterile distilled water. Ninety-six flat-bottom well microplates were used. One hundred microliters of diluted test samples were transferred, and serial dilutions (1-0.008 mg/mL) were prepared. A 100 μL of the 1:100 diluted bacterial suspensions, 1:10 diluted Helicobacter suspensions, and 1:1000 diluted Candida suspensions were then added to each well. After incubation at 37 °C for 24 h, for staining viable microorganisms, 20 µL 0.01% (w/v) resazurin solution was added and incubated again at 37 °C for another 3 h. The first well in which the blue color occurs was determined as the minimal inhibitory concentration (MIC, µg/mL). Clarithromycin, tetracycline, ketoconazole, and nystatin were used as standard antimicrobial agents at concentration ranges of 0.025 to 32 µg/mL. All experiments were repeated in triplicate, and average MICs are reported in Table 2.
Total Flavonoid Content Assay
Total flavonoid content was determined by the aluminum chloride colorimetric method. 39 A total of 500 µL test sample (in 75% ethanol) was mixed with 1500 µL 75% ethanol, 100 µL 10% aluminum chloride solution, 100 µL 1 M sodium acetate solution, and 2800 µL distilled water. After 30-minute incubation at room temperature, their absorbance at 415 nm was measured with a Varian Oxford AS400 spectrometer. The whole experiment was done in triplicate. Total flavonoid contents were calculated from the calibration curve obtained with the reference compound rutin.
Total Phenolic Content Assay
Total phenolic contents were determined by the Folin Ciocalteu method. 40 One hundred microliter of the test sample (in 75% ethanol) was mixed with 125 μL of Lowry C solution* and incubated at room temperature for 10 min. Then 12.5 μL of Folin Ciocalteu reagent (1/3 diluted with 96% ethanol) was added to the reaction mixture. The absorbance of the blue color that occurred after a 30-minute incubation at room temperature was measured with a Versa Max microplate reader at 750 nm. The whole experiment was made in triplicate. Total phenolic contents were calculated from the calibration curve obtained with gallic acid.
* Preparation of Lowry C solution:
Antioxidant Activity Assays
DPPH• (1,1-diphenyl-2-picrylhydrazyl) radical scavenging activity test: Serial dilutions were prepared from test samples in 75% ethanol. A 150 μL of test solution was incubated with 50 μL of 1 × 10−3 M DPPH• solution (in methanol) at room temperature in the dark. Then the absorbance was measured at 517 nm using a microplate reader (VersaMax). BHT (butylhydroxytoluene) was used as a reference, in triplicate experiments.
Percentage inhibitions (I) were calculated using the formula below
41
:
ABTS• [2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)] radical cation decolorization assay: Two mM ABTS• solution was prepared in 0.1 M phosphate buffer (pH 7.4). The formation of the ABTS• radical cation was achieved by adding potassium persulfate (2.45 mM) and stirring at room temperature in the dark for 12 h. Before usage, ABTS• solution was diluted in a 1:3 ratio with phosphate buffer, and serial dilutions were prepared from test samples in 75% ethanol. A 150 μL of sample solution was mixed with 50 μL ABTS• solution and incubated. After the 30-minute incubation in the dark at room temperature, absorbance was read at 734 nm with a microplate reader (Versa Max). Gallic acid was used as a reference, where the experiment was performed in triplicates, and % ABTS• scavenging activity was calculated using the formula below
27
:
Statistical Analysis
The data obtained from in vitro enzyme inhibition and antioxidant activity experiments were expressed as Standard Error Mean (± SEM). Differences between reference and sample groups were statistically evaluated using one-way ANOVA. Dunnett's multiple comparison tests were used as post-hoc tests. P < .05 was considered to be significant [*P < .05; **P < .01; ***P < .001, ****P < .0001].
Footnotes
Acknowledgments
This study was conducted as part of a doctoral thesis [Tuğba Günbatan, “Sideritis caesarea H. Duman, Aytaç & Başer bitkisinin antiülserojenik aktivitesi üzerinde araştırmalar,” Gazi University Institute of Health Sciences, Ankara, 2017].
Author Contributions
Research idea, İ. Gürbüz, T. Günbatan, and F. Demirci; Experiments, T. Günbatan, N. Karaca; Writing and interpretation of data, T. Günbatan, İ. Gürbüz, and F. Demirci; critical control, İ. Gürbüz, F. Demirci, and T. Günbatan.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly funded by the Scientific and Technological Research Council of Turkey [grant number TBAG-112S581]. This work is dedicated to those who lost their lives in the big devastating earthquakes of the sixth of February 2023, in Türkiye.
Ethical Approval
Not applicable.
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Statement of Human and Animal Rights
Not applicable.
